Abstract
Background:
Monitoring treatment response to preoperative chemotherapy is of utmost importance to avoid treatment toxicity, especially in non-responding patients. Currently, no reliable methods exist for tumor response assessment after preoperative chemotherapy. Therefore, the aim of this study was to evaluate dysphagia as a predictor of tumor response after preoperative chemotherapy and as a predictor of recurrence and survival.
Methods:
Patients with adenocarcinoma of the gastroesophageal junction, treated between 2010 and 2012, were retrospectively reviewed. Dysphagia scores (Mellow-Pinkas) were obtained before and after three cycles of perioperative chemotherapy together with clinicopathological patient characteristics. A clinical response was defined as improvement of dysphagia by at least 1 score from the baseline. The tumor response was defined as down staging of T-stage from initial computer tomography (CT) scan (cT-stage) to pathologic staging of surgical specimen (pT-stage). Patients were followed until death or censored on June 27th, 2014.
Results:
Of the 110 included patients, 59.1% had improvement of dysphagia after three cycles of perioperative chemotherapy, and 31.8% had a chemotherapy-induced tumor response after radical resection of tumor. Improvement of dysphagia was not correlated with the tumor response in the multivariate analysis (p = 0.23). Moreover, the presence of dysphagia was not correlated with recurrence (p = 0.92) or survival (p = 0.94) in the multivariate analysis.
Conclusion:
In our study, improvement of dysphagia was not valid for tumor response evaluation after preoperative chemotherapy and was not correlated with the tumor response. The presence of dysphagia does not seem to be a predictor of recurrence or survival.
Keywords
Introduction
The incidence of gastroesophageal junction cancer (GEJ cancer) has increased approximately sevenfold in the last three decades (1). However, only 40%–50% of patients are undergoing curative intended surgery (2, 3). Furthermore, survival remains poor after implementation of perioperative chemotherapy as a standard regimen (4). Although, up to 25% have been shown to have a histopathological response to neoadjuvant chemotherapy (5), some patients are non-responders and progress during therapy. Thus, monitoring chemotherapy response during treatment to detect non-responders seems prudent. At this time, postoperative histopathological staging is the gold standard in the assessment of treatment response (6) and is one of the strongest predictors of survival (7). Methods of accurate response evaluation during neoadjuvant treatment are lacking. In the MUNICON trials (the Metabolic response evalUatioN for Individualisation of neoadjuvant Chemotherapy in oesOphageal and oesophagogastric adeNocarcinoma), evaluation with Positron Emission Tomography (PET) before and after 14 days of neoadjuvant chemotherapy indicated that chemotherapy can be discontinued at an early stage in metabolic non-responders without compromising the prognosis (8). Interestingly, it was shown that the addition of neoadjuvant radiotherapy in metabolic non-responders did not lead to an improvement of their poor prognosis, thus indicating that early non-response indicates dismal tumor biology (9). The role of improvement in specific symptoms during chemotherapy has not been properly elucidated, but clinicians have taken the asymptomatic state of the patients as a promising sign.
Dysphagia is the cardinal symptom of patients with cancer in the GEJ. Up to 90% of the patients have dysphagia at the time of diagnosis (10–12). Moreover, GEJ tumors are associated with a higher degree of dysphagia than gastric tumors, which is dependent of the tumor stage at diagnosis (10). Therefore, the aim of the study was to examine, whether, an improvement of dysphagia after three cycles of perioperative chemotherapy was correlated with the tumor response in radically resected patients with adenocarcinoma of the GEJ. Furthermore, dysphagia was evaluated as a predictor of recurrence and survival.
Materials and Methods
Patients
All patients considered operable and resectable for GEJ cancer between 2010 and 2012 were retrospectively evaluated by two independent reviewers. Data were collected from a prospectively maintained database of all Danish patients treated for gastric, GEJ, and esophageal cancer with more than 7000 registered patient cases. This study was approved by The Danish Data Protection Agency and the Danish Health and Medicines Authority.
All patients were initially considered resectable and operable at the multidisciplinary team conference (MDT conference). The diagnostic workup included an upper endoscopy with biopsy, computer tomography (CT) of the chest and abdomen combined with ultrasound of the neck and/or PET-CT for tumor staging, and after the MDT conference, a staging laparoscopy to rule out peritoneal carcinomatosis. The inclusion criteria were age greater than 18 years, biopsy verified adenocarcinoma of the distal esophagus or the GEJ region, radical resection of tumor (R0 resection), and surgery performed as transthoracic esophagectomy with extended lymph node dissection.
Follow-Up
All patients alive were censored on June 27th, 2014 or at the date of death. This was cross-referenced with the Danish Central Population Registration (CPR-register), which contains updated information of, for example, gender, date of birth, place of birth, place of residence, and vital status of every Danish citizen. Survival was defined from the date of surgery to the date of death or last follow-up. Date of recurrence was obtained in patients’ medical records, either biopsy- or CT-verified.
Chemotherapy
The treatment consisted of six perioperative cycles of epirubicin, cisplatin or oxaliplatin, and capecitabine; three series administered preoperatively and three series postoperatively. Response to preoperative chemotherapy was measured as follows:
1. Dysphagia.
Dysphagia scores were retrieved from the patients’ medical records, primarily in the nurse records but also in the admission histories, before and after preoperative chemotherapy according to Mellow and Pinkas’ (13) dysphagia score. The score consists of levels 0–4 dependent on the type of food the patient is able to swallow. Level 0 indicates “no dysphagia” and level 4 indicates “complete food stop.” A response was defined as an improvement of dysphagia score from the baseline with at least − 1 level.
2. Tumor response.
The tumor response was defined as down staging of tumor stage (T-stage, TNM Seventh edition (14)) from the initial CT scan prior to surgery (cT-stage) to tumor assessment of surgical specimen (pT-stage).
3. Other variables.
Performance status was obtained in the oncological admission histories prior to induction of chemotherapy according to the Eastern Cooperative Oncology Group (ECOG) classification (15).
Statistics
Statistics was made with IBM SPSS® version 19.0.0 (SPSS, Inc, Chicago, IL). The distribution of different individual characteristics was evaluated with simple descriptive statistics. The univariate analysis was made with Kaplan Meier’s log rank. Pearson’s chi-square and Fisher’s exact test were used for nominal and ordinal variables. The prognostic value of dysphagia was evaluated with Cox proportional hazards regression. Results were considered significant if p ⩽ 0.05, two sided.
Results
In total, 158 patients were initially considered operable and resectable at the MDT conference. Of those, 48 patients were excluded (Fig. 1). The basis of the statistical analysis was 110 patients, who underwent R0 resection with transthoracic esophagectomy with D2+ lymphadenectomy. Of patients included, 85% were males with a median age of 67 (40–85) years.
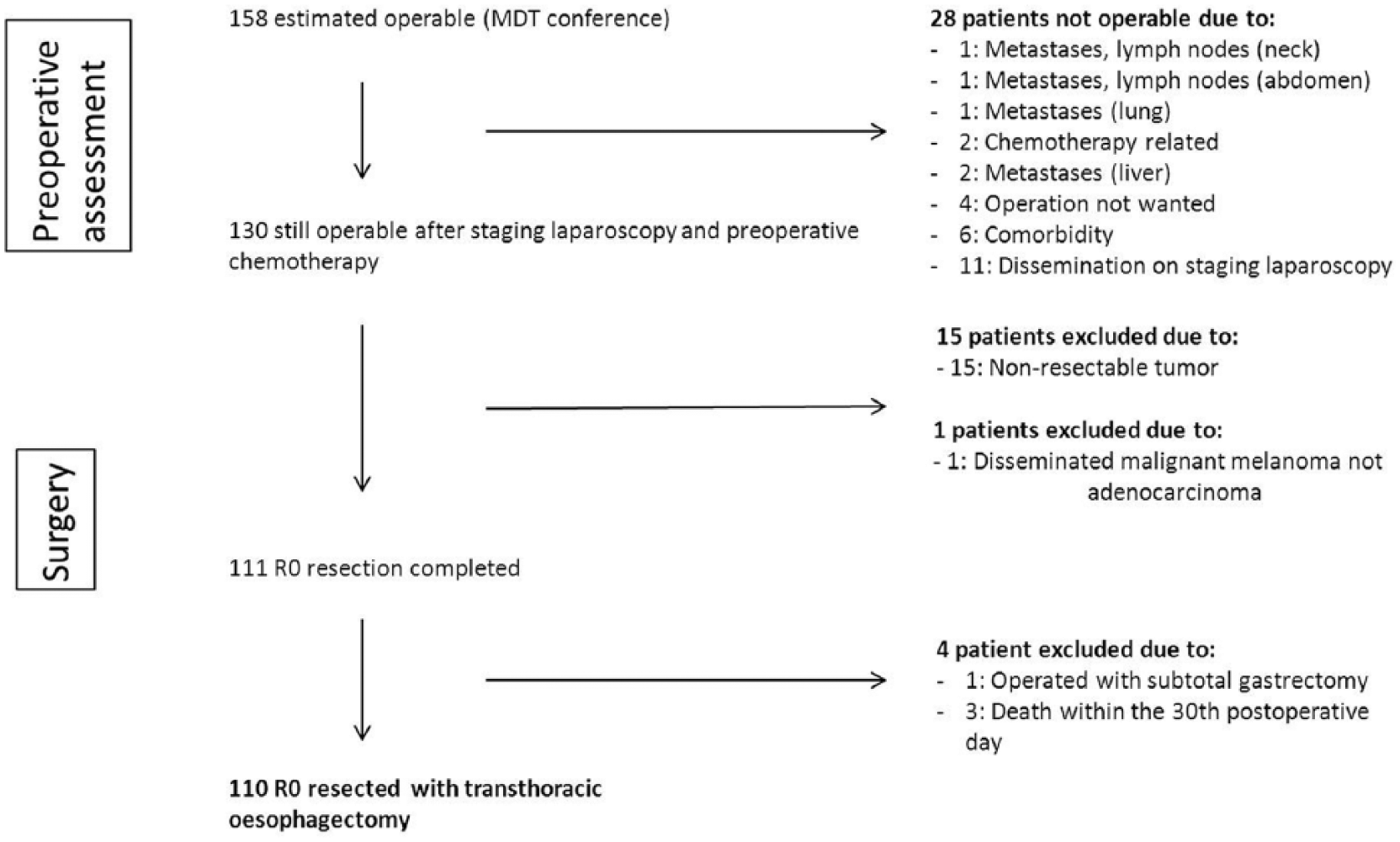
Overview of exclusion.
The median follow-up time was 20.6 months (1–43), and no patients were lost to follow-up. The median overall survival was 28.5 months (25–32) for radically resected patients. Furthermore, 40.9% of R0 resected patients had recurrence.
Predictors of Recurrence and Survival
Of the radically resected patients (n = 110), 80% had dysphagia at the time of diagnosis. Results of the univariate analysis are shown in Table 1. Dysphagia was not associated with survival, p = 0.12, but significantly associated with recurrence, p = 0.02, in the explorative univariate analysis. In multivariate analysis (Table 2), dysphagia was not significantly associated with either survival, p = 0.94, or recurrence, p = 0.92.
Results of univariate analysis.
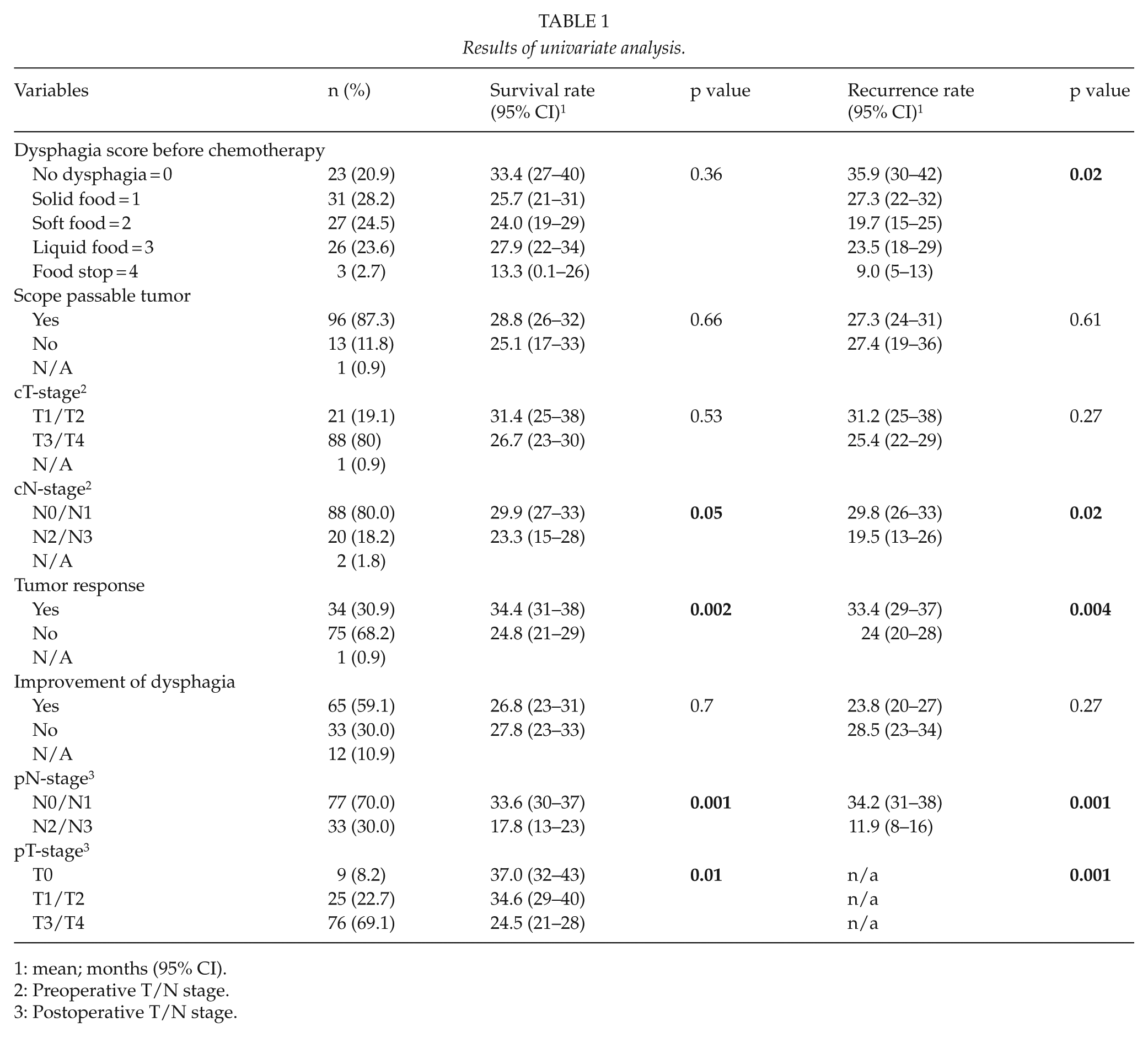
1: mean; months (95% CI).
2: Preoperative T/N stage.
3: Postoperative T/N stage.
Results of multivariate analysis.
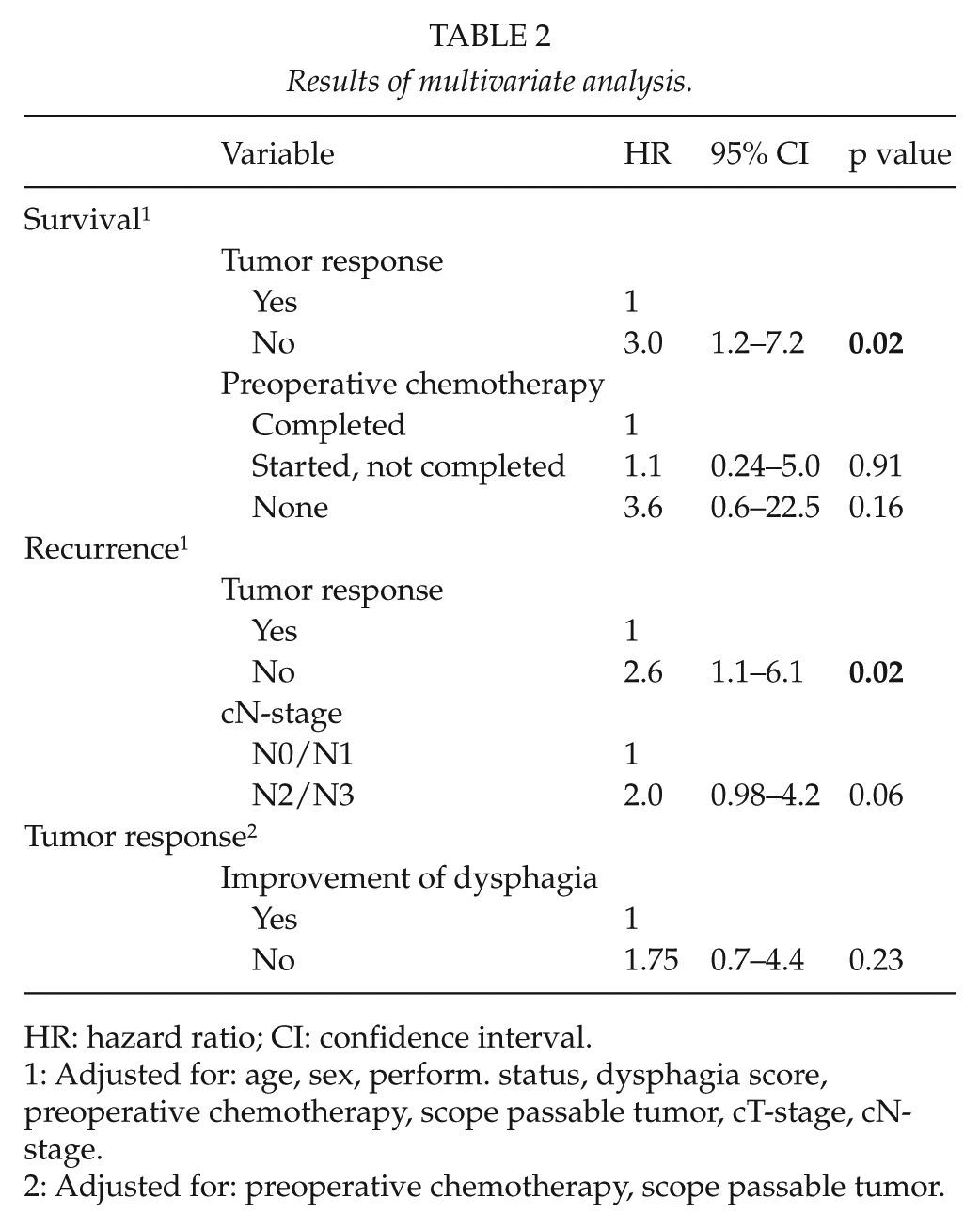
HR: hazard ratio; CI: confidence interval.
1: Adjusted for: age, sex, perform. status, dysphagia score, preoperative chemotherapy, scope passable tumor, cT-stage, cN-stage.
2: Adjusted for: preoperative chemotherapy, scope passable tumor.
Of other variables assessed, tumor response, cN-stage, pT-stage, and pN-stage were significantly correlated with survival and recurrence in the univariate analysis. The variables age, sex, performance status, preoperative chemotherapy, cT-stage, and improvement of dysphagia did not reach statistical significance (only improvement of dysphagia shown). The multivariate analysis showed a significant association between tumor response and survival and recurrence.
Improvement in Dysphagia and Tumor Response after Preoperative Chemotherapy
Of the cohort, 89.1% completed three cycles of preoperative chemotherapy. Furthermore, 59.1% of patients experienced an improvement in dysphagia, and 31.8% had a tumor response. Those patients who completed the chemotherapy treatment had a higher percentage of improvement of dysphagia (69.0%) and a higher rate of tumor responses (34.0%) than patients who received no chemotherapy (0.0% improvement of dysphagia and 0.0% tumor responses) or had an incomplete chemotherapy treatment (43.0% improvement of dysphagia and 29% tumor responses). However, this was not significant, p = 0.14 and p = 0.28. Moreover, improvement of dysphagia was not significantly correlated with the tumor response in the univariate (p = 0.2) or the multivariate analyses (p = 0.23).
Discussion
This study found that presence of dysphagia was not a predictor of recurrence or survival in the multivariate analysis. Furthermore, there was no correlation between improvement in dysphagia and the tumor response.
Relief from dysphagia is not equal to the shrinkage of the tumor during chemotherapy, which may explain our negative association to chemotherapy response. The law of Poiseuille states that resistance is dependent of radius in the fourth power (16). Hence, a small increase in radius has a great impact on loss of resistance, which means that a minor tumor response may lead to a significant improvement in dysphagia, theoretically (17). Thus, this could be a potential mechanism of bias. Moreover, there is a possibility that persistent dysphagia relates to decreased esophageal motility secondary to chemotherapy-induced fibrosis (12), which then would be incorrectly perceived as a non-response to chemotherapy. Furthermore, as this was a retrospective study, we had no accurate tumor size to correlate to dysphagia improvement.
Two other studies have examined this matter with similar conclusions (12, 18), which supports our findings. A study of 48 patients with squamous cell carcinoma or adenocarcinoma underwent neoadjuvant chemotherapy and surgery (12). The study found no association between improvement of dysphagia and tumor response. However, the response was defined as improvement or maintenance of baseline score, and moreover, the Mandard classification was used to assess the tumor response (19), both of which differ from our definition. Another study with 57 patients also found a negative association (18). On the other hand, a recent study found a significant correlation with improvement of dysphagia and R0-resection for advanced T4-tumors (20). Unfortunately, the association between improvement of dysphagia and tumor response was not evaluated. However, the study emphasizes that relief of dysphagia increases the possibility of a radical resection of tumor. Thus, the improvement of dysphagia as a marker for tumor response is still unclear.
Of our cohort, 80% had dysphagia at diagnosis, but no significant correlation with survival or recurrence was found in the multivariate analysis. In contrast, three studies (2, 21, 22) found dysphagia to be an independent predictor of survival after adjustment for confounders (Table 3). However, negative findings have also been reported in at least three studies (23–25). The three studies which found a significant association with survival were dissimilar to our study cohort regarding tumor site, tumor histology, methods of surgery, and chemotherapy regimen, which makes comparison difficult. One study included both operable and non-operable patients in the multivariate analysis (2), another was a small study with seven patients (22), and one study found only 5.7% of patients having dysphagia (21).
Dysphagia and survival.
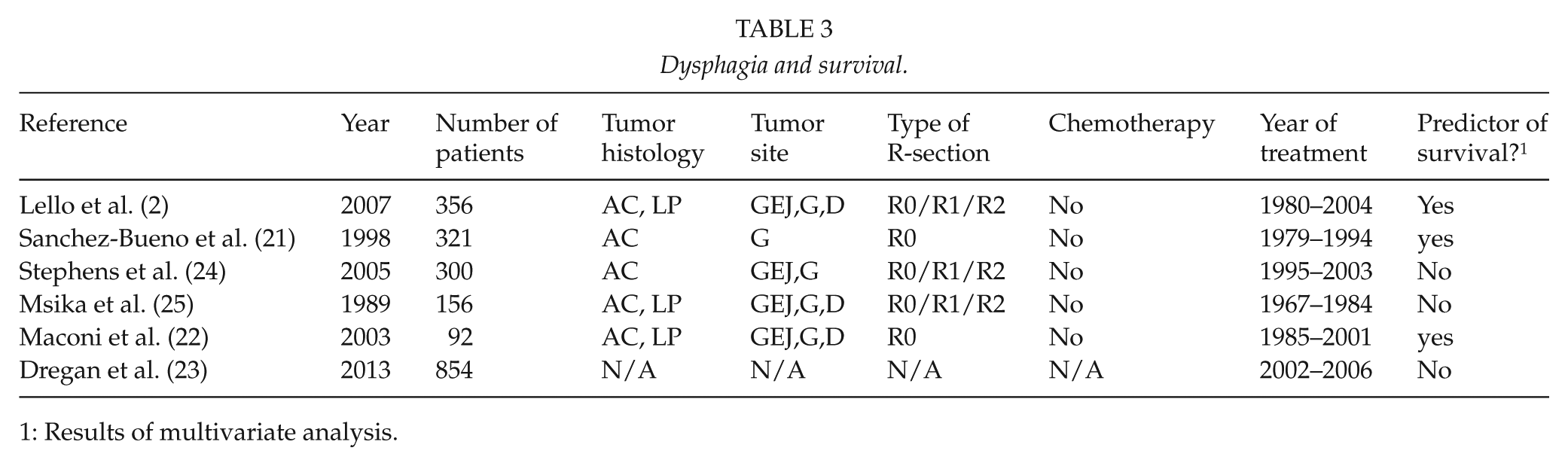
1: Results of multivariate analysis.
To our knowledge, only two studies have examined whether dysphagia is associated with recurrence. A recent study with 42 patients, found that 43% versus 19.6% had recurrence with the presence of dysphagia and no dysphagia, respectively (p < 0.05) (26). However, in the multivariate analysis, dysphagia was not a significant predictor of recurrence just as in the other study (27). This is similar to our findings, in which we did not find dysphagia to be a significant predictor of recurrence in the multivariate analysis, p = 0.92.
Other modalities, like CT and endoscopy, have been investigated in assessing response to chemotherapy. However, these are of limited use in response assessment and cannot stand alone in a clinical setting. Becker et al. (28) reported that tumor residual cells often were located in the periphery of the esophagus, and not in the luminal part, which explains one of the disabilities of endoscopy in this setting. Moreover, re-biopsies are only possible when tumor is passable by scope, which were non-evident in 12% of the patients in our study. CT scans have difficulties differentiating between residual tumor and edema or fibrosis after chemotherapy (6). A study from our institution with 28 patients examined CT-perfusion scans as an indicator of histopathologic response (29). The study found that the probability of a response increased with the reduction in tumor-permeability between baseline scan and after the third cycle of chemotherapy. However, CT-perfusion scan only yielded a sensitivity of 69% and a specificity of 58%, making the modality insufficient for clinical decision. Another well-used modality is PET-CT. One study found that PET-CT was not able to distinguish between complete pathological remission (0% residual tumor cells) and subtotal remission (<10% residual tumor cells), histopathologically (8). However, early termination of chemotherapy based on PET-CT did not negatively affect the clinical outcome in metabolic non-responders. Thus, PET-CT might seem to provide clinically useful information of future use.
Our study cohort was well defined regarding tumor site, histology, regimen of chemotherapy, and type of surgical procedure. This is particularly beneficial in the management of assessing chemotherapy response, which we consider a major strength in our study. Moreover, no patients were lost to follow-up, and the exact date of death was known due to the validity of the Danish CPR-register. Furthermore, the same multidisciplinary team evaluated all of our patients, which may reduce a potential selection bias. Despite these facts, we had 10.9% missing data for the dysphagia scores either before or after chemotherapy, which is a consequence of the retrospective design. This may give cause to bias in our results. Furthermore, our limited follow-up time of 20 months does not give us the option of drawing a definite conclusion regarding dysphagia and survival, which is why this should be interpreted with caution. Moreover, we acknowledge that our measure of tumor response does not include a repeat CT scan after preoperative chemotherapy or a histopathologic tumor regression grade (19), both of which would have been interesting variables to consider. Unfortunately, our institution does not perform repeat CT scans after preoperative chemotherapy or use the tumor regression grading system on surgical specimen. We used a pseudo measure of tumor response defined as down staging of tumor stage from initial CT scan to pathologic assessment of surgical specimen. Other authors have adopted similar methods of response assessment (7, 30). As CT-imaging has limited accuracy for assessing clinical tumor stage, any assessment of down staging by comparing post therapy pathological stage to pretreatment clinical stage may be biased. In this study, the tumor response measure was significantly correlated with survival and recurrence in the multivariate analysis (Table 2) and we consider this measure adequate for the purpose of our study.
In conclusion, in our study, dysphagia was not found to be a valuable parameter of tumor response assessment after preoperative chemotherapy for R0 resected patients with adenocarcinoma of the GEJ. Furthermore, dysphagia was not found to be an important predictor of survival and recurrence in radical resected patients with adenocarcinoma of the GEJ.
Footnotes
Declaration of Conflicting Interests
None of the authors have any conflicting interests to disclose.
Ethics Approval
This study was approved by The Danish Data Protection Agency (ID: 2007-58-0015) and the Danish Health and Medicines Authority (ID: 3-3013-603/1/).
Funding
This study was partial sponsored by The Danish Cancer Organization (grant number R84-A5558).
