Abstract
Introduction:
Burn anemia represents a common complication following a burn injury. Burn anemia etiology carries distinct features occurring at each stage of the post-injury and treatment periods resulting from different causes. We aimed to analyze the use of blood components in Finnish burn victims and to identify patient- and injury-related factors influencing their use.
Methods:
To study the use of blood products in burn patients, we used data collected from the Optimal Use of Blood registry, developed through co-operation between 10 major hospital districts and the Finnish Red Cross Blood Service. Burn patients ⩾18 years treated at the Helsinki University Hospital between 2005 and 2011 with an in-hospital stay ⩾1 day who received at least one transfusion during their hospital stay were included in this study.
Results:
Among all 558 burn patients, 192 (34%) received blood products during their hospital stay. The transfused cohort comprised 192 burn patients. The study cohort received a total of 6087 units of blood components, 2422 units of leukoreduced red blood cells, 1728 units of leukoreduced platelets, and 420 units of single-donor fresh frozen plasma or, after 2007, 1517 units of Octaplas® frozen plasma. All three types of blood components were administered to 29% of patients, whereas 45% received only red blood cells and 6% received only Octaplas. Transfused patients were significantly older (p < 0.001), experienced fire-/flame-related accidents and burns to multiple locations (p < 0.001), and their in-hospital mortality exceeded that for non-transfused burn patients fivefold (p < 0.05).
Discussion:
We show that Finnish adult burn patients received ample transfusions. The number of blood components transfused varied according to the anatomical location of the injury and patient survival. Whether the additional mortality is related directly to transfusions or is merely a manifestation of the more severe burn injury remains unknown.
Introduction
Anemia commonly occurs as a complication following a burn injury affecting patients with as little as >10% total burn surface area (%TBSA) affected (1). Burn anemia etiology carries distinct features occurring at each stage of the post-injury and treatment periods resulting from different causes. During the first 2 weeks post-burn, anemia mostly occurs due to blood loss directly through the burned area, dilution due to resuscitation, and from the surgical management of wounds or the collateral traumatic injury (2). Later, anemia expresses similar to critical illness anemia (3) originating from frequent dressing changes and the serial extraction of blood by venipuncture (4), inadequate nutrition (5), and diminished erythropoiesis (4, 6). Critical illness anemia accounts for 50%–70% of additional transfusions among burn patients (2, 7).
Burn patients frequently receive multiple transfusions, and 75% of those diagnosed with >20%TBSA receive packed red blood cells (RBCs) (7). Along with the increase in %TBSA, the number of patients transfused increases synchronically along with the RBC units transfused (7, 8). The occurrence of comorbidities (9) and the use of anticoagulants (10) further increase the chance of transfusions during the hospital stay. The increased rate of infections among transfused burn patients represents another adverse association (7, 11).
Between 2005 and 2013, 26,093 persons received in- and outpatient treatment in Finnish hospitals for burns (National Hospital Discharge Register, unpublished data, National Institute for Health and Welfare). The risk of a hospital-treated burn injury among adults varies between 38 per 100,000 individuals in the ⩽75 years age group and 54 per 100,000 individuals in adults aged 18–49 years. Approximately, 100 fire-related deaths occur annually in Finland, a rate higher than in the majority of high-income countries (12). In Finland, the treatment of severe burns is centralized to two university hospital burn centers: Helsinki University Hospital and Kuopio University Hospital. While the basis for admittance for a severe burn to either unit is based on geographical location, the mean %TBSA of patients at Helsinki University Hospital was higher compared with that of patients admitted to Kuopio University Hospital (13, 14).
The aim of this study was to analyze and report on the use of blood components among Finnish burn victims treated at Helsinki University Hospital between 2005 and 2011. We used the data (from our hospital) collected for the Optimal Use of Blood registry (VOK), developed through co-operation between the Finnish Red Cross Blood Service (FRCBS) and 10 (out of 21) of the largest Finnish hospital districts between 2002 and 2011. We further aimed to identify patient- and injury-related factors influencing the use of blood components.
Methods
The Helsinki University Hospital ethics committee approved this study. We obtained permission to use the registry information for scientific research from the National Institute of Health and Welfare (THL) after consulting the data protection authority.
The FRCBS supplies blood products in Finland through a centralized system. As a research and development program, FRCBS initiated the Program for the Optimal Use of Blood (VOK Program) in 2002. As a collaborative project between FRCBS and Finnish hospitals, this program provides commensurate data on blood use in participating organizations. The database aims to improve the safety and outcomes of patient care (15). The VOK database covered the use of about 68% of blood components in Finland between 2002 and 2011. At the Helsinki University Hospital, the VOK database covered 100% of blood products used during the study period.
Data for the VOK registry were collected from hospitals’ administrative laboratory and operating room registers as well as from the FRCBS’ Blood Donor Registry. Data were collected annually or bi-annually. Patient identifications are encrypted in the VOK registry and cannot be traced back to individual patients. The database includes information about diagnoses, procedures, laboratory results, and the transfused blood components. The registry includes up to five discharge diagnoses, the first diagnosis serving as the principal cause for the hospital stay, and up to three operation codes. This database allows for the analysis of differences in the use of blood and the evaluation of clinical practices. Participating hospitals may also conduct specific analyses of data pertaining to their own institution.
For this study, we queried the VOK database for adults aged ⩾18 years, patients with International Classification of Diseases, 10th Revision (ICD-10) burn diagnoses T20*, T21*, T22*, T23*, T24*, T25*, T29*, T30*, T31*, and T32* treated as inpatients for ⩾1 day at the Helsinki University Hospital between 2005 and 2011. The query resulted in a sample of 558 adult burn victims. The data were further sorted to include patients given at least one transfusion during their treatment. These patients formed the “transfused” study group. Their demographic data, diagnoses, surgical treatment, the total number of blood products received, and blood work results were retrieved from the VOK registry database for statistical analysis. We included only patients who met the inclusion criteria and for whom data were available for all parameters in the analyses.
Blood Components
Blood components were prepared using the buffy coat method from whole blood. Red cells were suspended in a saline, adenine, glucose, and mannitol (SAGM) solution and filtered to remove leukocytes. The shelf life of red cell components was 35 days. Platelets (PLTs; one clinical dose) were prepared from four buffy coats of the same ABO and RhD blood group, suspended in preliminary alcohol-screening (PAS) and subjected to filtration to remove leukocytes. The shelf life of PLTs was 5 days. Plasma was centrifuged twice to remove blood cells and fast frozen to prepare fresh frozen plasma (FFP). Since 2007, only pharmaceutically manufactured virus-inactivated pooled FFP has been used in Finland. Blood components complied with European Union Directives (Directive 2002/98/EC and Commission Directive 2004/33/EC) and with the Council of Europe Guide for the Preparation, Use and Quality Assurance of Blood Components.
Statistical Analyses
We used the chi-square test for homogeneity using the Yates continuity correction to compare two different groups. The Mann–Whitney U test was applied to compare the medians of continuous variables from two different populations. We considered values of p < 0.05 as statistically significant. All statistical analyses were performed using R software, version 3.1.2 (16).
Results
Patient Characteristics
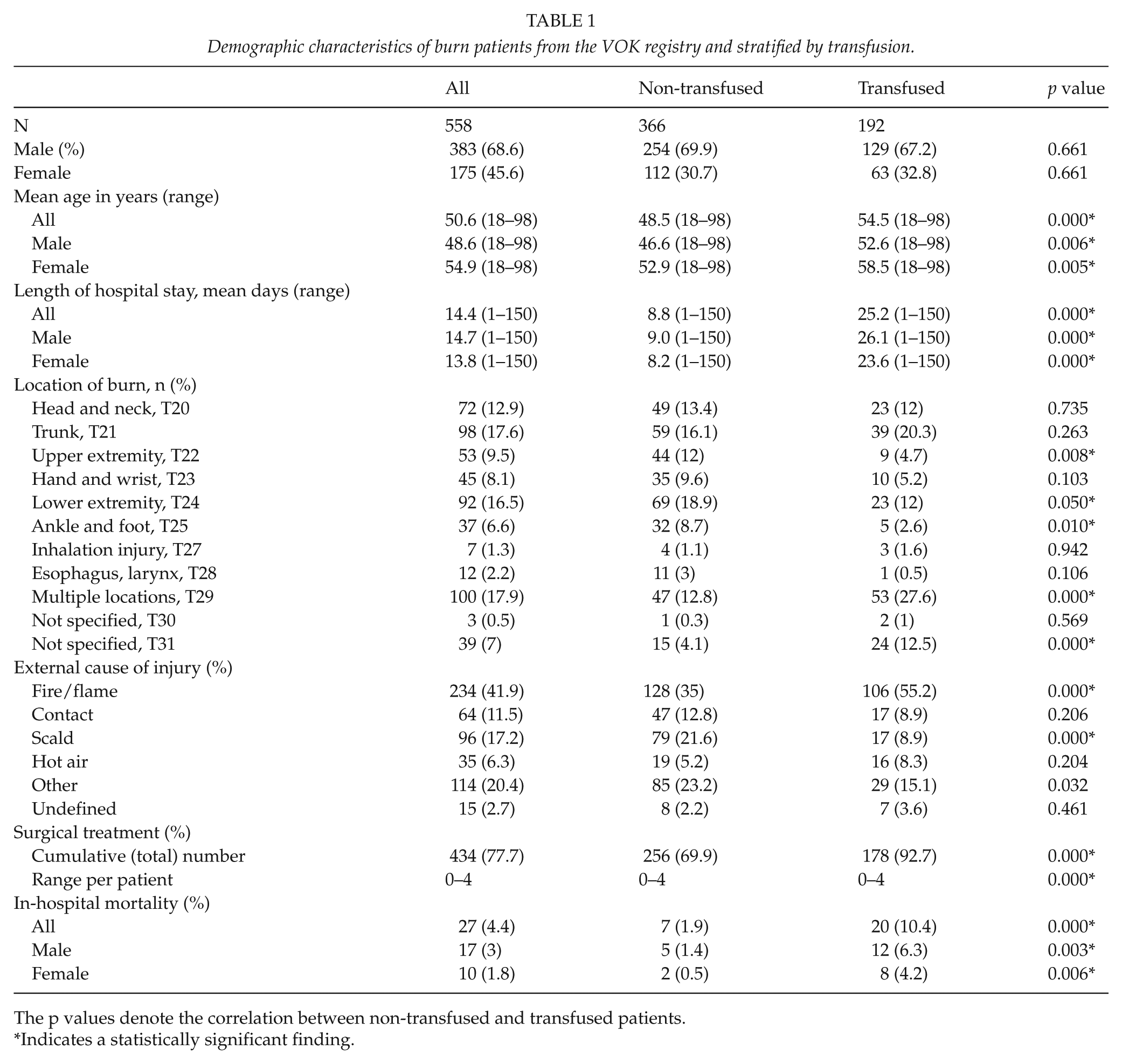
The study cohort comprised 192 burn patients who received at least one blood transfusion during their hospital stay. Table 1 provides the demographic data for all burn patients in the VOK registry and those included in this study.
Demographic characteristics of burn patients from the VOK registry and stratified by transfusion.
The p values denote the correlation between non-transfused and transfused patients.
Indicates a statistically significant finding.
Our study cohort included 129 (67%) males and 63 (33%) females with a gender ratio of 1:0.5. The median age of the study cohort was 54.5 years, where transfused patients were significantly older than those who did not receive transfusions (p < 0.001). The majority of cases were fire-/flame-related accidents (n = 105; 54.6%). In the transfused cohort, fire-/flame- and scald-related accidents predominated (p < 0.001).
Burns on multiple locations or unspecified burn locations were recorded more often among transfused patients compared with non-transfused burn patients (p < 0.001). Furthermore, the in-hospital mortality in the study cohort exceeded the hospital mortality of non-transfused burn patients nearly fivefold. Additionally, the excess hospital mortality stratified by gender among transfused patients was significantly higher (p < 0.05). Altogether, we recorded 178 burn operations in the study cohort (p < 0.001).
Blood Transfusions
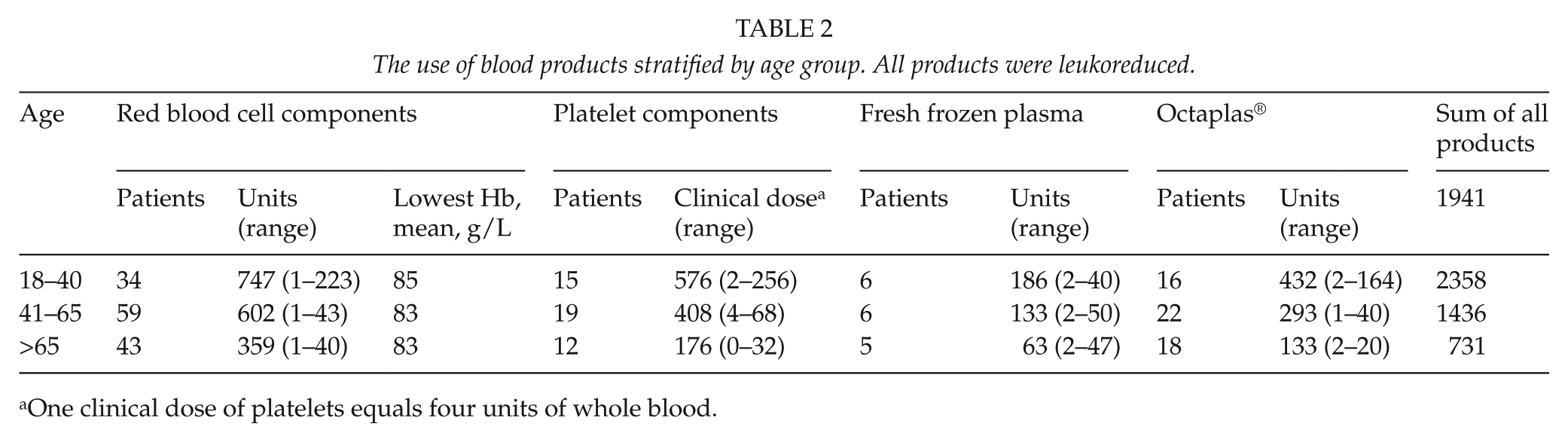
A total of 6087 units of blood components, including 2422 units of leukoreduced RBCs (one unit equals one bag or one clinical dose), 1728 units of leukoreduced PLTs (four units equal one bag or one clinical dosage), and 420 units of single-donor FFP or, after 2007, 1517 units of Octaplas® frozen plasma, were transfused during the hospital stay for the study cohort (Table 2). In further analyses, FFP and Octaplas were combined and labeled frozen plasma.
The use of blood products stratified by age group. All products were leukoreduced.
One clinical dose of platelets equals four units of whole blood.
All three types of blood components were administered to 55 (28.6%) patients, whereas 87 (45.3%) patients received only RBCs and 11 (5.7%) received only Octaplas. None received only PLTs.
Most of the patients in the study cohort (n = 180, 94%) were transfused with RBCs. The mean number of RBC units was 12.6 (median: 6, standard deviation (SD): ±21) per patient. PLTs were administered to 72 (38%) patients, receiving a mean of 24 units (median: 16, SD: ±33). Octaplas was administered to 93 (48%) patients, with a mean of 16 (median: 10, SD: ±22), while 21 (11%) patients received a mean of 20 units of (median: 12, SD: ±23) FFP.
By survival
Among patients who survived (n = 172), a total of 5130 units of blood components were administered, including 2096 units of RBCs (mean of 12 units per patient), 1448 units of PLTs (mean of 8.4 units per patient), 203 units of FFP (mean of 1.18 units per patient), and 1383 units of Octaplas (mean of 8 units per patient). The lowest mean hemoglobin for survivors was 83 g/L.
Among those patients who died (n = 20) during their hospital stay, a total of 957 units of blood components were administered. This included 326 units of RBCs (mean of 16 units per patient), 280 units of PLTs (mean of 14 units per patient), 217 units of FFP (mean of 17 units per patient), and 134 units of Octaplas. The lowest mean hemoglobin was 85 g/L.
By age group
Table 2 summarizes the use of blood components stratified by age group. We found no notable differences between age groups.
By principal diagnoses
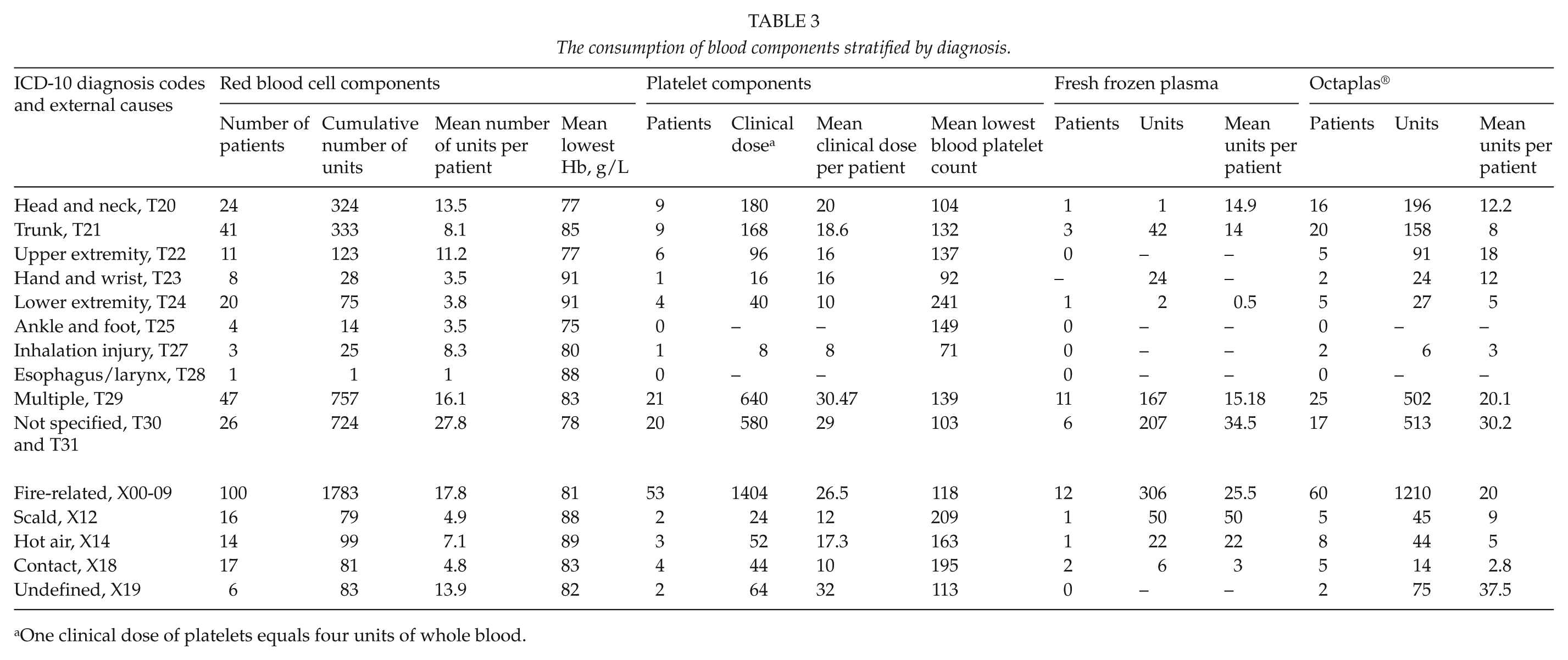
Table 3 provides a summary of transfusions by principal diagnoses and the use of the blood products during the study years. Patients diagnosed with burn injuries to multiple locations or unspecified and to the head and neck received the largest volume of blood components. The smallest anatomical entities—hands and wrists and ankles and feet—received the smallest quantities of blood components.
The consumption of blood components stratified by diagnosis.
One clinical dose of platelets equals four units of whole blood.
Stratification by external cause revealed that patients with fire-related burns received the highest volume of blood products. No trend in the change of transfusion practices during the study years could be noted (Supplementary Table 1, available online at: http://sjs.sagepub.com/).
Discussion
Here, we reviewed the VOK registry data based on the use of blood products, including RBC components, PLTs, and FFP, among Finnish burn patients treated at the Helsinki University Hospital between 2005 and 2011. Among all 558 burn patients, 193 (34%) received blood products during their hospital stay. In addition, 74% of those received transfusions of RBCs either alone or together with other blood products. We found that the percentage of patients receiving RBC transfusions was lower than previous reports. For instance, Palmieri et al. (7) concluded that up to 79% of the burn patients they studied received packed RBC transfusions. However, their sample differed from ours to include only patients with ⩾20%TBSA, whereas we included all patients admitted based on a burn injury diagnosis.
We found that the most common blood product transfused included RBCs. Nearly 95% of transfused patients received RBCs followed by FFP/Octaplas (60%) and platelet components (38%). These transfusion proportions for the different blood components are in line with previous reports (8, 10). Only 29% of patients in our study required transfusions of all three blood components. The probability of transfusion was significantly elevated as patient age increased and in the presence of a large cutaneous insult (p < 0.001). We found no association between inhalation injury and transfusion. Furthermore, we found that in-hospital mortality among transfused patients exceeded that of non-transfused patients fivefold.
Although numerous reports exist indicating that mortality rates are higher among transfused burn patients (7, 9, 17), most studies fail to show a significant association between transfusions and mortality (10, 11), in contrast to our findings. Earlier reports focused only on RBC transfusions. Quite recently, Lu et al. (10) showed that mortality was associated with plasma transfusions. Transfusions may carry an indirect effect on mortality by compromising the immune system and predisposing patients to severe infections leading to an increased mortality (18, 19). On the other hand, the larger number of transfusions may serve as a surrogate marker for the severity of the burn injury (20). The association between hospital mortality and transfusion found in this study should, therefore, be interpreted with caution, while other factors not included in our data or analysis may influence hospital mortality among patients receiving transfusions. These may include factors such as a higher %TBSA and multiple operations increasing the number of transfusions independently affecting hospital mortality.
Another possible complication or adverse effect associated with transfusions includes their potential to immunosuppress patients, shown to significantly increase infection rates among patients. The infection rate is substantially amplified with each unit of blood transfused (7, 11). Patients with large burns are by definition immunosuppressed, where the extent of immunosuppression appears greater following a burn injury compared to other forms of trauma (21). The impairment of the immune system depends on the extent of the cutaneous insult (22), particularly affecting patients with burns >30%TBSA where the immune system is more significantly affected. The overall incidence of infections is higher compared with other patients in intensive care units (23) or other trauma patients; thus, a burn victim is potentially at risk of acquiring a life-threatening infection (24). The additional effect of transfusions on the occurrence of infections is similarly difficult to precisely estimate.
In this study, we found that the trigger hemoglobin, that is, the lowest hemoglobin value before an RBC transfusion, varied considerably between the different principal diagnoses and anatomical burn locations (Table 3). In the critical care unit, the Transfusion Requirements in Critical Care trial and the American Society of Anesthesiologists Transfusion Guidelines recognize the benefits of restrictive transfusions (25). Trigger hemoglobin values between 92 and 100 g/L (7, 17) are currently considered conservative and high. Studies on restrictive policies for the threshold of transfusions resulted in better outcomes (17, 26, 27), with smaller amounts of blood components transfused. A large US-based multicenter study showed that the transfusion trigger hemoglobin did not vary based on the extent of the burn injury (7). In our study, the trigger hemoglobin varied according to the anatomical location of the burns. Patients whose burns were located in the smallest anatomical areas—hands and wrists or ankles and feet—received the lowest number of units of blood products. While the head and neck represent relatively small anatomical regions, the patients in this study with head and neck burns received relatively large volumes of blood products. One reason for this might be that isolated facial burns resulting from fire/flame injuries are rare, whereby the face is usually affected as a part of a larger burn (28–30).
The prognosis for a severe burn injury still relates to three major risk factors for death: an age ⩾60 years, ⩾40%TBSA, and the presence of inhalation injury (31). In addition, these factors relate to attempts at defining when burn care becomes futile. Ethical considerations in decisions determining at which point further treatment is futile remain challenging and may cause anxiety for the burn victim’s family and the burn care team (32). Such decisions based on medical facts are not straightforward. The higher trigger hemoglobin for non-survivors may depict the severity of the injury and the measures to apply to help save the burn patient. That said, based on our findings, we cannot exclude the direct effect of transfusions on hospital mortality.
Acute traumatic coagulopathy is a rare event affecting thermally injured patients (33, 34) unlike other trauma victims. A major burn injury is associated with early coagulopathy occurring within 24 h post-burn (33). By contrast, Sherren et al. (35) showed contradictory results, whereby nearly 40% of their burn victims were diagnosed with acute burn-induced coagulopathy upon admission. A closer look at these studies reveals that the definitions of coagulopathy vary between studies sufficiently to produce discrepancies across results.
Additionally, idioms used in the literature, including “burn anemia,” “anemia of thermal injury,” “anemia of thermal burns,” or “anemia in burns” (36), refer to anemia occurring at any time during burn care. Burn anemia correlates with %TBSA (1, 8, 36), and thus, a large burn injury is frequently followed by multiple operations resulting in an increase in the number of transfusions (37). Most clinical and research efforts focus on controlling blood loss in the operating room, including the use of tourniquets, epinephrine tumescence, topical hemostatic dressings such as epinephrine-soaked pads, thrombin and fibrin sealants, controlled intraoperative hypotension, and excision with a laser (38–40). Blood loss in the operating room during a burn surgery from the excised wounds and donor sites accounts for only about one-third of the transfusions burn patients received during a hospital stay (7).
We recognize certain limitations in our study. Based on the burn patients recorded in the National Hospital Discharge Registry, our cohort included only about 40% of all burn patients. FRCBS maintained the VOK registry together with participating hospitals between 2002 and 2011. The data were collected by identifying cases and, then, subsequently querying the relevant patient-associated data from hospitals’ electronic information systems. Despite our best effort aimed at 100% coverage, the VOK registry is incomplete. For instance, patients missed from the enrollment of births were previously noted; only 50%–75% of births were included in the registry (Jarno Tuimala, oral communication). Furthermore, data collected for research purposes regarding burn patients were incomplete, including the absence of %TBSA and the etiology of registered thermal injuries. In the registry data, %TBSA and burn injury etiology were missing. %TBSA represents an important factor determining the volume of transfusions burn patients receive (11, 41). Based on the discussion above, information on blood work guiding decisions such as transfusions, hemoglobin, platelet counts, and INR in the registry was far from complete. Therefore, our findings regarding these may be considered tendencies rather than generalizations. In this study, we addressed the most important question regarding the use of blood components in a large cohort, supporting the strength of this study. In addition, that the Helsinki University Hospital serves as a secondary referral center and a tertiary referral center for all severe burns in Finland represents a strength of this study, particularly given that the catchment area for this study is sufficiently large.
To conclude, in this study, we showed that Finnish adult burn patients received an ample volume of transfusions. The number of units of blood components varied according to the anatomical location of the injury and patient survival.
Footnotes
Acknowledgements
We thank Professor Mika Gissler of THL for providing us with up-to-date incidences of burn injuries in Finland.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Each author declares no financial conflicts of interest with regard to the data presented in this article. Funding for this article was derived from departmental sources only.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
