Abstract
Background and Aims:
Esophageal perforation is a rare diagnosis, which is associated with a high morbidity and mortality. There is only small scientific background regarding the best choice of treatment. Parameters indicating a good clinical outcome seem to be localization, depth of the defect, pre-existing risk factors, and time interval between the event and start of treatment.
Material and Methods:
We evaluate retrospective data from 39 patients who were treated with a esophageal perforation in our hospital between 2004 and 2012.
Results and Conclusions:
Our collected data agree with the available published literature. Endoscopic treatment seems to be favorable in early diagnosis.
Introduction
Esophageal perforation is a potential life-threatening diagnosis of various etiologies (1, 2). Due to a low number of cases describing such an event, an evidence-based guideline for treating esophageal perforation is not available. De Schipper et al. (3) tried to create a treatment algorithm for Boerhaave’s Syndrome by means of a retrospective analysis by collecting data from 1974 to 2008. In 2011, Soreide et al. established a therapeutic and diagnostic algorithm for the initial treatment in the trauma room and for the following 24 h. (4) Brinster et al. (5) were able to show that in 59% of the cases, the etiology is iatrogenic, in most cases due to upper endoscopy. The total risk of a perforation caused by a diagnostic flexible upper endoscopy is described as low as 0.03% but increases with further interventions (6).
Esophageal perforation tends to occur at four predisposing anatomical positions: (a) the Killian Triangle, a muscle-free zone of the proximal esophagus; (b) at the crossing at the aortic arch; (c) at the connection to the left main bronchus; and (d) at the gastroesophageal junction. Regarding the etiology, esophageal perforation can also be caused due to a malignancy or in the setting of the above-mentioned Boerhaave’s Syndrome. The latter describes a barotrauma due to regurgitation, which leads to left lateral perforation of the esophagus (3).
Due to the anatomical conditions in the mediastinum with absence of immunocompetent tissue, the body is unable to cover the defect, leading to consecutive mediastinitis with a mortality up to 20% (1). Thus, a prompt and targeted diagnosis and therapy are crucial.
The localization, extent of the defect, and the time gap between the event and the initiation of therapy can be used in the decision-making process, although no evidence-based recommendations are available in the published literature.
In this analysis, we updated the formerly published data (7) by complementing it with further collective data from 2008 to 2012. The aim of our study was to show that the early diagnosis seems to be the main impact factor for successful treatment. In fact, an early diagnosis also favors an interventional endoscopical treatment.
Methods and Material
Between 2004 and 2012, a total of 39 patients (20 women and 19 men) with the diagnosis of an esophageal perforation have been treated at the Klinikum Oldenburg. The mean age at diagnosis was 66.4 years (range: 16–91 years). The data were retrospectively evaluated (Table 1).
Retrospectively evaluated data of patients.
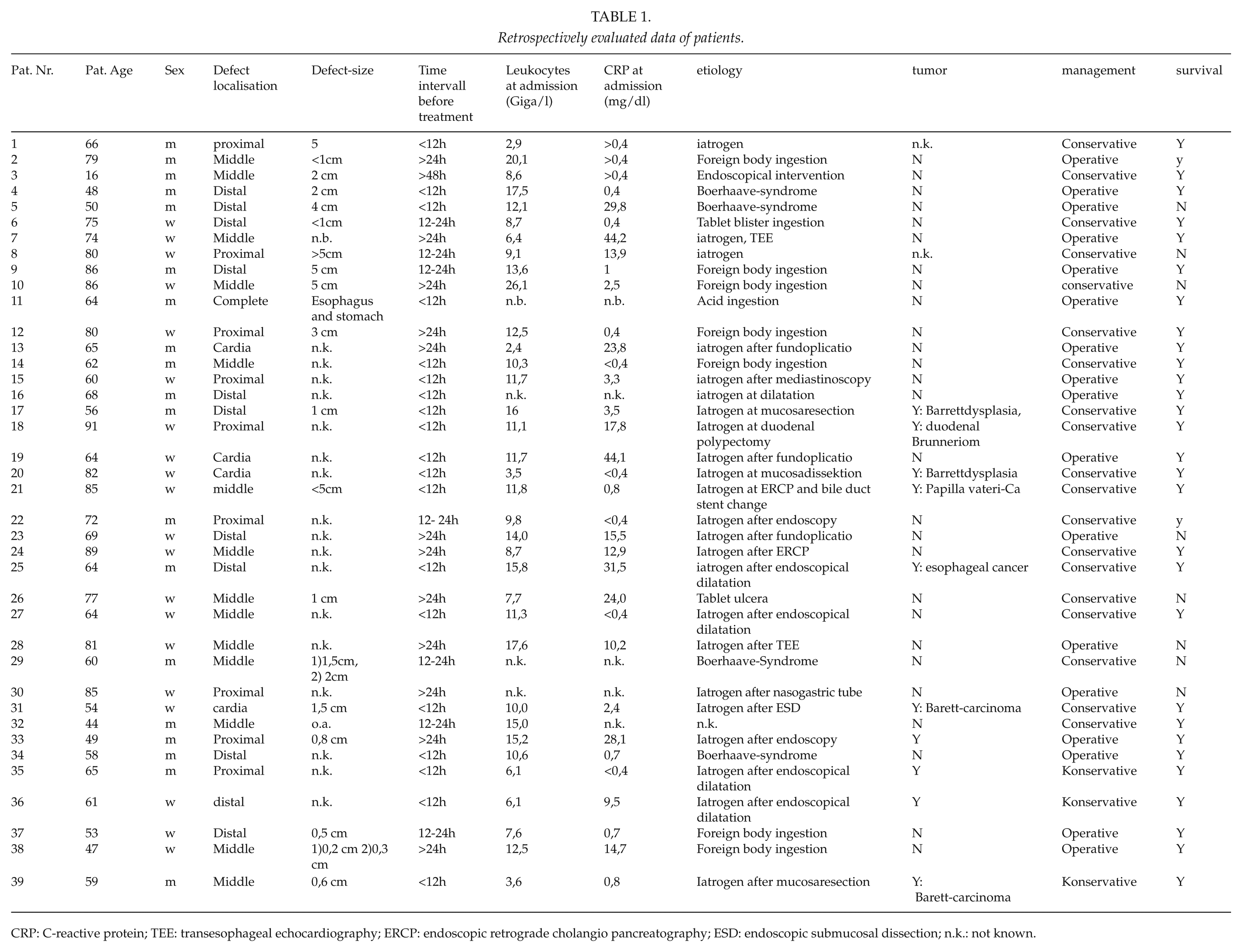
CRP: C-reactive protein; TEE: transesophageal echocardiography; ERCP: endoscopic retrograde cholangio pancreatography; ESD: endoscopic submucosal dissection; n.k.: not known.
Results
Etiology, Localization, and Diagnostics
Regarding the etiology of esophageal perforation, four groups exist: (a) 18/39 iatrogenic/interventional-associated (2 transesophageal echocardiography (TEE), 1 cystic duct stent replacement, 1 endoscopic retrograde cholangio pancreatography (ERCP), 1 polypectomy of the duodenum, 1 submucosal dissection of Barrett’s esophagus, 6 dilatations, 3 diagnostic endoscopies), (b) 9/39 accidental/foreign body ingestion, (c) 4/39 Boerhaave’s Syndrome, and (d) 3/39 postoperative and 5 events could not be classified.
A safe inquiry about the presenting symptoms could not be obtained from the available data. We refer to our initial analysis in which one-quarter of the patients presented with leading symptoms suggestive of esophageal perforation (7).
The most common localization of an esophageal perforation was the distal third with 16/29, followed by the middle third 14/39, and the proximal third with 9/39 patients. An upper endoscopy and a computed tomography (CT) scan with contrast were performed in all patients. The mean size of the defect was 2.4 cm (range: <1 cm to >5 cm, n = 19; however, size of the defect was not documented in all patients).
Treatment
Of the 39 patients, 17 were referred to us from the surrounding hospital which partially performed frustrating therapeutic attempts, 9 patients presented to our surgical emergency room, and 13 patients had already been treated internally.
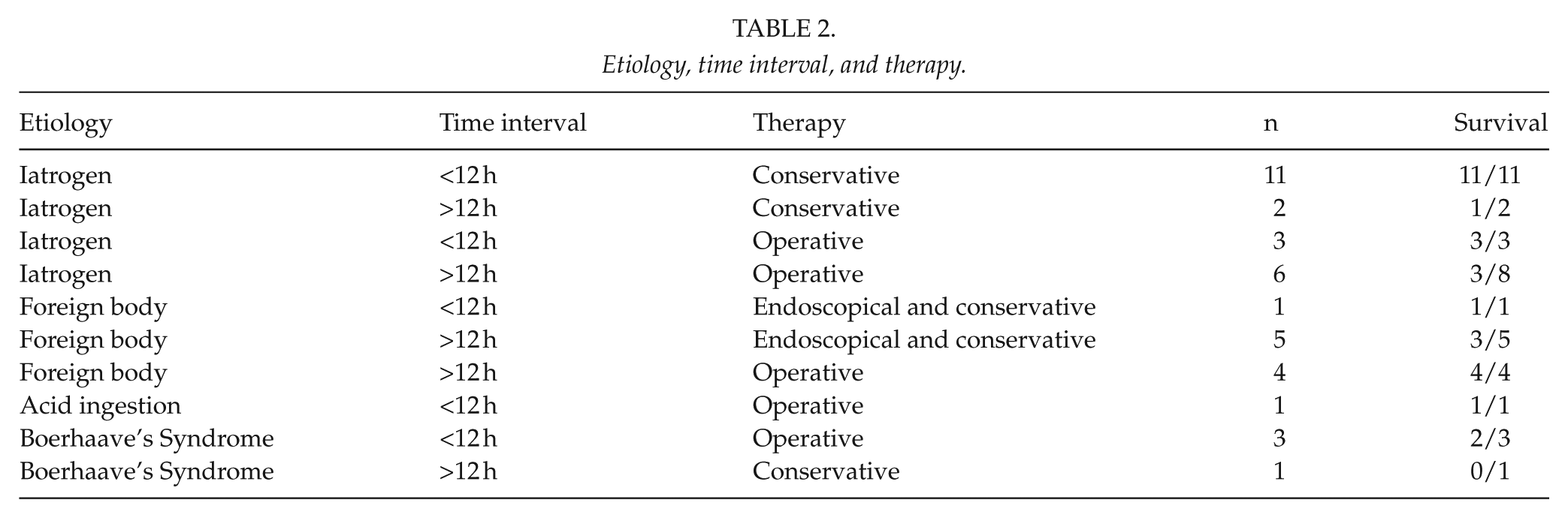
Treatment was initiated within 12 h of the event in 18/39 patients, at >12 h in 5/39 patients, and at >24 h in 16/39 patients (Table 2).
Etiology, time interval, and therapy.
Conservative/interventional treatment
A total of 22/39 patients were treated conservatively or interventional. Of the 22 patients, 7 were treated by stenting, 1 patient was treated by application of hemoclips, and 14 were treated by observation and/or recurrent endoscopic lavages. In five patients, an interventional procedure was followed by surgery. Out of those patients, two received surgery, as we were unable to resect (R0) the adenocarcinoma endoscopically. Thus, resection of the malignancy was performed after primary stabilization with a stent. In n = 1, the state of the patient deteriorated after initial stenting, leading to surgery. In n = 2, initial stenting with subsequent surgery was the treatment plan. Insertion of a thoracic drainage was not performed routinely in the interventional group, rather dependent on the CT result.
Surgical treatment
In n = 17 patients, an initial surgical procedure was performed. Different surgical techniques were used. In n = 5, stitching of the defect was performed; in n = 3, stitching of the defect with subsequent fundoplicatio was performed, of which n = 1 received interposition of the colon in a second setting, n = 1 received esophagectomy with gastric interposition in a second setting, n = 1 received esophagectomy with gastric interposition; n = 3 received esophagectomy with colon interposition; n = 2 received discontinuity resection of the proximal stomach and distal esophagus without reconstruction; in n = 1, abdominal approach through a right laparotomy, transhiatal lavage, and drainage were performed; and in n = 1, a collar approach with mediastinotomy and drainage was performed. The latter approach was used in treating a defect of the proximal esophagus after the extraction of glass fragments. In n = 3 patients who received an interposition of the colon, a gastric interposition was not performed because in n = 1, acid ingestions injured the stomach and in n = 1, gastric/esophageal gangrene occurred post fundoplicatio.
Outcome
A total of n = 31 survived after esophageal perforation and n = 8 passed away, correlating to a mortality of about 21%. While looking at the patient collective according to the time passed before treatment was initiated, one notices that the survival rate was at 94% within the early treatment group (<12 h) compared to 66% within the delayed treatment group (>12 h). This outcome correlates with available published literature. Out of n = 8 who passed away, n = 1 was unable to receive surgical treatment due to her reduced state of health. Another patient was referred to us with a fulminant sepsis who deceased shortly after. Another patient passed away due to an endocarditis and a pulmonary embolism secondary to Boerhaave’s Syndrome. N = 2 deceased postoperatively due to a reduced state of health although no clear focus of infection could be identified. One died spontaneously due to an underlying hematothorax secondary to a pulmonary artery bleeding. One patient passed away due to malignant cardiac arrhythmia.
Discussion
This retrospective analysis was done to characterize and describe patients with an esophageal perforation, who in part were referred, diagnosed, and received their initial treatment from surrounding hospitals.
Due to its etiology and acute clinical presentation, current data from random-control studies do not exist. Thus, a recommendation based on evidence-based medicine is restricted.
The esophageal perforation is a difficult diagnosis and needs to be diagnosed on time. Risk factors are (a) the size of the defect, (b) the localization of the defect, and (c) the time interval between the event and start of therapy (8, 9). One must assume that patients who suffer from an esophageal perforation secondary to an endoscopic procedure tend to have a better outcome, since the time passed between the event and initial treatment is relatively short. Several study groups tried to identify and describe an algorithm for the diagnosis and therapy of esophageal perforation but failed to do so due to the lack of evidence-based data (10). Proven predictors for a negative outcome are malignant associated perforations and an existing mediastinitis at the point of diagnosis (11, 12). Boerhaave’s Syndrome is difficult to diagnose leading to a time delay before treatment is initiated, thus increasing the risk of mortality compared to iatrogenic perforations (13, 14).
The diagnostic of choice must be able to illustrate the perforation in a safe and timely manner. The available published literature describes the barium swallow as the “gold standard” with a false negative ratio of 10% (15). In addition, the extent of a surrounding inflammatory process in terms of a mediastinitis is crucial and can guide the therapy toward a surgical intervention. Thus, from our point of view, the CT with contrast is the better choice, as it is able to illustrate the perforation as well as a surrounding inflammatory process. In addition to the CT, most authors also recommend an upper endoscopy (16), although some express the concern of increasing the size of the already existing defect since it is listed as one of the mentioned etiologies. In the analysis of Horwitz et al. (17), the endoscopy has a sensitivity of 100% with a specificity of 83%. Despite those concerns, we recommend performing an upper endoscopy in all patients, as one is able to illustrate the exact size of the defect and might even be able to treat it by primary stenting. Even the meta-analysis by Biancari et al. (18) published in 2013 described a lack of data for stent-grafting. Only 18 studies with overall 216 patients were available. The pooled mortality was 7.3%; a selection bias is even possible.
In our data collective, not a single endoscopic procedure led to a secondary injury; 55% of our patients (n = 22) received a successful conservative/interventional treatment. In n = 1, the regime of the therapy had to be converted from an initial conservative treatment to a surgical one due to arising complications. In n = 2 patients, a conservative procedure was followed by surgery to treat the underlying malignancy which caused the initial esophageal perforation. The range of conservative/interventional techniques range from pure observation, regular endoscopic lavage, and stenting up to the application of fibrin glue (19–22). Some authors favor interventional procedures (23), while others favor primary surgical treatment (1, 5, 24). Because of the fact that some patients already received therapeutical procedures in other hospitals with resulting time delay for further therapy, we were not able to provide every patient with interventional or conservative therapy. Regarding surgical therapy, the experienced surgeon has different operative options at hand. Localization of the defect and the presence of surrounding inflammatory processes are crucial in choosing the ideal surgical strategy. The localization defines the access to the cavity: cervical in the case of collar injuries, right-thoracic in thoracic defects, and abdominal or combined abdominothoracic in distal defects. Stitching the defect occurs most commonly when the present defect is small with a subsequent closure by pleura, pericardium, minor omentum, or musculature (25). In our patient collective, we performed stitching of the defect without any complications in n = 5 patients, and in n = 3, stitching of the defect with a subsequent fundoplicatio to cover the initial defect. An insufficiency was noted in one patient, which had been described as a complication in the available literature (26). A total of n = 2 patients passed away due to other complications. Primary esophagectomy was recommended in patients with extensive defects, extending surrounding inflammatory processes and other complications (3, 16). In most cases, an interposition of the stomach was performed in a second setting. Interposition of the colon is the exception to the rule and is only used when the gastric tissue is not viable such as in the case of gangrene or alkali burn.
A uniform recommendation on the basis of the available collective data cannot be made. The evaluation of our collective data does not show a statistical correlation between time of diagnosis, type of therapy, and type of lesion. Regarding the rate of survival subject to the time interval between event and initial treatment, our collective data show that the prognosis is defined by this factor. Furthermore, a primary conservative/interventional treatment seems to be a good treatment choice at early diagnosis in the group of iatrogenic dependent perforations (n = 11, survival 11/11). The meta-analysis with the largest collective data for surgery had a perioperative mortality of 18% (5) compared to 21% in our collective. Overall, this elucidates the interdisciplinary challenge and the necessity of prompt communications between and within hospitals to offer quick and efficient treatment in esophageal perforation.
Summary
Perforation of the esophagus is a rare event which is associated with a high mortality. Crucial factors that determine the clinical outcome are (a) general risk factors, (b) etiology, (c) localization and size of the defect, (d) time interval between perforation and diagnosis, and (6) prompt initiation of sufficient treatment.
Considering interdisciplinary diagnosis and therapy as well as respective published studies, it was shown that with early diagnosis, conservative or endoscopic treatment options represent a good therapy. Preferential surgical intervention depends on size of the defect, localization, and inflammatory changes of the defect. This could lead up to a two-session approach with an esophagectomy and a necessary reconstruction of the passage.
Footnotes
Declaration of Conflicting Interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
