Abstract
Background and Aims:
Although conventional laparoscopic surgery is less traumatic than open surgery, it does cause tissue trauma and multiple scar formation. The size and number of ports determine the extent of the trauma. Single-port laparoscopic surgery is assumed to minimize and perhaps eliminate the potential adverse effects of conventional laparoscopy. The aim of this study was to examine short-term outcomes of single-port laparoscopic surgery for rectal cancer.
Material and Methods:
Prospectively collected data from 25 consecutive patients who underwent single-port laparoscopic surgery for rectal cancer between January 2010 and March 2012. Perioperative data, oncological resections, and short-term outcomes were assessed.
Results:
Male:female ratio was 10:15. Of the 25 patients, 44% had previously undergone abdominal surgery. Median body mass index was 24 kg/m2 (range: 19–32 kg/m2). In all, four patients (16%) had neoadjuvant therapy. Median operating time was 260 min (range: 136–397 min). An additional port was needed in two patients, and one case was converted to hand-assisted approach. Median postoperative stay was 7 days (range: 4–39 days), and three patients (12%) were readmitted. Median lymph node harvest was 13 (range: 3–33). The surgical margins were clear in all patients. Complications were seen in six patients.
Conclusion:
Single-port laparoscopic surgery for rectal cancer can be performed in selected patients with rectal cancer without compromising oncological safety and with acceptable morbidity and mortality rates.
Keywords
Introduction
Laparoscopic procedures offer faster recovery, reduced hospital stay, fewer wound-related complications, and better cosmesis. Oncological outcomes do not differ from the traditional open procedures (1, 2). Although conventional laparoscopic surgery (CLS) is less traumatic than open surgery, it does cause tissue trauma and multiple scar formation (3). The size and number of ports determine the extent of the trauma (4, 5). Single-port (SP) or single-incision laparoscopic surgery (SILS) is assumed to minimize and perhaps eliminate the potential adverse effects of CLS. Several clinical series have demonstrated the feasibility and safety of SP surgery in different surgical procedures (6, 7). SP technique allows colorectal procedures to be performed as an almost scarless procedure either through the umbilicus or a stoma site. Two case reports have described SP right-sided hemicolectomy with laparoscopic instruments and transparietal sutures with acceptable oncological results (8, 9). Nevertheless, a few drawbacks hamper the implementation of the SP approach in colorectal surgery: longer procedure times, limited patient applicability, inadequate current technology, and difficulties with training result in a significant learning curve.
The SP literature on rectal cancer is very limited (10 –12). We found it to be interesting to report the preliminary experiences and short-term outcomes with single-port laparoscopic surgery for rectal cancer (SPLRS) at our institution.
Material and Methods
Patient Selection
The departmental review board approved this study. Patients referred to our institution for surgical treatment of rectal cancer were evaluated for SPLRS. Rectal tumors were staged according to national guidelines. The preoperative work-up included targeted biopsy from the rectal lesion, proctoscopy with a rigid rectoscope, colonoscopy, computed tomography (CT) with intravenous contrast, and magnetic resonance imaging (MRI) of the rectum. Tumors were considered rectal cancers if located less than 15 cm from the anal verge (AV) on examination with a rigid rectoscope. All patients were discussed at a multidisciplinary colorectal cancer team (MDT) meeting before and after surgery. The following variables were assessed: (1) demography: age, gender, body mass index (BMI), and American Society of Anesthesiologists (ASA) Score; (2) tumor characteristics on preoperative MRI: size, tumor, node, metastases (TNM) stage, distance from the AV, and circumferential resection margin (CRM); (3) perioperative data: operative procedure, operating time, blood loss, and conversion to CLS and hand-assisted laparoscopic surgery (HALS) or open surgery; (4) histopathology reports: TNM stage, quality of specimen, number of harvested lymph nodes, length of specimen, CRM, distal resection margin (DRM), and tumor size; (5) postoperative data: short-term complications, length of stay (LOS) in hospital, readmissions, and 30-day overall mortality.
Patients with CT- or MRI-evidenced tumor infiltration into adjacent organs and T4 obstructive cancers and patients with mid-rectal tumors located from 5 to 8 cm from AV were considered unsuitable for this SP feasibility study as technical difficulties with a limited space in narrow pelvis could be foreseen. For this reason, there is a predominance of upper and lower rectal tumors in this series. The distance from the AV, the size of tumor, and BMI together with anatomical shape of the patient had a substantial role in the selection. All patients were informed about the possible risks of SP and understood that they were having a nonstandard procedure. Informed written consent was obtained. Patients were also informed about the conventional CLS approach and that additional incisions and/or conversion to open surgery may be needed as warranted during the SPLRS procedure. Stoma sites were marked preoperatively. A Hartmann’s operation (HO) is the recommended procedure for elderly patients or patients with severe comorbidities in our country due to increased risk of anastomotic leakage. A phosphate enema was given prior to surgery. Oncological surveillance and follow-up after surgery were done according to Danish Colorectal Cancer Group (DCCG) guidelines (13).
Statistical Analyses
Proportions are given as percentages with 95% confidence intervals (CIs). Median values are reported with interquartile ranges and upper–lower ranges. Statistical analyses were done with the SPSS statistical package version 19 (SPSS, Inc., Chicago, IL, USA).
The SPLRS Technique
The patient was initially placed in the Lloyd-Davies position. Depending on operative procedure and the location of the rectal tumor, a right- or left-side pre-marked stoma site or umbilical site was used to access the abdomen. The camera operator was located on the right side of the patient together with the surgeon in all patients operated with transumbilical access and/or chosen stoma site on the right side of abdomen (Fig. 1A). The surgeon was located on the left side of the patient, while the chosen stoma and extraction site was located on the left side (Fig. 1B). A skin and facial incision of 2.5 cm was used to access the abdominal cavity. The abdomen was entered under direct vision and the SILS port (Covidien Ltd, Norwalk, CT, USA) was inserted. The armamentarium included a 5-mm straight 30° optic laparoscope, a 5-mm Harmonic ACE (Ethicon Endo-Surgery, Inc., Cincinnati, OH, USA), and a 5-mm curved grasper. The patient was placed in steep Trendelenburg position, and the operating table was rotated toward the right side. The sigmoid colon was suspended toward the abdominal wall with transparietal sutures (Fig. 2A). Mesocolic dissection and inferior mesenteric pedicle isolation were achieved with a medial or a lateral approach. The inferior mesenteric artery (IMA) was ligated close to its origin with clips or Endo GIA (Covidien Ltd, Norwalk, CT, USA). In cases undergoing abdominoperineal resection (APR) or HO, the superior rectal artery was divided just below the IMA after application of 5-mm clips (Endo Clip™ III 5 mm, Covidien Ltd). The splenic flexure was only mobilized in cases with a nonredundant sigmoid colon. The patient was repositioned to the Trendelenburg position, and the small bowel was reflected cranially. Movement of the rectum was achieved with transabdominal sutures, which are inserted through the abdominal wall, penetrate the mesenterium, make a loop around the rectum/mesorectum (half hitch), and are fixed with clips to both sides of mesorectum (Fig. 2B). Suture traction creates triangulation, enhances exposure, and improves margin clearance when performing deep pelvic dissection. The rectosigmoid colon was gradually retracted from the pelvis, retroperitoneum, and sacral promontory with a grasper and the transabdominal sutures. The posterior mesorectum was easily identified, and the mesorectal plane was dissected with harmonic scalpel, preserving the hypogastric nerves. Dissection was continued down to the pelvic floor. Further elevation of the upper rectum with the transabdominal sutures facilitates the posterior dissection. Anterior dissection between the rectum and the posterior vaginal wall (in women) or between the seminal vesicles and prostate (in men) was performed by decreasing tension of the transabdominal sutures and retracting the peritoneal reflection anterior to the rectum. Dissection proceeded laterally on both sides of the rectum until circumferential mobilization was accomplished. Digital examination was performed to verify the distance between the inferior margin of the tumor and the line of resection. The distal tumor margin was marked with a clip. A blue or green 45-mm Endo GIA roticulator stapler (Covidien Ltd) was fired twice to divide the rectum. A wound protector (Alexis O™, Applied Medical, Rancho Santo Margarita, CA, USA) was placed at the aperture of the SILS port. The specimen was extracted through the SILS aperture and resected. Extracorporal preparation of the proximal colon was completed with the placement of the anvil of a 29- to 31-mm circular stapler in position to perform a side-to-end or end-to-end colorectal anastomosis in case of low anterior resection (LAR). After pneumoperitoneum was reestablished, a circular stapler was inserted through the anus and a conventional intracorporal stapled colorectal anastomosis was made. The pelvic cavity was filled with water, and air was insufflated with a rigid rectoscope to test for anastomotic leakage. In cases needing a diverting stoma, the distal ileum 20 cm proximal to the ileocecal valve was extracted through the SILS aperture and a loop ileostomy was made. The umbilicus was used for both SP site and specimen extraction in four patients (Fig. 3). In 10 patients, who underwent LAR, the diverting ileostomy site was used both for SP placement and subsequent extraction. The left colostomy site was used for SP placement, extraction of the specimen, and formation of a sigmoidostomy in patients who underwent HO. Finally, patients who underwent APR had the single-port access (SPA) incision located at the left colostomy site, and specimen removal was accomplished through the perineal incision. A medial-to-lateral approach was used in most cases. In 11 patients (APR: n = 8; HO: n = 3), conventional lateral-to-medial dissection was preferred.
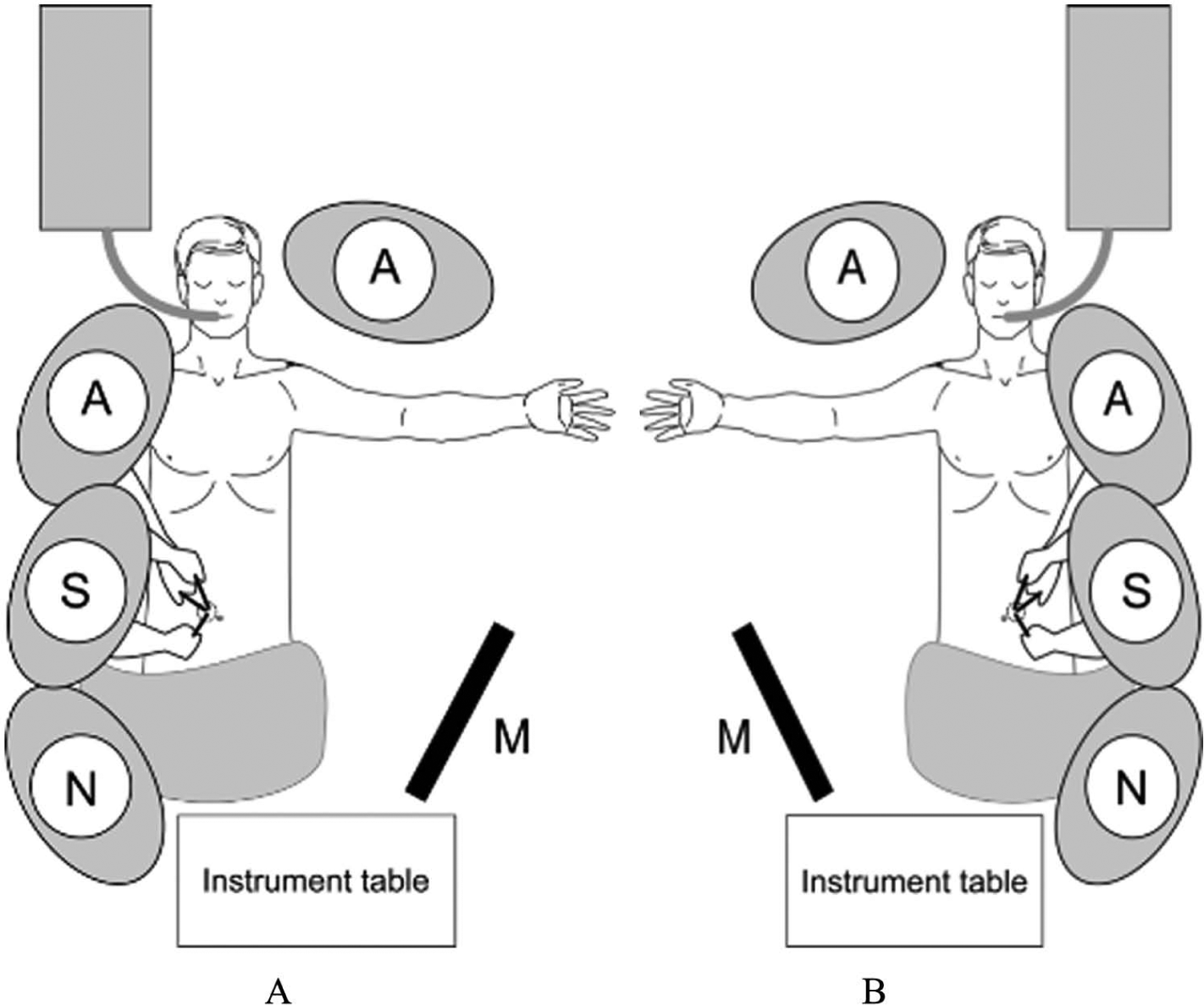
A) Patient positioning for low anterior resection. B) Operative setup for HO and abdominal phase of APR.
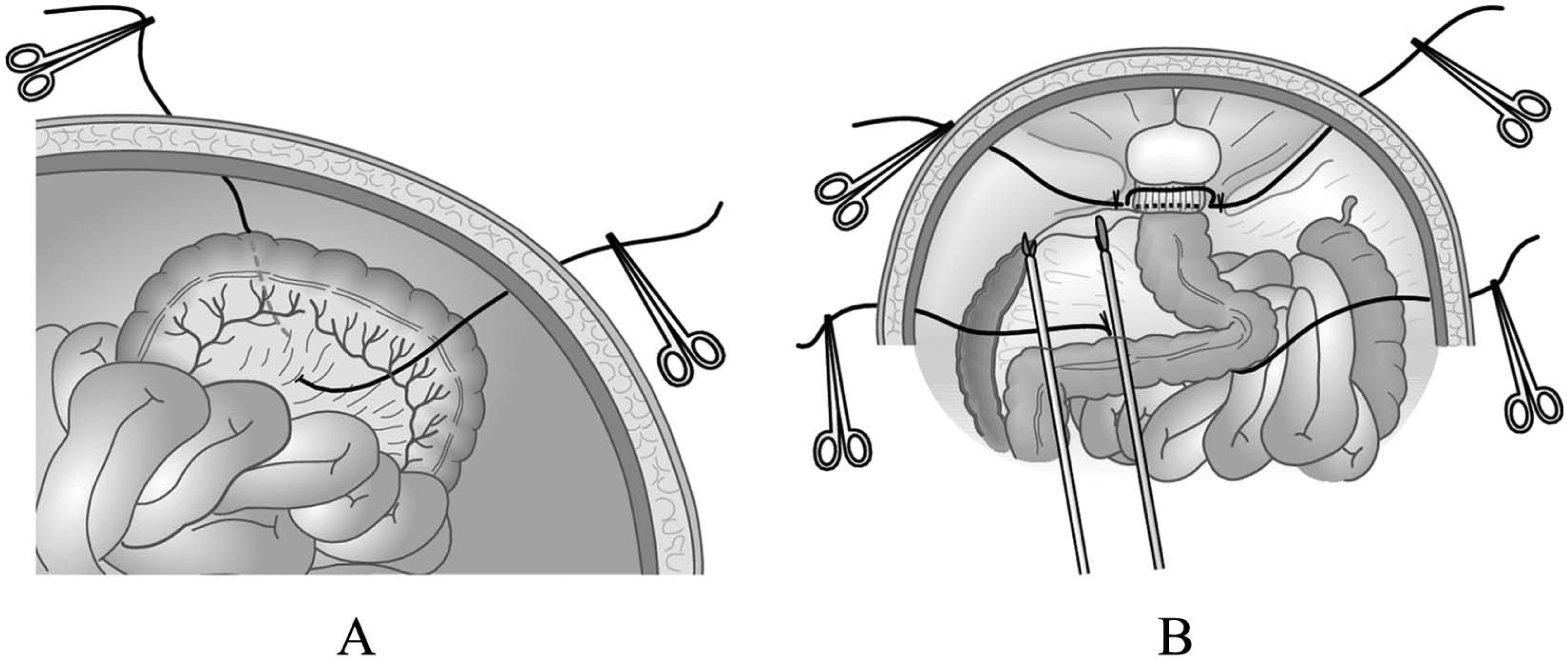
A) Illustration of the transparietal sutures of the sigmoid colon. B) Schematic intraoperative view of the transabdominal sutures suspending the rectum and sigmoid colon.
Appearance at 4 weeks following SPA low anterior resection with transumbilical access.
A protective loop ileostomy was done in accordance with national guidelines in all patients with an anastomosis less than 5 cm from the AV. Intestinal continuity was reestablished after 3 months or after completion of adjuvant therapy. In patients who had preoperative chemoradiotherapy (CRT), surgery was done 6–8 weeks after completion of treatment. A standardized perioperative care protocol was used. Placement of additional laparoscopic ports, use of HALS, or laparotomy was considered conversion to CLS.
Results
Between January 2010 and March 2012, 25 patients underwent SPLRS. Clinical and demographic data are shown in Table 1. Previous abdominal surgery, mostly appendectomy and hysterectomy, was reported in 44% (CI = 26.6, 63.1). Most patients were female. Median BMI was 24 kg/m2 (range: 19–32 kg/m2, interquartile range: 21.5–24.5 kg/m2). Most tumors were stage I/II and limited to the rectal wall. In all, four patients (16%; CI: 6.5, 34.9) had neoadjuvant CRT.
Patient characteristics and demography.
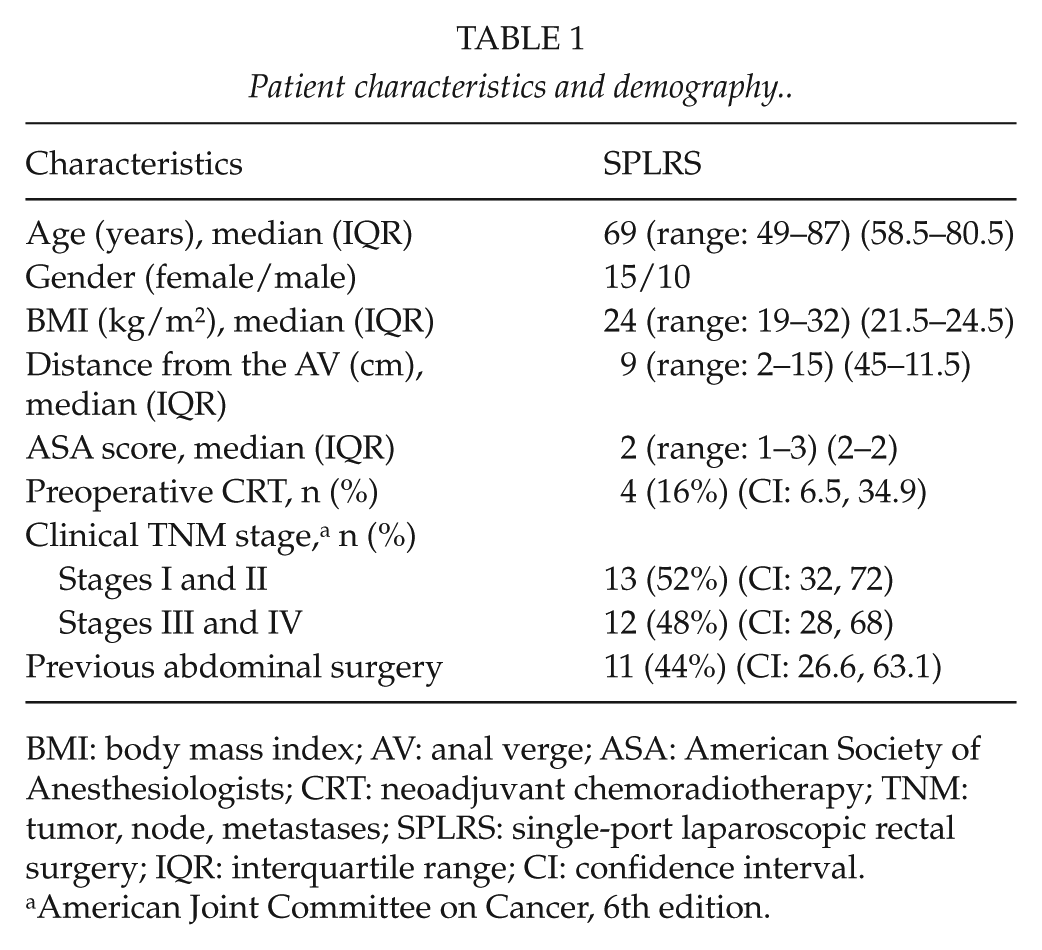
BMI: body mass index; AV: anal verge; ASA: American Society of Anesthesiologists; CRT: neoadjuvant chemoradiotherapy; TNM: tumor, node, metastases; SPLRS: single-port laparoscopic rectal surgery; IQR: interquartile range; CI: confidence interval.
American Joint Committee on Cancer, 6th edition.
Table 2 shows the operative procedures. The majority (88%) of the procedures were completed with a single incision. An additional laparoscopic 5-mm port was inserted in two patients for suturing an anterior anastomotic defect and for controlling pelvic bleeding. Conversion to HALS was done in one patient with a large bulky tumor (Table 2). Median duration of the procedure from skin incision to closure was 280 min, ranging from 136 to 397 min (interquartile range: 225–321 min). Median estimated blood loss (EBL) was 40 mL (range: 0–400 mL, interquartile range: 10–100 mL).
Procedural details and perioperative data.
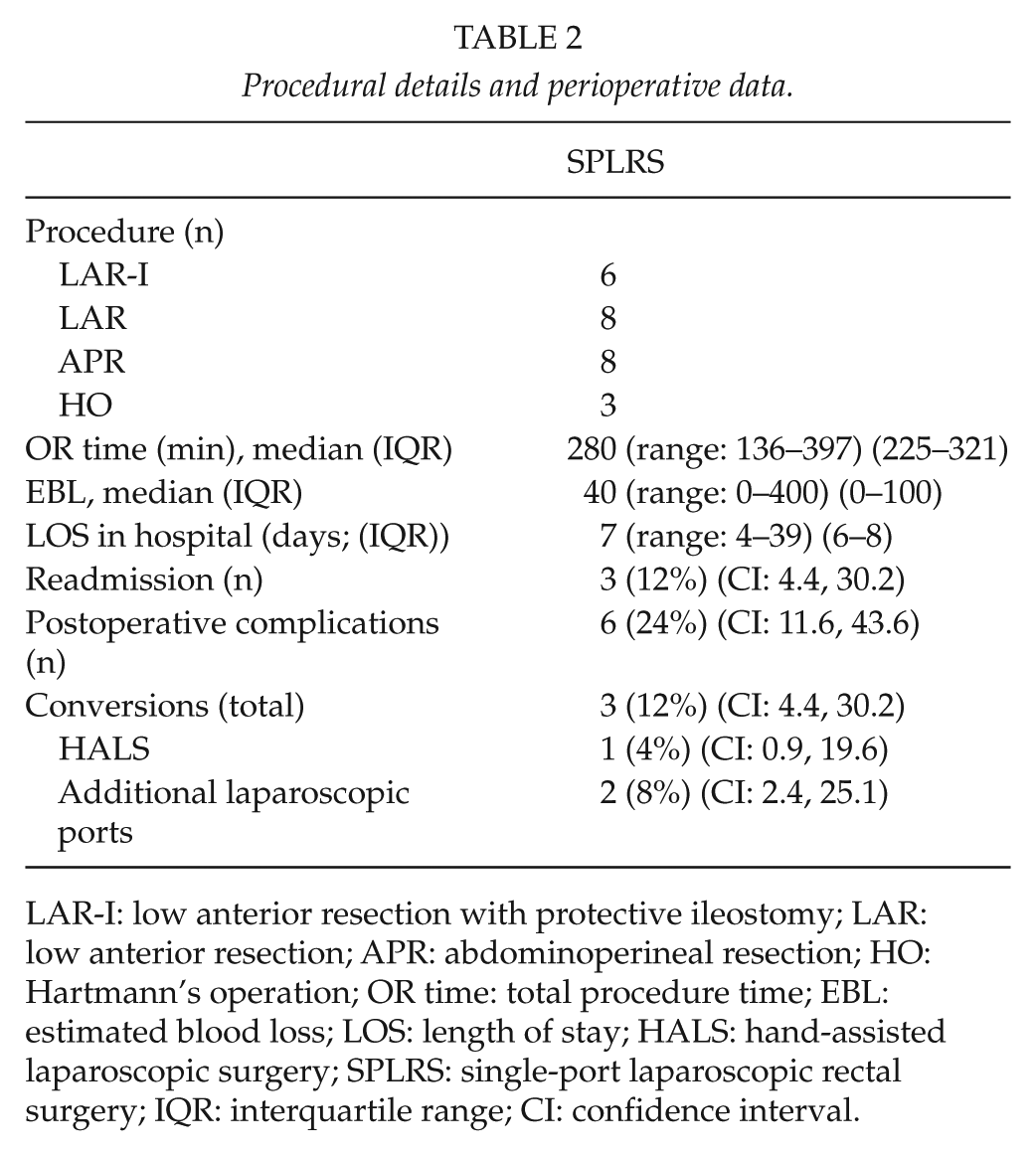
LAR-I: low anterior resection with protective ileostomy; LAR: low anterior resection; APR: abdominoperineal resection; HO: Hartmann’s operation; OR time: total procedure time; EBL: estimated blood loss; LOS: length of stay; HALS: hand-assisted laparoscopic surgery; SPLRS: single-port laparoscopic rectal surgery; IQR: interquartile range; CI: confidence interval.
A total of seven complications were seen in six patients (24%; CI: 11.6, 43.6). One patient suffering from lower limb arterial insufficiency developed compartment syndrome and underwent fasciotomy on the first postoperative day. Two patients with urinary tract infection were treated with antibiotics. One anastomotic leakage required reoperation. Median LOS was 7 days (range: 4–39 days, interquartile range: 6–8 days), that is, patients with a stoma stayed in hospital for median of 7 days (range: 4–7 days), whereas patients without a stoma were discharged on median of 4 days (range: 4–29 days). Three patients were readmitted (12%; CI: 4.4, 30.2). One patient had electrolyte and fluid derangement. Two patients developed an aseptic pelvic fluid collection, and one patient had leakage from rectal stump requiring ultrasound-guided drainage. The median follow-up period was 22 months (range: 6–31 months, interquartile range: 10–25.5 months). One patient aged 86 years, with ASA score of 3, and with known heart failure died 39 days after surgery from progressive heart failure and pulmonary edema. One patient with solitary lung metastasis was referred for further surgery. All the remaining patients are alive and disease free to date. No late wound complications have been reported. No patients had surgical site infections or incisional hernias within the follow-up period.
Histopathological and oncological results are shown in Table 3. Median number of harvested lymph nodes was 13 (range: 3–33, interquartile range: 11.5–16), median specimen length was 17 cm varying from 10 to 35 cm (interquartile range: 14–25.5 cm), and median tumor size was 32 mm (range: 10–82, interquartile range: 19.2–48 mm). Surgical margins were clear in all patients. Macroscopic quality assessment of the total mesorectal excision (TME) specimens was done routinely. Mesorectum was deemed complete or nearly complete in 92% of the patients.
Pathological details and follow-up.
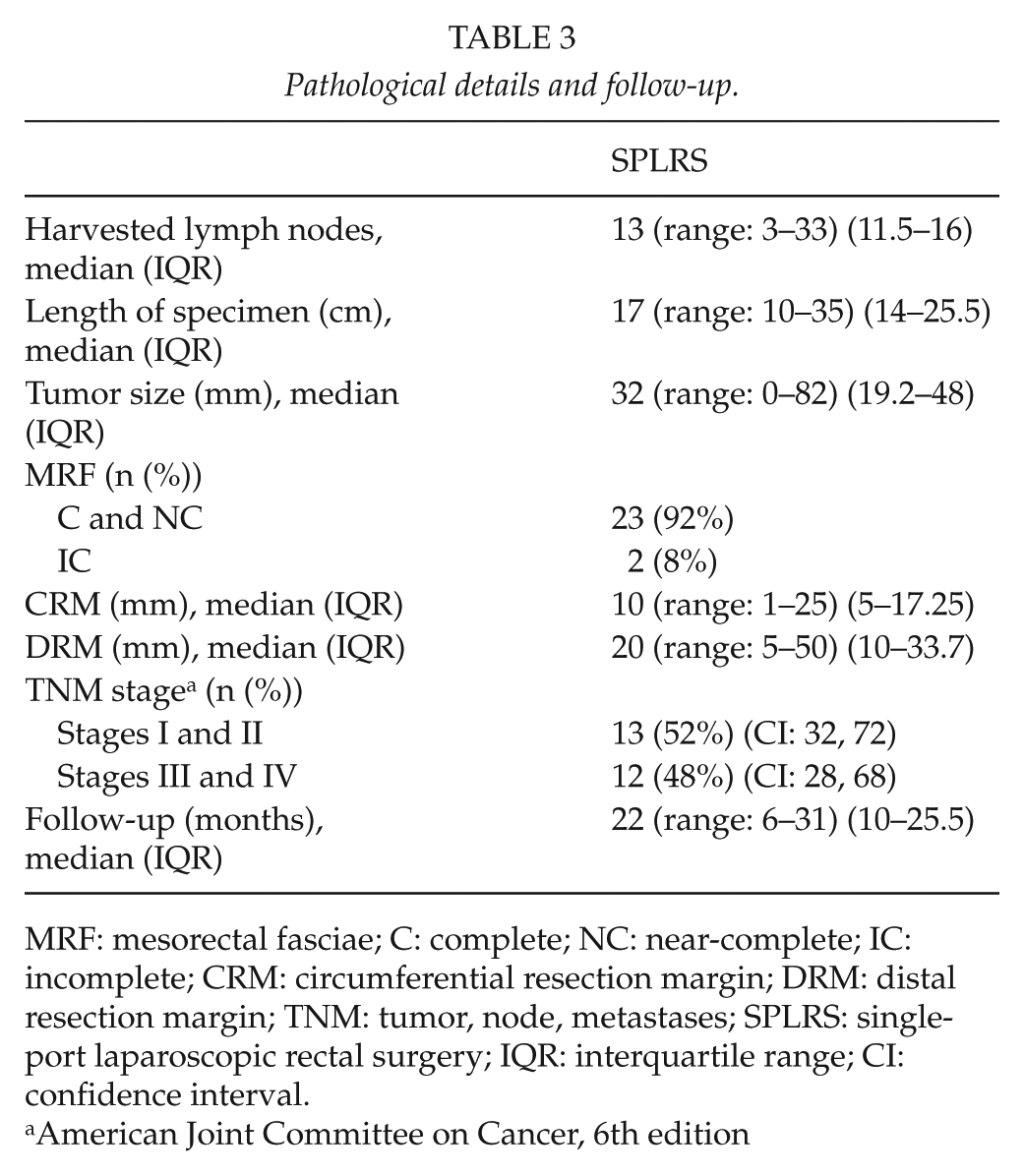
MRF: mesorectal fasciae; C: complete; NC: near-complete; IC: incomplete; CRM: circumferential resection margin; DRM: distal resection margin; TNM: tumor, node, metastases; SPLRS: single-port laparoscopic rectal surgery; IQR: interquartile range; CI: confidence interval.
American Joint Committee on Cancer, 6th edition
Discussion
This feasibility study focused solely on whether SP surgery can be performed safely in selected patients with rectal cancer. There were few serious complications and one anastomotic leakage. Blood loss, LOS, readmission rates, and postoperative morbidity were not different from CLS (14 –16).
Possible limitations of this study include a small and highly selected study population and a short follow-up. Patients were not randomized for SPLRS or CLS; postoperative pain, immunological activity, or quality of life was not assessed; and no attempts were made to determine whether SPLRS offers specific advantages compared with CLS. The procedure was performed by the same team of dedicated laparoscopic surgeons, which may limit the generalizability of the results to centers with less routine in laparoscopic procedures.
SPLRS takes more time than CLS. SP is a relatively new method, and extra time was needed to fine-tune the technique during the first cases. Technical difficulties include obtaining sufficient traction and countertraction through the single port, especially in patients with low rectal lesions and a narrow pelvis. Most available laparoscopic instruments are not produced for SPLRS, and there is a risk of hand and trocar struggle inside and outside the abdominal cavity. Taking down adhesions can also be a tedious procedure through the single port. These factors may all have contributed to the learning curve and prolonged operating time.
Previous studies on SP procedures in colorectal cancer have included heterogenous groups of patients with a mixture of both colonic and rectal lesions. Case series and nonrandomized comparisons between SPA technique and CLS for colon cancer have demonstrated the safety and feasibility of the procedure with similar outcomes as in CLS (17 –22). Only a few reports on SPA in rectal surgery have been published to date (10 –12, 23, 24).
The overall conversion rate was 12%. In two-thirds of the conversions, the placing of an additional 5-mm port was sufficient to finish the procedure. The conversion to HALS was required for mobilizing a distal rectal tumor with dense inflammatory perirectal reaction. These surgical difficulties are probably not related to the SPLRS procedure per se but apply to laparoscopic surgery in general. Although the initial length of the SPA incision was approximately 2.5 cm, further extension up to 5.5 cm was needed in eight patients to allow extraction of a bulky specimen. The median LOS and complication rates were similar to prior studies on conventional laparoscopic rectal surgery (14, 15, 25).
The median number of harvested lymph nodes exceeded the recommended number of 12 nodes and matched reports from other laparoscopic series and population-based studies (14, 26, 27). Macroscopic evaluation of mesorectal fasciae is considered an important marker for the quality of surgery and oncological safety. Laparoscopic surgery may cause more tears and breaks in the mesorectum than open surgery. SPLRS may potentially reduce the number of tears, as the colon is held in place with sutures. Nagtegaal et al. (28) showed that patients with an incomplete mesorectum have a significantly higher risk of local recurrence, whereas no difference was seen in local recurrence rates between patients with a complete and near-complete mesorectum. This series included 12 patients (48%) with stage III (11 patients) and stage IV (1 patient) cancers. In all, four patients (16%) had neoadjuvant CRT. It is generally agreed that neoadjuvant CRT is a good option for selected patients with a high risk of local recurrence, that is, patients with transmural tumor growth or node-positive tumors in the middle or lower part of the rectum. Nevertheless, the use of CRT for stage II and stage III rectal cancers varies considerably between countries. Most surgeons would only offer CRT that is in agreement with national or institutional guidelines.
National Danish guidelines (DCCG) for rectal cancer treatment recommend neoadjuvant CRT be offered only to patients with low rectal tumors (T3 or T4) and patients with mid-rectal tumors (T4 and advanced T3 (CRM < 5 mm) to avoid overtreatment of patients with early-stage disease. CRM measurement in this context seems to be more important than lymph node status. All patients in this study were treated according to these guidelines. Patients with rectal cancer are discussed at MDT meetings to allow tailored individual treatment for all patients. Consensus decisions to offer neoadjuvant CRT are made together with surgeons, oncologists, pathologists, and radiologists. Deviation from the standard treatment protocol is sometimes recommended for various reasons. In this study, three of four patients who had had neoadjuvant CRT were staged as stage I–II on their histopathologic examination probably due to downsizing of the tumor. Although Kapiteijn et al. (29) showed that a preoperative short course radiation in rectal cancer was associated with a lower 2-year local recurrence rate, neoadjuvant CRT to all patients with rectal cancer has not gained universal acceptance in the surgical community.
Better cosmetic outcome is seemingly one of the main benefits of the SP technique. Reducing the number of port sites may also lower the risk of accidental vascular or intestinal lesions resulting from trocar insertion. Furthermore, the risk of trocar site hernia, which amounts to 5%, may be diminished (30). The median follow-up of 22 months in this study, however, does not allow any definitive conclusions on cosmesis, local and port site recurrences, or late wound complications, for example, hernias.
It has been pointed out that the SP approach inevitably is a one-operating-surgeon technique, which may impose a negative impact on surgical education and training (20). This problem also applies for CLS. All 25 operations were performed by the same dedicated laparoscopic team. It is our experience that SPLRS requires substantial skills in two-handed laparoscopy. To optimize clinical outcome specialized training in advanced laparoscopy, for example, computer-based and clinical training is recommended before this technically demanding procedure is introduced in a general clinical setting. There are basic similarities between the SPA approach and transanal endoscopic microsurgery (TEM). Experience from TEM training courses may be useful for educating future colorectal surgeons in SPLRS.
As the possible advantages of SPLRS, for example, minimizing the surgical trauma and fewer wound-related problems, are probably discrete, large-scale comparative studies will be needed to demonstrate any significant differences. The surgical community should set up randomized multicenter studies to determine short- and long-term outcomes, quality-of-life differences, pain scores, and assessment of cosmetic outcomes. Robotic technology may also contribute to overcome the restrictions of limited space and instrument collision inherent to SPLRS. Eventually, patient preferences are more likely than physiological benefits to decide whether CLS or SPLRS will become the method of choice for the minimally invasive treatment of rectal cancer in the future.
Conclusion
In the largest series presented to date, SPLRS appeared to be safe and feasible in selected normal-weight patients with rectal tumors limited to the rectal wall. Adequate oncologic resections can be performed with acceptable morbidity and mortality by experienced colorectal surgeons. The procedure can be tedious and takes longer time than conventional laparoscopic rectal surgery.
Footnotes
Declaration of Conflicting Interests
The authors have no conflicts of interest or financial ties to disclose.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
