Abstract
Background and Aims:
The aim of the study was to clarify the frequency and the sequel of surgical complications occurring within 1 year after renal transplantation.
Patients and Methods:
Surgical complications after 1670 consecutive adult kidney transplantations performed between 2000 and 2009 were retrospectively analyzed. In 2%, a living-related allograft was used, and 10% were retransplantations. An intravesical technique without stenting was used for the ureteric implantation.
Results:
There were 282 surgical complications occurring in 259 (15.5%) transplantations. Ureteral obstruction occurred in 53 (3.1%), lymphoceles in 39 (1.5%), postoperative hemorrhage in 36 (2.1%), and renal vein thrombosis in 22 (1.3%) patients, respectively. Out of the 17 lung emboli, 4 were fatal. Male recipients had twice as much ureteral stenosis as female (2.4 vs 1.2%, p < 0.05), and the opposite was true of urinary leakage (1.8% vs 4.0%, p < 0.025). Five-year patient and graft survival was impaired in patients with complications compared with patients without complications. Five-year patient survival was 92% versus 88% and graft survival 87% versus 74%.
Conclusion:
Surgical complications impair patient and graft survival after kidney transplantation.
Introduction
Surgical complications after kidney transplantation continue to be a problem despite meticulous surgical technique and progress in the pre- and postoperative care of kidney transplant recipients. The complications are rarely life threatening at presentation, but may increase the length of hospitalization as well as costs. Timely diagnosis and appropriate surgical or percutaneous interventions are imperative to avoid an increased risk of morbidity and a threat to transplant outcome (1). The incidence of surgical complications following kidney transplantation ranges from 4.2% to 34% depending on series, and on the types of complications reported in the studies (2, 3).
Surgical complications after kidney transplantation have been mainly attributed to technical problems during organ procurement and transplantation. However, today the range of acceptance criteria for suitable kidneys for transplantation has been expanded, including a greater proportion of marginal donors and suboptimal grafts (4–6). In addition, atheromatosis and obesity are increasingly observed among kidney transplant candidates (7), which might increase the risk of vascular thrombotic events as well as ureteral complications after surgery.
The aim of the study was to clarify the frequency and the sequels of surgical complications occurring within 1 year after kidney transplantation in a series of 1670 consecutive transplantations from a single center.
Patients and methods
We retrospectively reviewed 1670 consecutive kidney transplantations performed in 1650 patients. The transplantations were performed between January 2000 and December 2009 at the Department of Transplantation and Liver Surgery at Helsinki University Central Hospital. The aim of this study was to assess the surgical complications occurring within 1 year after kidney transplantation. Of the transplants, 98% were obtained from deceased heart beating donors. In 2%, a living-related allograft was used, and 10% of the procedures were retransplantations. The demographics of the kidney transplantations are shown in Table 1. As a prerequisite for placing a patient on the kidney waiting list at our institution is that patients with gallstones should have their gallbladders removed and patients with symptomatic colon diverticula should have had a sigmoidectomy before becoming accepted on the kidney waiting list. Also, until 2007 for obese patients, a body mass index (BMI) ≤30 was required before the acceptance on the waiting list.
Clinical data of 1670 adult kidney transplantations with and without surgical complications.
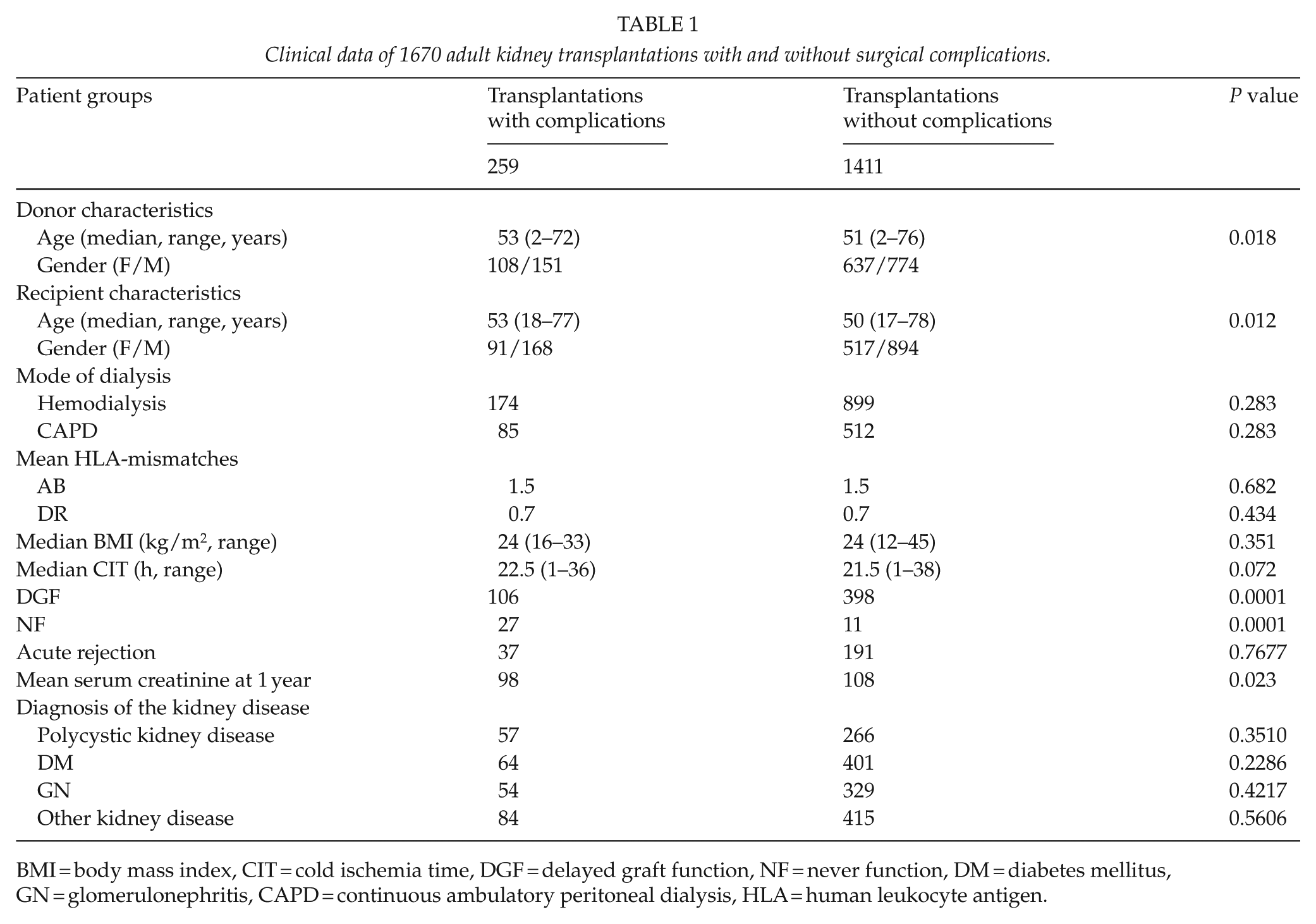
BMI = body mass index, CIT = cold ischemia time, DGF = delayed graft function, NF = never function, DM = diabetes mellitus, GN = glomerulonephritis, CAPD = continuous ambulatory peritoneal dialysis, HLA = human leukocyte antigen.
After transplantation, recipients were followed-up as in-patients for 3 weeks at the transplant unit. Thereafter, patients were followed in their local nephrology centers, from which follow-up data are routinely sent to the Finnish Kidney Transplant Registry. Complication data for this study were retrieved from the Finnish Kidney Transplant Registry and confirmed from clinical records covering the hospitalization period and local outpatient follow-ups.
The standard immunosuppression was a triple-drug regimen with Cyclosporine A, mycophenolate mofetil (MMF), and steroids. In immunologically high-risk patients (high level of panel reactive antibodies (PRA), previous graft lost in rejection, or long waiting time), cyclosporine was replaced by tacrolimus, and/or induction therapy with basiliximab.
Biopsy-proven acute rejections were treated depending on the type of rejection (cellular or antibody mediated) with high-dose intravenous corticosteroids and/or conversion of cyclosporine to tacrolimus and, in steroid-resistant rejections, with plasmapheresis or the monoclonal antibody OKT 3.
Cytomegalovirus (CMV) infections were treated with IV ganciclovir or high-dose valganciclovir (900 mg twice daily if normal renal function), followed by 1–6 months secondary oral valganciclovir prophylaxis in most patients.
Delayed graft function (DGF) was defined as described by Halloran et al. (8): oliguria less than 1 L/24 h for more than 2 days, or plasma creatinine concentration greater than 500 µmol/L throughout the first week after transplantation, or more than one dialysis session needed during the first week after transplantation.
Intravenous steroids were given to all donors before undergoing the organ retrieval operation. The University of Wisconsin solution was used for in situ perfusion and cold storage preservation of the kidneys.
The internal iliac artery was used as inflow vessel in living-related transplantations with end-to-end anastomosis, and the external iliac artery with end-to-side anastomosis in most of the kidney transplantations from deceased donors.
The vesico-ureteric anastomosis was fashioned after revascularization of the kidney and following distension of the bladder. The surgical procedure for the ureteric implantation was performed using modified Leadbetter–Politano intravesical procedure. Ureteric stents were not used. No suction drains were used. A Foley catheter was left in the bladder for 5 days.
Antibiotic prophylaxis consisted of two doses of cefuroxime administered prior to transplantation and 12 h after surgery. Renal function was monitored daily for 3 weeks after the transplantation by plasma creatinine levels and urine output. Ultrasound was employed routinely for follow-up at Day 1 and for special indications. Percutaneous nephrostomy and antegrade pyelography were readily done if indicated, and the nephrostomy was retained for securing renal function as needed. In case of a ureteral stricture, a JJ-stent was inserted if possible. If the treatment with nephrostomy or stenting failed, a surgical ureteric reconstruction was completed usually 3 to 6 months later. Lymphoceles causing symptoms were primarily treated with percutaneous drains. Surgical marsupialization into the abdominal cavity was performed laparoscopically or by open surgery in selected cases. All the transplantations were performed by or under the direct supervision of a consultant transplant surgeon.
Major complications in the present series are shown in Table 2. Miscellaneous surgical complications included, for example, wound infections, abscess formation in the proximity of the kidney, colon diverticulitis, renal artery stenosis, peptic ulcers, cholecystitis, ileus, non-infectious wound complications, bladder obstruction, arteriovenous fistula after core needle biopsy, and rupture of the kidney vein during transplantation.
Surgical complications in 1670 kidney transplantations.
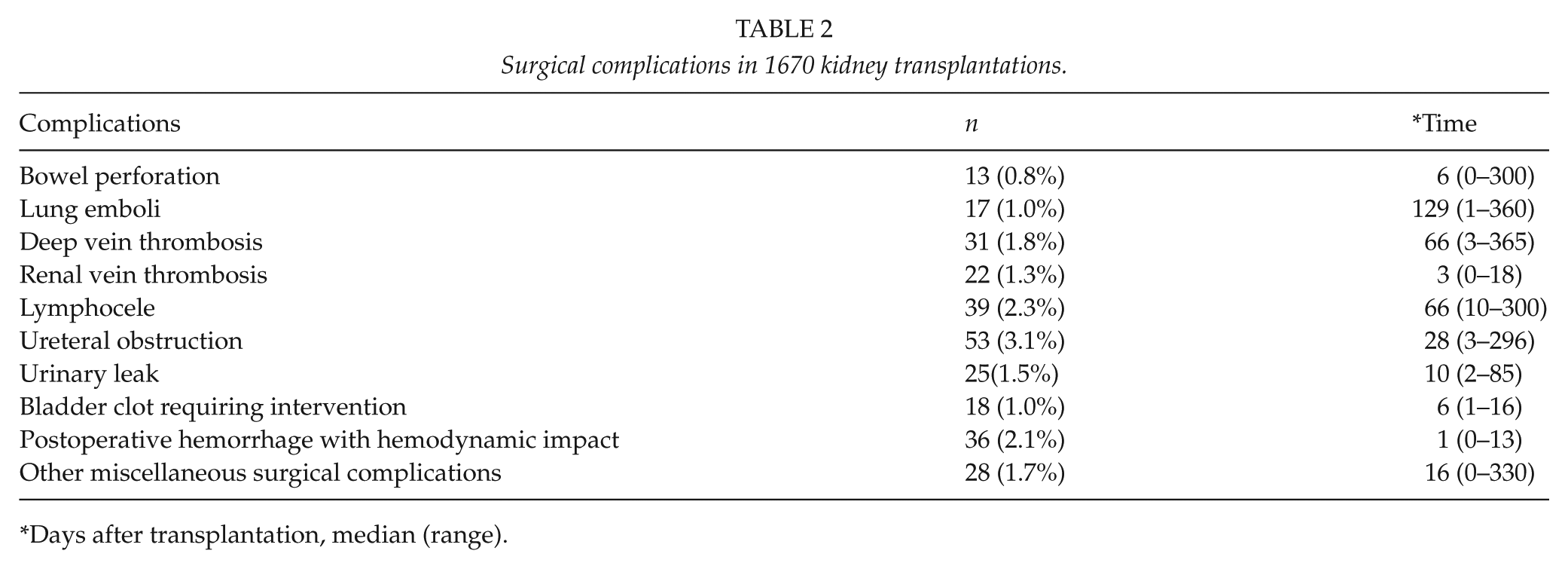
Days after transplantation, median (range).
Analysis Of Data
The variables were compared using the Mann–Whitney U test and the χ2 test. A p value of <0.05 was considered significant. Survival was univariately estimated by comparing survival probabilities according to the Kaplan–Meier product limit method and log-rank test. Correlations were assessed by the Spearman rank correlation test.
Results
All in all, there were 282 surgical complications during the first year after transplantation in 259 (15.5%) patients. The donors as well as the recipients who developed complications were markedly older than those who did not develop complications; no gender difference was, however, observed (Table 1). There were 175 transplant recipients older than 65 years. In the group without complications, 9% were >65 years old, whereas in the complication group 14% were >65 years (p = 0.025). There was no marked difference in the time on dialysis or on the waiting list between the patients with surgical complications and those without complications.
Table 2 shows the surgical complications after kidney transplantation. Ureteral obstructions were observed in 53 (3.1%) patients. These were primarily treated with pyelostomy and ureteral stenting and only 7 (13%) patients needed later surgical reconstruction. Urinary leakage occurred in 25 patients out of which 6 (24%) were surgically treated. Male recipients had twice as much ureteral stenosis as female (2.4 vs 1.2%, p < 0.05) and the opposite was true of urinary leakage (1.8% vs 4.0%, p < 0.025). The median donor age in the group with ureteral stenosis was significantly higher than in that without stenosis (p = 0.014). A combined leakage and stricture was observed in five cases.
A total of 39 (2.3%) lymphoceles needing interventions were encountered, out of which 12 (30%) required surgery (4 laparoscopically and 8 by open surgery); in the other 27 cases, percutaneous drainage was sufficient. In three patients, the lymphocele compressed the iliac vein causing vein thrombosis and a subsequent lung emboli, one of which was fatal. All in all, 4 (23.5%) of the 17 lung emboli were fatal.
Transplant vein thrombosis occurred in 22 (1.3%) cases, resulting in immediate transplantectomy in all but one case, in which a marginal graft function was obtained after thrombectomy. In 20 out of 22 cases with transplant vein thrombosis, the transplanted kidney was the right donor kidney. Deep vein thrombosis occurred in 31 patients. Renal artery thrombosis occurred in 5 (0.3%) cases.
Postoperative bleeding with hemodynamic impact was observed in 36 (2.1%) of the cases and a bladder clot needing intervention was seen in 18 (1.0%) patients (Table 2). Most postoperative bleedings were diagnosed on the first day after the transplantation.
Bowel perforations occurred in 13 (0.8%) patients. All but one of the bowel perforations occurred in the colon. In the group of miscellaneous surgical complications, there were also other gastrointestinal (GI) complications: one case of cholecystitis, two cases of colon diverticulitis, and four gastric ulcers cases. All in all, there were 21 (1.2%) GI complications.
The overall acute rejection rate was 14%, and the overall DGF rate was 30%. There was no marked difference in episodes of acute rejection between patients with or without surgical complications (Table 1); also, no significant difference in the diagnosis of the kidney disease could be seen between the groups (Table 1). Likewise, no marked difference in the human leukocyte antigen (HLA) mismatches or mode of dialysis could be observed between the patients with or without complications (Table 1).
There were 27 never functioning (NF) kidneys in the complication group, out of which 21 were due to renal vein thrombosis. In the group without surgical complications, there were 11 NF kidneys.
There were moderate correlations between BMI and bleeding (ρ = 0.078, p < 0.001), BMI and deep vein thrombosis (ρ = 0.082, p < 0.001), and BMI and renal vein thrombosis (ρ = 0.058, p < 0.018). Only 86 (5%) patients in the whole cohort had a BMI >30 kg/m2. Worth to observe is that the patients with renal vein thrombosis were leaner than those without. There were moderate correlations between age and bowel perforation (ρ = 0.054, p < 0.026), age and pulmonary emboli (ρ = 0.067, p < 0.006), and age and other miscellaneous complications (ρ = 0.051, p < 0.039).
According to the Clavien–Dindo classification of surgical complications (9), 1% of the complications were Grade I, 23% Grade II, 42% Grade IIIa, 31% Grade IIIb, 1% Grade Iva, and 2% Grade V.
Kaplan–Meier overall patient and graft survival curves for patients with and without surgical complications are shown in Fig. 1A and B. Both 5- and 10-year patient and graft survival were impaired in patients with surgical complications compared to those without complications. Five- and ten-year patient survival was 92% versus 88% and 79% versus 73%, respectively. Five- and ten-year graft survival was 87% versus 74% and 72% versus 60%, respectively.
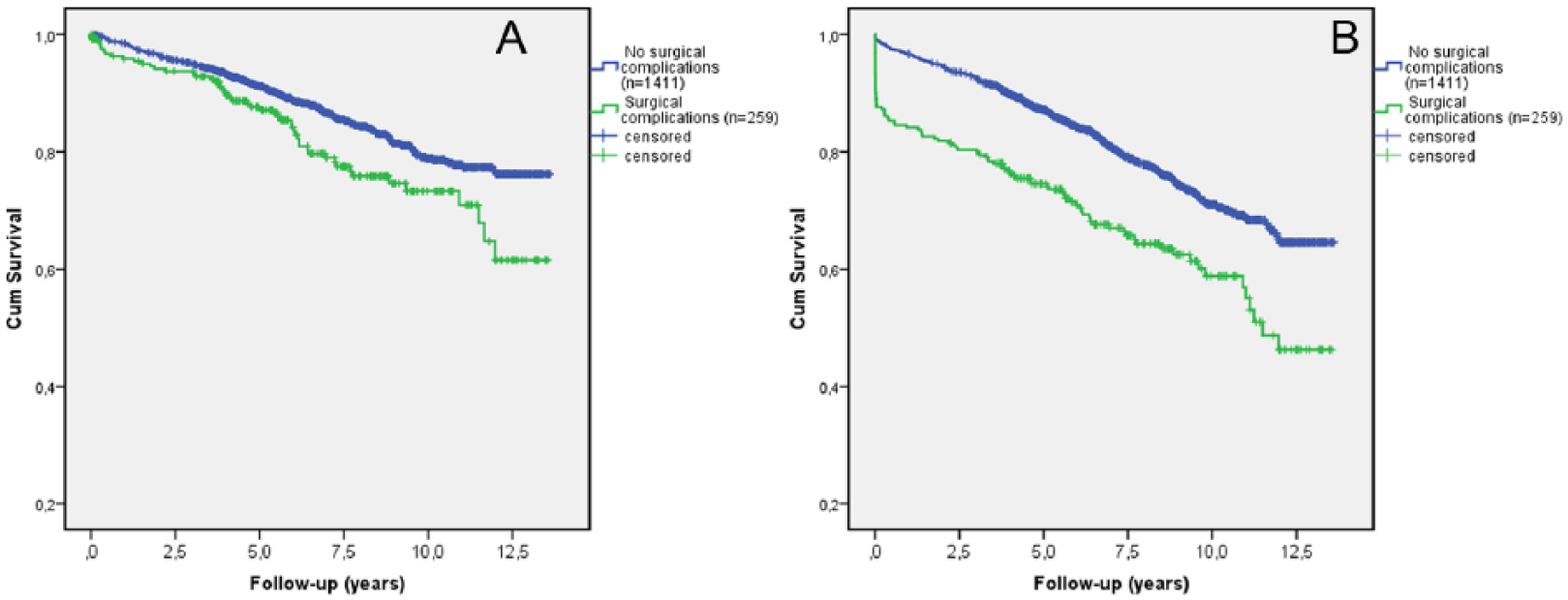
Kaplan–Meier survival curves. (A) Patient survival curves in patients with no surgical complications and in patients with surgical complications. (B) Graft survival curves in patients with no surgical complications and in patients with surgical complications.
Discussion
In this retrospective study, the overall incidence of surgical complications was 15.5%. We showed that both patient and graft survival was impaired in patients with surgical complications compared with those without complications. In previous studies, the range of surgical complications has varied between 4.2% and 35%. In a previous study from our department looking at urological complications, the complication rate was 4.2% (2). In this study, when looking at the same “urological complications” the complication rate was 8%. The increase in complications in this study compared with the previous one from our department might be explained by the fact that there was a clear increase in the mean age of the recipients and the donors. The impact of older age is in accordance with the recent study by Barba et al. (10). The retrospective nature of the study and the fact that only complications needing interventions were collected resulted in the low percentage of surgical complications of Grades I and II according to the Clavien–Dindo classification system (9) in this study.
The two major surgical/urological complications after kidney transplantation are urinary leaks and collecting system obstruction. The transplant ureter is highly susceptible for damage due to reduced blood supply. Ureteric ischemia, in the absence of technical complications, is thought to be mainly responsible for the complications (11). In this study, the method for the ureteroneocystostomy was a two-incision intravesical technique without stenting, a modified Leadbetter–Politano procedure. This technique has been superseded by the Lich–Gregoir extravesical ureteroneocystostomy technique in many centers. There are few studies comparing these two techniques, but it seems as the implantation technique of the transplanted ureter does not have a significant impact on the ureteral stenosis rate (12). In this study, the overall stenosis rate was 2.0%, which is markedly higher than in the previous study from our institution, but in the same range as in previous studies using either the extravesical or intravesical technique with or without stenting (12). Over the recent years, there has been debate as to whether stenting the ureterocystostomy results in a reduced rate of leaks and strictures postoperatively; some studies are advocating a routine use of stents (13) and some do not (14). The leakage rate in this study is in accordance with previous studies (15). Karam et al. (16) found that donors over 65 years represented an independent risk factor for ureteral stenosis. In this study, the median age of donors with ureter strictures was higher than in the group without stricture. However, we could not find any good reason for why male kidney recipients had twice as much ureteral stenosis as female and why the opposite was true of urinary leakage.
In 28% of the patients, diabetes was the indication for transplantation in our study. However, it did not correlate with the occurrence of surgical complications, which is in accordance with the study from Minnee et al. (17). However, in the earlier study from our institution, a significantly higher incidence of diabetes was observed in the group with urological complications compared with those without complications (2). One possible explanation might be that diabetic care and dialysis are better in the current era making the diabetic recipients less prone to excessive complications.
There are a growing number of obese patients receiving kidney transplants (18). Obesity has been linked to complications during kidney transplantation, because of increased difficulty in surgery, longer operation times, and delayed wound healing (19). A BMI over 30 has been associated with an increased risk of patient and graft loss (20, 21). However, in a recent study by Zrim et al. (22), a higher BMI was not associated with surgical or urological complications. Out of the recipients reported here, 86 had a BMI of more than 30 at the time of transplantation. High BMI correlated with the risk of deep vein thrombosis, again a risk factor for lung emboli.
Lymphocele, a well-known complication, has the incidence of 1%–26% after kidney transplantation (23). Most lymphoceles are asymptomatic and are easily noticed during routine ultrasound examination. Symptomatic lymphoceles can cause hydronephrosis, pain, ipsilateral leg swelling, and deep vein thrombosis (24, 25). The incidence of symptomatic lymphoceles in this study is in accordance with previous studies (26) but more than double that of the previous study from our institution (2). More regular use of ultrasound during recent times has made the diagnosis and follow-up of lymphoceles easier. Nevertheless, although infrequent, lymphoceles may have fatal consequences, causing even one death in this study. The management of lymphoceles after kidney transplantation is variable. Laparoscopic fenestration of symptomatic lymphoceles has been associated with the lowest risk of lymphocele recurrence (23), but treatment with percutaneous drainage and some sclerosing agent is far less invasive. In this series, most of the lymphoceles could be treated successfully with percutaneous drainage, however.
Incidence rates of postoperative bleeding between 6% and 12% (3) have been reported, and they usually occur early after the transplantation. Factors promoting bleeding are, for example, high urea levels, antiaggregant and anticoagulant therapies received by many of the transplant recipients. Unfortunately, the use of any antiaggregant or anticoagulant therapy was not recorded in this study.
Severe gastrointestinal complications after kidney transplantation have been found in up to 10% of patients (27). In the era before the use of proton-pump inhibitors and H2 blockers, peptic ulcers accounted for up to 4% of deaths after transplantations (28). In the study by Sarkio et al. (27), the frequency of gastric ulcers was 4% during a follow-up time of 6 years in median. In this study, only four gastric ulcers were observed during the first year after the transplantation. The incidence of colon perforations after kidney transplantation has been around 1%–2% (27, 29), which is in accordance with this study. Particularly aged patients and patients with polycystic kidney disease have been reported to have an increased risk of colonic complications (27, 28). In this study, polycystic kidney disease was not associated with bowel perforation, but the patients diagnosed with perforation were markedly older than those without.
Renal allograft vascular thrombosis is a serious complication following kidney transplantation that ultimately leads to graft loss. The incidence of renal venous thrombotic complications in our study was within the range previously reported (3). In this study, they occurred early after the transplantation, in median on the third day, as in most previous studies. Causes might be kinking of the vein, compression by hematomas, anastomotic stenosis, extension of an underlying deep venous thrombosis, and a hypercoagulable state (1). In this study, no correlation with the aforementioned causes could be observed. There have been reports that thrombosis is more common in right grafts because of the shorter length of the vein, which could cause its mechanical collapse (30).
Our retrospective study design may be regarded as a limitation concerning, for example, the registration of mild Grades I and II surgical complications. It is also very difficult or almost impossible to judge which of the complications are patient or surgical related especially in a retrospective study like the present one. As, for example, in the cases of the transplant kidney vein thrombi, where in 20 out of 22 cases, the right donor kidney was used, here you might presume that the graft plays some part, but of course the surgical technique has as much impact on the outcome as the kidney graft. The strength of the study is, however, that the transplant procedure has remained unchanged during the study period as well as the immunosuppressive protocols.
In conclusion, our findings indicate that surgical complications, still in the modern era, impair graft as well as patient survival after kidney transplantation. Old age is a risk factor for complications and thus, increasing age of both kidney recipients and donors will challenge the illusion of ever improving results of kidney transplantation.
Footnotes
Acknowledgements
This study was supported by grants from Helsinki University Hospital Funds (EVO).
Declaration of Conflicting Interests
None declared.
