Abstract
The authors describe their experience in the treatment of 83 Boerhaave patients. During the last few years the mortality of the disease has decreased. A successful treatment requires good treatment resources and experienced team work. The tailored open primary repair technique with fundic reinforcement, developed by the authors, is described in detail. This technique has decreased the amount of postoperative fistulation and esophageal resection. The mortality after stenting was 20%.
Boerhaave’s syndrome, an esophageal rupture caused most often by emetic barotrauma, is rare. To date, only small case series with empiric treatment recommendations have appeared (1). The rupture is mostly situated left posterolaterally in the distal esophagus. Without treatment, this disease is fatal, mostly due to septic complications caused by esophageal leakage.
Diagnosis
Classically, patients present with severe retching and vomiting followed by heavy retrosternal or upper abdominal pain. However, the clinical diagnosis is often elusive and is confused with more common acute cardiovascular, abdominal, and lung diseases.
Early diagnosis and early initiation of appropriate treatment are important. Diagnostic procedures and treatment must begin simultaneously. Intravenous antibiotics are started, and the patient ingests nothing by mouth. Esophagography with water-soluble contrast agent is a classic diagnostic tool, but we prefer to perform an emergency computed tomography (CT) scan of the thorax and upper abdomen and an upper gastrointestinal endoscopy. Besides diagnosis of the rupture, the CT allows assessment of the extent of pleural, mediastinal, and abdominal contamination. CT also directs the performance of interventional radiology allowing precise allocations of drains and punctures. Emergency endoscopy, preferably under sedation, helps in the diagnosis and selection of treatment modalities. However, endoscopy should be performed only by experienced endoscopists to avoid more damage to the esophagus, which has already been ruptured. Lack of bright bleeding after biopsy indicates vitality problems in the esophageal tissue. Besides the extent and localization of the rupture, the pathology and vitality of the esophageal wall can be estimated. In cases where the esophageal wall is necrotic, the possibility of esophageal resection has to be considered.
Treatment
Treatment should always be tailored to the patient’s general condition, the vitality of the esophageal wall, the rupture’s size and location (thoracic, abdominal, or both), and any delay in diagnosis. Conservative treatment with antibiotics is possible for small ruptures in patients with minimal or no sepsis at all. For successful conservative treatment, the rupture should be contained in the mediastinum and be drainable back into the esophagus without pleural fistula. The healing of the rupture in stabile patients should be followed weekly using esophagography and esophagoscopy. The infectious collections in thorax and abdomen, if diagnosed, should be drained.
All other cases require surgical intervention. An endoscopically replaced covered esophageal stent, increasingly recommended in other esophageal perforations, can sometimes be successful (2). However, stenting is usually a very time-consuming treatment and usually demands several debridement operations (thoracoscopy/thoracotomy or laparoscopy/laparotomy) and the resources of interventional radiology. Stented patients may die from complications even several months after rupture. For late removal, totally covered metal stents with a large diameter are the best choice. Stents often dislocate and may penetrate into the surrounding organs, requiring mandatory repeat checking of their position and conformation to ensure the absence of any esophageal fistula. We use stents mostly in patients who cannot stand more invasive treatment. A well-placed stent prevents fistulation of gastric and duodenal contents and saliva into the mediastinum and allows time for resuscitation of the vital functions in compromised patients for whom surgery may be too stressful. The possibility of replacing a stent after migration and post-stenting strictures caused by pressure necrosis should however be kept in mind.
Primary esophageal repair with fundic reinforcement is the gold standard in Boerhaave’s syndrome and should always be considered. The approach is usually left thoracotomy, sometimes with the addition of phrenolaparotomy. This allows good control of infection by cleansing of mediastinal and pleural collection from decortication of a lung. If the rupture reaches into the abdominal esophagus, the distal esophagus and fundus may easily be approached through short phrenolaparotomy performed parallel to the thoracic wall.
Our technique is presented in Figs 1 to 6. Reinforcement of the primary repair with the fundic wall prevents pleural leakage in those cases in which the repaired esophageal wall has not healed but opened up. This technique, first used in 1989, has reduced the number of esophagectomies even in delayed cases. After this reinforcement technique, only one patient had to be resected and one stented.
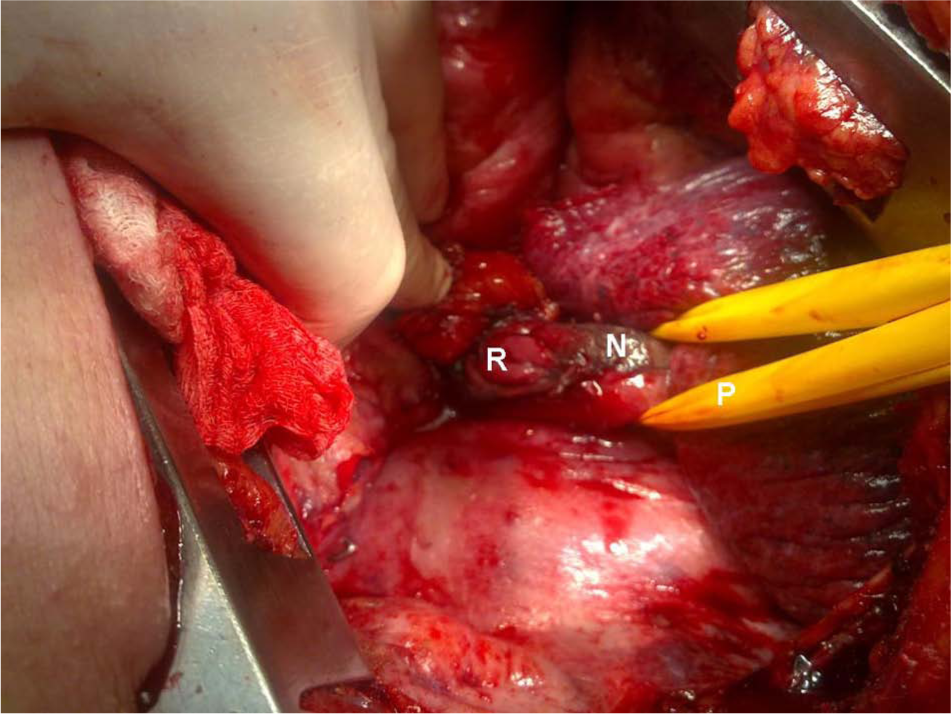
Mobilized esophagus after mediastinal cleansing and resection of the necrotic pleura covering a 4-day-old Boerhaave’s rupture (R); some necrosis (N) is still left on the esophageal wall, and Penrose drain (P) is around the esophagus. The approach is left posterolateral thoracotomy. The latissimus dorsi muscle can be mobilized and saved.
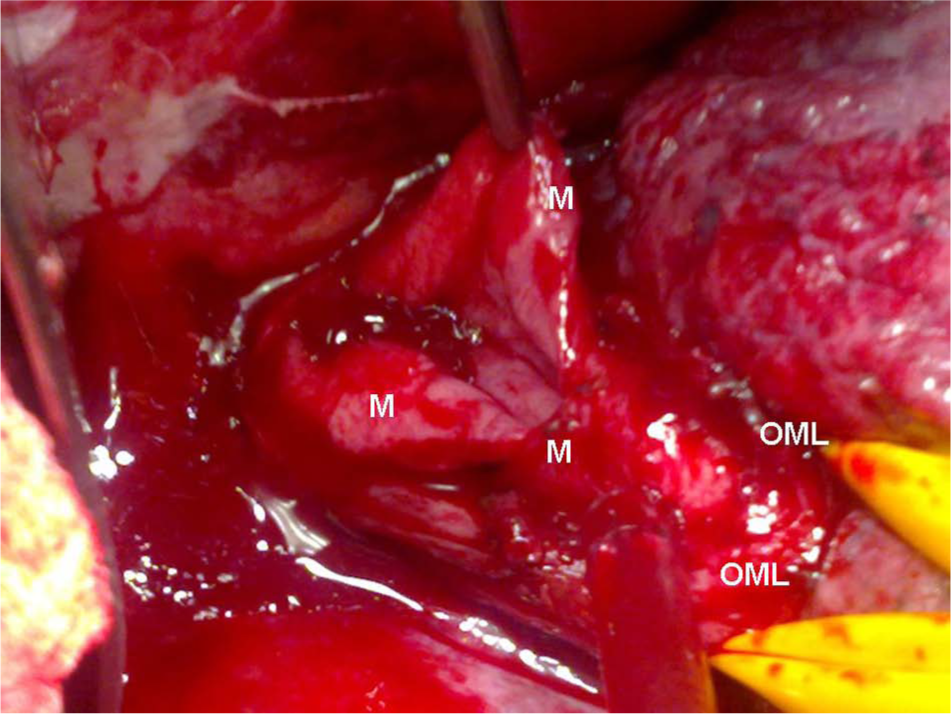
Esophageal wall after resection of the necrotic areas is vital, showing bleeding from its mucosal margins (M). Both ends of the mucosal rupture must be identified by means of mobilization of the esophageal muscular layer (OML).
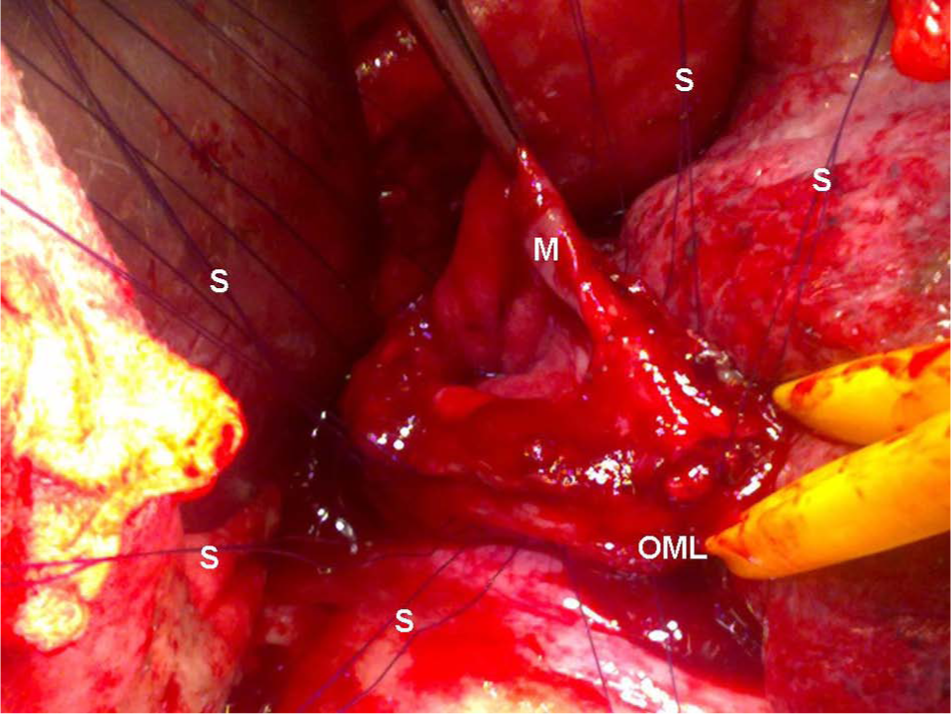
Resorbable 4-0 monofilament sutures (S) with two needles are first placed from the inside of the esophagus through all layers surrounding the whole rupture, including the distal portion, for firm reinforcement of the repaired rupture with the fundus. Note the sufficient lateral extension sutures from rupture borders.
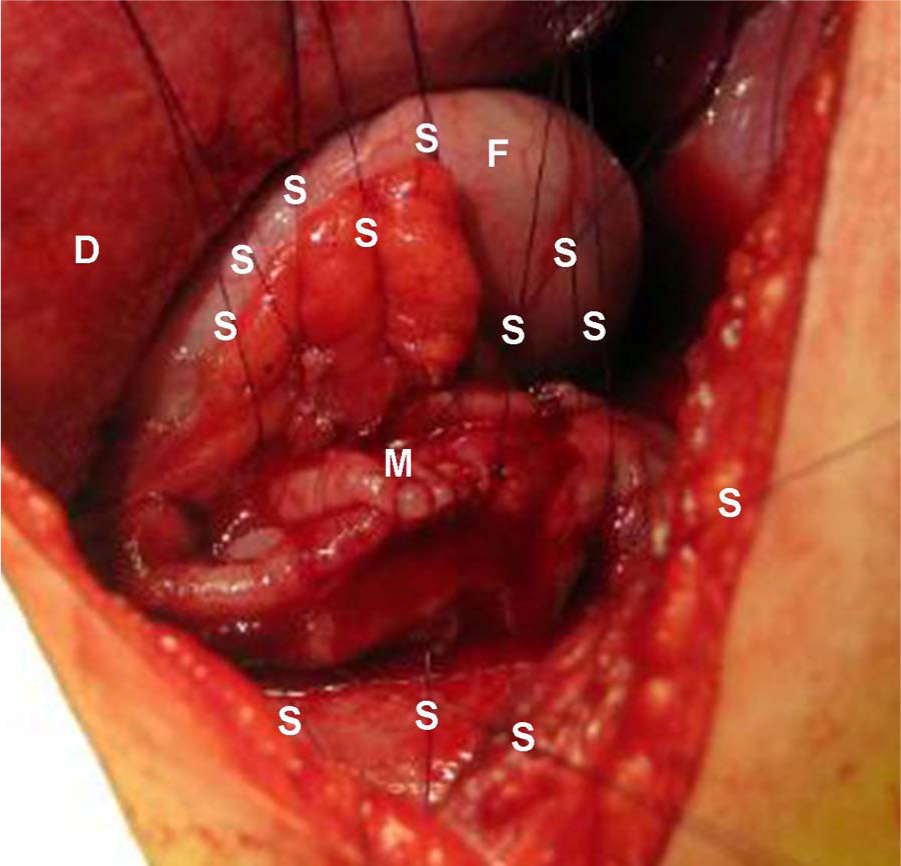
Repair of esophageal mucosa (M) with a running watertight suture (resorbable 4-0 or 5-0 monofilament) after placement of reinforcement sutures. The fundus (F) is mobilized dividing the short gastric vessels via esophageal hiatus or via a separate short phrenotomy made circularly about 3 cm from the thoracic wall. The fundus is pulled through the esophageal hiatus into the mediastinum.
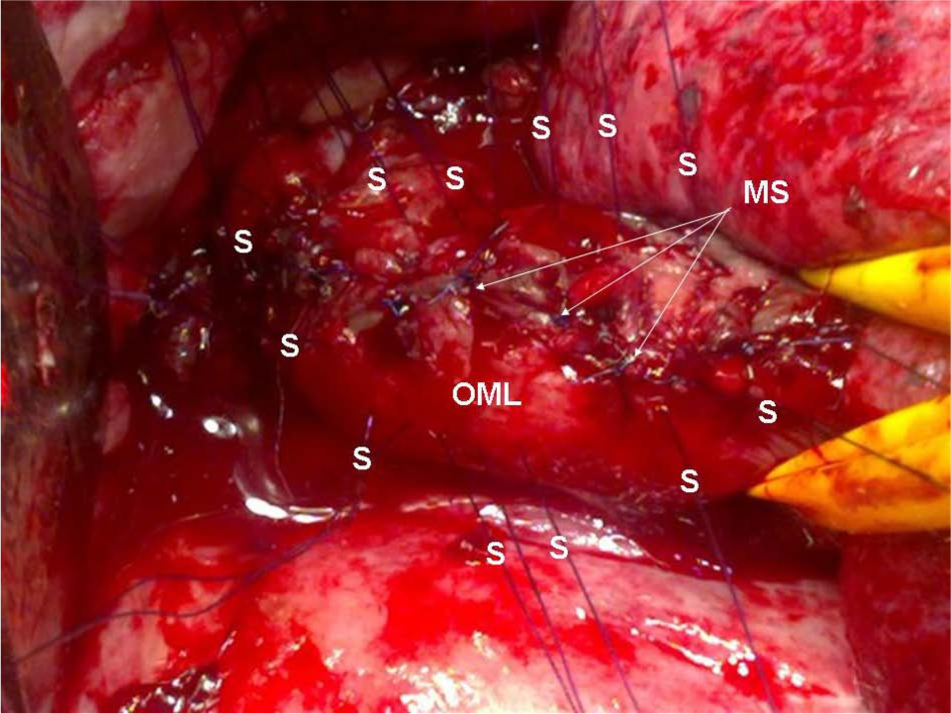
After mucosal repair, the esophageal muscular layer (OML) is separately closed by use of single 4-0 resorbable monofilament sutures (MS).
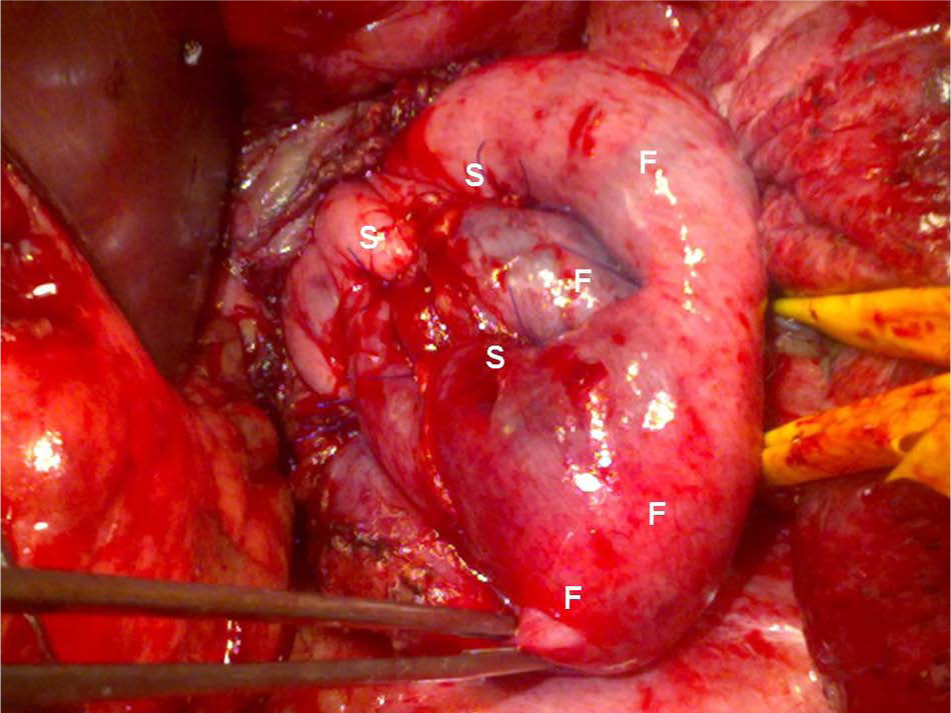
The rupture is reinforced with gastric fundus (F). The reinforcement sutures (S) go through the esophageal wall and both fundic walls building an “extra wall” over the repaired rupture. A partial or total fundoplication is performed. The distance between reinforced sutures throughout the esophageal and fundic layers is about 1 cm. The important fixation of the intrathoracic fundus into the esophageal hiatus is performed with nonresorbable 3-0 suture material.
Esophagectomy is indicated in patients whose esophageal wall is widely necrotic. Esophagectomy is still a lifesaving procedure. In comparison to figures during the last millennium, the number of esophagectomies in our division has decreased. The gastrostomy tube (peg) can be inserted at endoscopy before stabilizing the distal esophagus during surgery. The oral part of the esophagus is mobilized via left cervicotomy and slipped subcutaneously for esophagocutaneostomy preferably some centimeters caudal to the left clavicle, thus allowing stable fixation of the esophagostomy bag. In rare cases (with limited infection), the maximal length of the thoracic esophagus with vagal nerves can be preserved. The distal stump is closed in the mediastinum, and a side-to-side cervical esophagostomy is performed. In these cases, the reconstruction of the esophageal anastomosis in the posterior mediastinum may be challenging because of adhesions. Because of severe infection and edematous tissues, the reconstruction of the alimentary passage is usually worth doing not until after 6 months or later. Reconstruction of the alimentary tract (mostly with colon) routinely requires a retrosternal route. A subcutaneous route is possible, in patients who have undergone surgery such as coronary bypass, in the anterior mediastinum.
Results
The experience of the first author (J.S.) includes 83 patients with Boerhaave’s syndrome treated over a period of more than 35 years. Mortality has decreased from 50% (>24-h delayed cases at the beginning) (3) to the present 5%. No single reason explains this decrease over this lengthy period. Improvements in intensive care treatment and anti-infectious medication, earlier diagnosis, and more tailored treatment strategies have all contributed to this improved outcome for Boerhaave’s syndrome.
The number of resections has decreased as well. Many delayed cases earlier treated with esophagectomy have been successfully treated with primary repair by these reinforcement/techniques with the gastric fundus. The mortality in stented cases is 20%, probably because almost all stented patients had severe underlying disease. Overall delay over 24 h was observed in 46% of patients. The delay was 27% in stented patients, 55% in the esophagectomy group, and 43% in the primary repair group.
