Abstract
Surgical wound infection is one of the most common complications after peripheral vascular surgery. It increases the affected patient’s risk for major amputation as well as mortality. Furthermore, surgical wound infection is an additional cost. Wound infections after vascular surgery are of multifactorial nature and generally result from the interplay of patient- and procedure-related factors. The use of systemic antibiotic prophylaxis may be the most important method in preventing surgical wound infections. In this review article, we report the current literature of surgical wound infections after peripheral vascular surgery.
Keywords
Incidence
The majority of arterial surgery procedures are classified as clean surgery by the National Research Council because the operative exposure and revascularisation are performed in uninfected tissues without inflammation and the alimentary, respiratory, or urinary tract is not entered (1). Lower limb revascularisation surgery has been widely proven to be effective for the management of both claudication and critical limb ischemia. According to the National Nosocomial Infections Surveillance (NNIS) report, with almost 85,000 surgical procedures, the incidence of surgical wound infection (SWI) after clean surgery is 2.1% (2). However, the incidence of surgical wound complication, either infection or failure to heal, after lower limb revascularisation surgery remains high compared with other clean-surgery procedures.
Infection involving a vascular surgical wound may be superficial or deep, or the infection may involve vascular graft or operated native artery. For autogenous arterial revascularisations, only infections occurring within 30 days are classified as SWI, but when prosthetic graft is implanted, the incidence of SWI is calculated for 1 year (1).
There is an extensive variation in the reported incidence of SWI after lower limb vascular surgery. In prospective studies with the number of patients ranging from 69 to 561, the incidence of SWI was found to be between 3.5% and 32% (3–13).
The rate of SWI varies among different types of vascular-surgery procedures. The incidence of SWI, including patch infection after carotid surgery, is 0.2%–0.5% (14, 15). Open abdominal aortic surgery is associated with 2.4%–6.3% risk of 30-day SWI (16) and 0.44%–1.9% risk of graft infection (17, 18). In a retrospective analysis with 12,626 open abdominal aortic aneurysm reconstructions, the risk of developing graft infection was highest during first postoperative year: 32% of all graft infections developed during the first year after surgery (17). For vascular access created for hemodialysis, infection risk depends on the access material. Infection risks related to arteriovenous fistula and arteriovenous prosthesis are 0.9% and 9.1%, respectively (19). In prospective studies, the incidence of SWI after varicose vein surgery has been reported to vary between 5% and 14% (20, 21). It seems plausible to draw a conclusion that vascular-surgery procedures including lower limbs are associated with a relatively high risk of SWI. SWI rates after vascular-surgery procedures reported by various studies are presented in Table 1.
Reported rates of surgical wound infection after different types of vascular-surgery procedures.
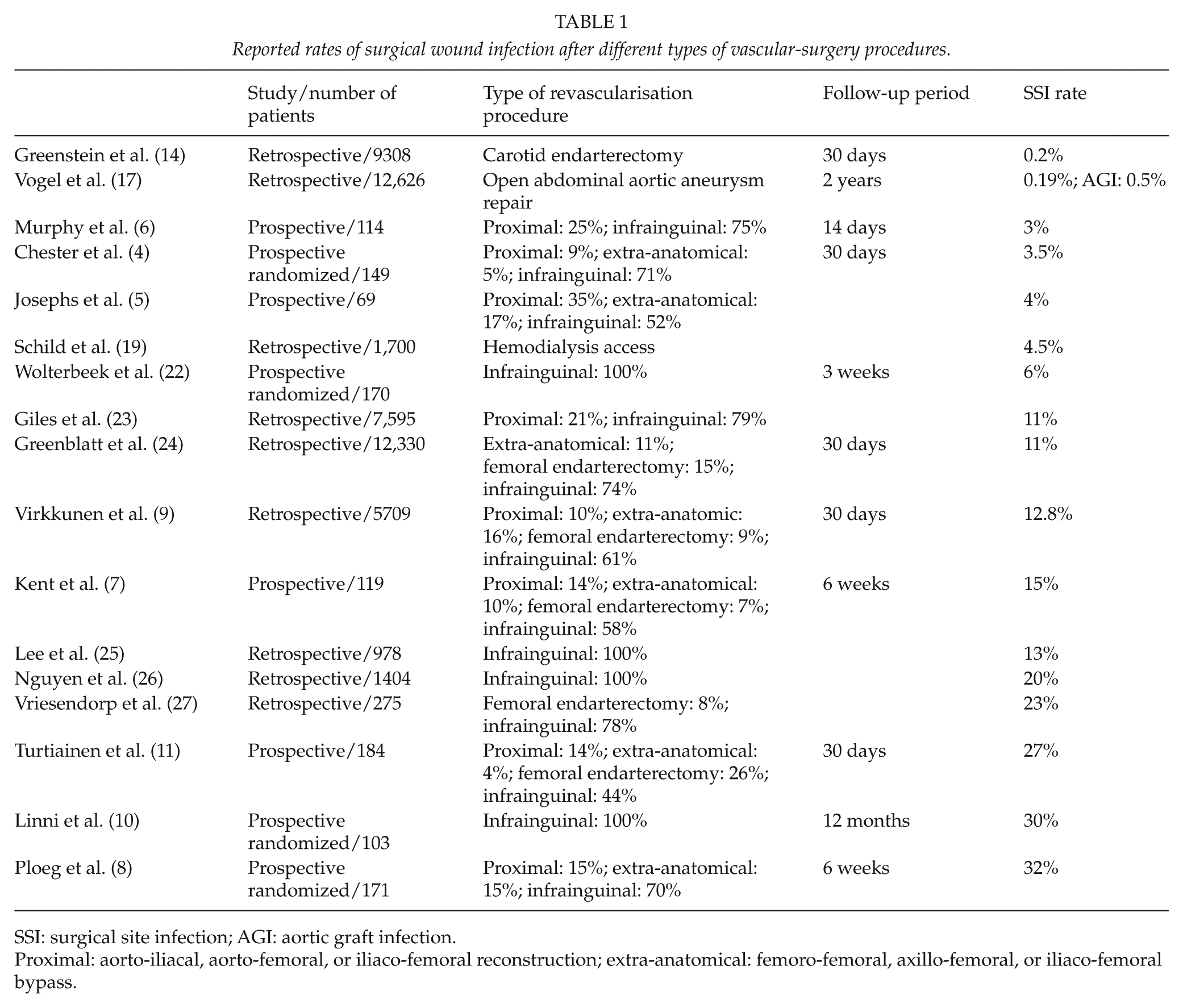
SSI: surgical site infection; AGI: aortic graft infection.
Proximal: aorto-iliacal, aorto-femoral, or iliaco-femoral reconstruction; extra-anatomical: femoro-femoral, axillo-femoral, or iliaco-femoral bypass.
Risk Factors for SWI after Lower Limb Vascular Surgery
The greater likelihood of SWI occurrence in the peripheral vascular-surgery patients compared with other clean-surgery patients is due to both procedure- and patient-related factors. The risk of deep graft infection following vascular prosthesis implantation is 1%–5% (28). However, the use of a vein graft has remained to be associated with increased risk of SWI compared with use of prosthetic graft or use of no graft (29). That is most probably related to long exposures needed for vein harvesting. Furthermore, graft placed mainly in the subcutaneous location instead of subfascial location increases the risk of SWI, with relative risk (RR) of 11.6, in patients undergoing infrainguinal bypass surgery (30). Prolonged operation time is associated with SWI (24, 31), which is speculated to be due to patient hypothermia, inadequate redosing of antibiotic, retractor-related tissue trauma, and breaks in sterile technique (24). Previous arterial surgery in the groin, earlier arteriographic procedure performed via operation site, and the use of wound drainage have also shown to increase the risk of SWI (11, 32, 33).
Groin incisions for vascular access may be either vertical crossing through the lymphatic tissue or oblique sparing the lymphatic tissue. Many surgeons prefer a vertical incision because it offers a direct approach and full exposure of the femoral artery. Two retrospective studies (n = 149, n = 196) have revealed that oblique groin incision for vascular access significantly decreased the incidence of SWI compared with vertical incision (4, 34). However, a later study using prospective collected data (n = 171) showed no difference between the two incisions (8). Furthermore, a prospective study with 116 groin incisions revealed that transverse incision may increase the risk of postoperative lymphatic leak, but does not increase the risk of SWI compared with oblique incision (35).
In a cohort study with 1880 patients undergoing lower limb bypass surgery, perioperative transfusion of packed red blood (PRB) cells was an independent risk factor for SWI. The incidence of SWI in patients receiving 0 units, 1–2 units, or ≥3 units of PRB cells was 4.8%, 6.5%, and 14.0%, respectively, p = 0.0004 (36).
Infection of vascular prosthetics implanted for arterial occlusive disease occurs in approximately 1%–5% of patients. The incidence of infection depends on the anatomical site, with the highest rate occurring in vascular access grafts placed for hemodialysis and in inguinal and lower extremity incisions in patients undergoing bypass procedures for femoral, popliteal, or tibial occlusion (28). No difference has been found in the incidence of SWI when using either polyester (Dacron) or expanded polytetrafluoroethylene (ePTFE) vascular prostheses (37).
Factors that increase the risk of aortic graft infection are graft contamination from skin flora during surgery, groin incision, graft revision, superficial SWI, emergency operation, and postoperative bacteremia (38).
A study with 12,330 patients who underwent open surgery for lower limb peripheral vascular disease found obesity to be an independent risk factor for SWI development (odds ratio (OR) = 2.1, 95% confidence interval (CI) = 1.8–2.4) (24). Furthermore, obesity has increased the risk of SWI after peripheral artery surgery in other studies with smaller number of patients with an OR varying between 1.7 and 6.08 (11, 13, 23, 25, 39). In addition, morbid obesity (body mass index (BMI) ≥ 40 kg/m2) has been found to be an independent risk factor for SWI after open aortic aneurysm repair with an OR of 2.6 (16).
The reasons for higher risk of SWI in obese patients may be tissue hypoxia and low antibiotic concentration. Wound and tissue oxygen tension has been found to be reduced during surgery among obese patients, and this is associated with increased SWI risk (40). Pharmacokinetic analysis has revealed that Cefazolin, used as prophylactic antibiotic, has a reduced concentration within adipose tissue in obese patients compared to lean patients (41).
Critical limb ischemia manifests as chronic rest pain, non-healing ulcers, or gangrene. Patients with contaminated ulcers remote from the surgical wound may be at risk of endogenous bacterial contamination via bloodstream or lymphatics. A retrospective study of 756 patients showed that critical ischemia with rest pain, an ulcer, or gangrene (Fontaine III-IV) independently increased the risk of SWI after lower limb vascular surgery (OR = 4.1, 95% CI = 1.88–8.88) (42).
Other patient-specific risk factors for SWI in lower limb vascular surgery include diabetes mellitus, postoperative hyperglycemia, chronic obstructive pulmonary disease (COPD), asthma, coronary artery disease, female gender, dialysis, and current use of corticosteroids (3, 5, 11–13, 24, 25, 27, 39). COPD and asthma may be risk factors to SWI due to a lowered oxygen delivery. An explanation of the association between female gender and inguinal SWI is thought to be gender-related differences in the amount and distribution of body fat and native skin flora (24). Patients with chronic renal failure and uremia have impaired host defenses and wound healing that can lead to increased risk of SWI (43).
Causative Agents
The most common causative agent associated with SWI after lower limb arterial surgery is Staphylococcus aureus which has been isolated from 29%–60% of infected wounds (11–13, 44, 45). Methicillin-resistant S. aureus has been reported to be responsible for 30%–50% of S. aureus wound infections in the United States (25, 44, 45). The other causative bacteria are as follows: Staphylococcus epidermidis 17%–24%, Streptococcus 19%, Pseudomonas 3%–20%, Enterococcus 6%–21%, Escherichia coli 2%–9%, and Enterobacter 3%–9% (11–13, 25, 44, 45–46). Cultures taken from wound with clinical signs of infection do not exhibit bacterial growth in 10%–17% of the cases (12, 13).
Early prosthetic vascular graft infections are usually owing to S. aureus or, in the case of aortoenteric fistulae, gram-negative species (47). Late infections often involve more indolent species such as S. epidermidis (38).
Impact on Patient Outcome
Although most of the SWIs after vascular-surgery procedures heal with treatment (11–13, 44, 46), they may be a significant source of morbidity. SWI increases the risk of graft failure requiring intervention (23, 24). Reported incidence of major amputation owing to SWI ranges from 2% to 25%, being highest as a result of vascular graft infection (26, 45). Several studies have not found association between SWI and 30-day mortality (24, 46, 48).
Epidemics of multidrug-resistant bacteria are particularly problematic; a matched case-control study revealed that an outbreak of multidrug-resistant Pseudomonas aeruginosa in a vascular-surgery ward was associated with decreased short-term amputation-free survival after infrainguinal bypass surgery in patients with critical limb ischemia (49).
Mortality owing to prosthetic vascular graft infections has been variously reported to be 11%–20% and 6.5%–11% owing to lower limb prosthetic vascular graft infection (29, 44, 45, 47). Aortic graft infection increases the mortality with OR of 5.6 (95% CI = 1.1–28.7) (47).
Impact on Resource Utilization
SWI causes additional costs of vascular-surgery procedures by delaying recovery, increasing the length of stay (LOS), and necessitating the need for additional treatment. LOS is increased by 2–14 days because of SWI (16, 29), and it has been calculated, in 1996, that cost of one SWI is €405 per infected patient (7). A database register study, conducted in the United States, with 870,000 elective vascular surgical procedures, including aortic, endovascular, and lower limb revascularisation procedures, was performed to describe the infectious complications after elective vascular surgery. In that study, nosocomial infections increased the LOS from 4.2 to 13.8 days and the hospital costs from US$12,000 to US$38,000 (€9000–€29,000) (50). A recent prospective study conducted in Finland showed that the average cost attributable to SWI after lower limb vascular surgery was €3320 (11).
Treatment of SWI after Vascular Surgery
The basic treatment of SWI is to open and drain the wound, and to remove the necrotic tissue. Culture should be performed to identify the source of infection, and antimicrobial therapy should be adjusted. Superficial incisional infections can often be treated with oral antibiotics only, without surgical debridement (51).
Treatment of vascular prosthetic graft infection remains a challenging problem. Traditional management of infected arterial grafts included total graft revision and complex extra-anatomical or venous revascularisation procedures. Since such management is associated with high mortality (26%–34%) and limb loss rate (23%–36%) (52, 53), techniques for partial and total graft preservation have been developed. However, removal of the entire infected graft is still essential in patients who present systemic sepsis or with infected disrupted anastomoses or anastomotic bleeding (44).
In a study of 120 patients with infected extracavitary prosthetic arterial grafts (95 ePTFE and 25 Dacron), complete or partial graft preservation was attempted in 94 (78%) patients. The hospital mortality rate was 12%, and the hospital amputation rate among survivors was 13% (14/106). In patients treated by complete graft preservation, the long-term complete graft preservation was successful in 71% (32/45) of cases. Partial graft preservation was successful in 85% (34/41) of surviving patients with occluded grafts (44). Another study with 40 grafts (24 prosthesis, 3 vein, and 13 biological) and 9 native arteries reported 91% complete wound healing rate when, after radical debridement and graft preservation, negative pressure wound therapy was applied directly on the infected graft/wound. There was no difference in outcome between the various graft types involved (54). Coverage of exposed prosthetic material in the groin with a muscle flap may be an effective option for decrease morbidity, and to increase limb and graft salvage (55, 56).
Prevention
There is a clear evidence of the benefit of prophylactic broad-spectrum antibiotics for vascular reconstruction (57). A meta-analysis of 34 studies was conducted to evaluate the evidence for effectiveness of interventions to prevent wound and graft infection in peripheral vascular reconstructions. Of these trials, 22 were trials of systemic antibiotics. The meta-analysis demonstrated that prophylactic treatment with systemic antibiotics administered immediately preoperatively reduces the risk of wound infection and early graft infection between two-thirds and three-quarters (57). Broad-spectrum cefalosporins, penicillin/β-lactamase inhibitors, or aminoglycosides would appear to confer similar benefits, and a 24-h regimen of antibiotics appears to be an effective prophylaxis (57).
The effect of local vancomycin on inguinal wound complication rates after vascular procedures was investigated in a retrospective study with 454 patients. The study found a significant decrease in the 30-day incidence of overall wound infections in patients receiving local vancomycin treatment intraoperatively (25.1% vs 17.2%; p = .049) (58).
The effect of the use of supplemental inspired postoperative oxygen on the incidence of SWI after lower limb vascular surgery was tested in prospective randomized study with 274 patients. The SWI rate was 18% in patients receiving supplemental postoperative oxygen and 28% in patients of the control group (p = 0.07). In the subgroup analysis of 103 patients with inguinal incisions only, the use of supplemental oxygen independently reduced the risk of SWI (12).
Suction drains are commonly used to prevent postoperative hematoma formation. However, the use of drainage has shown to increase the risk of SWI (32, 33), and a meta-analysis of four trials revealed that the use of drainage confers no benefit following lower limb revascularisation (59). On the other hand, a postoperative incisional hematoma has been associated with SWI development (25). Thus, it seems plausible that both meticulous hemostasis and avoidance of surgical drainage would decrease the risk of SWI. In addition, efforts to reduce operation time, for example, by using a “double-team,” would be beneficial since prolonged operation time is associated to SWI (24, 31).
A meta-analysis of 21 studies with 2799 patients undergoing coronary artery bypass graft (CABG) surgery showed that open saphenous vein harvesting increases the risk of SWI (OR = 3.85, 95% CI = 2.53–5.86) compared with minimally invasive vein harvesting (60). Another meta-analysis of 43 studies with 27,789 patients revealed that endoscopic vein harvesting reduces the risk of SWI in patients undergoing CABG compared with open technique (OR = 0.27; 95% CI = 0.22–0.32) (61). Furthermore, ultrasound vein mapping of the great saphenous vein reduced the risk of major SWI (1.9% vs 21.2%, p = 0.004) compared with no vein mapping in infrainguinal bypass surgery in a prospective randomized study of 103 patients (10). Using ultrasound vein mapping of great saphenous vein and minimally invasive vein harvest technique for lower limb revascularisation surgery may be beneficial in order to avoid SWI.
The effect of triclosan-coated sutures on the incidence of SWI after lower limb vascular surgery was tested in a prospective randomized study with 274 patients. The study showed that triclosan-coated sutures do not reduce the risk of SWI (13). Another randomized trial with 374 patients reported that leg-wound closure with triclosan-coated sutures does reduce the SWI rate in CABG patients after open vein harvesting (62). In this study, there was no difference in the incidence of SWI at 30 days postoperatively. However, the authors reported the diagnoses that were made at day 60 postoperatively. The primary diagnosis of SWI at 60 days was made by the patient, not by the surgeon. Furthermore, no multivariate analysis was done. Closer analysis may have led to the conclusion that the use of triclosan-coated sutures does not reduce the risk of wound infection in CABG after open vein harvesting.
Conclusion
SWI after vascular surgery is a frequent complication associated with significant additional costs. The most important risk factors for SWI are obesity and surgical infrainguinal incisions. The use of systemic antibiotic prophylaxis may be the most important factor in the prevention of SWI. The other methods to decrease the incidence of SWI include the use of preoperative ultrasound vein mapping, minimally invasive vein harvesting techniques, and the use of inspired supplemental oxygen postoperatively.
Footnotes
Declaration of Conflicting Interests
No conflict of interest.
