Abstract
Aim:
To analyze our experience in translating the concept of total mesorectal excision to “no-touch” complete removal of an intact mesocolonic envelope (complete mesocolic excision), along with central vascular ligation and apical node dissection, in the surgical treatment of right-sided colonic cancers, comparing “mesocolic” to less radical “non-mesocolic” planes of surgery in respect to quality of the surgical specimen and long-term oncologic outcome.
Method:
A total of 115 patients with right-sided colonic cancers were retrospectively enrolled from 2008 to 2013 and operated on following the intent of minimally invasive complete mesocolic excision with central vascular ligation.
Results:
Morbidity and mortality were 22.6% and 1.7%, respectively. Mesocolic, intramesocolic, and muscularis propria planes of resection were achieved in 65.2%, 21.7%, and 13% of cases, respectively, with significant impact for mesenteric plane of surgery on R0 resection rate (97.3%), circumferential resection margin <1 mm (2.6%), and consequent survival advantage (82.6% at 5 years) when compared to muscularis propria plane of surgery, with R0 resection rate and overall survival falling to 72% and 60%, respectively, and with circumferential resection margin <1 mm raising to 33.3%, all being statistically significant. Stratifying patients for stage of disease, laparoscopic complete mesocolic excision with central vascular ligation significantly impacted survival in patients with stage II, IIIA/B, and in a subgroup of IIIC patients with negative apical nodes.
Conclusion:
In our experience, minimally invasive complete mesocolic excision with central vascular ligation allows for both safety and higher quality of surgical specimens when compared to less radical intramesocolic or muscularis propria planes of “standard” surgery, significantly impacting loco-regional control and thus overall survival.
Keywords
Introduction
Emil Theodor Kocher first theorized that oncologic resection is removal not only of the organ but also of its lymphatic drainage; this concept was shortly after substantiated by Miles (1) and Jamieson and Dobson (2) for rectal and colonic cancers, respectively. After 70 years, a new revolution in radical surgery was performed by Heald et al. (3), who introduced the concept of total excision of the mesorectum (total mesorectal excision (TME)), the primitive embryological dorsal mesentery of the organ, reaching significant local control and, for that time, unbelievable 5-year overall survival (OS) and disease-free survival (DFS) so as to promptly become the central part of any multimodal rectal cancer treatment (4, 5).
In 2009, Hohenberger et al. (6) translated the concept of TME to colonic cancer, stressing that the oncologic outcome of traditionally more favorable colon neoplasia was eventually overtaken by rectal cancer, both for multimodal strategies not yet applied to colon tumors and for a more radical surgical approach performed along embryonic planes of development by means of TME; thus, complete mesocolic excision (CME) with central vascular ligation (CVL) was theorized, standardized, and soon validated by several studies (7, 8).
The concept of complete excision of the organ within its primitive mesentery associated with central transection of the supplying blood vessels is today considered the step toward a modern surgical oncology, as the no-touch resection of the primitive embryological dorsal mesenterium is pivotal in achieving optimal local clearance: this anatomical structure is the embryological envelope where the neuro-lymphovascular structures develop within a double-layered mesenchymal fibrofatty tissue and, consequently, the route for cancerous diffusion from the primitive tumor; its intact, complete, “no-touch” excision is thus crucial to minimize residual disease in the surgical field, consequently impacting local control and oncologic outcome. Furthermore, CVL allows for an extensive lymph node clearance along the feeding vessels of the colon, with definitive effect on regional recurrence and systemic dissemination, as suggested by relevant studies showing that enhanced lymph node harvesting significantly improves survival in stage I–III colonic cancers (9, 10).
The aim of this study is to discuss our experience with laparoscopic CME and CVL in cancers of the right colon, analyzing the quality of surgical specimen and 5-year oncologic outcome related to the plane of resection achieved, comparing mesocolic to less radical non-mesocolic planes of surgery.
Materials and Methods
From July 2008 to March 2013, 115 patients with stage I/IIIC right colonic cancers (classified according to the latest tumor–node–metastasis (TNM) classification, Seventh Edition, 2010) were operated on with laparoscopic CME and CVL; all patients were preoperatively investigated with colonoscopy, diagnosed with biopsy, and staged by contrast-enhanced thoracic and abdominal computed tomography (CT) scan along with carcinoembryonic antigen (CEA) blood levels; postoperative oncologic follow-up was based on biannual clinical evaluation with CEA testing and yearly thoraco-abdominal CT scan.
Lesions suspected for recurrence were further studied with positron emission tomography (PET)–CT scan when indicated and histologically confirmed if surgically resectable and patient fit for operation.
Operative Technique
The patient is placed in reversed Trendelenburg position with 30° left tilt, the pneumoperitoneum is induced by open technique with placement of three additional trocars semi-circumferentially around the umbilicus, the abdominal cavity is explored, and cytological washing and intra-operative ultrasound (IOUS) are performed. Once the “working space” is created, no-touch medial to lateral technique is always adopted: the ileocolic vessels are stretched so as to delineate the Treves’ arcade, and peritoneal incision is commenced at the base of the created peritoneal fold; dissection of the anterior peritoneal sheet is performed along the left margin of the superior mesenteric artery (SMA), with transection of the ileocolic and the inconstant right colic vessels at their roots, and “en bloc” lymphadenectomy of the anterior aspect of the superior mesenteric vein (SMV) from the ileocolic vessels to the gastro-colic trunk of Henle is preformed; at this point, the anatomo-embryological plane along the Toldt’s fascia is sharply developed from medial to lateral and from bottom to top, without mobilizing the duodenum, as suggested by Hohenberger et al. (6), but dissecting along the plane between the intact dorsal mesocolon of the hepatic flexure and the Fredet preduodenopancreatic fascia.
In case of cecum or ascending colon cancer, the stretched transverse mesocolon is progressively transected with central ligation of the right branch of the middle colic vessels, and the colon is stapled 10 cm off the tumor (right hemicolectomy); for hepatic flexure or proximal colon transversum cancers, middle colic and right gastroepiploic vessels are ligated at their roots, subpyloric lymph nodes are removed, 10–15 cm of greater omentectomy off the tumor is performed, and colon stapling is carried out just proximal to the splenic flexure (extended right hemicolectomy). Hepatic flexure is mobilized by severing the lateral peritoneal fold, the superior attachment (right phrenocolic ligament), and the medial attachment (cholecysto-duodeno-colic ligament). The cecal ligaments and the lateral attachment are progressively severed to obtain complete mobilization of the specimen; the ileum is stapled at 10–15 cm from the ileocecal valve, and the specimen is extracted within a plastic bag through a protected mini-Pfannenstiel incision; side-to-side mechanical intracorporeal anastomosis is fashioned, with final drainage of the perianastomotic area.
Pathology Protocol and Tissue Morphometry
All cancer specimens were left intact, both anterior and posterior aspects of the morphometric measurement were photographed, and then fixed in 10% formalin for subsequent serial slicing with 3–5 mm intervals through the tumor segment. Proximal and distal marginal clearance, the closest mesocolic and retroperitoneal (if present) resection margin (circumferential resection margin (CRM)), length of specimen, distance from the nearest bowel wall to high tie, distance from the tumor and high tie, area of mesentery, number of lymph nodes harvested, and quality of the specimen according to the grading system of the CLASICC trial (11) were recorded.
Statistical Method
Data are expressed as a mean ± standard deviation. The Student t test was used to analyze quantitative variables, while the chi-squared test was used for the qualitative ones. Survival curves were calculated according to the Kaplan–Meier method, and the statistical differences were confronted by the log-rank test. A p < 0.05 was considered statistically significant. All statistical analyses were performed using dedicated software (MedCalc®) on Windows Vista®.
Results
The mean age of patients (75 males and 40 females with a male to female ratio of 1.8) was 65 ± 1.3 years. The mean operative length was 179 ± 39 min, and the mean intra-operative blood loss was 115 ± 23 mL.
Staging of disease was according to the latest TNM Edition (Seventh Edition, 2010): 25 cases stage I (21.7%), 26 cases stage II (22.6%), 18 cases stage IIIA (15.7%), 22 cases stage IIIB (19.2%), and 24 cases stage IIIC (20.8%).
Mortality was 1.7% (two patients): one for pneumonia and one for pulmonary embolism. Morbidity was 22.6% (26 cases: 7 wound infections, 6 patients with urinary tract infection, 5 patients with anastomotic leakage, 5 patients with pneumonia, 2 deep venous thrombosis of the lower limb, and 1 pleural effusion). Flatus passage and resumption of oral intake were 2.3 ± 1.1 and 3.3 ± 0.5 days, respectively; hospital stay was 10.5 ± 1.9 days (Table 1).
Safety parameters.
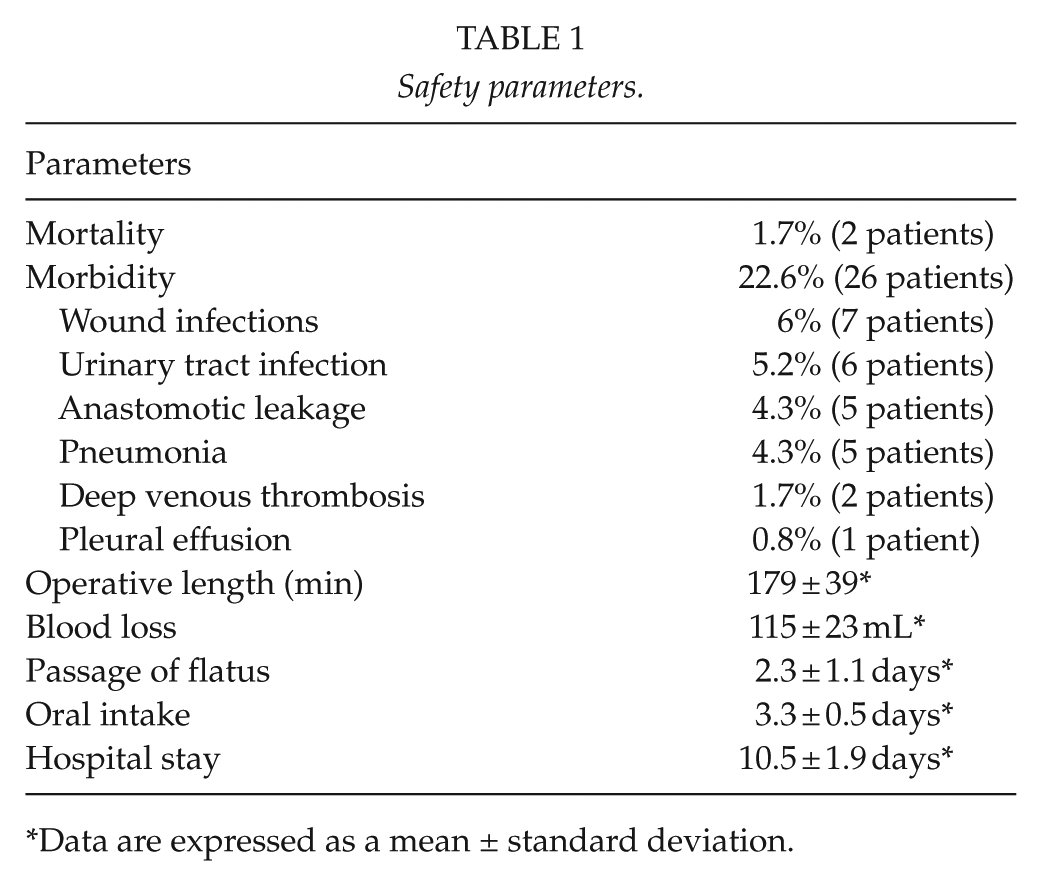
Data are expressed as a mean ± standard deviation.
Colonic resection was classified as mesocolic plane in 65.2% (75 cases), intramesocolic plane in 21.7% (25 cases) and muscularis propria plane in 13% (15 cases).
For mesocolic plane of surgery, mean ileocolic segment length was 23.5 ± 3.7 cm and resection margins were all free of microscopic disease; distance from the nearest bowel wall to high tie was 83 ± 7 mm, distance from tumor and high tie was 103 ± 5 mm, and area of mesentery was 15.350 ± 1.570 mm2; mean lymph nodes harvested were 29 ± 5. For non-mesocolic (i.e. intramesocolic and muscularis propria) planes of resection, mean ileocolic segment length was 21.3 ± 2.5 cm and resection margins were all free of microscopic disease; distance from the nearest bowel wall to high tie was 67 ± 5 mm, distance from tumor and high tie was 87 ± 9 mm, and area of mesentery was 14.135 ± 1.150 mm2; mean lymph nodes harvested were 19 ± 7. As shown in Table 2, all morphometric data and the mean number of lymph nodes harvested reached statistical difference in the two different planes of surgery (Table 2).
Tissue morphometric data from all fresh specimens.
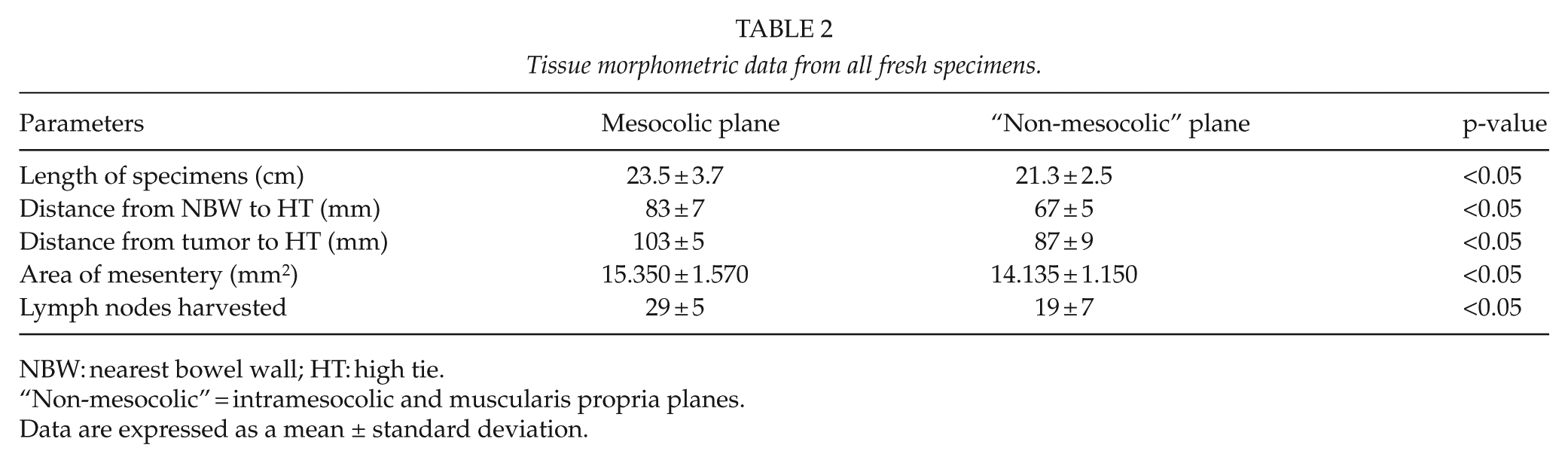
NBW: nearest bowel wall; HT: high tie.
“Non-mesocolic” = intramesocolic and muscularis propria planes.
Data are expressed as a mean ± standard deviation.
Pathologically, R0 resection (absence of macro-microscopic residual disease in the radial, proximal, and distal margins of the specimen) was achieved in 97.3% of mesocolic plane specimens, 88% of intramesocolic plane resections, and 66.6% of muscularis propria resections, all statistically significant (p < 0.05). CRM < 1 mm was present in 2.6% of mesocolic plane, 12% of intramesocolic plane, and 33.3% of muscularis propria resections, all being statistically significant (Table 3).
Oncologic outcome.
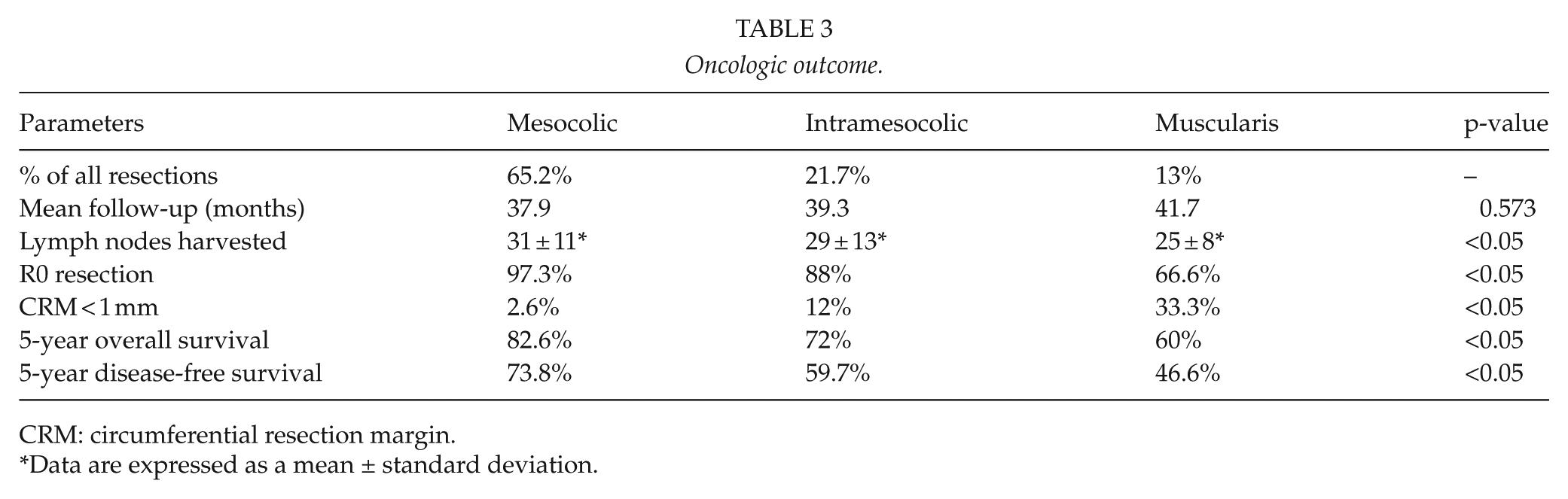
CRM: circumferential resection margin.
Data are expressed as a mean ± standard deviation.
Mean follow-up was 39.5 months: 68.9% (79 patients) were followed up at least 5 years, with none lost to follow-up. Five-year OS and DFS survival were 79.1% and 69.5%, respectively (Fig. 1): mesocolic plane had significantly better OS when compared to both intramesocolic and muscularis propria planes of resection (82.6%, 72%, and 60%, respectively; p < 0.05; Fig. 2 and Table 3), particularly for stage II and stage IIIA/B (93.7% and 76.9% for mesocolic plane vs 80% and 57% for “non-mesocolic,” that is, intramesocolic and muscularis propria planes of resection, respectively, all statistically significant; Fig. 3).
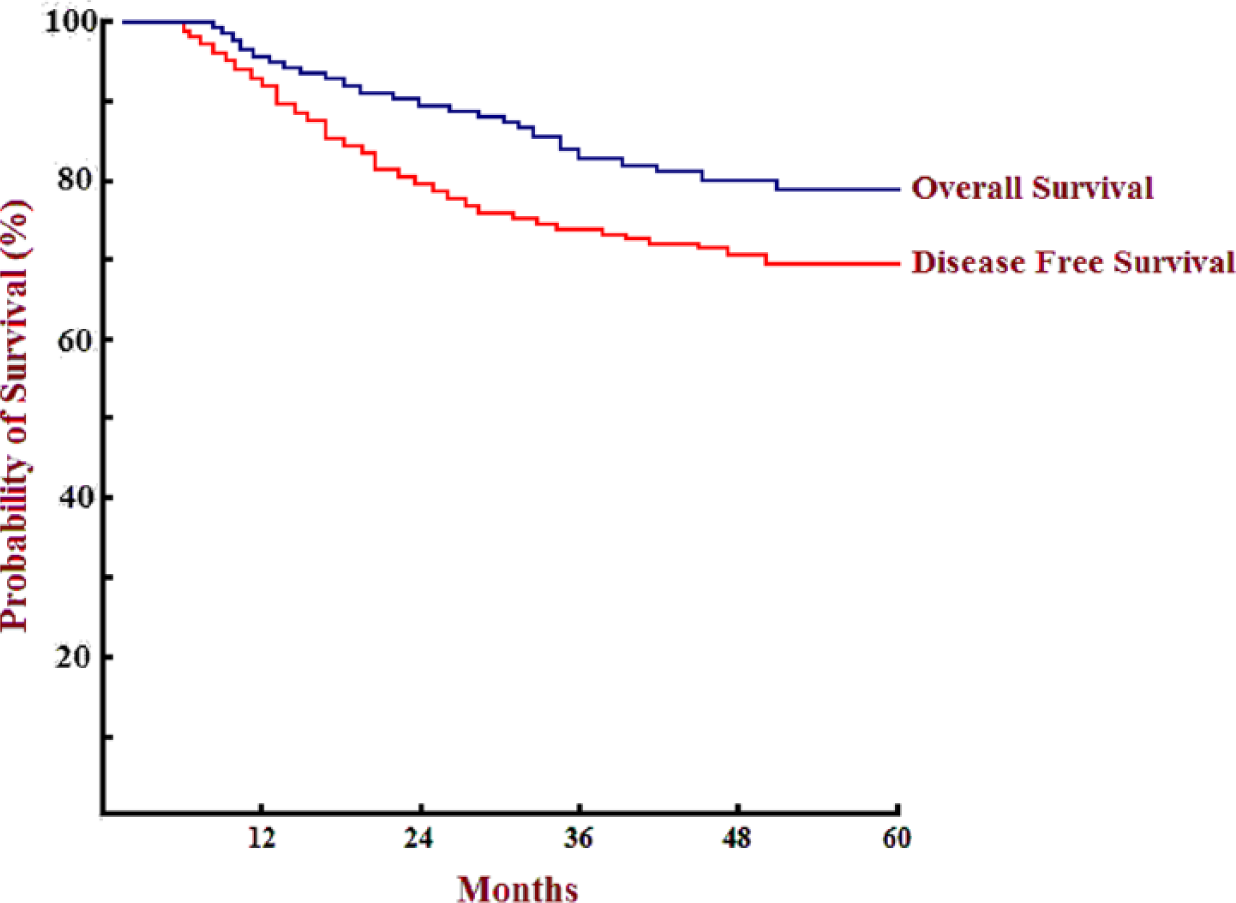
Overall (blue line) and Disease Free Survival (red line).
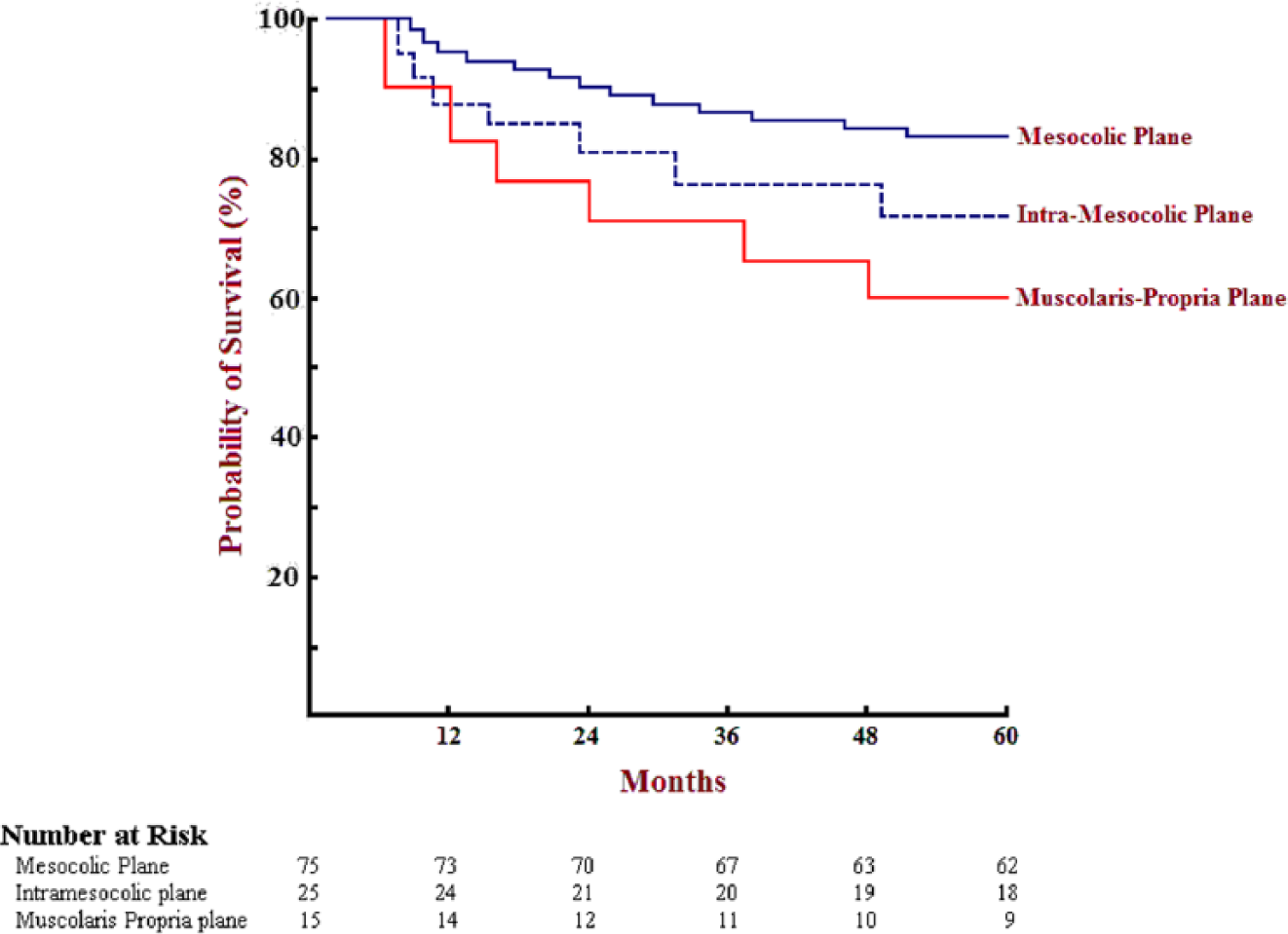
Non-stratified Survival Probability according to the plane of surgery: difference statistically significant for all planes of resection (p<0.05).
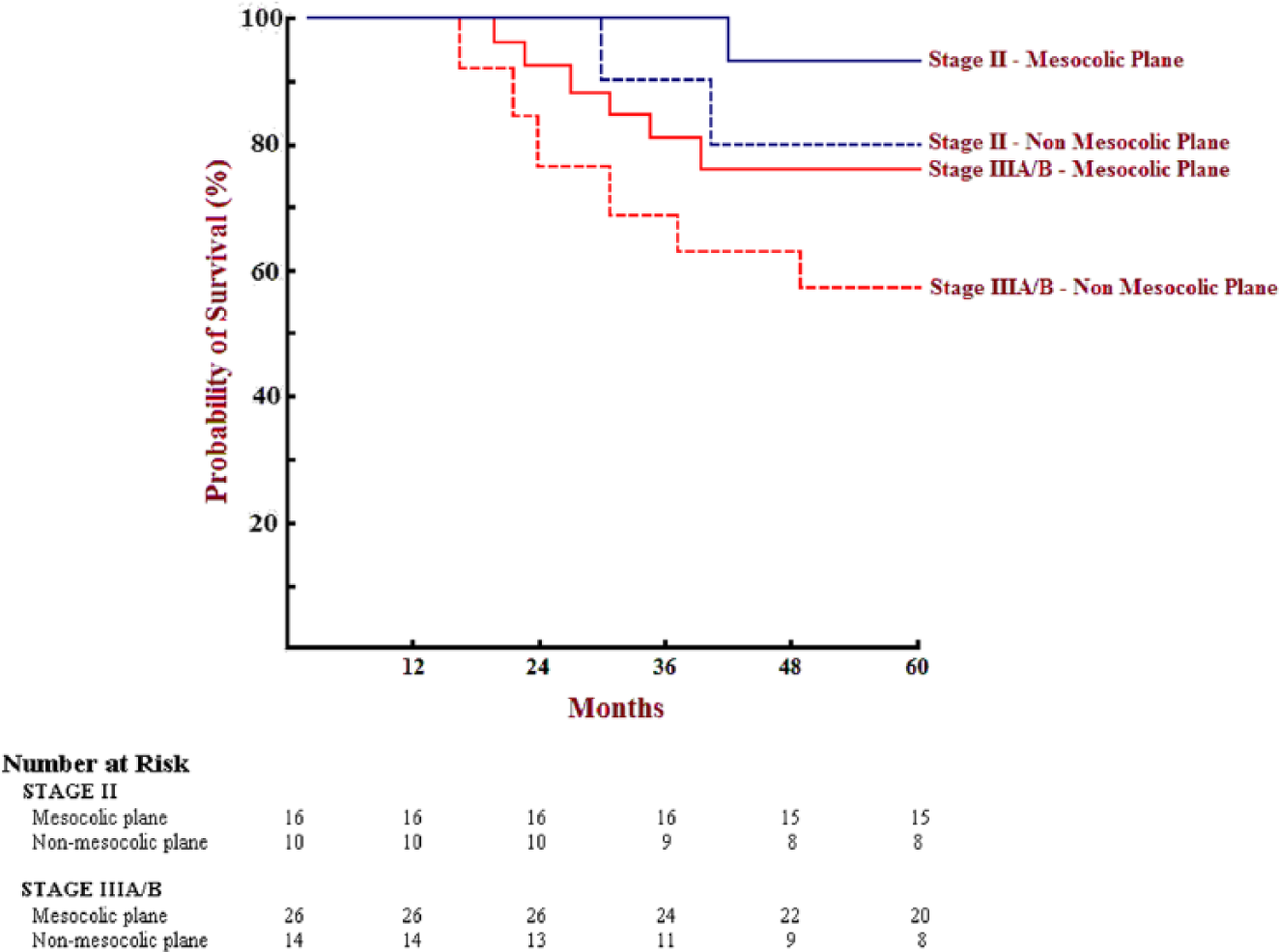
Stratified Survival Probability between mesocolic and non-mesocolic (intra-mesocolic and muscolaris-propria) planes of surgery for stage II and IIIA/B, all statistically significant (p<0.05).
Stage IIIC showed statistical difference in survival between N3 apical node–negative mesocolic resections compared to N3 apical node–negative “non-mesocolic” planes of surgery (62.5% vs 33.3%; p < 0.05; Fig. 4); differently, survival of stage IIIC N3 apical node–positive patients was 20% for both mesocolic and “non-mesocolic” surgery, not reaching statistical difference for plane of resection achieved (p = 0.957; Fig. 4). No statistical difference in survival was also noted for stage I, being 100% for both mesocolic and “non-mesocolic” planes (p = 0.995).
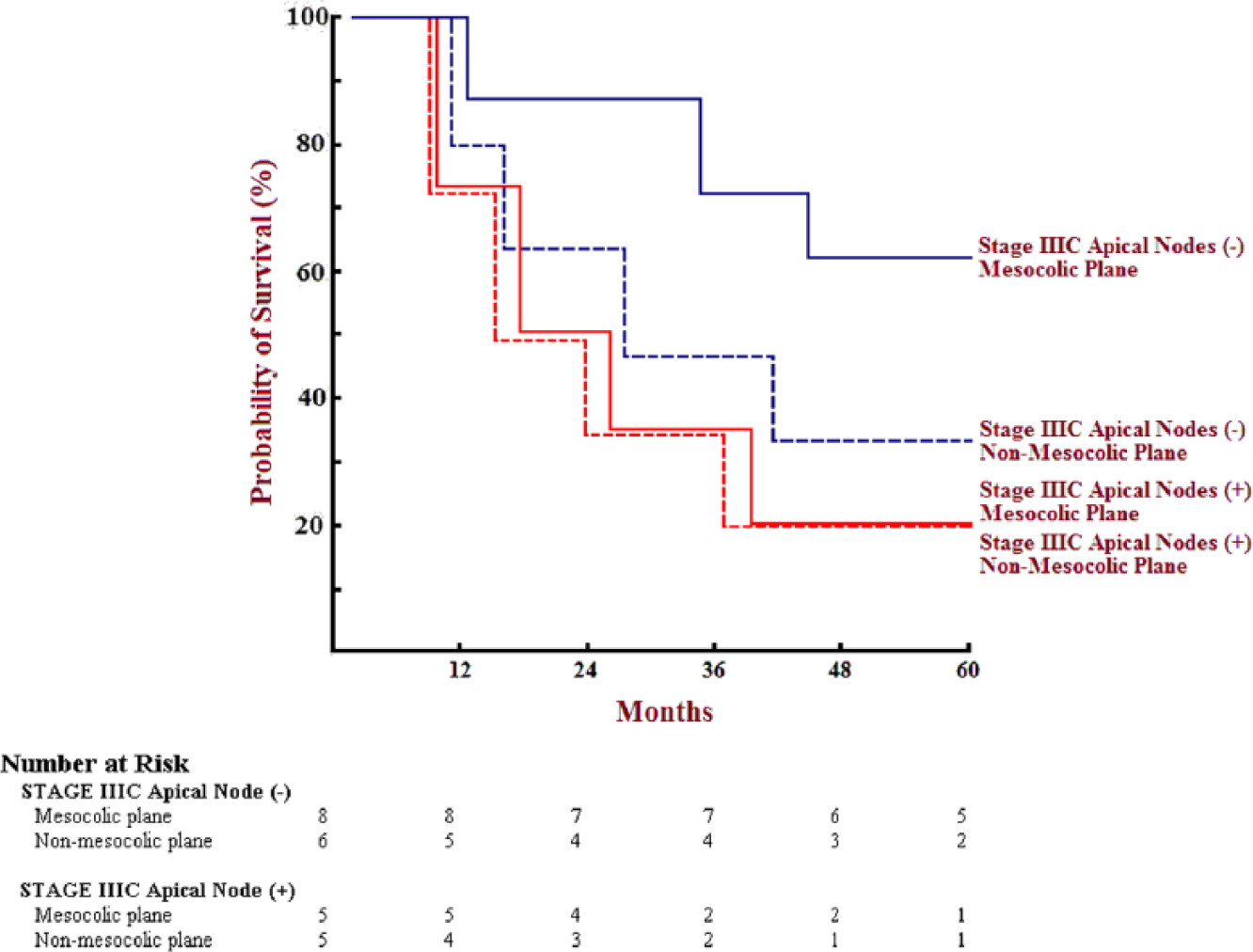
Stratified Survival Probability between mesocolic and non-mesocolic planes of surgery for stage IIIC: N3 apical nodes negative (-) outcome shows significant better survival when resection is performed in mesocolic plane (p<0.05). N3 apical nodes positive (+) outcome is poor, independently from plane of resection achieved (mesocolic not statistically different from non-mesocolic resections; p = 0.957).
Recurrence occurred in 35 patients (30.4% of cases), prevalently within 3 years: 19 patients (16.5%) developed liver metastases, 7 (6.1%) lung metastases, 4 (3.4%) peritoneal carcinomatosis, 4 (3.4%) para-aortic lymph node metastases, and 1 (0.8%) bone metastases. A total of 80% of N3 apical node–positive patients developed recurrence, mainly to para-aortic nodes (50%), to the lung (30%), and both to the lung and to the liver in 20%.
Discussion
The mesocolon, likewise the mesorectum, is the adult remnant of the primitive dorsal mesentery, by which the colon is suspended to the posterior abdominal wall during embryogenesis (12–17).
In the 5-mm embryo, the colon develops within a dorsal mesentery all along its course; the 270° counterclockwise rotation of the primitive mid-gut along the axis of the SMA determines the folding of the dorsal mesentery, giving rise to the future mesocolon (14–17): Sir Frederick Treves (18) affirmed that right (and left) mesocolon “fuses” with the primitive posterior parietal peritoneum, obliterating the space between these embryonic structures; yet, Goligher (19) demonstrated the feasibility of stripping back the colon and its meso toward the midline (its primitive embryological disposition before rotation), confirming the studies of Carl Toldt (20) and Congdon et al. (21), who claimed that mesentery of the right (and left) colon persists in adulthood not only at the level of the transverse and sigmoid colon but also all along its length, being separated from the posterior parietal peritoneum by a loose areolar connective tissue plane referred to as Toldt’s fascia.
The concept of CME is based on sharp division of the primitive mesocolon, deriving from splanchnopleuric layer of lateral mesoderm, and the primitive parietal peritoneum, derived from somatopleural layer of mesoderm, developing the avascular plane between the mesofascial and the retrofascial interface (22), along the plane of Toldt (the same concept of the Holy plane introduced by Heald et al. (3) for TME): this latter is not thus simply confined to the lateral peritoneal attachment (white line of Toldt), but occurs as a result of condensation of mesofascial interface fibers, all along the mesocolon course, up to its mesenteric insertion.
The no-touch resection of all the envelope of the primitive dorsal mesentery along the anatomo-embryological avascular cleavage planes is therefore fundamental for a true optimal R0 resection, as the meso contains all potential routes of initial metastatic spread through lymphovascular, neuroperineural, and fibrofatty tissues.
The quality of the specimen is thus classified in relation to the intactness and amount of mesocolic tissue removed, following the same criteria adopted for TME, according to the CLASICC trial grading system (11): specimens with very little or no attached mesocolic tissue and areas of visible muscularis propria are defined as muscularis propria plane (“poor” plane) of surgery, those with a moderate amount of mesocolic tissue with irregularity but without areas of visible muscularis propria as intramesocolic plane (“moderate” plane) of surgery, and those with mesocolic tissue totally covering the muscular tube in a continuous smooth peritoneofascial surface as mesocolic plane (“good” plane) of surgery.
In addition to CME, radical lymphadenectomy, comprising apical lymph nodes, proper of CVL, is of paramount importance in obtaining adequate regional control and impact on survival: the latest 2010 Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines recommend D2 dissection for clinically early stages of colorectal cancers and D3 dissection for more advanced disease, reaching impressive results in terms of local recurrence and patients’ survival (23, 24); yet, similar results have also been reported by Western authors, who claim CME with CVL as oncologically effective for right colonic cancer as D3 right hemicolectomy performed in Eastern countries (8, 25, 26). In fact, even if not yet completely established, CVL is crucial in micrometastatic clearance of central nodes, which are frequently missed by routine histological examination (27) and thus responsible for loco-regional recurrence and systemic dissemination (24); furthermore, for cancers located in the hepatic flexure and proximal transverse colon, there is a metastatic nodes incidence of about 5% for subpyloric station and about 4% for right gastroepiploic arcade (28): thus, central transection of middle colic vessels, ligation of right gastroepiploic vessels at the origin, 10–15 cm of greater omentectomy off the tumor, and removal of subpyloric nodes are all mandatory, especially in advanced stages (8).
Complete excision of the primitive dorsal mesenterium along the anatomo-embryological surgical planes by means of TME is today the standard of care for rectal cancers, but its role remains less defined in affecting local recurrence rates and survival in colon cancer: in our experience, the consequent specimen results in significantly increased length, wider mesocolic area, higher vascular tie, and more lymph node retrieved when compared to historical and less radical standard resections reported both in the literature (29, 30) and in our initial experience (Table 2): our performance of 34.7% of “non-mesocolic” resection was mainly concentrated when we started with laparoscopic CME and CVL and was virtually absent in the last 3 years.
Not stratifying patients for stage of disease, 5-year OS and DFS were 79.1% and 69.5%, respectively (Fig. 1); R0 resections were obtained in 97.3% of cases for mesocolic plane of surgery, but fell to a significant 88% and 66.6% for intramesocolic and muscularis propria surgical plane, respectively; CRM < 1 mm was common in muscularis propria (33.3%) and intramesocolic resections (12%) and rare in mesocolic plane surgery (2.6%), all statistically significant (Table 3).
Analyzing the OS curves stratified for plane of surgery but not for stage of disease, our data clearly show a significant better oncologic outcome for mesocolic plane of resection in respect to both intramesocolic and muscularis propria planes of surgery (82.6%, 72%, and 60%, respectively, all statistically significant; Fig. 2 and Table 3).
Stratifying the patients for stage of disease, the impact on survival is definitively evident in stage II and stage IIIA/B, with 5-year OS of 93.7% and 76.9% for mesocolic plane of resection versus 80% and 57% for “non-mesocolic,” that is, intramesocolic and muscularis propria planes of surgery, respectively, all being statistically significant (Fig. 3): these relevant improvements in survival stress the absolute necessity for removal of an inviolate mesocolic package in preventing loco-regional recurrence from inadequate radial clearance, possible peritoneal seeding due to mesocolon shears, and residual metastatic lymph nodes left in the surgical field for insufficient harvesting, especially for intermediate stages.
Differently, in stage IIIC our analysis identifies two subgroups of patients: N3 apical node–negative patients, for whom CME with CVL confers a significant survival advantage when compared to less extensive planes of surgery (62.5% for mesocolic vs 33.3% for non-mesocolic planes of resection; p < 0.05; Fig. 4), and N3 apical node–positive patients, characterized by survival not different from the historical figures of standard resection reported in the literature, independently of the plane of surgery achieved: at 5 years, only 20% of patients are alive for both mesocolic and non-mesocolic planes of resection, not reaching statistical difference (p = 0.957; Fig. 4). This could be justified by the fact that stage IIIC is characterized by pathologic disease far gone beyond the original primitive mesenterial diffusion and not any longer controllable with more extensive surgery, especially when apical nodes are macroscopically involved. Similarly, no survival advantage was observed in stage I patients, who benefit little from the extent of surgery because disease has limited local spread, with no regional metastasis.
Collectively, our experience demonstrates that CME with CVL is oncologically superior to the standard planes of resection, especially in intermediate stages of disease (stage II, IIIA/B, and apical node–negative IIIC): in these cases, cancer is still confined within the primitive meso-structures so as to obtain, with their intact removal, a significant survival advantage, not evident in extreme stages (I and IIIC apical node–positive), in which, either for exclusively local disease or because of diffuse spread beyond any surgical possibility of radical R0 resection, prognosis is probably independent from the extent of surgery.
Laparoscopic CME with CVL demonstrated not only superior oncologic efficacy but also to be absolutely safe, with the addition of all the advantages of minimally invasive techniques: mortality was in line with experiences of international series (8, 10, 23–26), and morbidity was lower than the classical open approach, with less intra-operative blood loss, quicker passage of flatus and oral intake resumption, less wound infections, and shorter hospital stay (see Table 1); anastomotic leakage rate was 4.3%, comparable to other international reports (8, 10, 23, 24).
Our analysis shows that laparoscopic CME with CVL is safe, without increase in morbi-mortality, and oncologically superior, with improvement in loco-regional control as shown in better 5-year OS and DFS, when compared to “non-mesocolic” (intramesocolic and muscularis propria) standard resections, paralleling worldwide results on the topic (8, 23–26).
Yet, given the study design, error and bias are statistically possible: the relatively small sample size increases the risk of type II error, and because of the retrospective, nonrandomized setting, the selection bias, that is, that patients with a “per se” better prognosis had a higher chance of being treated with CME and CVL in a mesocolic plane, is plausible; thus, evidence from prospective randomized trial is needed to recommend CME with CVL as the definitive surgical treatment in resectable colonic cancer.
Despite these statistical limits, it is anyhow the opinion of the author that a modern oncologic surgery should be based on the anatomo-embryologic concept of meso-resection and loco-regional cancerous spread: complete removal of the primitive mesenterium containing the organ and its vasculolymphatic and neuroperineural structures as an intact package, along with transection at the origin of supply and backflow vessels, becomes cardinal in obtaining a true extensive radical R0 resection, with specimens of higher quality and significant impact on survival when compared to less radical planes of surgery.
Thus, laparoscopic CME with CVL, maintaining intact the embryological envelope of the mesocolon and allowing high tight ligation, should be pursued in the surgical management of any advanced colonic tumor exactly the same way modern surgeons perform TME in rectal cancer: laparoscopic meso-resectional surgery is the novel key factor in the multimodal management of colonic cancer, determining a significant improvement in loco-regional control of tumors not spreading beyond the primitive meso-structures and improving long-term oncologic outcome, especially in intermediate stages of disease.
Footnotes
Acknowledgements
L. M. Siani contributed to study conception and design, acquisition of data, analysis and interpretation of data, and writing the article. C. Pulica contributed to study conception and design.
Declaration of Conflicting Interests
The authors declare no conflict of interest or any financial support.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
