Abstract
Background and Aims:
Gastrointestinal stromal tumors are rare neoplasms of the gastrointestinal tract. These lesions are characterized by different levels of malignancy. Only radical surgery offers a chance of curing the disease. The aim of this study is to present the results of gastrointestinal stromal tumor treatment with minimally invasive surgery.
Material and Methods:
The study group included 27 patients operated laparoscopically on for gastrointestinal stromal tumor with laparoscopic surgery between September 2009 and December 2013. The most common location of the tumor was the stomach (21 patients, 77.8%) and the small intestine (4 patients, 14.8%). We analyzed early surgery results, the number and character of complications, lengths of hospital stays, histological types of the removed tumors, and long-term results of treatment.
Results:
There was no need for conversion to open surgery in any patient from the study group. Post-surgery complications occurred in 2 patients (7.4%). The median duration of the hospital stay was 4.5 days; none of the patients had to be readmitted to the hospital in the first 30 days after the procedure. In 26 out of 27 patients, microscopic examination confirmed the radicality of the surgical procedure (R0 resection). The mean size of the removed lesions was 4.1 cm. Using the Joensuu malignancy classification model, it was established that in 6 (22.2%) patients gastrointestinal stromal tumor was characterized by a very low level of malignancy, in 11 patients (40.7%) a low level, in 4 (14.9%) a medium level, and in 6 (22.2%) a high level. The average duration of follow-up was 13 months. During the observation period, there was no recurrence of the disease.
Conclusion:
Minimally invasive surgery in the treatment of gastrointestinal stromal tumors is possible and allows for satisfactory results both in terms of the postoperative course and the oncological quality of the procedure.
Over 30 years have passed since the first gastrointestinal stromal tumor (GIST) was described by the pathologists Mazur and Clark (1). Today we know that these tumors are very rare, representing an estimated 1% of malignant tumors of the gastrointestinal tract (2). Although they are most common in the stomach (60%–70% of cases), they can also occur in other parts of the gastrointestinal tract (esophagus, small and large intestine), and even outside of it (greater omentum, retroperitoneal space, gallbladder, pancreas) (3). These lesions are characterized by different levels of malignancy, which depend on the size of the tumor, its location, and its mitotic rate (number of mitosis per 50 high-power microscopic fields (HPF)) (4). The most common symptoms reported by patients are uncharacteristic upper abdominal pain, nausea and vomiting, weight loss, anemia (associated with chronic bleeding from the tumor), and sometimes of acute gastrointestinal bleeding or mechanical obstruction (5).
Only radical surgery offers a chance of curing the disease (6, 7, 8). Today we are witnessing a tendency to increase the use of minimally invasive techniques in oncological surgery. They are coming to be used also for patients with GISTs (9). A strong argument in favor of using laparoscopic surgery in the treatment of GISTs is that since they very rarely metastasize to the lymph nodes, unlike other malignant abdominal tumors, it is possible to perform relatively less extensive procedures, limited to a local excision of the tumor within healthy tissue, without the need for routine extensive lymphadenectomy (7, 10). It has been suggested that laparoscopic surgery allows for similar oncological results as traditional surgery, but with less perioperative trauma, less blood loss, and faster recovery of normal gastrointestinal function. This in turn reduces the length of the patient’s hospitalization and risk of complications (11). Reports of laparoscopic surgery being applied in the treatment of GISTs are scarce. There is a lack of clear and generally accepted guidelines for this technique in the treatment of rare neoplasms.
Aim
The aim of this study is to evaluate the results of the application of minimally invasive techniques to the management of GISTs.
Patients and Methods
The study design included a retrospective analysis of all the consecutive cases of GISTs operated at one tertiary reference center (Second Department of General Surgery, Jagiellonian University—Medical College in Krakow) between September 2009 and December 2013. According to the policy of the institution, all procedures were begun laparoscopically. There were no open operations for GIST during this period.
The study group included 27 patients operated laparoscopically on for GIST between September 2009 and December 2013. There were 18 women and 9 men in the group. The median age was 62 years (26–86 years). In this group 10 patients had a history of previous surgical interventions within the abdomen (3 laparoscopic cholecystectomy, 1 open cholecystectomy, 3 open appendectomy, 2 hysterectomy, and 1 nephrectomy).
All patients were in good general status but had some comorbidities (12 patients had arterial hypertension, 6 had coronary heart disease (Canadian Cardiovascular Society grading of angina pectoris [CCS] 2), 3 had diabetes mellitus type treated with insulin therapy, 1 had chronic pulmonary disease).
The most common location of the tumor was the stomach (21 patients, 77.8%) and the small intestine (4 patients, 14.8%). Additionally, there was one patient with a GIST in the gallbladder and another patient with a lesion located in the uncinate process of the pancreas. Information about patients, localization of the tumor, and type of operation is presented in Table 1.
Patients, localization of the tumor, and type of operation.
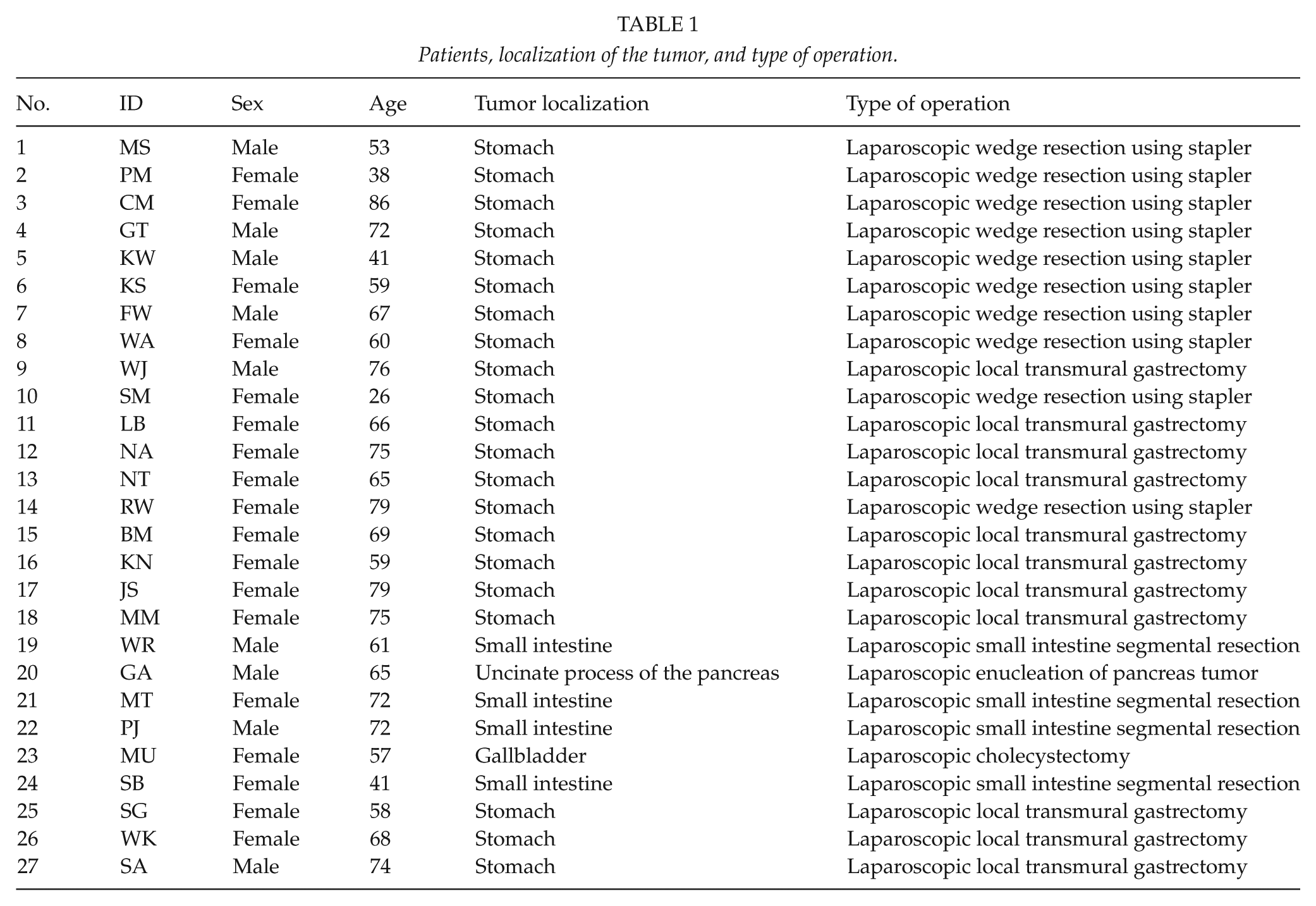
Preoperative verification during endoscopic examination was obtained for 18 patients. In the case of the 9 remaining patients, for 5 of them, we had strong clinical suspicion of GIST based on imaging and in the other 4 cases the indication for surgery was the presence of an intraabdominal tumor, and GIST diagnosis came after a pathological evaluation of the operative specimen. Diagnostic imaging ruled out the presence of metastases or locally advanced tumors in all patients.
We performed 21 local resections of the stomach (10 with the use of a stapler and 11 with a harmonic scalpel), 4 small intestine segmental resections, 1 cholecystectomy, and 1 tumor enucleation in the uncinate process of the pancreas. Intraoperative gastroscopy has been used in 15 cases for precise localization of the tumor during an operation. At the beginning, we used to apply stapled resection in all patients. Later, with growing experience in manual suturing, we changed the procedure to local excision with a harmonic scalpel and hand suture of the defect. The adoption of the later approach was also supported by the occurrence of difficulties in gastric passage observed in some patients after stapled resection.
When dissecting the tumor, we adhered to the no-touch principle, grasping only neighboring tissues, which allowed us to avoid rupturing the tumor. A plastic bag was used to remove the specimen from the abdominal cavity in order to isolate the tumor from contact with the margins of the minilaparotomy.
In four of the patients with stomach GISTs, tumors were removed through gastroscopy (NOSE, Natural Orifice Specimen Extraction), and in the remaining patients, through minilaparotomy.
We analyzed early surgery results, the number and character of complications, lengths of hospital stays, histological types of the removed tumors, and long-term results of treatment.
All GISTs were analyzed and classified using the Joensuu (12) scale. The Joensuu classification includes four factors: tumor size, tumor site, the mitotic count, and presence of tumor rupture.
None of the patients was submitted to postoperative adjuvant therapy.
All numbers are given in median (range).
Results
There was no need for conversion to open surgery in any patient from the study group. Postoperative complications occurred in two patients (7.4%). In one patient, after stapled resection, we observed narrowing of the gastric lumen causing an impairment of the passage of gastric contents almost to the level of complete obstruction, and reoperating was necessary. The other patient experienced impaired gastric emptying which subsided after noninvasive treatment. There were no other postoperative complications in the study group. The median duration of the hospital stay was 4 days (3–6 days); none of the patients had to be readmitted to the hospital within the first 30 days after the procedure.
In 26 out of 27 patients, microscopic examination confirmed the disease-free margin of the surgical specimen (R0 resection), and in the remaining patient with a stomach GIST, tumor tissue was found in one of the surgical margins (R1 resection). Given the tumor’s low malignancy (a lesion smaller than 1 cm located in the stomach, 1 mitosis per 50 HPF), a decision was made to limit intervention to close monitoring only. The mean size of the removed lesions was 4.1 cm (0.7–9.5 cm). In almost one-third of the patients, the tumors were larger than 5 cm. In 20 patients (74.1%), the mitosis rate in the removed tumors did not exceed 5 per 50 HPF, and in 7 (25.9%), the tumors were characterized by higher mitotic activity.
In the majority of patients, the tumors were composed of epithelioid cells (20 patients, 74.1%), in 11.1% (3 patients) spindle cells, and in 14.8% of the group (4 patients) mixed. In all patients, immunohistochemical examination showed the positive expression of cluster of differentiation (CD) 117, in 17 patients (62.9%) of CD 34, in 6 patients (22.2%) of SMA (smooth muscle actin), and in 3 patients (11.1%) of S100 (Table 2). Using the Joensuu malignancy classification model, in 6 (22.2%) patients GIST was characterized by a very low level of malignancy, in 11 patients (40.7%) a low level, in 4 (14.9%) a medium level, and in 6 (22.2%) a high level. Detailed information about tumor size, histological type, and malignancy grade is shown in Table 3.
The removed tumors according to histological type.

HPF: high-power microscopic fields; CD: cluster of differentiation; SMA: smooth muscle actin.
Tumor size, histological type, and malignancy grade.
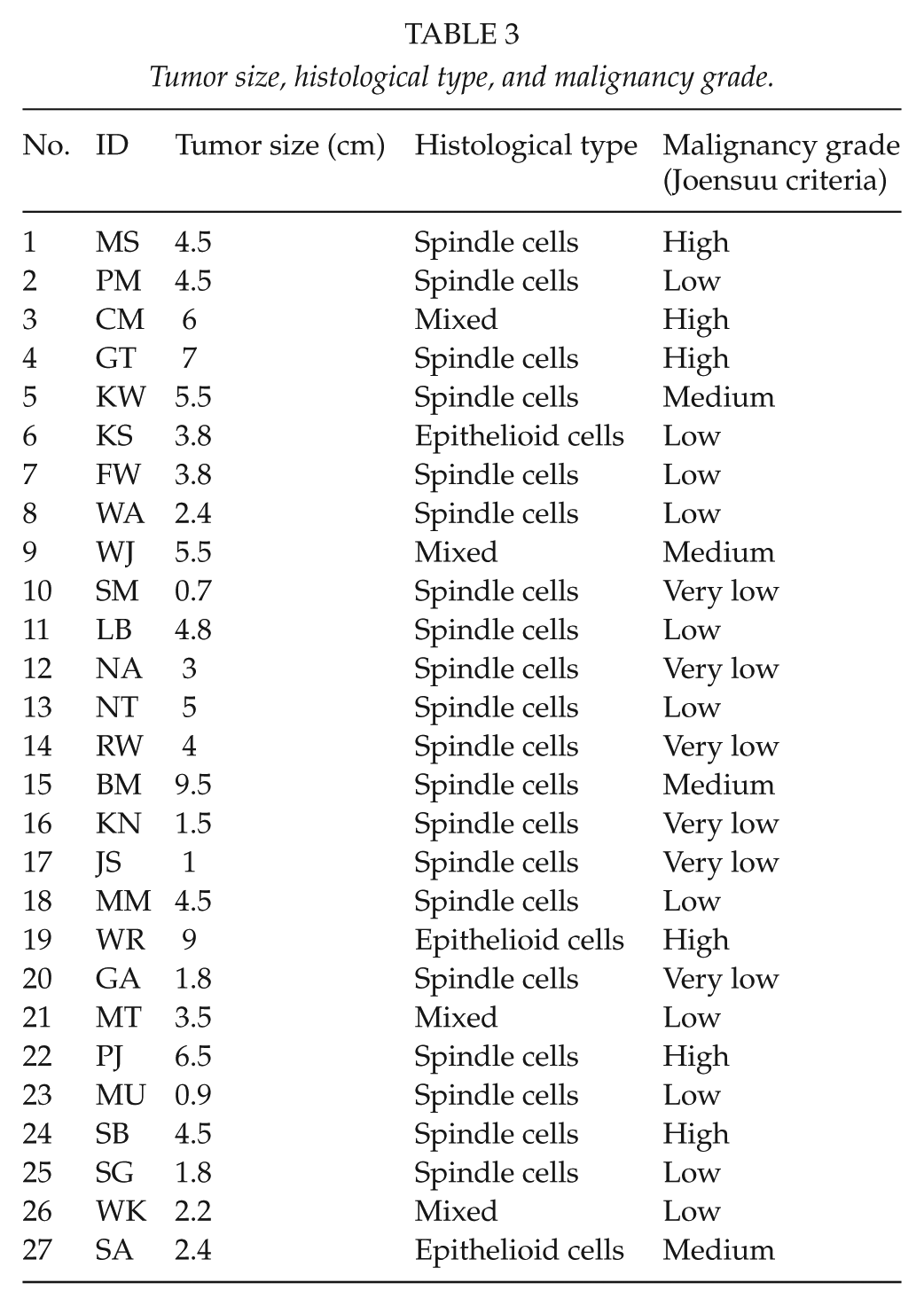
The median duration of follow-up was 13 months (3–53 months); however, in the case of 10 patients it was shorter than 6 months. During this period, there was no recurrence of the disease. There were no port-site metastases observed in this period of time. One patient died of reasons unrelated to the primary disease; the rest of the study group remains in follow-up.
Discussion
The first references to laparoscopic operations in patients with GISTs were made toward the end of the 20th century (13). From the very introduction of these operations, most authors agreed that minimally invasive methods allow for adequate oncological surgery, that is, complete removal of the tumor within healthy tissue, that is, R0 resection with appropriate margin (14, 15). Although it has not been proven that the presence of GIST tissue in the surgical margin in microscopical examination affects patient survival rates, it is an important risk factor for relapse of the disease in the peritoneal cavity (16). In 26 patients from the study group, histopathological examination confirmed the full local radicality of the procedure. In one patient with a stomach GIST, tumor tissue was present in one of the surgical margins (R1 resection), but no recurrence was noted during follow-up.
There was no need for conversion to open surgery for any patient in the study group. In his report, Roggin and Posner compared the results of five studies (over 270 patients) on laparoscopic surgery of GISTs. The average conversion rate was very low—it was necessary in only 3% (0%–6.5%) of cases (3). This is probably due to the fact that this type of surgery involves mostly local excision of the tumor without lymphadenectomy and could be regarded as relatively simple. The analysis did not show a predominant reason for conversion. Among the reasons listed there were the following: bleeding, inability to locate the tumor, adhesions related to previous surgeries, and need for more extensive surgery than expected (17).
Another strong point in the discussion about the accuracy of laparoscopic surgery in GIST is the fact that no port-site metastases were observed in follow-up. These findings are similar to those of other authors, who also did not observe such phenomena (18, 19). In four patients, given the small size of the tumor, the specimen was removed through the mouth, with the aid of intraoperative endoscopy (NOSE). Similarly, in these patients no local relapse was observed either. We suggest that the NOSE technique can constitute a valuable alternative to minilaparotomy necessary for removing the specimen; however, it is applicable only in the case of small lesions, which are possible to remove through the esophagus and throat (20).
Post-surgery complications occurred in two patients (7.4%), and in both these cases they were related to problems of gastric content passage. One patient required reoperation on the second postoperative day due to almost complete obstruction at resection site. We have observed that removing the lesions with a harmonic scalpel and manual stitching allow for better control of the resection margins and diameter of gastric lumen as compared to staplers. A 7% complication rate is acceptable and comparable with other reports, while significantly lower than in open resections (21). The mean hospital stay was 4.5 days. Like the other authors, we believe that the application of minimally invasive surgery enabled the shortened length of hospitalization (21, 22).
All the patients were subject to regular postoperative follow-up. The average follow-up duration was 13 months. This average time is low because 10 of the patients were operated on within the last 6 months. In the case of the remaining 17 patients, the follow-up time was longer, and in this group it averaged 23 months. None of the patients has been diagnosed with recurrence of the disease.
In 2011, Ohtani’s metanalysis proved that the laparoscopic technique allows for similar results as open surgery in terms of oncological radicality and disease-free survival time. Very important findings were published in 2013 by Ohtani et al. (23), who compared 12 studies (overall over 600 GIST patients) and observed that laparoscopy was associated with a lower rate of local relapse and metachronic distant metastasis. This finding is furthermore supported by other authors, who provide sufficient evidence that patients treated with laparoscopic surgery benefited from a better postoperative course, suggesting that minimally invasive techniques should be the treatment of choice in patients with GISTs.
The size of GISTs removed laparoscopically remains the subject of some debate. The average size of the excised lesions in our study group was 4.01 cm (0.7–9.5 cm). Tumor size over 5 cm is, according to some recommendations, an absolute contraindication to minimally invasive surgery, suggesting its use to lesions under 2 cm (16). However, recent research clearly indicates that, under certain conditions, laparoscopic surgery is safe even in the case of lesions larger than 10 cm (24–27). In our material, good results in treatment of GISTs larger than 5 cm and smaller than 10 cm support the thesis that lesions up to 10 cm can be safely operated laparoscopically.
The rupture of the tumor capsule is a proven risk factor for local relapse (8). Particular care should be taken during surgery of large tumors, because rupture of the tumor capsule occurs much more frequently in these cases. Laparoscopy is not contraindicated; however, it is important to remain especially cautious (22). Rupture of the tumor capsule did not occur in any of our patients; hence, we were able to avoid increasing the risk for the potential spread of cancer cells into the peritoneum.
An important aspect of the treatment of GISTs is the possibility of a multidisciplinary approach. Thanks to molecularly targeted drugs like imatinib, sunitinib, or sorafenib, it is possible to improve the results of surgical treatment. In this group of patients, adjuvant treatment with imatinib could have been considered in those patients who had very high malignancy according to the Joensuu scale. To our regret, since adjuvant treatment in this group of patients is not reimbursed in Poland, none of our patients received such therapy. We did not have any candidate for the neoadjuvant approach with kinase inhibitors as none of the patients on whom we operated had lesions that were locally advanced.
Two cases of patients with extragastrointestinal stromal tumors (EGIST) required additional comment. It is very rare for the primary tumor to be located in the pancreatic parenchyma. Only about 20 such cases have been documented (28). In the case of a 55-year-old man operated on at our center, a tumor 18 mm in diameter was initially described in diagnostic imaging as a hormonally inactive neuroendocrine tumor, and thus we planned enucleation. No metastases were found in the abdominal cavity, and laboratory tests did not show elevated levels of tumor markers. Preoperative verification was not possible due to the location of the lesion. The patient has been under follow-up for 6 months and as of now has not been diagnosed with disease progression. The second patient with an EGIST was a 57-year-old woman operated on for an asymptomatic gallbladder tumor. A routine ultrasonography (USG) examination showed a polipoid thickening of the wall of the fundus. The patient was submitted to laparoscopic cholecystectomy; postoperative diagnosis confirmed the presence of GIST tissue. Additional diagnostic imaging did not show metastatic lesions, and no relapse was observed in the follow-up. Stromal tumors of the gallbladder are very rare, with only a few described cases, and are characterized by poorer prognosis, with the majority of cases showing an aggressive course of disease (29). We decided not to extend the scope of resection after the laparoscopic cholecystectomy due to the disease-free margins, the location of the tumor in the fundus of the gallbladder, and low mitotic index. No relapse was observed during 2 years of follow-up.
Conclusion
We are convinced that the use of minimally invasive surgery in the treatment of GISTs is feasible and allows for satisfactory results both in terms of the postoperative course and the oncological quality of the procedure. It is necessary that the personnel performing laparoscopic surgery be experienced and proficient, as disease-free margins and an intact tumor capsule are mandatory for adequate oncological operation.
Footnotes
Declaration of Conflicting Interests
The authors declare that there is no conflict of interest.
Funding
The publication was supported by Faculty of Medicine, Jagiellonian University Medical College, Leading National Research Centre (KNOW) 2012–2017.
