Abstract
Introduction
Breast cancer requires coordinated care from multiple specialists which can lead to delays in diagnosis and treatment.1–3 Treatment delays have both psychological and physical repercussions for patients such as elevated levels of anxiety and depression, decreased quality of life, and increased mortality.4–9 Given this, it is critical to understand the cause of these delays. In general, reasons for delay can be broken into system, provider, and patient factors.3,4,10 Our study further explores system factors in the form of treatment order and reconstructive modalities.
A plastic surgeon is essential to comprehensive breast cancer care given the recognized benefits of breast reconstruction which has been shown to improve patients’ quality of life and have positive effects on their psychological well-being.11–17 Whereas mastectomy alone reduces quality of life and has a negative impact on body image. 18 The timing and type of breast reconstruction surgery can vary based on patient factors, surgeon preferences, and adjunct treatment sequencing.12,19 Therefore, it is imperative the surgeon has a good understanding of reconstructive and microsurgical techniques to provide the patient with the best possible outcome.10,13,16,17 However, this additional surgical intervention can increase complications and recovery time thereby impeding subsequent therapies.20–23
Currently, for patients undergoing immediate breast reconstruction (IBR), it remains unknown whether treatment order and/or reconstructive modality have an impact on wait times to downstream adjuvant therapies. To evaluate this, wait times between care pathway milestones were compiled and compared to national standards. 24
Methods
We conducted a retrospective chart review to identify patients undergoing IBR in conjunction with total mastectomy for therapeutic breast cancer treatment. The patients included in the study were 18 years of age or older and underwent alloplastic or autologous IBR between May 31, 2010, and April 4, 2021, within two regional jurisdictions, Vancouver Coastal Health or Providence Health Care. The study excluded patients with missing data on diagnostic workups, consultations, or operative reports, and if parts of their treatment were completed elsewhere.
The list of eligible patients was identified through the medical records of breast reconstructive surgeons affiliated with the University of British Columbia Breast Reconstruction program. Patients’ electronic medical records were used to extract demographic and clinical data. Patient characteristics, including age, body mass index (BMI), Charlson comorbidity index (CCI), and smoking status were collected. Type of breast/axillary resection and types of alloplastic or autologous reconstruction were recorded. Alloplastic reconstruction included tissue expanders/acellular dermal matrix and direct-to-implant reconstruction. Autologous reconstruction included deep inferior epigastric perforator, superficial inferior epigastric artery, transverse rectus abdominis, and latissimus dorsi flaps. Data for balancing procedures (ie, mastopexy, mammoplasty, and fat grafting) performed on the contralateral side for unilateral procedures was also collected. Indications for unplanned operations were recorded, such as hematoma, seroma, flap vascular compromise, revision of MFN, removal of a tissue expander or implant due to exposure/infection, and oncologic re-excision for positive margins. To examine time delays for key diagnostic and therapeutic procedures, wait times between care pathway milestones were collected and compared for SF and NC cohorts. The pretreatment phase included the final diagnostic biopsy, referral to plastic surgery, first consultation (from an oncologic service such as medical oncology, surgical oncology, or radiation oncology), plastic surgery consultation, and the start of the first treatment modality. The treatment phase included transitions between treatment modalities (Table 1).
Summary of the 2019 Pan Canadian Breast Cancer Surgery Standards 24
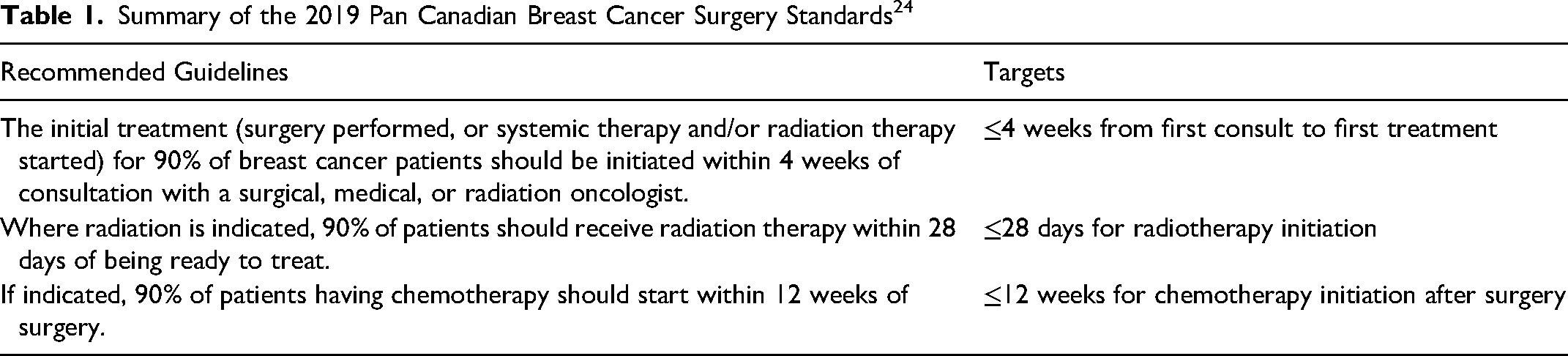
To analyze the effects of IBR on treatment delays, patients were classified based on their treatment sequence—surgery first (SF): undergoing surgical resection and IBR as first treatment; or neoadjuvant chemotherapy (NC): undergoing systemic chemotherapy as the first treatment. Subgroup analysis based on alloplastic or autologous IBR was conducted to examine the effects of the reconstructive modality on wait times to downstream ablative therapies. Wait times to a third treatment modality, namely postmastectomy radiation therapy, was also evaluated when relevant.
Chi-square (or Fisher Exact when n < 5) and Mann-Whitney U tests were used to compare proportions and median waits (days) to standards. All tests were two-tailed and statistical significance was defined as P<0.05. Statistical analyses were performed with IBM SPSS Statistics, Version 29.0 (Armonk, NewYork: IBM Corp).
Results
Study Population
A total of 459 eligible patients were identified; 22 patients were excluded as they received prophylactic surgery only, 55 patients were excluded as they had oncoplastic or combined alloplastic and autologous reconstructive methods, and 45 patients were excluded due to missing data. Of the 337 included patients, 249 (74%) were classified as SF and 88 (26%) as NC. Within SF, there were 173 (69%) patients classified as SF alloplastic and 76 (31%) as SF autologous. Within NC, there were 60 (68%) patients classified as NC alloplastic and 28 (32%) as NC autologous. Study population and disease characteristics are defined in Table 2.
Study Population Characteristics
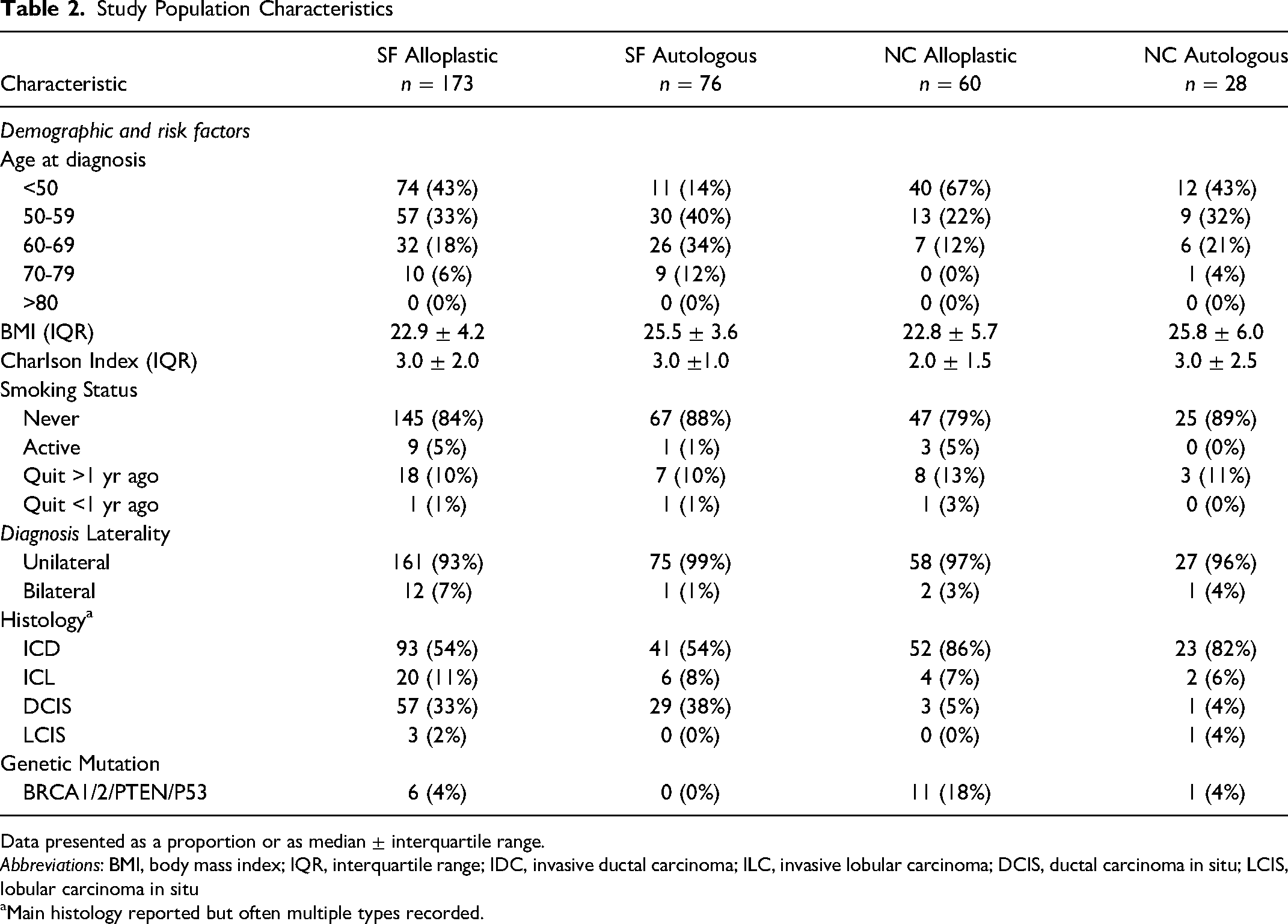
Data presented as a proportion or as median ± interquartile range.
Abbreviations: BMI, body mass index; IQR, interquartile range; IDC, invasive ductal carcinoma; ILC, invasive lobular carcinoma; DCIS, ductal carcinoma in situ; LCIS, lobular carcinoma in situ
Main histology reported but often multiple types recorded.
Care Pathway Comparisons
In the pretreatment phase, SF patients were seen 11 days sooner for plastic surgery consultation after referral from a medical or surgical oncologist (13 ± 14 days vs 24 ± 22, P<.001 days) compared to NC patients. There was no difference whether it was a SF alloplastic or SF autologous patients (12 ± 15 days vs 14 ± 14 days, P=.348). However, this additional consultation led to SF patients waiting 25 days longer between first consult and treatment initiation (47 ± 52 days vs 22 ± 22 days, P<.001). When comparing reconstructive modalities, SF alloplastic patients received their initial treatment 10 days sooner than SF autologous patients (43 ± 52 days vs 53 ± 59 days, P=.030). There were no differences between any pretreatment phase wait times for NC alloplastic versus NC autologous patients.
In the treatment phase, SF patients progressed from first to second treatment modality 23 days slower compared to NC patients (62 ± 35 days vs 39 ± 17 days, P<.001). However, they advanced 13 days faster than NC patients from second to third treatment modality (38 ± 20 days vs 50 ± 31 days, P<.001). Within reconstructive modalities, there were no significant differences seen between treatments for either SF alloplastic versus SF autologous (first to second treatment, 63 ± 41 days vs 58 ± 23 days, P=.236; second to third treatment, 36 ± 17 days vs 40 ± 24 days, P=1.00) or NC alloplastic versus NC autologous (first to second treatment, 40 ± 17 days vs 39 ± 23 days, P=.844; second to third treatment, 57 ± 39 days vs 47 ± 32 days, P=.100). A summary of pretreatment and treatment phase wait time comparisons are displayed in Table 3 with subgroup comparisons in Table 4.
Comparison of Wait Times Between Milestones in Breast Cancer Care Pathway for Surgery First as Compared to Neoadjuvant Chemotherapy
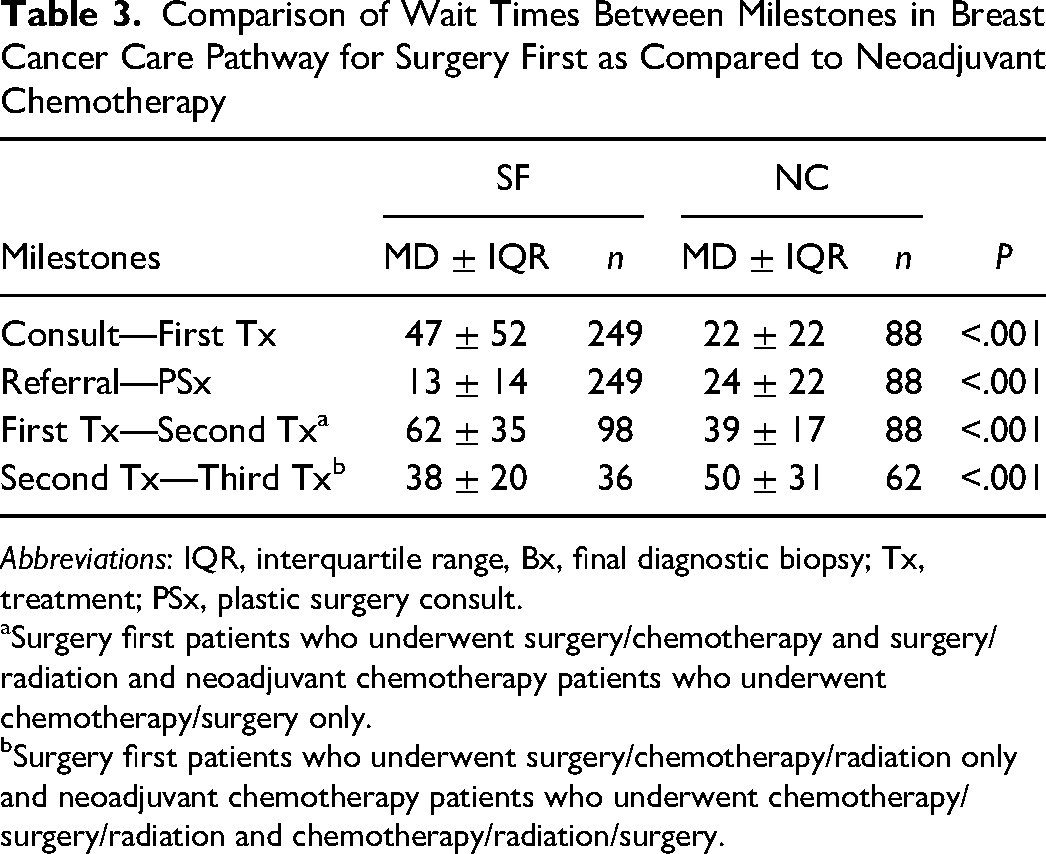
Abbreviations: IQR, interquartile range, Bx, final diagnostic biopsy; Tx, treatment; PSx, plastic surgery consult.
Surgery first patients who underwent surgery/chemotherapy and surgery/radiation and neoadjuvant chemotherapy patients who underwent chemotherapy/surgery only.
Surgery first patients who underwent surgery/chemotherapy/radiation only and neoadjuvant chemotherapy patients who underwent chemotherapy/surgery/radiation and chemotherapy/radiation/surgery.
Comparison of Wait Times Between Milestones in Breast Cancer Care Pathway for Reconstructive Subgroups

Abbreviations: MD, median days; IQR, interquartile range, Bx, final diagnostic biopsy; Tx, treatment; PSx, plastic surgery consult.
Surgery first alloplastic and autologous patients who underwent surgery/chemotherapy and surgery/radiation. Neoadjuvant chemotherapy alloplastic and autologous patients who underwent chemotherapy/surgery only.
Surgery first alloplastic and autologous patients who underwent surgery/chemotherapy/radiation only. Neoadjuvant chemotherapy alloplastic and autologous patients who underwent chemotherapy/surgery/radiation and chemotherapy/radiation/surgery.
Comparison to Standards
Initiation of Treatment. SF patients experienced greater delays than NC patients and were less likely to achieve the targets set by the 2019 Pan Canadian Breast Cancer Surgery Standards. 24 For first consultation to treatment initiation, SF patients had a lower success rate in meeting this standard compared to NC patients (29% vs 63%, P<.001). Within reconstructive modalities, both SF alloplastic patients and NC alloplastic patients had better rates of treatment initiation within 4 weeks of consultation relative to their autologous counterparts (31% vs 24%, P=.227; 68% vs 50%, P=.098, respectively).
Initiation of Radiation. The 2019 Pan Canadian Breast Cancer Surgery Standards recommends patients receive postoperative radiation no more than 28 days after being deemed “ready to treat” (RTT) after surgery or chemotherapy. 24 RTT is decided by a radiation oncologist and is defined as the date on which all pretreatment testing/interventions are complete, any planned delays are over, and the patient is ready to begin treatment from both a social/personal and medical perspective. Overall, SF patients had a lower success rate in achieving this standard compared to NC patients (39% vs 56%, P=.048). Within reconstructive modalities, both SF alloplastic and NC alloplastic patients were less likely to meet this standard compared to their autologous counterparts (49% vs 69%, P=.193; 51% vs 67%, P=.195, respectively). However, across all groups there was a difference seen between patients transitioning from surgery versus chemotherapy with those being deemed RTT after chemotherapy being more likely to meet this standard.
Initiation of Chemotherapy. The 2019 Pan Canadian Breast Cancer Surgery Standards recommends that 90% of patients having chemotherapy should start within 12 weeks of surgery when indicated. 24 This was achieved by our group with 90% of SF patients meeting this standard. Within reconstructive modalities, 90% of SF alloplastic patients and 95% of SF autologous patients were successful in meeting this standard. A summary of standard comparisons is displayed in Table 5 with subgroup comparisons in Table 6.
Proportion of Patients That Meet National Standards by Treatment Sequence
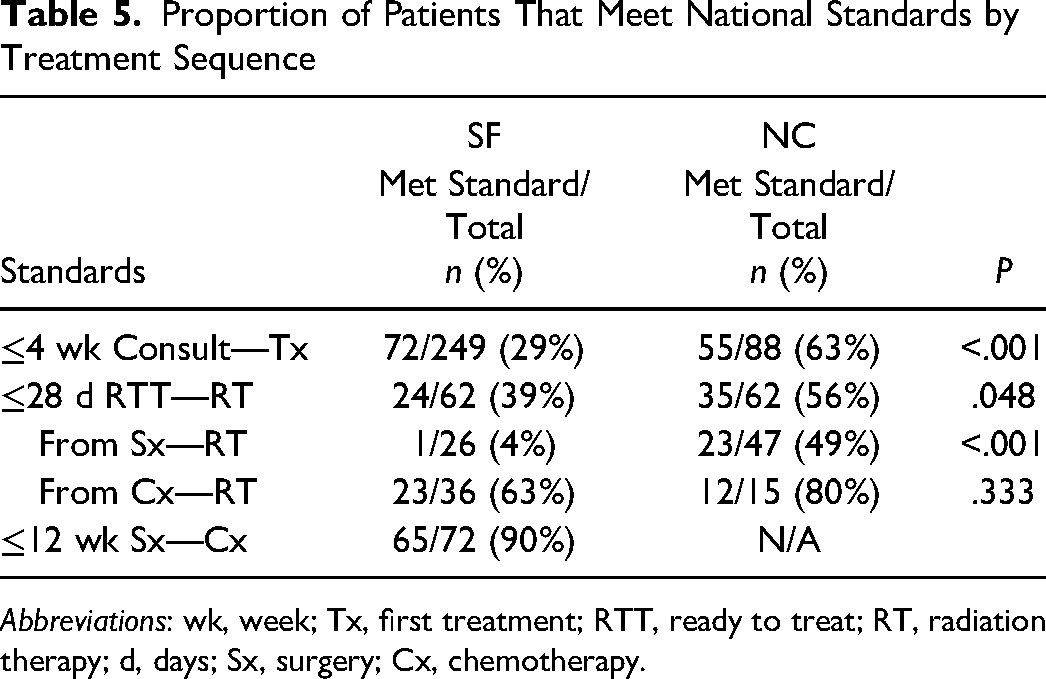
Abbreviations: wk, week; Tx, first treatment; RTT, ready to treat; RT, radiation therapy; d, days; Sx, surgery; Cx, chemotherapy.
Proportion of Patients That Meet National Standards by Reconstruction Type

Abbreviations: wk, week; Tx, first treatment; Sx, surgery; RT, radiation therapy; d, days; Cx, chemotherapy.
Delays in Care
Complications and Unplanned Operations. Complication rates were not markedly different between SF and NC patients (28% vs 38%, P=.086) with the most common complication for both groups being mastectomy flap necrosis. Additionally, both groups experienced a similar rate of unplanned operations (20% vs 25%, P=.333). Within reconstructive subgroups, there were slightly lower complication rates for both SF alloplastic patients compared to SF autologous patients (25% vs 32%, P=0.084) and for NC alloplastic patients compared to NC autologous patients (35% vs 43%, P=.478). Yet again, there were similar rates of unplanned operations between SF alloplastic and SF autologous patients (21% vs 18%, P=.665) and between NC alloplastic and NC autologous patients (25% vs 25%, P=1.00). A summary of these comparisons is shown in Tables 7 and 8.
Complication and Unplanned Operation Rate for Surgery First as Compared to Neoadjuvant Chemotherapy

Abbreviations: MFN, mastectomy flap necrosis.
Total counts individual cases while the complication breakdown accounts for cases having multiple complications
Complication and Unplanned Operation Rate for Reconstructive Subgroups
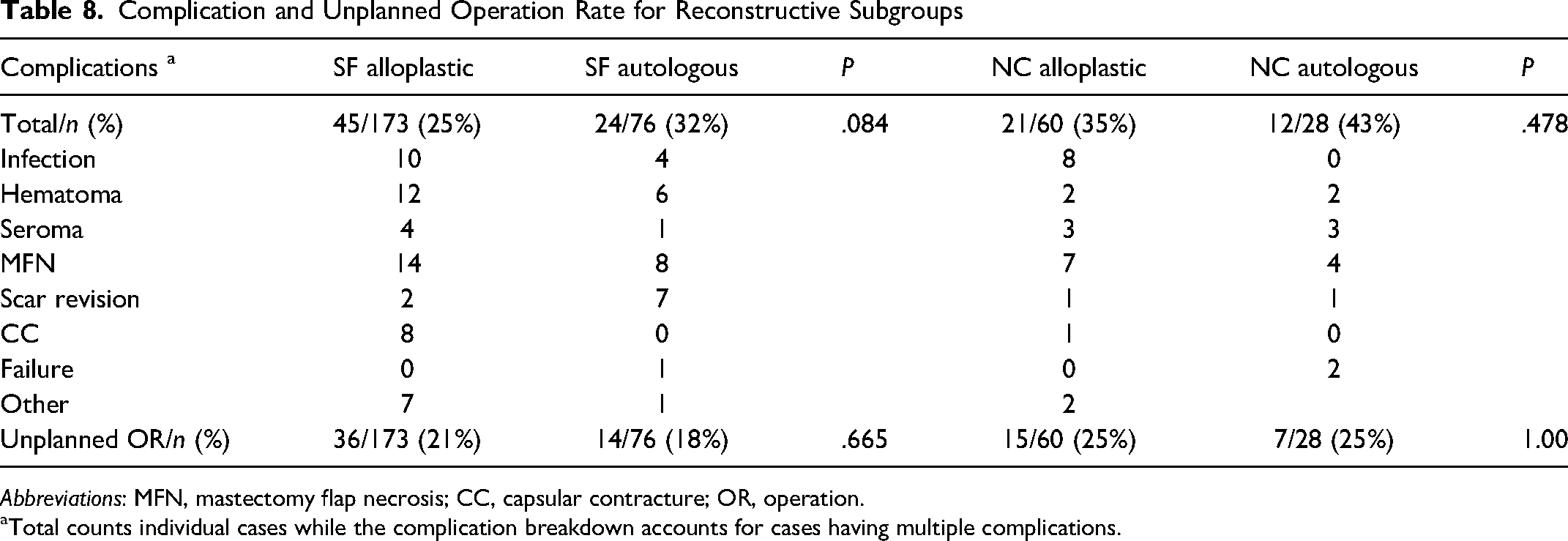
Abbreviations: MFN, mastectomy flap necrosis; CC, capsular contracture; OR, operation.
Total counts individual cases while the complication breakdown accounts for cases having multiple complications.
Discussion
In this study, patients undergoing surgery first (SF) experienced longer pretreatment phase wait times compared to neoadjuvant chemotherapy (NC) patients. Additionally, those undergoing alloplastic reconstruction tended to have shorter wait times compared to those undergoing autologous reconstruction. These results suggest that the availability of surgical resources is limited and may delay treatment initiation, with greater delays for patients seeking autologous reconstruction. Despite this, autologous reconstruction remains an important reconstructive choice for women given its superior patient-reported and aesthetic outcomes in many patients.11,25–27
Within the treatment phase, patients receiving surgery first required an extra 23 days to progress from first to second treatment modalities, while neoadjuvant chemotherapy patients required an extra 13 days to move from second to third treatment modalities. These time points represent the transition from surgery to subsequent therapies for the respective cohorts. To further explore why there are delays following surgery, we tracked complication rates and number of unplanned ORs. Complication rates were 28% and 38% for surgery first and neoadjuvant chemotherapy patients, with 20% to 25% of complications resulting in an unplanned operation. While there were no significant differences seen between the two primary groups, there was a trend of higher complication rates for autologous reconstruction compared to alloplastic.
Compared to Canadian standards, 24 our study identified that only 29% of patients receiving surgery first and 63% of patients receiving neoadjuvant chemotherapy met the standard of treatment initiation within 4 weeks of consultation with a surgical, medical, or radiation oncologist. Additionally, only 39% of SF patients and 56% of NC patients received radiation within 28 days of being deemed RTT. For those receiving radiation directly after surgery, patients experienced greater delays as compared to those undergoing chemotherapy than radiation. The majority of our patients successfully achieved the standard of initiating chemotherapy within 12 weeks after surgery.
When looking at other Canadian provinces, comparative data is sparse, however the most recently published report from Alberta in 2000 outlines a mean wait time of 20 days between definitive diagnosis and treatment initiation. 28 In 2001, data from Quebec reports an overall median wait time of 34 days between diagnosis and treatment and data from Ontario reports a median wait of 20 days between treatment decision and treatment initiation.29,30 In 2005, Manitoba published their time from consultation to first surgery being only 15 days with times from chemotherapy to radiation and surgery to radiation being 8 days and 24 days, respectively. 31 In 2006, data published from Ontario reports a wait time of 84 days for adjuvant radiotherapy following surgery. 32 In 2016, data from Newfoundland and Labrador reports a wait time of 22 days between first consultation and surgery. 33 While definitions of the wait time vary between published reports, these studies highlight existing variations between provinces in achieving national benchmarks for care delivery.
Within reconstructive subgroups, it was more challenging for surgery first patients undergoing autologous reconstruction to meet treatment standards compared to those receiving alloplastic reconstruction, likely due to increased operative resources required.15,19,34 After this initial delay, these patients were more successful in meeting the standard for initiating radiotherapy compared to those undergoing alloplastic reconstruction. This may be a result of the providers’ and patients’ preference for autologous reconstruction in the preoperative setting of a known need for post-mastectomy radiation.15,16,19,35,36 In these cases of advanced disease, these patients would have had a preoperative consultation with a radiation oncologist in anticipation of their post-mastectomy radiation.
When considering these delays, it is also important to note how histopathologic status may influence the urgency of treatment initiation. For example, surgery first patients had a greater incidence of noninvasive cancers, such as DCIS or LCIS, which can tolerate a prolonged wait for treatment initiation without a significant impact on patient outcomes.21,37–39 Alternatively, neoadjuvant chemotherapy patients tended to have more invasive cancers where delays in receiving chemotherapy or radiation can result in an increased risk of local recurrence and decreased chance of survival.40–44 Therefore, these patients are prioritized when there is limited access to radiotherapy and other resources.
Our findings are consistent with previous literature and expand upon our knowledge of wait times according to reconstructive type. 45 By identifying these “at risk” patient groups, we can better triage care to reduce discrepancies in wait times between the initiation of treatments and time intervals in between.
Breast reconstruction may be essential to a patient's quality of life, body image, and physical and mental well-being.4,11,13,19 For the provision of comprehensive breast cancer care, plastic surgeons provide reconstructive clinical expertise for patients seeking reconstruction. While this added component of cancer care may increase the demands for coordination of resources, it is essential for patient satisfaction in survivorship, with IBR affording patients the most cohesive treatment with improved psychosocial well-being.16,17,46,47 To address delays experienced by our patients, we are strategizing to align the allocation of resources with the clinical need in real time with simulation modeling to alter allocation according to fluctuating clinical needs. Multidisciplinary clinics also may be employed to streamline patient care and improve treatment outcomes by centralizing care providers.12,48–55 Benefits of this model have been shown in British Columbia with a Rapid Access Breast Clinic, providing superior care compared to traditional siloed models of care delivery.56,57 Increased collaboration and resource utilization are key drivers for optimal delivery of comprehensive breast cancer care. 46
Study Limitations
This study was limited geographically to a single center, potentially impacting the generalizability of the results to other regions. Furthermore, our data was restricted to IBR patients only. Therefore, all surgical delays are assumed to be due to reconstructive surgery as there is no comparison to mastectomy alone or delayed reconstruction. Future work will include this population of breast cancer patients to better delineate attributable wait times to processes of care delivery. Additionally, our cohort size and event number were inadequate to further explore the timing of complication rates for patients undergoing neoadjuvant chemotherapy.
Conclusion
Wait times for downstream ablative therapies are most prolonged for IBR patients receiving surgery first, especially if they undergo autologous reconstruction. To improve wait times, alternative strategies may be employed to optimize resource utilization.
Footnotes
Acknowledgments
Dr Kathryn Isaac is the holder of the Dr Patricia Clugston Chair in Breast Reconstruction Surgery at the University of British Columbia and would like to acknowledge the support of VGH & UBC Hospital Foundation's donors and partners who made this Chair possible.
Author Contributions
DC–Conceptualization, Data collection, Data analysis, Manuscript Writing, Review and Editing Manuscript. SV– Conceptualization, Data collection, Data analysis, Manuscript Writing, Review and Editing Manuscript. KSR–Conceptualization, Data collection, Data analysis. MMN–Data collection. ESB–Project administration, Supervision, Conceptualization, Manuscript editing, and Care Provider. CD–Project administration, Supervision, Conceptualization, Manuscript editing, and Care Provider. SAM–Project administration, Supervision, Conceptualization, Manuscript editing, and Care Provider. NVL–Project administration, Supervision, Conceptualization, Manuscript editing, and Care Provider. KVI–Project administration, Supervision, Conceptualization, Manuscript Writing, Review and Editing Manuscript, and Care Provider.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Ethics approval was obtained from the University of British Columbia's Research Ethics Board (ID: H21-00122). All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. Informed consent was waived by the University of British Columbia's Research Ethics Board.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Presentations
Poster presentation at the Canadian Society of Plastic Surgery Annual Meeting 2023.
