Abstract
Background and Aims:
Risk-reducing mastectomy of BRCA1 and BRCA2 gene mutation carriers is known to significantly reduce lifetime risk of breast cancer. Our aim was to study the frequency and outcome of risk-reducing mastectomies performed in Helsinki University Central Hospital during 1997–2010.
Material and Methods:
In testing for mutations in BRCA1 and BRCA2, 136 female carriers had been identified and followed up in Helsinki University Central Hospital.
Results:
A total of 69 breasts in 52 women were operated on for risk-reduction, including 28 (54%) bilateral mastectomies at mean age of 43 years. Autologous tissue was used for reconstruction in 40 (50%) and implants in 31 (39%) of the breasts, respectively. In all, 8 patients (15%) chose to have no reconstruction. Minor or major complications were recorded in 21 (40%) patients. Five reconstructions failed and were corrected with re-reconstruction.
Conclusions:
In this series of Finnish BRCA1 and BRCA2 mutation carriers, a high percentage 52 (41%) chose risk-reducing breast surgery. Autologous tissue was favored in breast reconstructions. Immediate breast reconstructions were associated with a relatively high risk of complications in free flaps and in implant reconstructions, but not in latissimus dorsi reconstructions. It is mandatory that patients are informed about the risks associated with risk-reducing operations.
Introduction
The cumulative lifetime risk of having breast cancer is 65% (44%−78%) in BRCA1 gene mutation carriers and 45% (31%−56%) in BRCA2 carriers. For ovarian cancer, the risks are 39% (18%−54%) and 11% (2.4%−19%) for BRCA1 and BRCA2 carriers, respectively (1). Risk management strategies in women with BRCA1 or BRCA2 mutation include cancer screening, chemoprevention, or risk-reducing surgery (2). Cancer screening by different modalities aims at early detection of breast cancer, when it still is curable by appropriate treatment. Present recommendations for surveillance include annual breast magnetic resonance imaging (MRI), mammography, and ultrasound in addition to clinical breast examination and breast self-examination. The combination of MRI, mammography, and ultrasound has a sensitivity of 95% versus 45% for mammography as the only modality (3).
In risk-reducing surgery, the goal is to prevent cancer prior to its existence. A meta-analysis suggests an 80% reduction in ovarian or fallopian tube cancer risk and a 50% reduction in breast cancer risk associated with risk-reducing salpingo-oophorectomy (RRSO) (4). Breast cancer risk-reduction in BRCA1 and BRCA2 mutation carriers after bilateral prophylactic mastectomy is estimated to be ≥90% (5, 6). The frequency of risk-reducing mastectomy (RRM), however, varies remarkably internationally. In the study by Metcalfe et al. (7), the rates for RRM were highest in North America (United States) and lowest in East Europe (Poland). At present, choices for risk-reducing operations on the breast include simple mastectomy (i.e. mastectomy without axillary lymph node dissection), skin-sparing mastectomy (SSM), and nipple-sparing mastectomy.
RRM is usually performed with immediate breast reconstruction (IBR). Reconstructive techniques can be classified into autologous and implant-based reconstruction as well as a combination of the two. A wide scale of differing complication rates has been reported in various studies (5, 8–10).
Genetic testing for mutations in BRCA1 and BRCA2 started at Helsinki University Central Hospital (HUCH) in 1997. Since then, mutation carriers have been offered surveillance and, alternatively, RRM and RRSO. Chemoprevention has not been used in Finland for carriers without a diagnosis of breast cancer. The aim of this study was to collect the data of all known BRCA1/2 gene mutation carriers in HUCH and to study the frequency and outcome of risk-reducing breast operations.
Material and Methods
In HUCH, approximately 700 at-risk individuals had been tested for BRCA1/2 mutations between 1997 and March 2010. In order to obtain complete identification of mutation carriers, we used three different sources of information. First, the mutation carriers were identified from the patient records of the Department of Clinical Genetics. Second, we identified all mutation-positive individuals from the two laboratories performing testing for BRCA genes in Helsinki. Third, we used the patient records of the Department of Plastic Surgery and the Breast Surgery Unit.
Using this strategy, 197 mutation carriers were found: 105 BRCA1 mutation carriers and 92 BRCA2 mutation carriers. Of these, 162 (84%) were women, and 136 of the 162 either had follow-up or were operated on in HUCH and were included in the study. The remaining 26 had follow-up and treatment in other hospital districts. We then studied the patient files of these 136 female BRCA1/2 carriers for possible risk-reducing operations during the study period January 1997 to March 2010. The data on surgical method of RRM, the method of reconstruction, and surgery-related complications were collected. All patient files were also examined for possible recurrences and new primary cancers.
Results
Cancer History of the Mutation Carriers
Of the 136 female BRCA1/2 mutation carriers, 86 (63%) had breast or ovarian cancer or both, while 49 patients (36%) were cancer free. One mutation carrier had metastatic adenocarcinoma of pancreas. Of those with diagnosed breast or ovarian cancer, 75 had breast cancer (16 bilateral), and 21 had ovarian cancer including 10 women who also had breast cancer. There were 61 (45%) female mutation carriers who had not had breast cancer. Breast or ovarian cancer was diagnosed before genetic testing in 34 women, 48 had the initiation of genetic testing at cancer diagnosis, and 4 had cancer diagnosis after being tested positive.
Risk-Reducing Surgery and Reconstruction
Ten mutation carriers had previously had bilateral mastectomy due to breast cancer and were excluded from the potential risk-reducing mastectomy group. RRM was performed on 52 (41%) of the 126 remaining mutation carriers. In all, 28 operations were bilateral and 24 unilateral. Of the 49 cancer-free carriers, 17 (35%) chose RRM. Of the 65 mutation carriers with a history of unilateral breast cancer, 35 (54%) chose contralateral RRM.
Preoperative imaging with mammography and ultrasound was performed on all patients undergoing RRM. One mutation carrier, who was scheduled for risk-reducing surgery, was diagnosed with breast cancer in the preoperative examinations and was thus treated as a breast cancer patient and not included in the risk-reducing surgery group in this study.
Altogether, 80 RRM were performed, comprising 57 with skin-sparing technique, 14 with nipple-sparing technique, and 9 simple mastectomies (Table 1). Mean age at surgery was 43 years (range: 27–64 years) (Fig. 1). Nine (11%) mastectomies in 8 women were performed without breast reconstruction, while 44 patients (85%) had their breasts reconstructed (Fig. 2). RRSO had been performed on 70 (51%) of the 136 carriers.
Risk-reducing mastectomies (RRM) of 80 breasts in 52 women in a cohort of 136 BRCA1/2 mutation carriers.

NAC: nipple–areola complex.
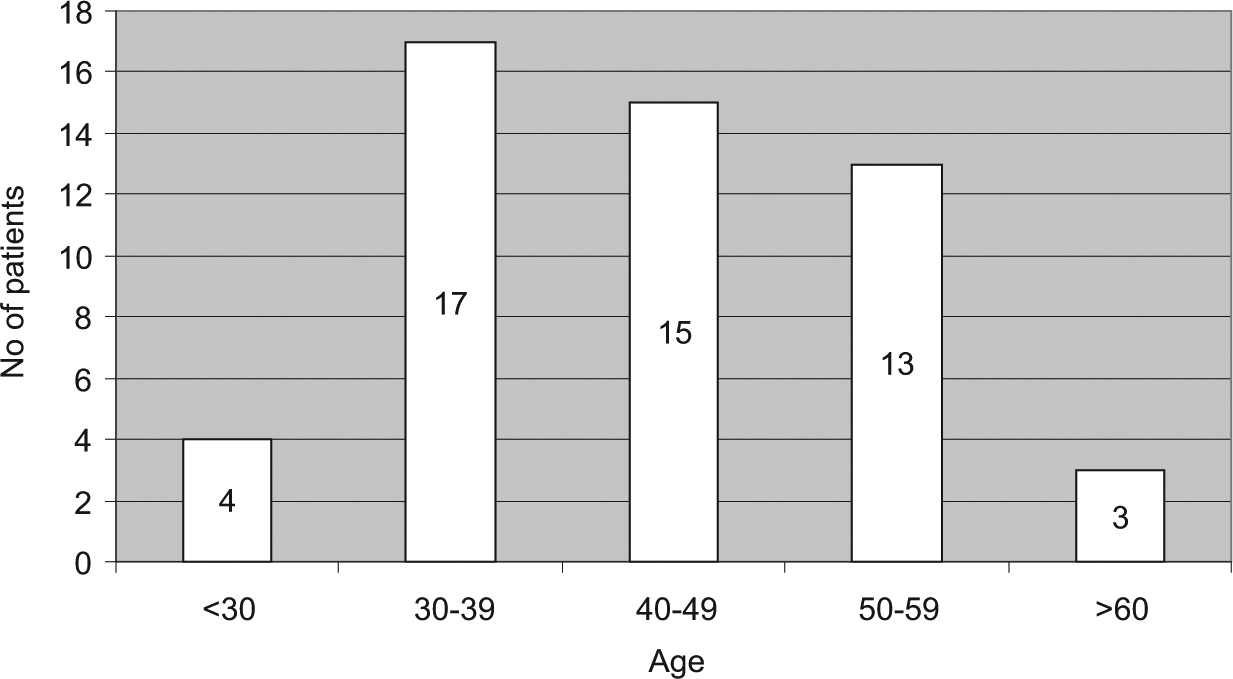
Age at RRM.
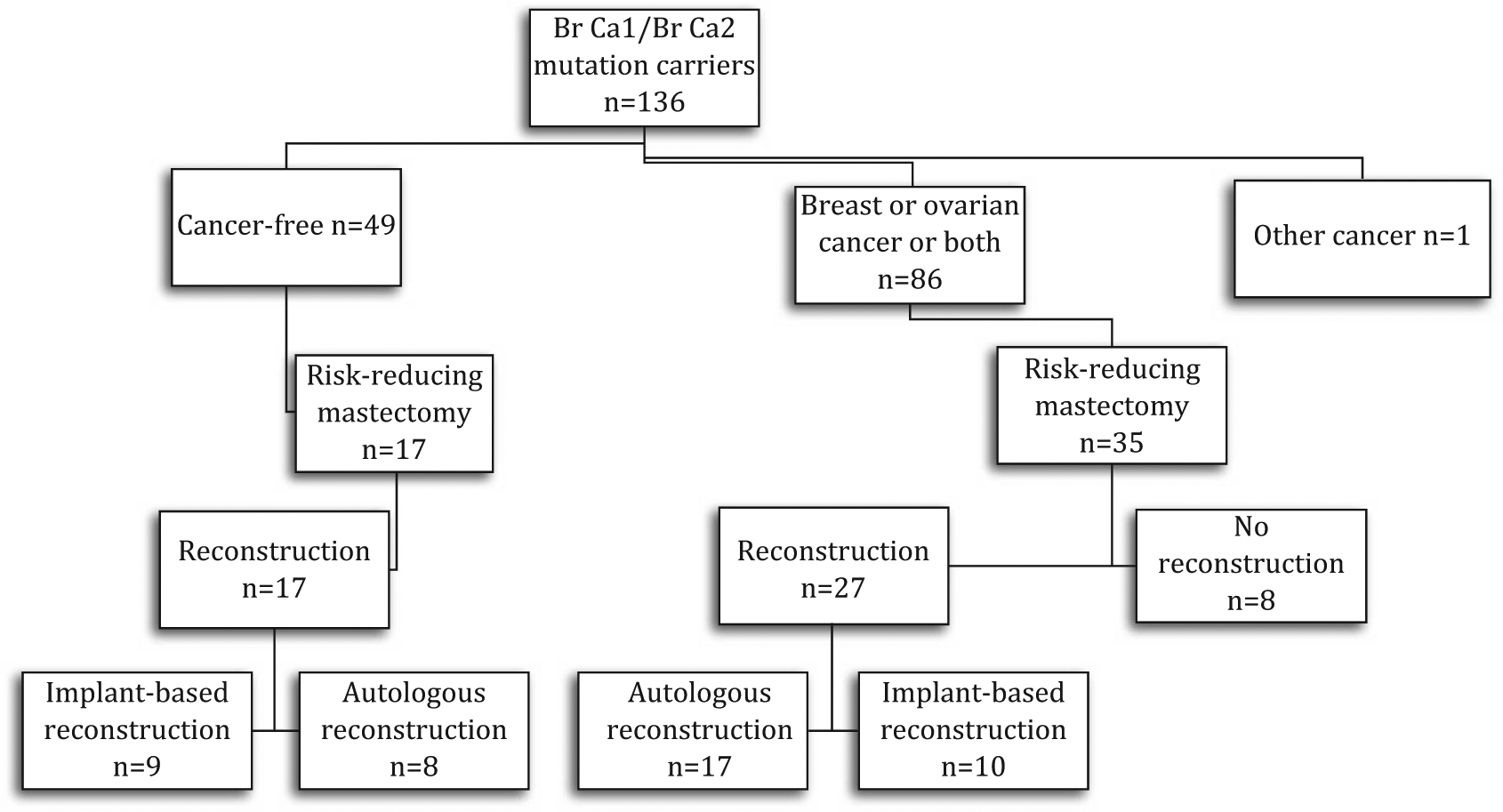
Choices of reconstruction for the 52 BRCA1/2 mutation carriers who had risk-reducing breast operations in Helsinki University Central Hospital during the period 1997–2010.
Ten patients had earlier undergone unilateral breast conserving surgery for cancer treatment and wished to have this breast removed for risk-reduction. In three patients, adjuvant radiotherapy was not yet given, and the RRM occurred shortly after primary cancer resection.
IBR was performed for the majority of the patients (85%) following RRM. An autologous flap reconstruction was the most frequent choice, in 40 breasts of 25 women (59% of all reconstructed breasts). Latissimus dorsi (LD) flap with or without an implant was used in 52% of the flap reconstructions (Fig. 3). Transverse rectus abdominis muscle (TRAM), deep inferior epigastric perforator (DIEP), superficial inferior epigastric artery (SIEA), and transverse musculocutaneous gracilis (TMG) flaps were also used for reconstruction. When implant-based reconstruction was chosen, this was usually done with a two-stage technique using a tissue expander initially with subsequent exchange for a permanent silicone implant in the second operation. Implant-based reconstruction was used in 44% of all the reconstructed breasts. The reconstruction methods are presented in Table 2.
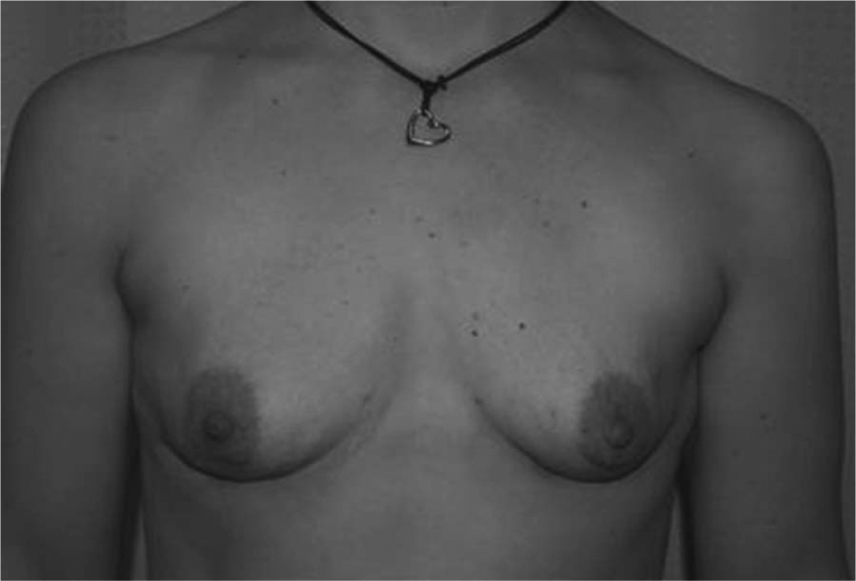
A 34-year-old woman with BRCA2 gene mutation viewed 6 months post bilateral nipple-sparing risk-reducing mastectomy and immediate latissimus dorsi reconstruction.
Reconstruction methods of the 80 breasts of 52 women undergoing RRM.
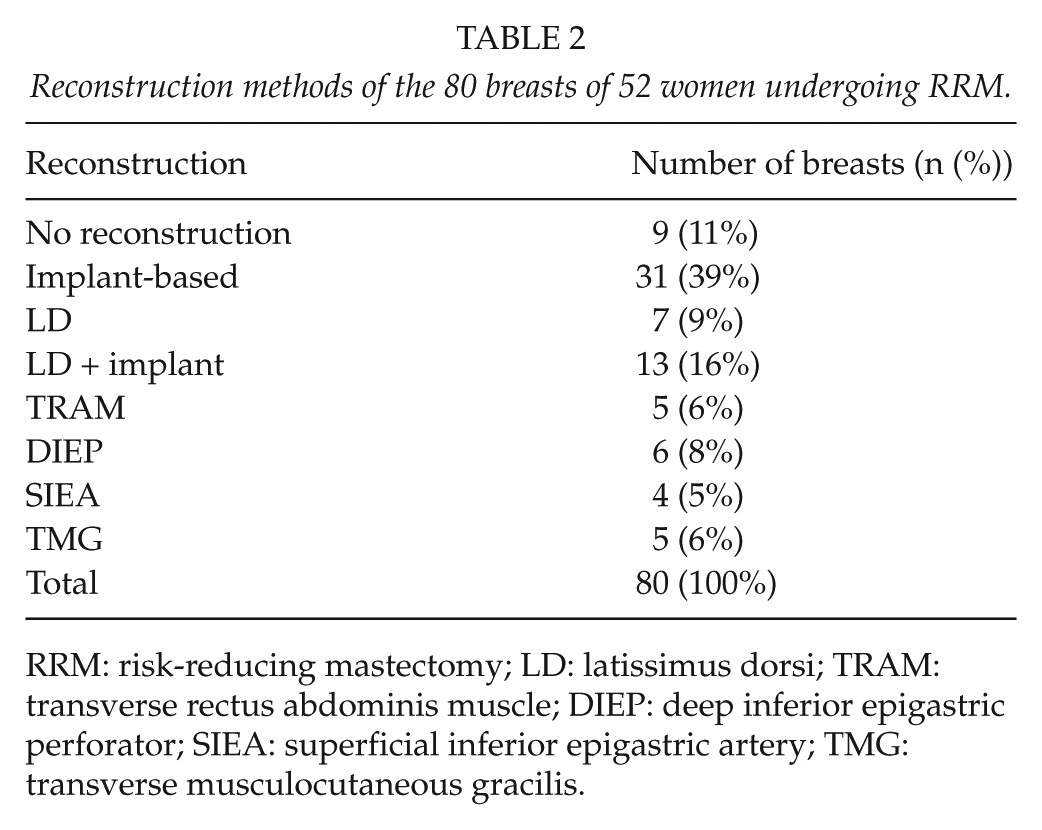
RRM: risk-reducing mastectomy; LD: latissimus dorsi; TRAM: transverse rectus abdominis muscle; DIEP: deep inferior epigastric perforator; SIEA: superficial inferior epigastric artery; TMG: transverse musculocutaneous gracilis.
Pathology
All removed breasts were examined histopathologically. The examination of mastectomy specimens from risk-reducing operations revealed atypical ductal hyperplasia (ADH) in seven breasts (9%) of five women (10%); ADH was bilateral in two cases. No preinvasive or invasive lesions were found. Sentinel lymph node biopsy was not used in this series of RRMs.
Complications
Altogether, 26 surgical complications in 21 patients necessitating 20 reoperations were recorded (Table 3). The frequency of complications was 33% (26/80) per operated breast and 40% (21/52) per patient.
Complications in RRM.
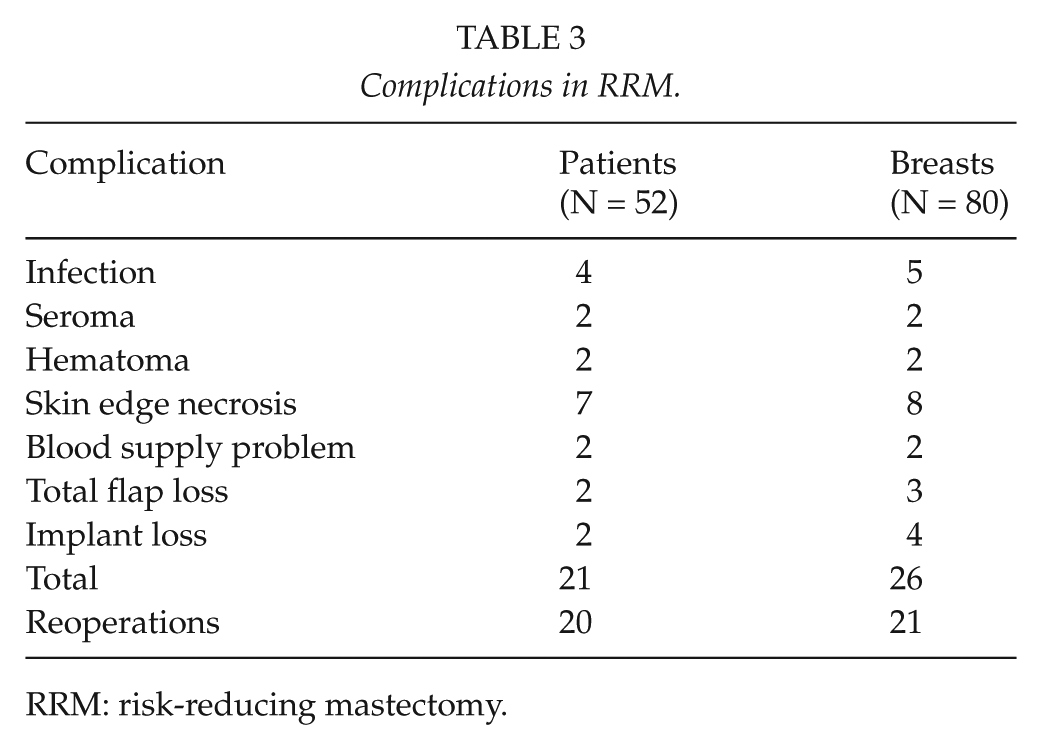
RRM: risk-reducing mastectomy.
Wound edge necrosis and wound infection were the most common complications in our patients. In two patients, a reoperation for hematoma was needed. In addition, two persistent seromas of the mastectomy site were recorded; one settled down in repeated aspirations, and the other needed an operation. The majority of procedures for complications were minor wound revisions.
In the flap reconstruction group, there were five cases of blood supply problem. Venous congestion was recorded in three flaps, which were re-operated. Two flaps were saved after reanastomosis. However, there were three flaps, which were eventually lost. Two were SIEA flaps and one was TMG flap, that is, all three were free microvascular flaps. These breasts were re-reconstructed afterward with other autologous flaps. The other complications in the flap reconstruction group were minor donor site problems, for example, seroma formations and one infection in the TMG donor site. In the group of reconstructions with autologous flaps, there were 11 (28%) complications in total.
In the implant-based reconstruction group, the most common complication was wound infection, which required a reoperation in most cases. Two major complications were due to infection of prosthesis on both sides and required a new bilateral reconstruction with autologous flaps, which succeeded expectedly. In other words, four implants were lost (13%). In the group of implant-based reconstructions, complications were recorded in 13 (42%) breasts.
In those 10 patients who had previously had their breast in question resected, there were altogether four cases (40%) of minor complications: one wound infection and three skin edge necroses. Three (30%) of the complications required revision including one skin grafting. Of these patients, two (20%) had previously had radiotherapy. No thromboembolic complications were detected.
Follow-Up
Median follow-up time after RRM was 52 months (range: 1–133 months). During the postoperative follow-up, one patient had cancer diagnosed with the removal of 24/24 metastatic axillary lymph nodes 45 months after risk-reducing surgery and 14 years after the treatment of contralateral breast cancer. Furthermore, one patient was operated on for ADH in the residual breast tissue under the nipple–areola complex (NAC) 7 years after a bilateral nipple-sparing risk-reducing mastectomy. She had also been diagnosed with ADH in her original risk-reducing mastectomy specimen.
Discussion
This series of 136 BRCA1/2 mutation carriers is the first study on RRM in Finland. In this study population, 75 women (55 %) had earlier had breast cancer, and in 16 women (21%), the breast cancer had been bilateral. Altogether, 41% of all female BRCA1/2 mutation carriers underwent RRM. In all, 35% of unaffected carriers chose RRM, and 54% of affected women underwent RRM. These are high figures compared to previous publications. In the Irish study by Carroll et al. (11), the frequency of RRM was remarkably lower, that is, 12.1%. In a questionnaire study of 1383 unaffected women from 9 countries, the overall rate of RRM was 18%, but with great variation from one country to another. In Poland, the rate of BRCA1/2 mutation patients who had chosen RRM was only 2.7%, whereas the rate in the United States was 36.3% (7). Gronwald et al. (12) analyzed 414 Polish BRCA1 carriers and noticed the same: only 5% underwent RRM. In a multicentre study by Friebel et al. (13), the overall rate of bilateral RRM was 21%, while the rate was 20% in the United States and 31% in the United Kingdom. In an older series by Meijers-Heijboer et al. (14), 51% of unaffected mutation carriers opted for bilateral RRM. In their study, there was a tendency toward mastectomy in younger women, and having children was a predictor of RRM (14). In addition, a history of previous cancers inside the family has been shown to raise the frequency of RRM (7, 15).
For mutation carriers with treated breast cancer, Metcalfe et al. (16) reported higher figures of contralateral RRM internationally (27.3%), but the variation between countries was remarkable. In their study, opposite to our results, the proportion of European affected women undertaking RRM was only 5% (16), but the number of European women in the study was low. In an Australian study by Kiely et al. (17), 15% of 1239 women with prior breast cancer opted for contralateral RRM.
In our series, the majority of patients (67%) had a unilateral breast cancer diagnosis before the decision of risk-reducing surgery was made. These breast cancers had mostly been detected before genetic testing was available. Our RRM rate is high compared to other published series. Although the cohort is small, it covers all mutation-positive women in Finland’s largest population-based breast cancer clinic. The possibility for IBR may increase the number of women opting for RRM. Availability of chemoprevention may also influence the rates of risk-reducing operations; in Finland, it is not presently used (18).
Of the 136 women, 70 (51%) underwent RRSO, including 33 women who also had RRM. In the study of Metcalfe et al. (7), 57.2% of all mutation carriers chose RRSO. In the study by Friebel et al. (13), the proportion opting for RRSO was higher than RRM, 51% and 21%, respectively. Meijers-Heijboer et al. (6) have reported 64% of affected women expressing interest in RRSO. RRSO is a safe operation with not only a remarkable reduction in the risk of ovarian cancer, but also a reasonable reduction in the risk of breast cancer. Its benefit is that RRSO does not have such an effect on body image as RRM (at least when done without immediate reconstruction). The disadvantage of RRSO is a premature menopause and iatrogenic fertility, and therefore, it is recommended not to be performed earlier than the age of close to 40 years (4).
Young mutation carriers benefit most from risk-reducing surgery (19). In older age groups, the breasts are generally easier to follow up, and the patients are less likely to choose surgical risk-reduction (20). In our series, the average age at RRM was 43 years, which is in concordance with previously published data. Average age ranges from 36 to 42 years at RRM and from 40 to 47.3 years at RRSO (2, 5, 7, 13). In the first few years after genetic testing became available in our unit, the majority of patients were older women with previous breast cancer history. In the past few years, the average age has slowly been going down. The ratio of healthy mutation carriers is expected to rise as increasing numbers of relatives of cancer patients are being tested.
In our unit, SSM was commonly used for RRM. Generally SSM has been shown to be an oncologically safe procedure and is comparable to simple mastectomy, but minor wound edge necrosis is common (21, 22). In nipple-sparing mastectomy, there remains a small risk of cancer arising beneath the nipple and areola if breast tissue is left behind. However, a review article on nipple-sparing mastectomy suggests that it is an acceptable technique for women undergoing RRM (23). Metcalfe et al. (24) suggest that subcutaneous mastectomy (nipple-sparing mastectomy) should be considered in risk-reducing surgery. They estimated the lifetime risk for breast cancer to be 4% for mutation carriers after subcutaneous mastectomy (24). In the current series, there were only a few nipple-sparing mastectomies. Currently, nipple-sparing mastectomy is increasingly being used for risk-reduction in many centers and also in our unit.
Preoperative MRI is currently used to exclude occult malignancy before RRM. In our patient series, MRI was not yet used for all early patients. Preoperative mammography and ultrasound detected one breast cancer. No preinvasive or invasive lesions were found in the histopathological examinations of mastectomy specimens. However, ADH, which is considered to be a premalignant lesion, was found in 10% of our patients. In a recent Swedish study of Arver et al. (25), premalignant or malignant lesions were found in 10% of patients, which coincides with the results of our study; in addition, there were also 2% of invasive cancers in their series.
In the study of Heemskerk-Gerritsen et al. (5), the proportion of invasive occult cancers was 3%. Goldflam et al. (9) reported occult premalignant or malignant findings in 23% of the cancers. Of these, 18.4% were moderate- to high-risk lesions such as ADH. Their preoperative management included recent mammogram but not MRI.
In the literature, the benefits and disadvantages of sentinel node biopsy have been debated. Nasser et al. (26) reported a positive sentinel lymph node in 2% of women who underwent a risk-reducing mastectomy. Sentinel node biopsy is associated with some morbidity, such as possible arm swelling, pain, numbness, and restriction of arm movement, which argues against it in risk-reducing operations (26). The one case in our series with massive axillary nodal metastasis during follow-up would most probably have been diagnosed by sentinel node biopsy at the time of risk-reducing mastectomy. The cancer was not, however, found in the breast specimen, and it is possible that the metastatic disease in the axilla had arisen from the contralateral breast cancer that had been treated 14 years earlier.
An immediate reconstruction following risk-reducing surgery is almost always feasible because there is no need for oncological treatments. In our series, 85% of the operated mutation carriers chose to have IBR. In the study by Heemskerk-Gerritsen et al. (5), 94% of unaffected women opted for breast reconstruction. If a woman had had an earlier breast cancer, only 61% opted for a breast reconstruction. In another study, only 49% underwent breast reconstruction after contralateral RRM. The patients who underwent reconstruction were, on average, 6 years younger than those not having reconstruction (17). In the study of Arver et al. (25), there was a high proportion of reconstructions (99%), but mainly implant-based (93%). In contrast, in the study of Goldflam et al. (9), from the United States, the percentage of flap reconstructions was shown to be high (65%). In our study, the proportion of women choosing an autologous tissue reconstruction was 56%, which is higher than in many previously published reports. A flap reconstruction creates more natural look and touch than implant.
The complication rates vary considerably, 8%−64%, in previously published reports. This may depend on patient selection for RRM or the definition of complications. In some studies, only complications that have led to reoperation have been reported (5, 8, 27–29). In the study by Heemskerk-Gerritsen et al. (5), the complication rate was 49.6%. This is close to our complication rate. Proportion of women with complication has been shown to be lower in groups of no reconstruction, but even then there are complications (8–10). No difference was noted in reoperations between bilateral and unilateral RRM in the studies of Heemskerk-Gerritsen et al. (5) and Zion et al. (10). In our study, 40% of the operated patients had some complications, and most of these required a reoperation. The complication rate calculated per operated breast was 33%. The complication rate was the highest in patients having implant reconstructions, but most of these complications were minor. Most of the reconstructions were carried out with an implant or an LD flap with prosthesis. LD flap turned out to be the safest reconstruction technique in our series. LD flap was introduced a long time ago, and reconstruction with LD flap is known as a straightforward technique to carry out.
Total flap failure is a severe but possible complication. There is a risk of flap loss in microsurgery and a risk of infection with implants. Altogether, four implants (in two patients) and three autologous flaps were lost in our series. All flap losses occurred with microvascular flaps. New operative techniques have their learning curve, and SIEA and TMG flaps were introduced to practice during the study period. Computed tomography (CT) angiography was not yet used to evaluate the anatomy preoperatively. The cases with complications deserve careful examination to reduce the high number of complications and to improve the quality of risk-reducing surgery.
There are several studies that have shown the efficacy of risk-reducing mastectomy to be 90% or more (5, 6). During the median follow-up time of 52 months, no other breast cancer cases were detected in our series, except the discussed case with axillary metastases.
In this series of Finnish BRCA1 and BRCA2 mutation carriers, a high percentage (41%) had risk-reducing breast surgery, which is known to be effective in reducing the risk of breast cancer. It is also known to reduce anxiety and the fear of dying of breast cancer (30). The selection of optimal reconstructive techniques for each patient includes both implant and autologous tissue transfer techniques. These operations are prone to complications, which should be carefully explained to the patient beforehand.
Footnotes
Funding
This study was supported by the grant from Jyväskylä Central Hospital research funds.
