Abstract
Aims:
Acute acalculous cholecystitis can be treated with percutaneous cholecystostomy in critically ill patients unfit for surgery. However, the evidence on the outcome is sparse. We conducted a retrospective analysis of acute acalculous cholecystitis patients treated with percutaneous cholecystostomy during a 10-year study period.
Methods:
An observational study of 56 consecutive patients treated with percutaneous cholecystostomy for acute acalculous cholecystitis was conducted in the period from 1 June 2002 to 31 May 2012. All data were obtained by review of medical records.
Results:
A total of 56 consecutive patients were treated with percutaneous cholecystostomy for acute acalculous cholecystitis. Six patients (10.7%) died within 30 days after the procedure. Percutaneous cholecystostomy could serve as a definitive treatment option in 45 patients (80.4%), whereas 1 patient (1.8%) required cholecystectomy due to recurrence of cholecystitis. Four patients (7.1%) were treated with percutaneous cholecystostomy as a bridging procedure to subsequent elective laparoscopic cholecystectomy within a median of 8.8 months (range: 7.7–33.4 months). There was no significant difference in the risk of cholecystitis recurrence between patients with (6/37) and without (2/3) contrast passage to the duodenum on cholangiography (p = 0.096).
Conclusion:
Percutaneous cholecystostomy is successful as a definitive treatment option in the majority of patients with acute acalculous cholecystitis. It is associated with a low rate of mortality and subsequent cholecystectomy.
Introduction
Early cholecystectomy is the standard treatment in patients with acute cholecystitis and carries a low rate of morbidity and mortality (1–3). However, patients with acute acalculous cholecystitis (AAC), which generally occurs in the course of a serious illness or following major surgery (4), are often unsuitable for general anesthesia. Thus, more appropriate treatment modalities for these patients should be considered.
Percutaneous cholecystostomy (PC) offers an alternative treatment option in critically ill patients with cholecystitis (5). In PC, a catheter is placed in the gallbladder lumen under ultrasound guidance, resulting in decompression of the gallbladder and thus relief of the inflammatory condition (6). PC is mainly used as a bridging procedure followed by delayed cholecystectomy when the patient is stabilized, although it can also be used as a definitive treatment option for patients considered unfit for surgery (6).
However, the outcome of PC as a definitive treatment option in patients with AAC unfit for surgery is unclear and has only been investigated by few studies (7–10). Although the series were small, results are promising. In addition, two major studies comparing PC with other treatment modalities have also presented beneficial results (11, 12). However, to date, only one study of a sufficient size has evaluated PC as a definitive treatment in an exclusive population of AAC patients (13).
To examine the effectiveness of PC as a definitive treatment option in patients with AAC unsuitable for surgery, we conducted a retrospective analysis of all patients treated with PC as intended definitive treatment for AAC at our institution during a 10-year study period.
Methods
We conducted a retrospective study of all consecutive patients treated with PC for AAC at our institution in the period from 1 June 2002 to 31 May 2012. During the study period, no patients with AAC were treated by other measures than PC.
Patient Selection
By searching the hospital’s patient administrative system, we identified 60 consecutive patients with AAC treated with PC during the study period. The patients were identified using the following procedure codes from The Danish Classification of Surgical Procedures and Therapy: KJKA16 (percutaneous gallbladder drainage), UXRD46 (cholangiography through a catheter), and UXRD40 (cholangiography). We excluded 2 patients, who were lost to follow-up and 2 patients, who both underwent early cholecystectomy during the index admission, leaving us with a final study population of 56 patients (Fig. 1).
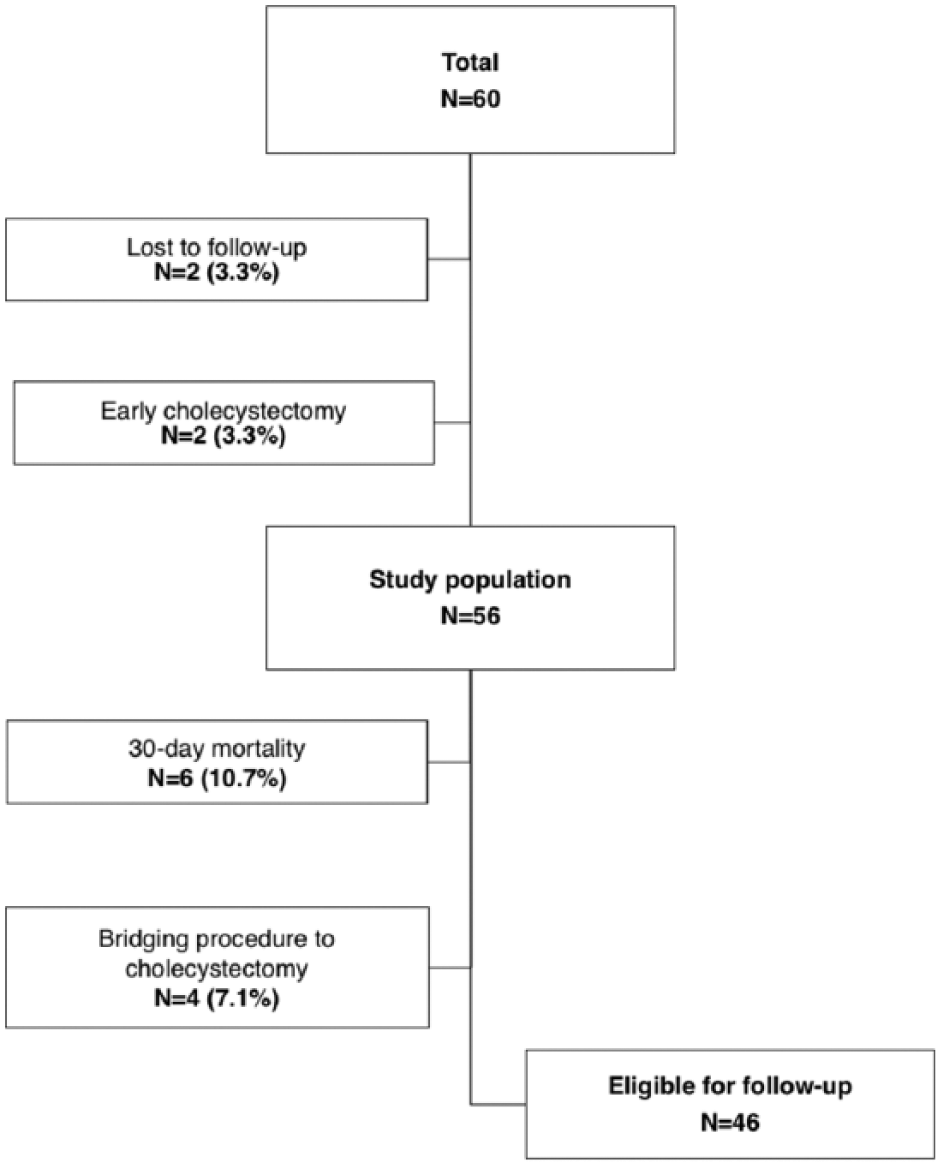
Flowchart of 60 consecutive patients treated with percutaneous cholecystostomy for acute acalculous cholecystitis.
Data on Patients and Procedures
To obtain data on patient demographics, symptom duration, treatment modality and outcome, cholecystectomy and other operations performed during the index admission, recurrence of cholecystitis, and 30-day mortality, all patients’ medical records were reviewed. Any subsequent cholecystectomy at our institution was recorded. We identified patients undergoing subsequent cholecystectomy at another Danish hospital using the Danish National Patient Registry (DNPR), which comprises information on all in-hospital admissions since 1977 and all outpatient clinic and emergency department visits since 1995 in Denmark (14). Linkage to the DNPR was possible through the civil personal registration number, a unique identification number assigned to all Danish residents at birth or immigration.
Diagnosis of Aac and Treatment Algorithm
The diagnosis of AAC was based on symptomatology and objective examination (abdominal pain in the upper right quadrant, a positive Murphy’s sign, and fever) plus biochemical tests (elevated blood levels of C-reactive protein or leukocytes and possibly affected liver function tests). The clinical evaluation was supplemented with ultrasonography or, in rare instances, computed tomography (CT) imaging or magnetic resonance cholangiopancreaticography (MRCP). On imaging, absence of gallstones in combination with enlargement of the gallbladder wall (≥5 mm), probe tenderness, and pericholecystic fluid were regarded as signs of AAC. Complicated symptomatology, for example, suspected cholangitis, prompted an endoscopic retrograde cholangiopancreaticography (ERCP) before a PC was performed.
Cholangiography
Following PC, an antegrade cholangiography through the catheter was performed on postoperative day 3. If no contrast passage to the duodenum was visualized, the procedure was repeated on postoperative day 5. In case of contrast passage to the duodenum and a satisfactory clinical response, the catheter was removed. The presence of common bile duct stones prompted an ERCP.
Technique of Pc
PC was performed under ultrasound guidance by a trained interventional radiologist. The choice of either a transperitoneal or transhepatic approach varied according to personal preference of the radiologist and availability of the gallbladder under the given circumstances. The catheter was placed under local anesthesia using an aseptic technique. In the case of a transhepatic placement, the gallbladder was always punctured using Seldinger’s technique, and a 7-Fr pigtail catheter (Skater, Single Step Drainage Z-locking; PBN Medicals, Stenløse, Denmark) was placed in the gallbladder lumen. In the case of a transperitoneal placement, a one-step method using a 7-Fr pigtail catheter was also used. The catheter was subsequently fixed to the skin using a patch and flushed up to three times daily.
Statistical Analyses
Numerical data were described using medians with ranges as measures of variation, and comparisons of numerical data were carried out using a nonparametric median test. Categorical data were compared using a chi-squared test. Any two-sided p-value less than 0.05 was considered statistically significant. All statistical analyses were carried out using Stata 13 (StataCorp LP, College Station, TX, USA). This study was approved by the Danish Data Protection Agency (j.nr. 2007-58-0010).
Results
We included 56 consecutive patients treated with PC for AAC (Table 1).
Descriptive characteristics of 56 consecutive patients treated with percutaneous cholecystostomy for acute acalculous cholecystitis.
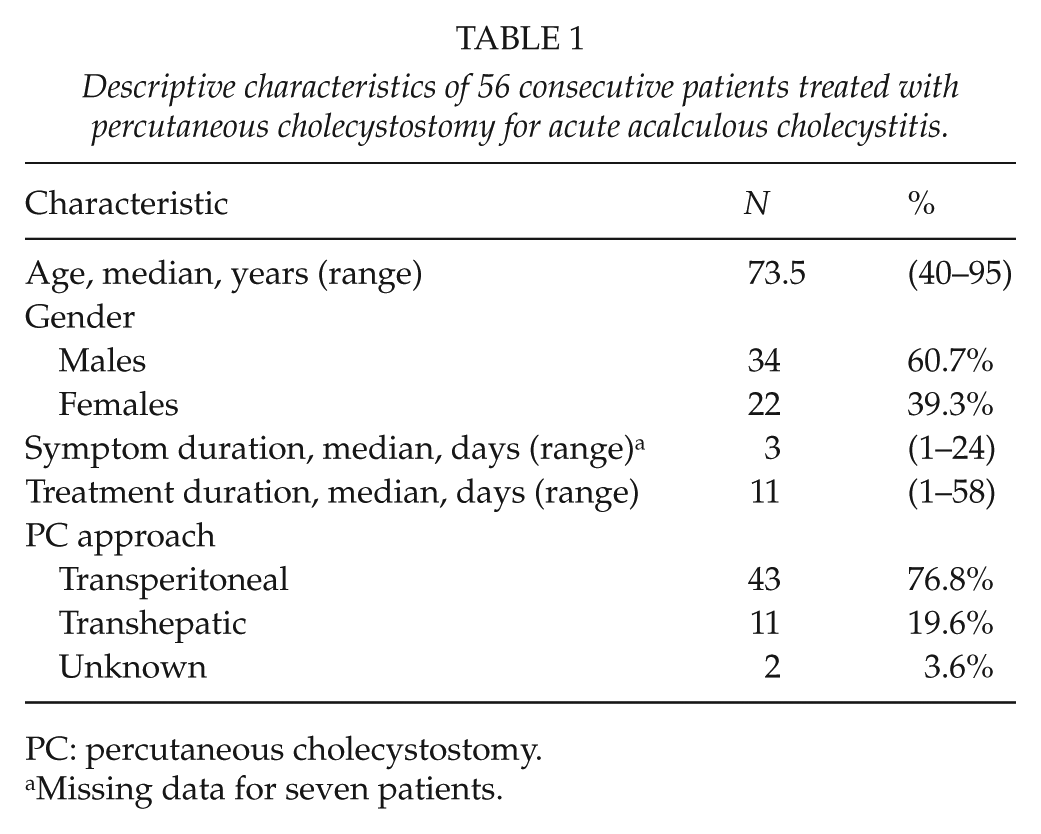
PC: percutaneous cholecystostomy.
Missing data for seven patients.
Treatment Outcome and Recurrence of Cholecystitis
We experienced a 30-day mortality of 10.7% (N = 6). Median duration from PC to death was 8 days (range: 4–24 days). In 4 patients (7.1%), the initial PC served as a bridging procedure to subsequent cholecystectomy within a median of 8.8 months (range: 7.7–33.4 months), leaving us with 46 patients eligible for follow-up. Of these, 38 patients (67.9%) were successfully treated with the initial PC as the definitive treatment strategy, whereas 8 patients (14.3%) experienced recurrence of cholecystitis. All 8 patients were subsequently successfully treated with repeated PC (N = 5), cholecystectomy (N = 1), or conservative treatment (N = 2). Thus, in total 45 patients (80.4%) required no further intervention than PC as a definitive treatment option for AAC. Patients with recurrence of cholecystitis did not differ from patients without recurrence of cholecystitis (Table 2).
Descriptive characteristics of 46 consecutive patients eligible for follow-up after treatment with percutaneous cholecystostomy for acute acalculous cholecystitis according to recurrence.
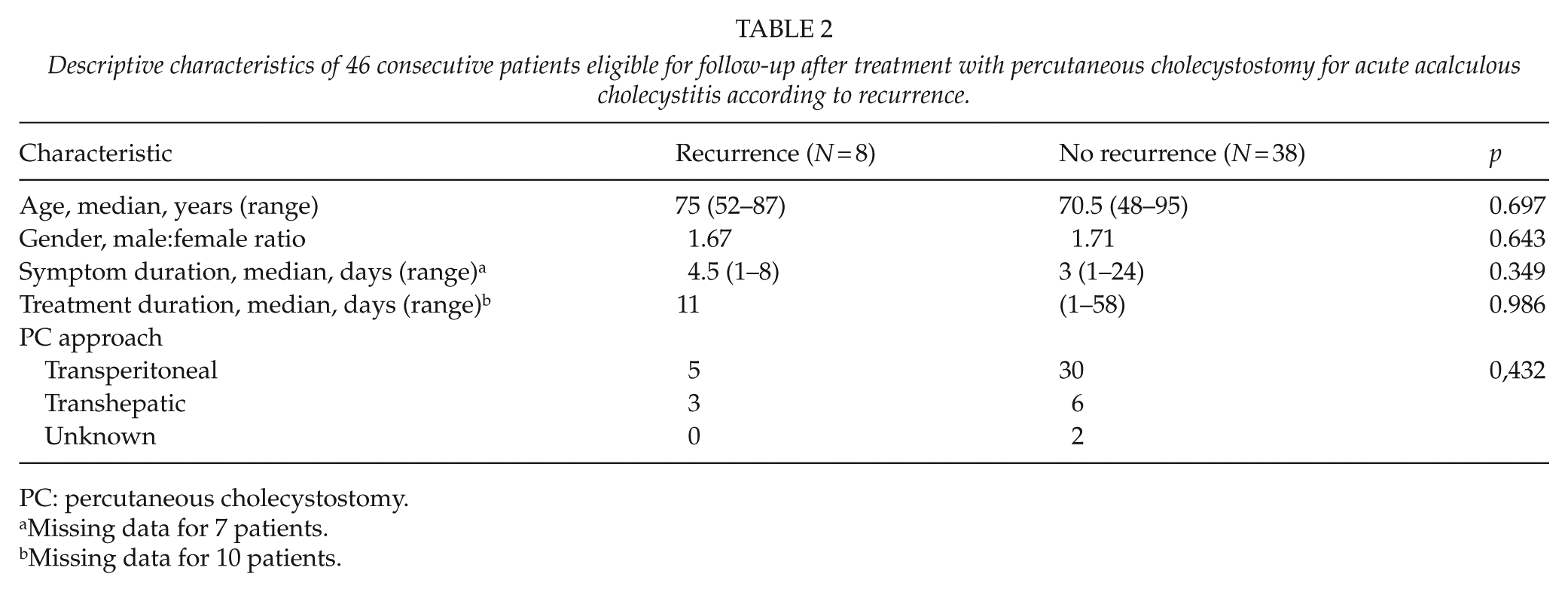
PC: percutaneous cholecystostomy.
Missing data for 7 patients.
Missing data for 10 patients.
Complications
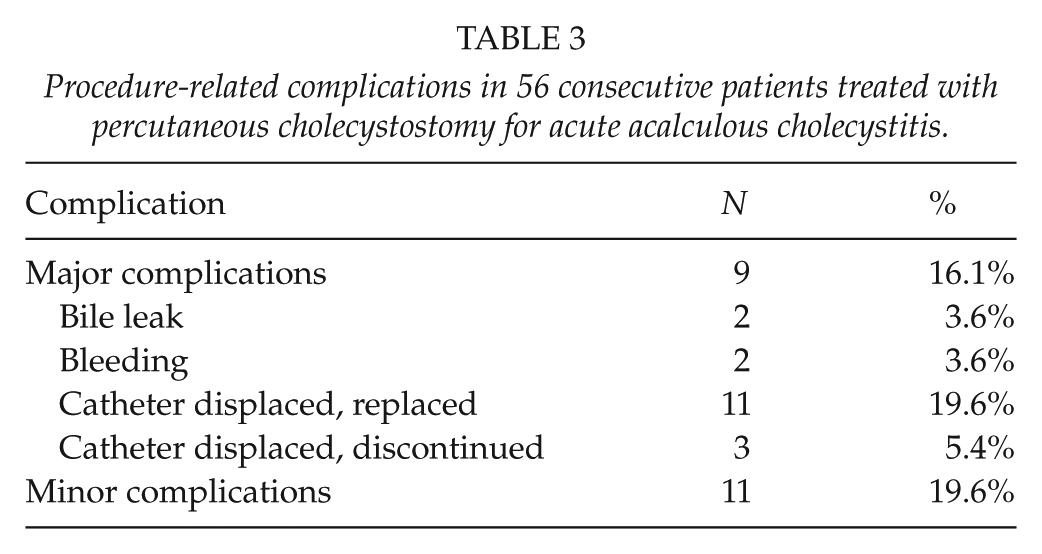
In total, 9 (15.8%) patients experienced major complications, of which the majority was related to displacement of the catheter (Table 3). The catheter was only replaced in the absence of clinical improvement. A total of 11 patients (19.6%) experienced minor complications. There seemed a tendency toward fewer complications in patients treated with the transhepatic approach compared to patients treated with the transperitoneal approach, but our sample was too small for statistical analyses (data not shown).
Procedure-related complications in 56 consecutive patients treated with percutaneous cholecystostomy for acute acalculous cholecystitis.
Cholangiography
Of the 46 patients scheduled for follow-up, 40 underwent cholangiography to examine contrast passage from the gallbladder to the duodenum (Fig. 2). There was no significant difference in the risk of cholecystitis recurrence between patients with (6/37) and without (2/3) contrast passage to the duodenum (p = 0.096).
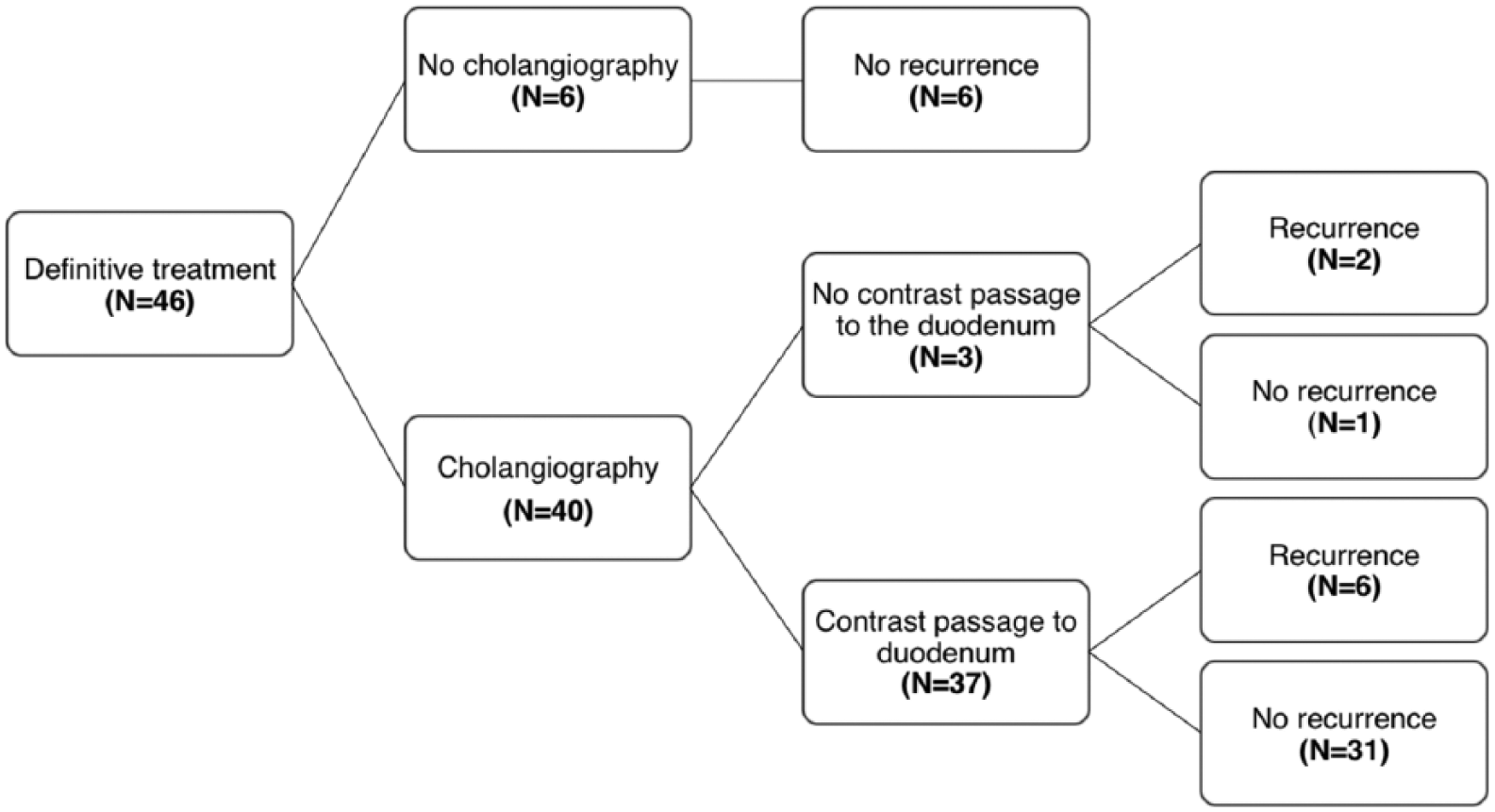
Results of cholangiography in 46 patients definitively treated with percutaneous cholecystostomy for acute acalculous cholecystitis.
Discussion
In this study of 56 consecutive patients with AAC over a 10-year period, we demonstrated that PC could serve as the definitive therapy in the majority (80.4%) of the patients, of which 5 patients needed a repeated procedure. In addition, we present a 30-day mortality rate of 10.7% reflecting the burden of comorbid diseases in these patients.
Previous minor studies including 30 patients or less have examined the outcome of PC in patients with AAC (10, 15–18). However, the majority of these studies did not distinguish between acalculous and calculous cholecystitis in their analyses. As these conditions differ in pathophysiology and outcome, comparisons across studies are difficult. Other studies with the same lack of distinction between acalculous and calculous cholecystitis have suggested that PC could be a definitive treatment approach in patients with cholecystitis unfit for surgery in around 80% of the patients, which corresponds with our findings (19, 20).
In a study of 57 patients, which is similar to our population, Chung et al. (13) investigated the long-term outcome of PC as a definitive treatment in patients with AAC. They experienced an in-hospital mortality of 21%, which is twice as high as our finding. The majority of their deaths were due to underlying diseases. Our study contained no information on the cause of death or severity of the patient’s illness. Thus, in theory, the discrepancy in mortality rates could be attributable to a possibly more critically ill population in the study by Chung et al. However, as all patients in our study were unsuitable for surgery, the diverging mortality rates are unlikely to be explained by differences in the severity of the patients’ illnesses.
In our study, the majority underwent cholangiography to ensure contrast passage to the duodenum. There was a tendency toward a higher rate of cholecystitis recurrence among patients without contrast passage compared with those with contrast passage on the cholangiogram. Although numbers are too small for statistical analyses, our findings are in line with findings by Chung et al., reporting that none of the two patients with recurrence in their study had a cholangiography. Furthermore, Chung et al. report that they left the catheter in situ for a median of 51 days, which is much longer than in our study (median 10 days). There is conflicting evidence on the optimal time to remove the catheter. Some authors suggest a minimum duration of 6 weeks to ensure resolution of inflammation (21), although others successfully removed the catheter after 1 week (16).
We experienced a rate of major complications such as bile leak and catheter displacement in 15.8% of our population. This is higher compared to findings by Melloul et al. (22) but lower compared to a study by Nikfarjam et al. (17). However, once again, these studies did not distinguish between calculous and acalculous cholecystitis in their analyses. In the literature, the rate of major complications associated with PC for both acalculous and calculous cholecystitis is reported between 3% and 8% (6), which is lower than in our study. The discrepancies could be attributable to a high proportion of patients with calculous cholecystitis, and thus possibly fewer critically ill patients, in the studies by Melloul and Nikfarjam.
Several limitations must be taken when interpreting our findings. Due to the retrospective design, we had no fixed algorithm on the diagnosis of AAC. However, as we found no gallstones on any imaging modality, we expect our diagnoses to be correct. Although our study is large compared to most previous studies, our population size is a limitation. In addition, we had no information on the severity of the patients’ comorbid burden or primary condition, which limits the possibility of comparing results with other studies. However, no patient was initially suitable for surgery, which indicates that the patients were all critically ill. Furthermore, we had no information on long-term mortality, and we cannot preclude the possibility that patients had recurrence of cholecystitis and were admitted to another hospital. However, we were able to check the DNPR for any subsequent cholecystectomy performed at another hospital. Thus, only patients undergoing a cholecystectomy at another hospital could be recorded.
Our study was confined to include AAC patients only, which we consider a major strength. Most previous studies examining PC as a definitive treatment option consisted of patients with both calculous and acalculous cholecystitis, and the majority did not take this difference into account in their analyses. Our series consisted of consecutive patients with AAC, which were all treated with PC, with a very limited number lost to follow-up, which limits the possibility of selection bias. However, we cannot exclude the possibility that this disorder may be under-diagnosed.
For clinical implications, our study shows that PC can be an effective treatment approach in patients with AAC unfit for initial cholecystectomy. The procedure can be used as a definitive treatment option with a low rate of subsequent cholecystectomy, although our high rate of complications must be taken into account. We suggest that future studies focus to investigate the optimal time of catheter removal and to further examine the use of cholangiography, which seems to decrease the risk of recurrence of cholecystitis. In conclusion, PC is successful as a definitive treatment option in the majority of patients with AAC and associated with a low rate of mortality and subsequent cholecystectomy.
Footnotes
Acknowledgements
None.
Declaration of Conflicting Interests
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
