Abstract
Background and aims:
Several studies have demonstrated that breast cancer survival rates differ with ethnicity. Most of these studies analyzed discrepancies between African-American and Caucasian-American women and were performed in the United States. There are increasing concerns about differences in breast cancer survival among immigrants from Asia and Africa living in Europe, including those living in Scandinavian countries. There are few data on breast cancer survival in relation to race or ethnicity in Scandinavian countries, even though immigrants from Asia and Africa have lived in Scandinavian countries for decades. The aim of this study was to identify variations in breast cancer incidence, treatment modalities, relapse, and survival among women from Pakistan, Sri Lanka, and Somalia compared to ethnic Norwegian women.
Material and methods:
The incidence, treatment modalities, relapse, and survival of breast cancer were analyzed in women from Pakistan, Sri Lanka, and Somalia in a nation-based study over a period of 7 years. Results for women from Pakistan, Sri Lanka, and Somalia were compared with those from a group of ethnic Norwegian women during the same period. In our study, 63 patients from Pakistan, Sri Lanka, and Somalia were diagnosed with breast cancer during the period 2002–2009 in Norway.
Results and conclusion:
Comparison between women from Pakistan, Sri Lanka, and Somalia and ethnic women from Norway revealed significant differences in cancer stage at the time of diagnosis, age at diagnosis, type of surgical treatment, and relapse and breast cancer mortality rates. The findings of this study demonstrate that the outcome after a breast cancer diagnosis is significantly worse for women from Pakistan, Sri Lanka, and Somalia than for ethnic Norwegian women. In addition, the mean age at the breast cancer diagnosis was lower for women from Pakistan, Sri Lanka, and Somalia, especially those from Sri Lanka and Somalia, than for ethnic Norwegian women.
Introduction
Several studies have revealed that breast cancer survival rates differ between minority populations and the majority population. Most of these studies are from the United States and include African-American, Hispanic, or South-Asian women in comparison with Caucasian patients (1–7). A few studies have focused on breast cancer survival differences between immigrant women and the majority population in European countries (8).
Many of the previous studies have demonstrated that women from ethnic minority groups have a younger median age at cancer presentation, a larger tumor size with a more advanced stage/grade, and a higher number of affected regional lymph nodes. Ethnic minorities also tend to have triple-negative (estrogen, progesterone, and Her-2 negative) tumors, which are associated with a poor prognosis (9, 10). Some studies have also revealed that ethnic minority groups, and specially African-Americans, experience greater delays in breast cancer diagnosis and treatment (4, 11). These differences are difficult to explain, but socioeconomic status (SES) is one of the parameters that has received considerable attention as a potential correlate of breast cancer survival. Some studies have shown that a low SES is associated with more advanced breast cancer (12, 13), while other studies have demonstrated the important role of ethnicity in a late stage at diagnosis (14). Biological differences and comorbidities have also been discussed as contributing factors (15).
The strongest determinant of cancer survival is the stage at diagnosis. The overall 5-year breast cancer survival rate in Norway is 88.7% (16) and is 98.8% for localized disease, 88% for regional disease, and 21.4% for metastatic breast cancer (16). During the last two decades, there have been considerable improvements in breast cancer survival in Norway, with the total breast cancer survival rate increasing from 74.3% in 1990 to 98.7% in 2010. Screening mammography and new protocols for surgical and oncological treatments were introduced during the period 1990–2010, leading to the earlier detection of breast cancer.
In Norway and other Scandinavian countries, information concerning ethnicity and breast cancer survival is sparse, even though immigrants from Asia and Africa have been living in Norway for several decades. The largest group of immigrants from Asia and Africa in Norway come from Pakistan, Sri Lanka and Somalia (PSS).Presumably due to religious and cultural differences, immigrants from these countries typically experience the greatest difficulty integrating into the Norwegian society. The utilization of health-care services by these groups may be suboptimal, thus contributing to their inferior outcomes for breast cancer compared to the majority population. The literature on ethnic minority groups shows that these women lack knowledge regarding the incidence of and risk factors for breast cancer. It is necessary to identify cultural beliefs and values relating to breast cancer in order to provide appropriate information to these women (17–19).
It has been reported that immigrants living in Norway are overrepresented among those with a lower SES (20). Some international studies have demonstrated a link between low SES and more advanced breast cancer (12). Such findings may also apply to Norway.
Breast cancer is the most frequent cancer in women, and one of the cancer forms for which survival is greatly affected by cultural, economic, and social factors. The aim of this study was, therefore, to determine whether there are ethnicity differences in breast cancer survival in Norway. We choose women from PSS, because women from these countries represent most of the challenges related to integration caused by cultural, religious, and socioeconomic factors.
Material and methods
The Norwegian Knowledge Center for the Health Services has collected discharge data/patient administrative data from all Norwegian hospitals for patients discharged during the period 2002–2009. The discharge data provide information about age, gender, diagnoses, procedure codes, length of stay, hospital department, municipality of residence, type of admission (i.e. acute or elective), and in-hospital mortality. The database was linked to the National Population Registry and the Norwegian Causes of Death Registry to obtain the exact date of death and birthplace information.
All women with a primary diagnosis of breast cancer were identified. The total number of patients with a primary diagnosis of breast cancer during 2002–2009 was 20,499. Women living in Norway, but born in PSS, with a primary diagnosis of breast cancer were identified as a subpopulation. This subpopulation comprised 63 women, and medical records were available for 60 of these patients. Each of these 60 women was matched with at least 1 (and usually 2) ethnic Norwegian women with breast cancer, who had surgery at the same hospital on the same day and by the same surgeon. The ethnic Norwegian women were assigned as the control group and consisted of 101 women. The follow-up time was the same in the two groups. All women in both groups got adjuvant treatment according to the guidelines for breast cancer treatment in Norway. There were no differences in compliance between these groups.
Statistical methods
Statistical analyses were initially performed using PASW Statistics for Windows 18 (SPSS). Simple correlations between the covariates were estimated using Pearson’s correlation. Mortality, local relapse, and operation method were used as separate outcomes in different univariate logistic regression estimations where ethnicity was the single covariate. A multivariate logistic regression model with several covariates was then established for each of the outcomes. Statistical analysis using the multivariate regression model and stepwise analysis was performed with R version 3.0.2 (2013-09-25, Foundation for Statistical Computing). The cutoff for statistical significance was set at p = 0.05.
Results
Of the 63 women living in Norway but born in PSS, 42, 13, and 8 were born in Pakistan, Sri Lanka, and Somalia, respectively. Medical records were available for 60 of these patients.
Breast cancer was diagnosed by screening in only 15% (9/60) of the patients in the PSS group, compared to in 67.3% (68/101) of those in the control group (Fig. 1). The mean age at diagnosis was 44.5 years in the PSS group and 51 years in the control group. Furthermore, 66.7% (40/60) of the women in the PSS group had diabetes mellitus (DM; either type I or type II), compared to only 7.9% among the control group. Stage II or III cancer was diagnosed in 70.0% (42/60) of the PSS women, while only 29.7% (30/101) of those in the control group were diagnosed with stage II or III cancer. When regarding the histological grade, 40% of the women in the PSS group had grade III cancer while only 20% of the women in the control group had histological grade III tumor. Furthermore, about 32% of the tumors in the PSS group were estrogen receptor negative, while 24% of the tumors in the control group were estrogen receptor negative. The information about Her-2 and Ki-67 status was not available for more than 50% of the tumors, and these parameters are therefore not included in the study.
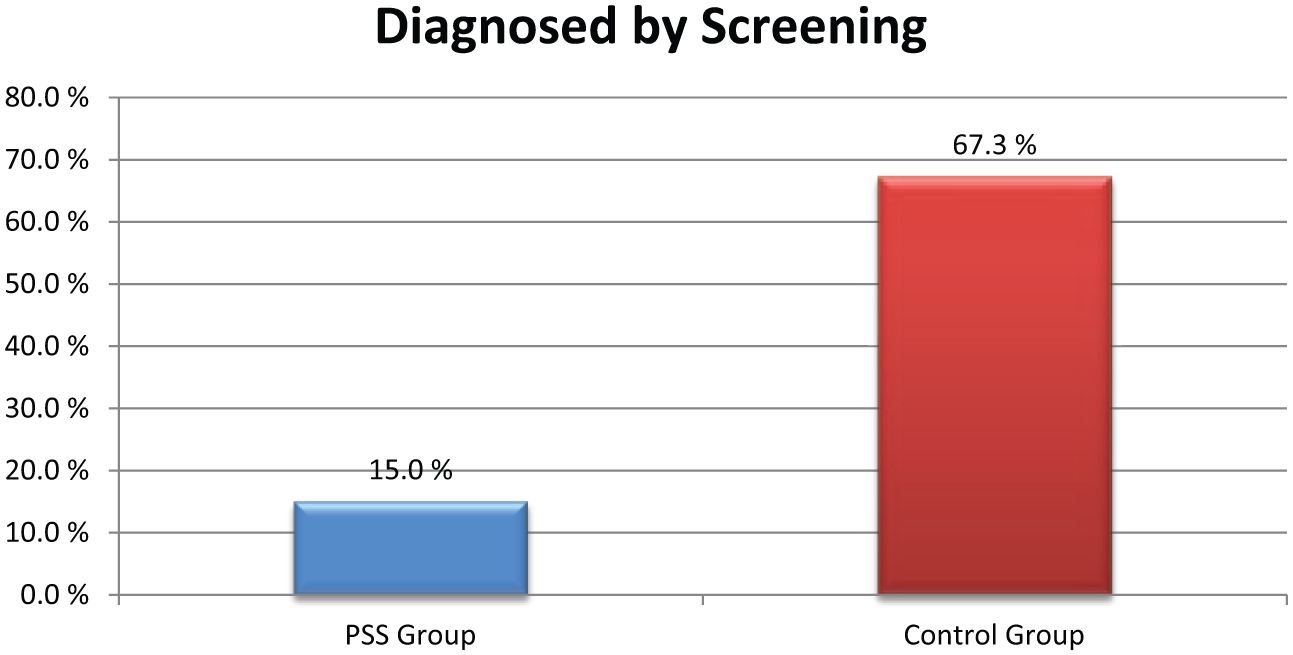
Participation in the mammography screening among patients in the PSS group and the control group.
In the PSS group, 30% of the women had locally advanced disease, compared to none in the control group at the time of the diagnosis (Fig. 2). Ethnicity was strongly correlated with mortality (Pearson’s correlation = −0.309, p < 0.0001), local relapse rate (Pearson’s correlation = −0.501, p < 0.0001), and the type of operation and prevalence of diabetes.
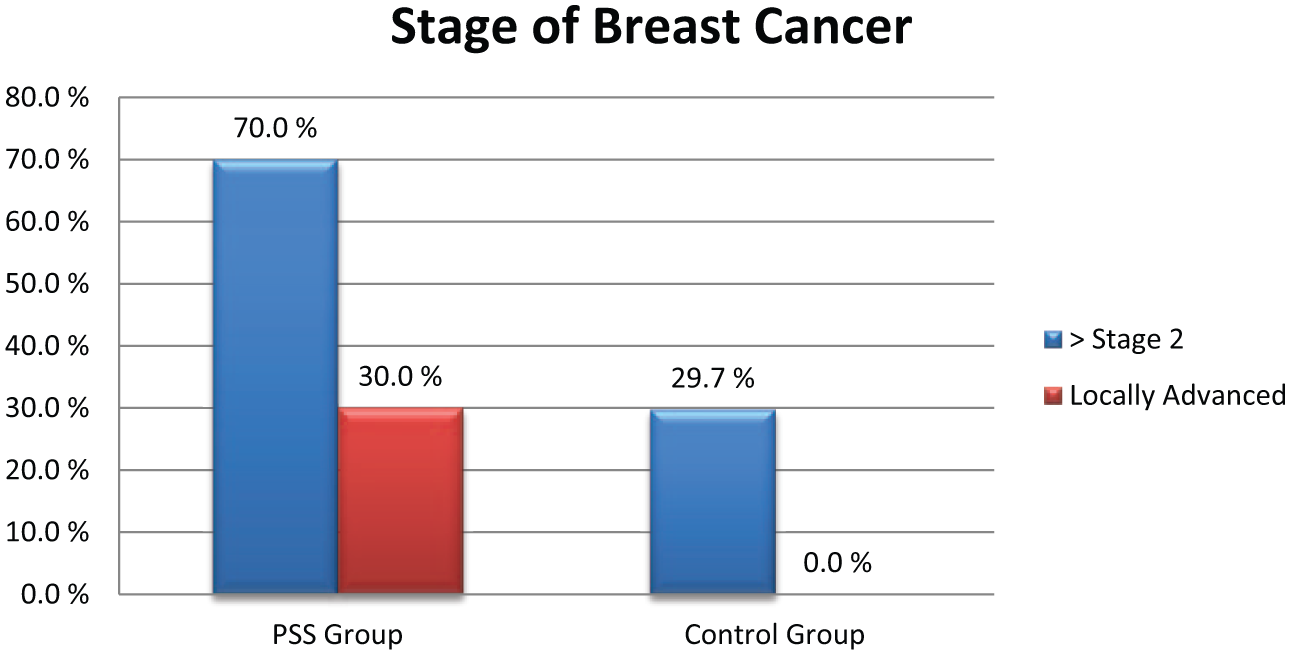
Stage of the breast cancer and locally advanced breast cancer when diagnosed among patients from the PSS group compared to the control group.
The breast-cancer–related mortality rate was 23.3% in the PSS group but only 7.9% in the control group (Fig. 3). In the univariate logistic regression model, ethnicity was significantly associated with mortality (odds ratio (OR) = 2.2, confidence interval (CI) = 1.4–3.5, p = 0.001). In addition, age (OR = 0.95, CI = 0.91–0.99, p = 0.01), stage (OR = 5.6, CI = 2.4–13.1, p = 0.00006), participation in a screening program (OR = 0.12, CI = 0.033–0.41, p = 0.001), and diabetes (OR = 3.3, CI = 1.3–8.5, p = 0.013) were also significantly associated with mortality. Both screening and stage at diagnosis can be related to ethnicity, and can be important factors to explain the effect of ethnicity. To examine whether these other ethnicity-related factors could explain the effect of ethnicity, we tested this in a stepwise multivariate logistic regression model. In this model, screening (OR = 0.19, p = 0.016) and stage at diagnosis (OR = 0.57, p = 0.002) remained as significant explanatory variables for mortality, indicating that the significance of ethnicity can be explained by late diagnosis and low participation in screening programs.
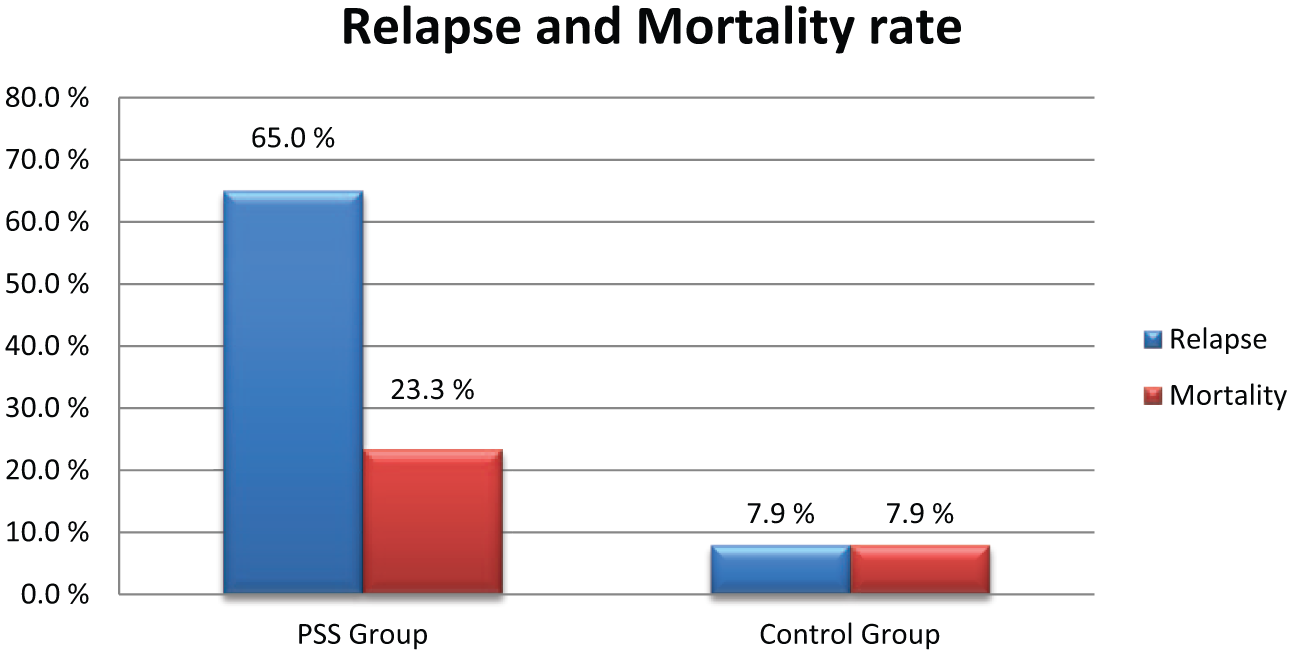
Relapse rate and mortality rate among patients in the PSS group and the control group.
Breast cancer relapse occurred in 65% (39/60) of those in the PSS group but only 7.9% of those in the control group. Also, local relapse was significantly higher in the PSS group (OR = 19.9, 95% CI = 8.1–49.0, p = 7.4 × 10−11). When we examined to look for factors that could explain the difference between the PSS group and the control group, stage (OR = 5.90, 95% CI = 1.4–24.2, p = 0.014), screening (OR = 0.18, 95% CI = 0.05–0.60, p = 0.007), age (OR = 0.93, 95% CI = 0.88–0.98, p = 0.006), and diabetes (OR = 5.3, 95% CI = 1.6–17.3, p = 0.005) remained in the model.
Of the PSS group, 68.3% (41/60) underwent mastectomy and 66.7% (40/60) underwent axillary-lymph-node dissection; the corresponding proportions in the control group were 36.6% (37/101) and 19.8% (20/101), respectively (Fig. 4).
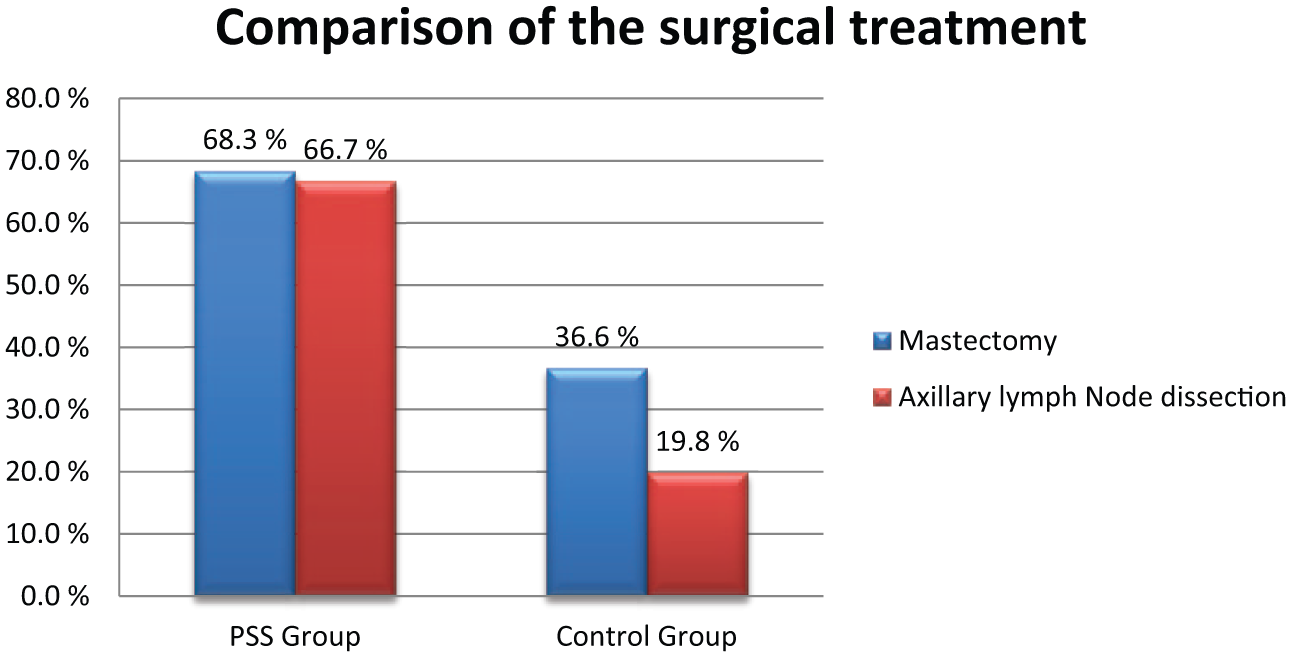
Comparison of the surgical treatment among the PSS group and the control group.
Discussion
In this pilot study, we have shown that mortality of breast cancer and the risk of breast cancer relapse is significantly higher in the PSS group than in ethnic Norwegians. Although this is a national study including all persons with breast cancer in Norway, the PSS group remains small and results should be carefully interpreted. The results are, however, not surprising. Cancer stage at the time of diagnosis and differences in participation in the national screening program were the explanatory variables for higher mortality rate observed in this study. However, the biological differences of the tumors may also play a significant role regarding the prognosis after diagnosis. We observed a higher rate of histological grade III, and estrogen receptor negative tumors in the PSS group compared to the control group, although it is impossible to explore in this study whether the differences in the histological grade and receptor status may reflect the size of the tumor.
Several recent studies have compared breast cancer among the majority and minority populations within a particular country (1, 3, 5, 6, 21). All of these studies (mostly from the United States) found that breast cancer survival differed with ethnicity. The explanations and reasons for these differences are complex and include biological parameters, SES, and access to health services.
All women in Norway between the ages of 50 and 69 years are offered a free examination that includes mammography screening biennially, with the intent of improving breast cancer survival. This study found that the participation rate in this screening program was much lower among women from PSS than among ethnic Norwegian women. The age range of 50–69 years for screening program in Norway was chosen because the incidence of breast cancer among Norwegian women is highest in this age group. The results obtained in this study indicate that breast cancer is diagnosed before the age of 50 years in many women from PSS, and especially those from Sri Lanka and Somalia. However, neither ethnicity itself nor age appeared to have a significant impact on the differences in mortality. Although being young and having a lower age at diagnosis was significantly associated with relapse (OR=0.93, p=0.006), which might indicate that patients in the PSS Group could not fully benefit from the Norwegian Screening program because of their age.
Participation in the screening program was one of the significant explanatory variables. This finding is in accordance with those international studies showing that the participation rate in mammography screening is lower among women from PSS living in Western countries (22). Similar results have been shown among Somali women living in the United States (23). Our study indicates that the most important reason why women from PSS do not attend screening program in Norway is because the screening program starts later than the age when women from PSS develop the breast cancer.
The obtained results clearly point to delayed diagnosis as the most important single factor contributing to relapse and death. There are several possible explanations for this finding that could not be fully addressed in this study. However, it is natural to assume that the women from the PSS population have a reduced access to the health-care diagnostic service. This would include several elements such as general communication barriers with the Norwegian health services system including language barriers. There may also be elements of a lower priority being applied to members of the PSS population.
Another element of diagnostic activity for breast cancer in Norwegian health-care system is self-examination of the breasts, which is important for detecting breast cancer at an early stage. Cultural barriers among women from PSS could result in a low rate of self-examination, which could have contributed to more advanced stage of breast cancer at the time of diagnosis. Forbes et al. observed that black women in East London and especially women from South Asia were much less likely to report self-examination of their breasts compared to Caucasian women (24).
Metabolic syndrome has been linked to breast cancer prognosis (25). This study found significant correlations of diabetes with relapse and mortality. In the multivariate logistic model, diabetes was no longer a significant explanatory variable for mortality, but it remained significantly associated with local relapse (OR = 5.3, p = 0.005). We observed that many of the women from PSS had diabetes as comorbidity, and many were overweight. Diabetes is a contributor to metabolic syndrome. Several studies, both in vitro and in vivo, have demonstrated a strong link between metabolic syndrome and the incidence of breast cancer. However, there is accumulating data showing that metabolic syndrome may also have an impact on the prognosis after breast cancer diagnosis. Erickson et al. (26) demonstrated a reduced overall survival of early-stage breast cancer in association with chronic hyperglycemia. They also found that women with breast cancer who had chronic hyperglycemia were often less educated, had nonwhite race, and had breast cancer that was more advanced at diagnosis. Kabat et al. (27) also found that an elevated serum insulin level was a risk factor for breast cancer. Dankner et al. (28) showed that there may be a negative role of elevated basal insulin levels in cancer prognosis including breast cancer.
In our study, we found that the rate of breast cancer mortality was approximately 50% among women from Somalia, although few such women were included in this study. There is limited information in the literature about breast cancer survival among women from Somalia living in European countries. Our study indicates that women from Somalia may be at particular risk of a poor prognosis, after breast cancer diagnosis.
In summary, this study demonstrates significant differences in breast cancer survival between various ethnic groups in Norway. Immigrants from Asia and Africa experience higher relapse rates and mortality from breast cancer compared to ethnic Norwegian women. Breast cancer is diagnosed at an earlier age in women from PSS (especially in women from Sri Lanka and Somalia) than in ethnic Norwegian women. Therefore, many women from PSS miss the window of opportunity to benefit from mammography screening in Norway. Finally, the incidence of DM is high in the immigrant group, but the impact of DM on the patient prognosis is difficult to know, since there is missing information about metabolic syndrome among patients included in the study. The results from this study demonstrate that special attention should be paid to women from Somalia and Sri Lanka with respect to breast cancer care. Efforts should be made to detect tumors at early stage, for example, by offering mammography screening at a younger age, and by increasing awareness about breast cancer and self-examination in this group.
Footnotes
Declaration of conflicting interests
The authors declare that they have no conflict of interest.
Funding
This work was supported by the University of Oslo.
