Abstract
Background and Aims:
Octreotide is suggested to harden the pancreas, thus facilitating the construction of a pancreatic anastomosis and lowering the risk of postoperative fistula. We tested the hypothesis that intra-arterial application of octreotide in the gastroduodenal artery during pancreatectomy may increase pancreatic hardness.
Material and Methods:
A single-center, prospective, double-blinded, randomized controlled trial with parallel assignment was conducted. Patients planned for a pancreatoduodenectomy or a total pancreatectomy, who had a palpatory and durometer proven (<40 Shore units) soft pancreas, were assigned to receive intraoperatively either 5 mL 500µg octreotide or 5 mL 0.9% saline solution as a bolus injection in the gastroduodenal artery. Pancreatic hardness was measured before, early, and late after intervention. The investigator performing the durometer measurements and pathologist were masked to group assignment. The primary outcome was increased pancreatic hardness. Analysis was by intention to treat. This trial is registered at http://www.clinicaltrials.gov (ID NCT01400100).
Results:
A total of 12 patients received octreotide and 13 received saline solution. Pancreatic hardness marginally increased in the octreotide group: 0.67 ± 2.3 Shore units, whereas it decreased in the control group: −2.15 ± 2.7 Shore units. The difference was statistically significant, p = 0.029 (95% confidence interval = −4.87 to −0.77). Histology did not find any correlate for this clinically irrelevant hardening effect.
Conclusions:
A single bolus application of octreotide did not deliver a clinically relevant increase in pancreatic hardness. Future studies on the hardening effect of octreotide should employ repeated or continuous preoperative administration of this drug.
Keywords
Introduction
Pancreatic texture has a substantial influence on the incidence of postoperative complications after pancreatoduodenectomy (PD) (1). Soft pancreas is generally accepted as a risk factor for the development of postoperative pancreatic fistula (POPF) (2–5). Somatostatin and its analogue octreotide have been used for more than three decades in the prevention and treatment of POPF (6–10). Although the exact mechanism of this protective action has never been fully clarified, octreotide is still widely used by pancreatic surgeons worldwide. Most proponents of octreotide use believe that it reduces fistula formation and promotes fistula closure in two different ways: it inhibits exocrine pancreatic secretion and it hardens the pancreatic tissue. While the first mechanism was scientifically proven in numerous studies (8, 11, 12), the theory about the hardening effect of octreotide on pancreatic texture is based on the results of a single animal experiment and a small case series in humans (13, 14). The first study reported a significant hardening effect after a single injection of octreotide in the gastroduodenal artery (GDA) in humans undergoing PD (13). However, pancreatic texture was assessed not quantitatively, but just by palpation, and the authors did not have any explanation for the immediate effect of the drug. The second study quantitatively confirmed the hardening effect of octreotide on pancreas; however, pancreatic texture was measured only after the procedure, and conclusions were based on the assumption that all pancreata had similar hardness before octreotide administration (14).
This randomized controlled trial aimed to prove whether the intraoperative administration of octreotide as a single bolus injection in the GDA may lead to a relevant increase in the hardness of human pancreatic tissue.
Methods
Trial Design and Participants
This efficacy study was performed as a single-center, randomized, controlled trial with a two-group parallel assignment at the Pancreas Center at St Josef Hospital, Bochum, Germany. Patients eligible for participation were those planned for a pancreatic resection including ligature of the GDA and having a risky soft pancreas. Thus, all patients who were aged between 18 and 90 years, signed informed consent, had intraoperatively a soft pancreas by palpation and proven by a durometer measurement, and underwent either a PD or a total pancreatectomy (TP) were included in the study. A threshold of <40 Shore units (SU) was accepted for soft pancreas, based on the results of previous studies on pancreas durometry (3, 4, 14). Exclusion criteria included a planned pancreatic resection without transection of the GDA (e.g. distal or central pancreatectomy); emergency pancreatic resections; anatomical variation of the vascular supply of the pancreas or liver, posing an increased risk of octreotide distribution in organs other than pancreas; acute pancreatitis at the time of surgery; baseline pancreatic hardness ≥40 SU; intraoperative complications or unstable patient; active infectious disease such as hepatitis or HIV infection; known or suspected allergy toward octreotide or its analogues; previous pancreatic surgery; participation in another clinical trial; and technical difficulties such as impossible cannulation of the GDA. Based on the volume of pancreatic surgical procedures at our institution and a retrospective analysis of the proportion of patients having soft pancreas, an estimated recruitment time of 6 months was expected.
All patients provided written informed consent for study participation. The study protocol was approved by the ethics committee of the Ruhr University of Bochum, Germany (Nr. 4033-11) and internationally registered (ID NCT01400100 at ClinicalTrials.gov).
Randomization and Blinding
Patients were assigned to octreotide or saline solution administration through a simple random sampling procedure. After confirming that all inclusion criteria were fulfilled and after intraoperative assessment of baseline pancreatic hardness (≤40 SU), the surgeon made a phone call to the study office, where a study nurse randomly drew a piece of paper from a box and disclosed the allocation group. Durometric measurements were performed by a second surgeon who was blinded to the kind of drug applied by the first surgeon. The patient and the pathologist were also blinded to the applied drug.
Interventions
The surgical technique of PD and TP used at our department is a standard one and is described elsewhere in detail (3, 5, 15). At our institution, all patients undergoing pancreatic surgery receive at induction to anesthesia 100 µg octreotide subcutaneously (s.c.) along with the antibiotic prophylaxis. All 60 patients who signed informed consent and were evaluated as possible candidates for the study did not receive octreotide preoperatively, in order to eliminate the chance of interference with the effect of intraoperative octreotide injection. Those patients who were excluded intraoperatively and were not randomized received their octreotide s.c. during the operation. All of the 25 patients included in this study postoperatively received octreotide s.c. 100 µg three times a day (t.i.d.) for at least 7 days, according to the standards at our department.
The first technical step intraoperatively after proving resectability included palpatory assessment of the pancreas by the surgeon in order to detect soft pancreatic tissue in the region of the pancreatic isthmus, where the transection of the pancreas was to be performed later. At this moment, a sterile Shore durometer was used to measure the initial baseline value of pancreatic hardness before transection of any important blood vessels in the region (M1).
Tissue hardness was measured using a handheld sterile Shore durometer (Schmidt Control Instruments, PHPSO-C0609, no. C316-07620 and no. C316-07621; Hans Schmidt and Co. GmbH, Waldkraiburg, Germany) according to a technique described earlier (4) (Fig. 1), with higher values indicating a harder material.
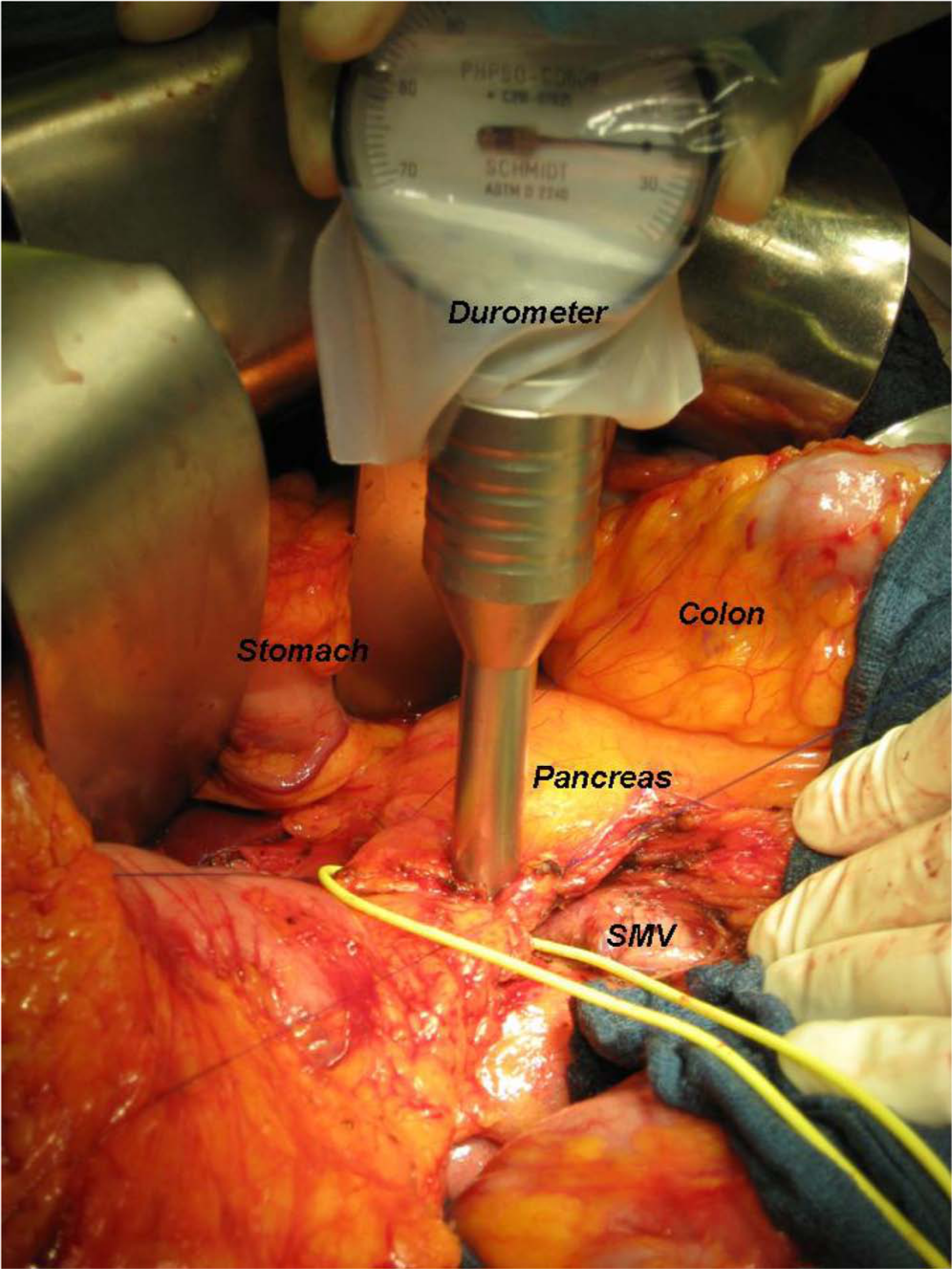
Technique of intraoperative durometer measurement of pancreatic hardness.
After group allocation, the GDA was ligated proximally with a 3-0 polypropylene suture; blood flow to the liver in the hepatic artery (HA) was controlled at palpation, and a single bolus of either 5 mL 500 µg Sandostatin® (octreotide acetate 0.1 mg/mL by Novartis, Basel, Switzerland) or 5 mL NaCl 0.9% solution was injected in the GDA in distal direction using a 5-mL syringe and a 24G needle (Fig. 2). Immediately after injection of the solution, the GDA was suture-ligated distally, the artery was transected between the ligatures, and the pancreas was carefully observed for any macroscopically detectable changes in appearance and texture. Then, a second durometer measurement (M2) was performed to get an early postinterventional value. Preparation was continued, duodenum and common hepatic ducts were transected, and just before transection of the pancreas, around 60 min after injection, a late third durometer measurement was performed (M3). All measurements were performed in one and the same area of the pancreatic isthmus by a second surgeon, blinded to the intervention. The resected specimen was then sent to the Institute of Pathology, where an experienced pathologist, blinded to intervention, performed a histological analysis. A histological score was calculated based on the properties of the pancreatic tissue at the resection margin in order to stratify the risk of the development of a POPF and prove the inclusion of patients with high-risk pancreas. The exact calculation of the histological score is described elsewhere (3). In brief, it presents a sum score of five separately scored histomorphological parameters including fibrosis grade, inter- and intralobular fat content, signs of chronic pancreatitis, and pancreatic main duct size. The score ranges from 0 to 14, whereas the lower the score, the softer the pancreas and the higher the risk of a POPF.
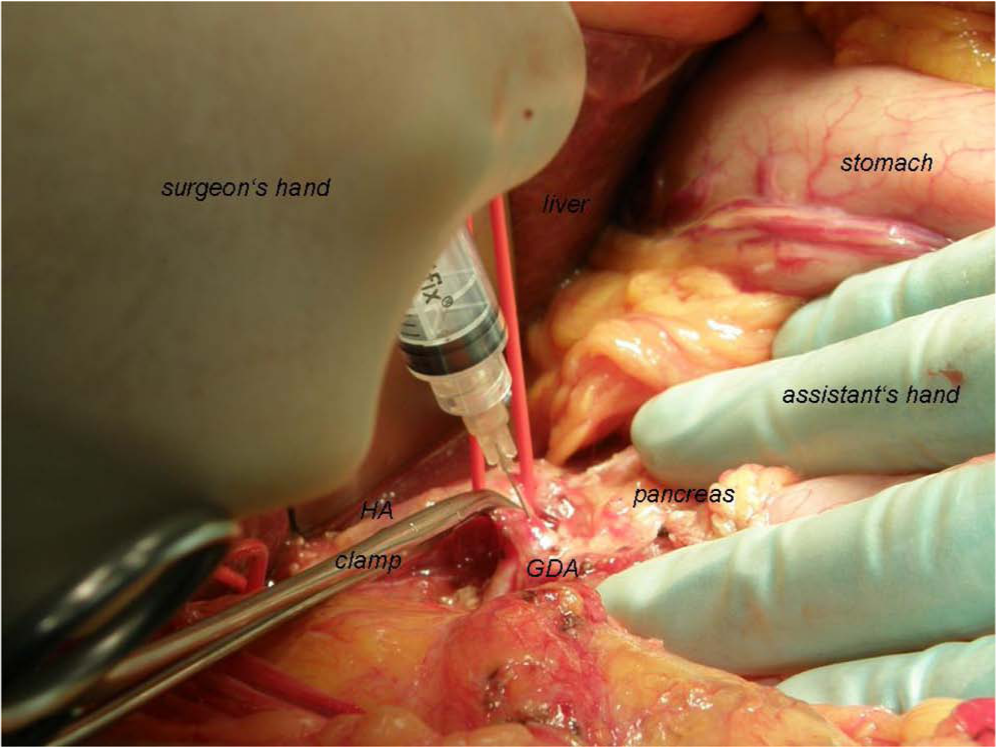
Technique of intraoperative octreotide application in the GDA.
Outcomes
The primary outcome measure of the study was a clinically relevant quantitative change in the hardness of pancreatic tissue detected after the local application of octreotide during the resectional phase of a PD/TP. Secondary outcome measure was the rate of POPF, as defined by the International Study Group on Pancreatic Fistula (16). Postoperative hemorrhage and delayed gastric emptying (DGE) were defined according to the established international classifications (17, 18).
Sample Size and Statistical Methods
Based on a sample size calculation for two independent samples, with a power of 90% and an alpha error of 0.05, assuming a mean baseline pancreatic hardness of 35 SU with a standard deviation of 10 SU, and setting an increase of 15 SU in pancreatic hardness as clinically relevant, at least 12 patients per group were necessary to detect statistically significant differences between the groups. Unpaired t-test, Mann–Whitney test, and exact Fisher’s test were used where appropriate. All statistical tests were two-tailed. Significance was defined as p < 0.05. Statistical analyses were performed using SPSS 18.0 for Windows (SPSS Inc.).
Results
Between 21 July 2011 and 26 January 2012, a total of 26 patients with soft pancreas were included. One patient of the octreotide group received only 200 µg of the drug because of a technical mistake and was excluded from further analysis due to violation of study protocol. The flow diagram of the study is presented in Fig. 3. Table 1 presents a summary of patients’ characteristics, diagnosis, and performed surgery in both groups. A significant difference between the groups existed for none of the parameters, except for age. Operative times and the time interval between injection and the last measurement of pancreatic hardness, that is, the median time for which the applied drug was allowed to exert its supposed effect were identical in both groups.
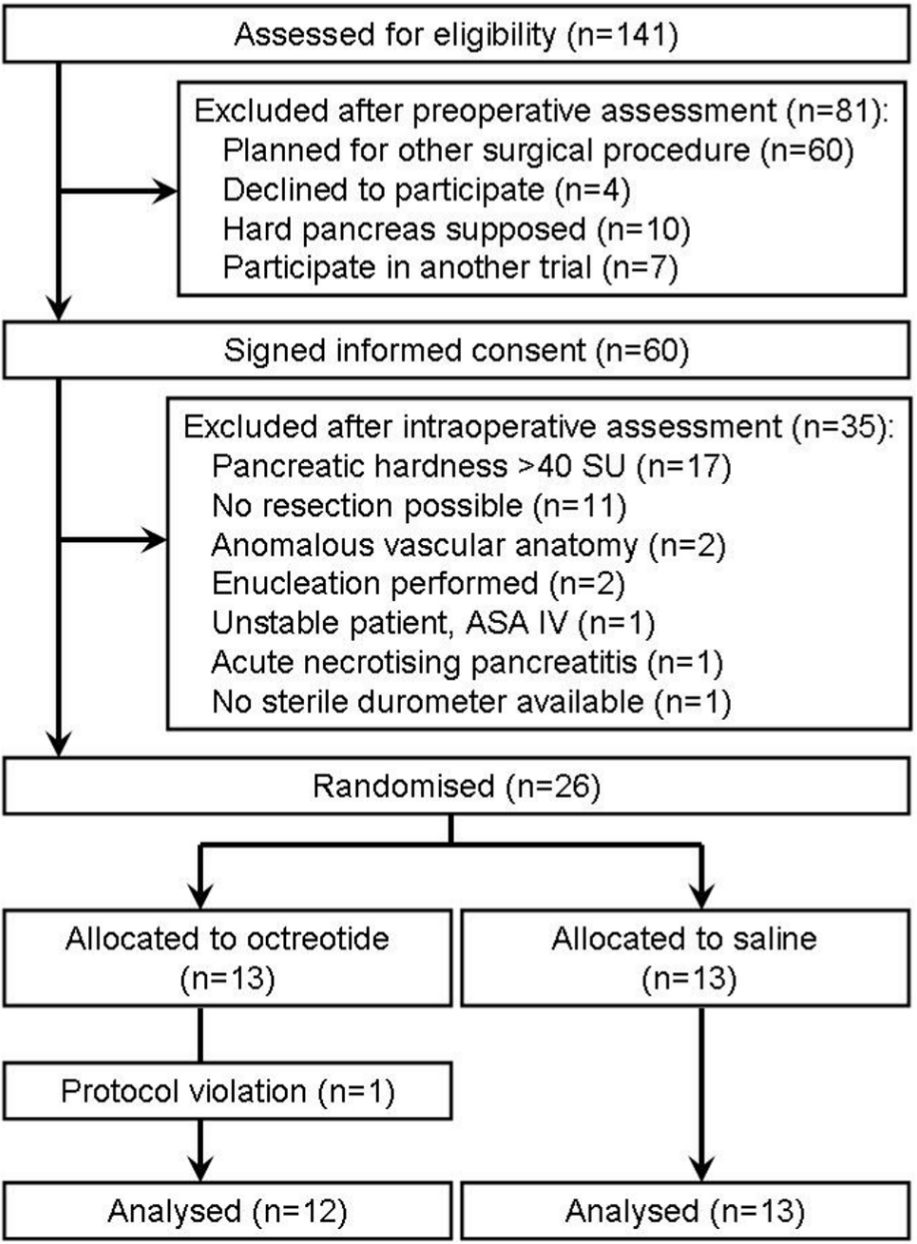
Flow diagram of the study.
A summary of patients’ characteristics.
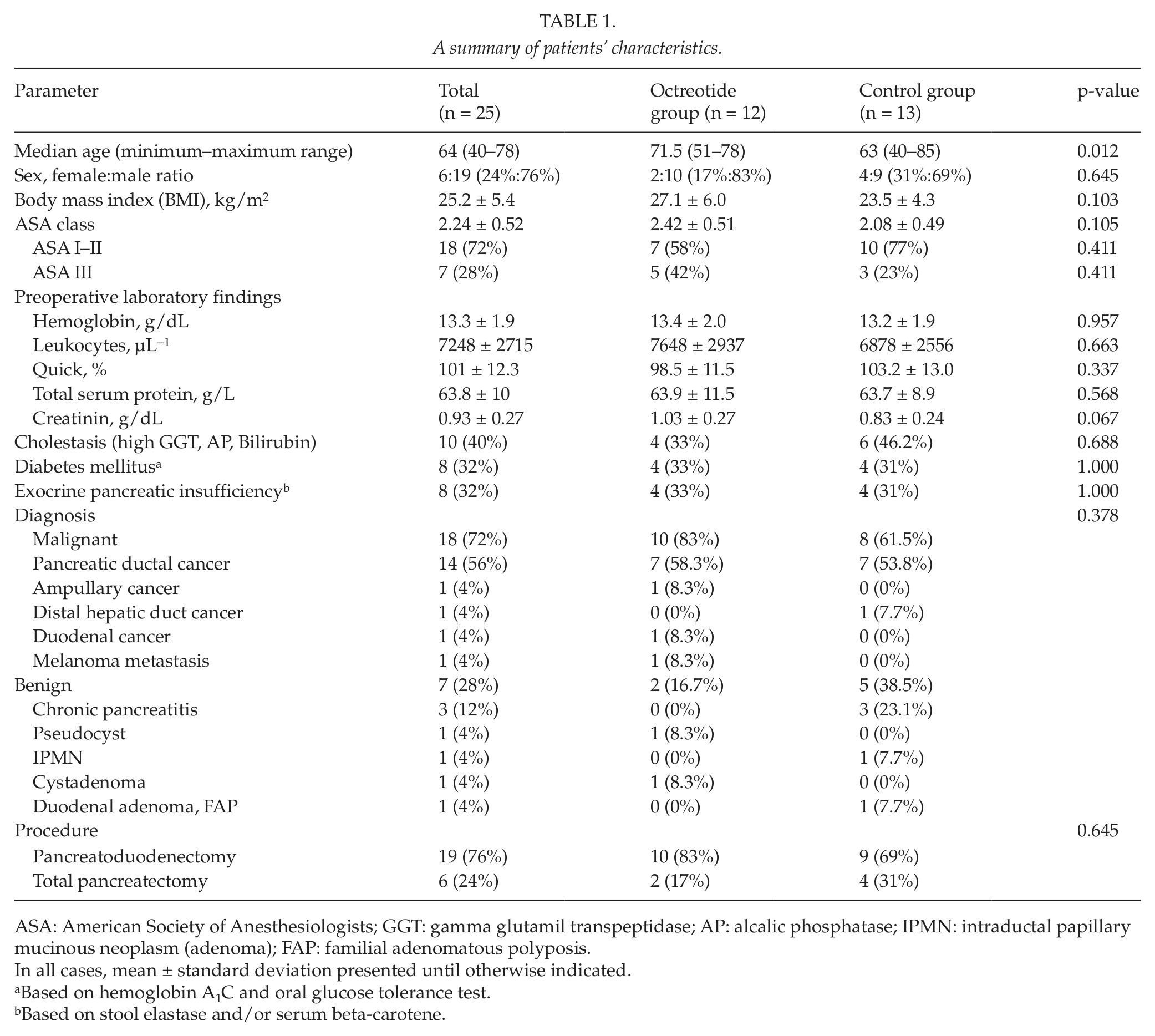
ASA: American Society of Anesthesiologists; GGT: gamma glutamil transpeptidase; AP: alcalic phosphatase; IPMN: intraductal papillary mucinous neoplasm (adenoma); FAP: familial adenomatous polyposis.
In all cases, mean ± standard deviation presented until otherwise indicated.
Based on hemoglobin A1C and oral glucose tolerance test.
Based on stool elastase and/or serum beta-carotene.
The first assessment of pancreatic texture included both inspection and palpation by the surgeon and quantitative validation by a Shore durometer in order to allow randomization. All 25 pancreata were assessed as soft/normal or very soft by the surgeon, and all of them showed values of hardness less than 40 SU (minimum 18 SU; maximum 39 SU). Baseline hardness of the octreotide group was practically the same as that of the control group (28 SU). There was a trend of continuously decreasing pancreatic hardness in the control group, whereas a slight increase was observed in the octreotide group. The absolute intraoperative change in pancreatic hardness was significantly different between the octreotide and control groups, 0.67 ± 2.3 SU versus −2.15 ± 2.7 SU, p = 0.029 (95% confidence interval (CI) = −4.87 SU to −0.77 SU) (Table 2). However, the median increase of pancreatic hardness in the octreotide group compared to the control group accounted for a mere 2.5 SU, far beneath the expected 10–15 SU of increase. Respectively, neither visual nor palpatory change in pancreatic texture was observed by the surgical team in any of the 25 patients. A transient and slight pallor and swelling of the pancreatic tissue in the pancreatic head were observed in some, but not all, of the patients in the octreotide group.
Results of intraoperative durometric measurements and points of time at which they were performed.
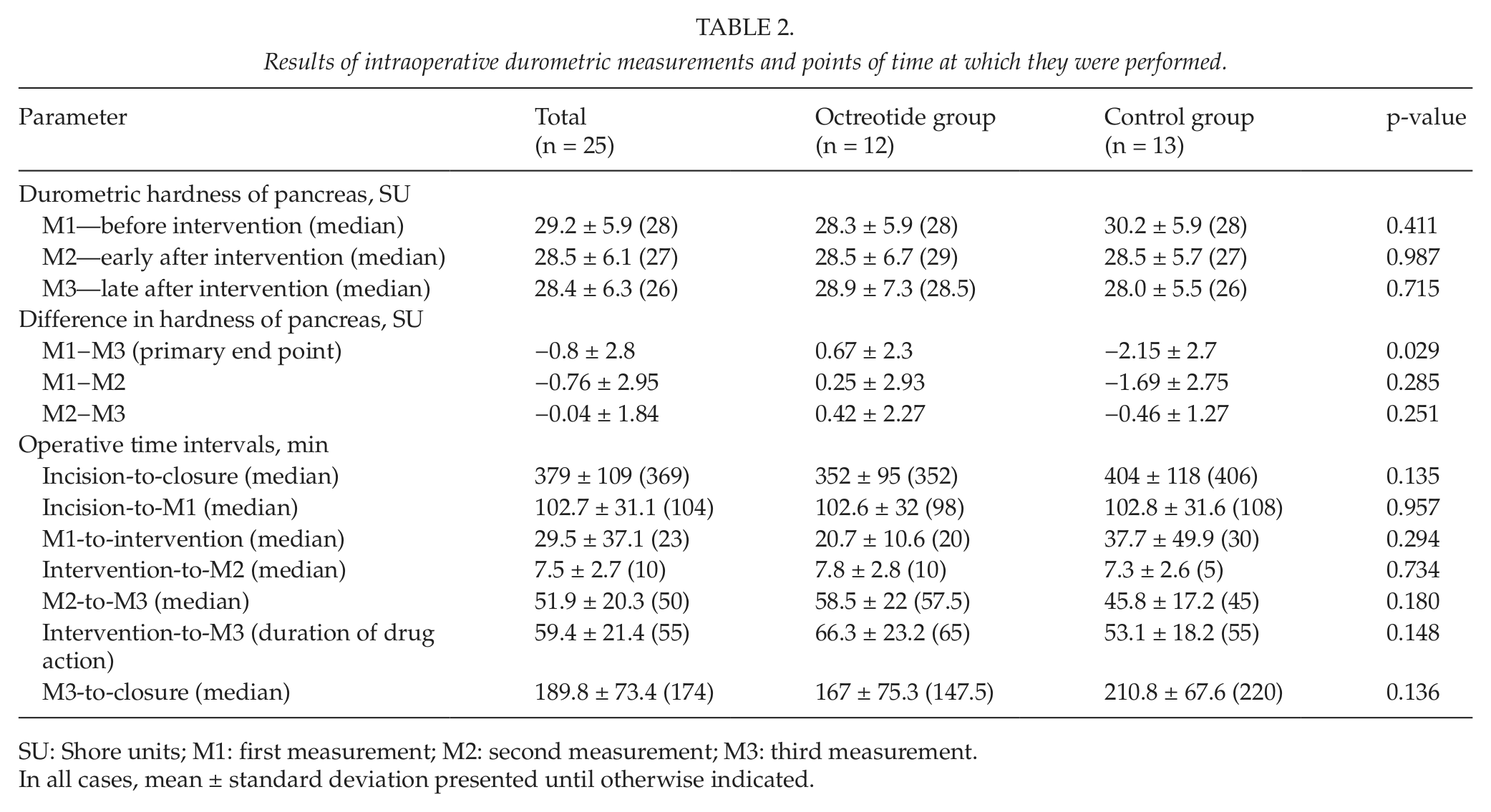
SU: Shore units; M1: first measurement; M2: second measurement; M3: third measurement.
In all cases, mean ± standard deviation presented until otherwise indicated.
A summary of the major postoperative findings is given in Table 3. One patient died in the octreotide group. He underwent a PD for a solitary metastasis of a malignant melanoma in the pancreatic head and postoperatively developed a grade C pancreatic fistula with delayed bleeding from the right HA with two consecutive reoperations. After an initial stabilization, the patient later developed a severe bilateral pneumonia with multiresistant germs and died on the postoperative day 61. There were two grade C pancreatic fistulas in the octreotide group. One grade C pancreatic fistula and one grade A fistula were observed in the control group. The rate of POPF was identical in both groups. It was 16% based on the total collective of patients. After exclusion of 5 TPs, the fistula rate was 21% (4/19) related to those pancreata at risk.
Postoperative findings.
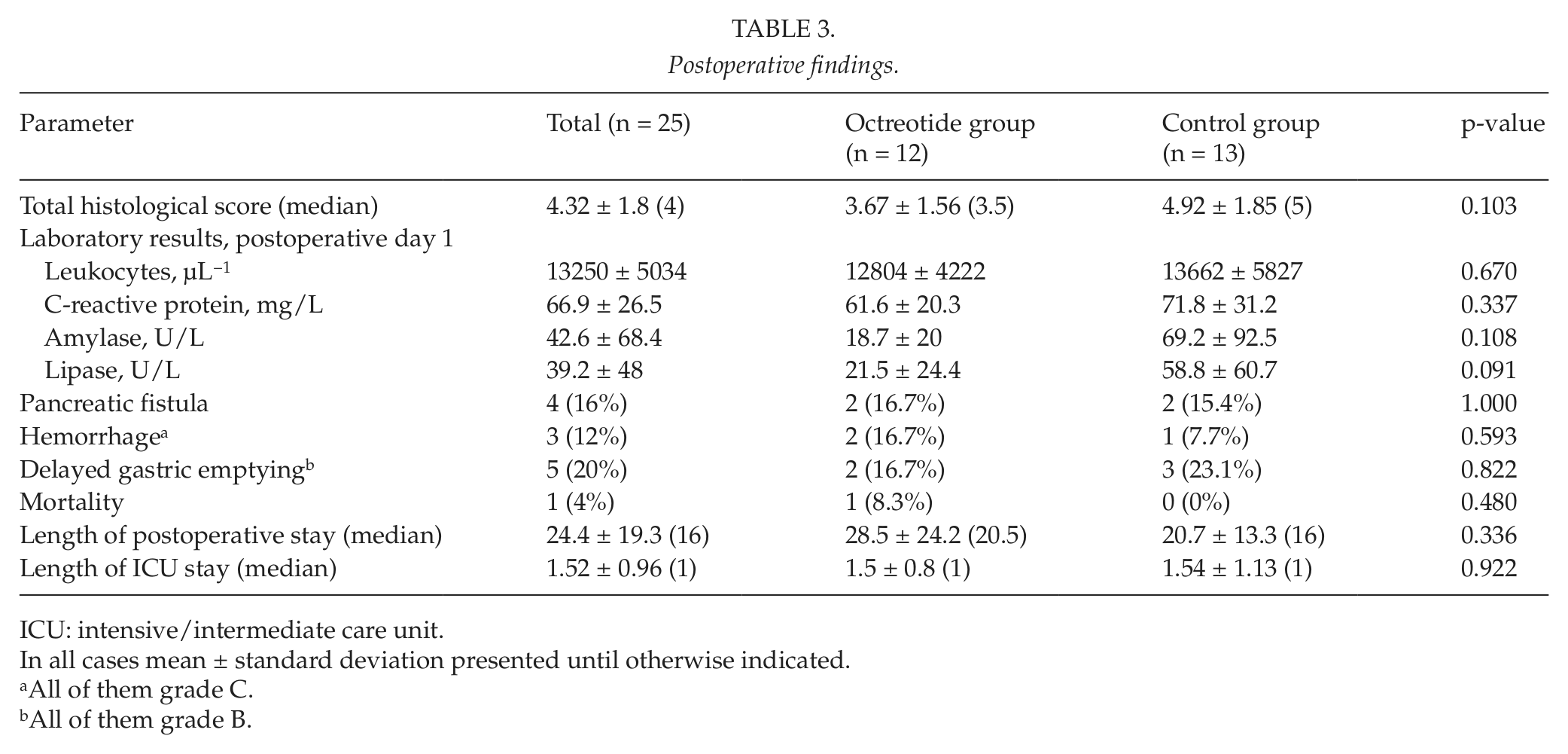
ICU: intensive/intermediate care unit.
In all cases mean ± standard deviation presented until otherwise indicated.
All of them grade C.
All of them grade B.
Two postoperative hemorrhages occurred in the octreotide group and one case in the control group. One was treated angiographically with a covered stent, and the other two required relaparotomy. In the octreotide group, two cases of DGE occurred after PD for malignancy. In the control group, there were two cases of DGE after TP for duodenal adenoma and multifocal intraductal papillary mucinous neoplasm (IPMN). The third case appeared after a PD for pancreatic cancer.
However, this study was primarily experimental and focused solely on intraoperatively measured changes in pancreatic hardness. The number of patients does not allow any significantly sound conclusions regarding the clinical results.
Octreotide-treated pancreata were indistinguishable from those of the control group at histology. Calculation of the histological score confirmed post factum that all recruited patients had high-risk soft pancreata, characterized by a low mean score of 4.32 ± 1.8.
Discussion
Octreotide is the most widely used somatostatin analogue. It has been used to prevent and treat pancreatic fistula for more than 30 years because of its suppression of exocrine pancreatic secretion (6, 7, 10, 17). Two recent reports in the surgical literature supposed that octreotide might have a beneficial effect not only on pancreatic function but also on pancreatic texture (13, 14).
Konstadoulakis et al. (13) reported in 2005 that intra-arterial bolus administration of 500 µg octreotide in the GDA during PD in a small case series of 4 patients immediately hardened the pancreatic remnant, and thus allowed an easier construction of the pancreaticoenteric anastomosis. According to the authors, all pancreata were considerably harder at palpation just within minutes after octreotide application. In two of the patients, intraoperative ultrasound was used to verify the observed hardening effect, which was explained through the local vasoconstriction caused by the drug (13).
In 2006, Foitzik et al. (14) confirmed the observation that octreotide hardened the pancreas in the setting of an animal experiment. Using durometer and dynamometer measurements, they found significantly higher values of pancreatic hardness and suture holding capacity in octreotide pretreated pigs compared to control animals. Furthermore, the longer the octreotide pretreatment, the harder the pancreas. The authors supposed that the hardening effect was due to water and protein redistribution caused by octreotide in the intra- and extracellular compartments of the pancreas (14).
Despite their ingenious design, both the above-mentioned studies suffered some major limitations. Konstadoulakis et al. (13) did not assess pancreatic texture quantitatively, but only at palpation, and used ultrasound to verify their observations, although increased opacity in ultrasound is not necessarily equal to increased tissue hardness. Foitzik et al. (14) performed an animal experiment the design of which is not reproducible in humans. Their conclusion that octreotide hardened the pancreas was based on the assumption that pancreatic hardness of control animals at the end of the experiment was equivalent to the baseline pancreatic hardness of the treated pigs before drug application.
The present experimental study was designed to overcome the mentioned limitations, combining the best features of both studies and adding an evidence-based approach in order to answer the question whether octreotide hardens the pancreas. Its aim was definitely not to find any difference in the clinical outcome between octreotide-treated patients and controls, since such a hypothesis would have meant a much larger number of included patients. Being a clinical trial in humans with pre- and postinterventional assessment of pancreatic hardness, it hurdled the obstacles of the Foitzik’s experiment. At the same time, the use of a sterile durometer to quantify the results, the standardized histological examination, and the randomized controlled character of the trial eliminated the major flaws of the Konstadoulakis’ case series. Further advantages of this trial, which assured objective and reproducible results, included standardized pre-, intra-, and postoperative patient care, surgical technique, and complications management, guaranteed by the high-volume of the institution. It allowed also the fast recruitment of the required number of patients despite stringent inclusion criteria. The wide range of exclusion criteria aimed at patient safety and protection of neighborhood organs from possible harms due to drug application. Finally, only patients with risky soft pancreas were included in the study, and the success of the selection process was unequivocally proven by the histological score. The use of a sterile durometer allowed precise assessment of pancreatic hardness through direct and immediate measurements in situ.
The study has some potential limitations. Inclusion of TPs in the study might have affected the results in at least the M3 measurements because of the worse vascularization compared to PD. The fact that all three patients with chronic pancreatitis were in the control group may be misleading. All of them had soft pancreatic tissue, and many of the remaining patients had signs of chronic pancreatitis at histology, despite other leading diagnosis. The way and site of drug application as well as the dosage of octreotide were chosen to be identical with those of the Konstadoulakis study, in order to test the generalizability of their positive results. Nevertheless, whether intra-arterial application of octreotide is better than subcutaneous or intravenous application and whether the GDA is the best injection site remains unclear. Whether the volume of the injected solution as well as the total amount of octreotide were optimal is also questionable. These parameters should be dependent on the mechanism of tissue hardening, which remains further unclear. It seems that octreotide could harden the pancreas mainly through temporary vasoconstriction. Whether such a transient effect would suffice for a safer anastomosis to the same extent as chronic changes in pancreatic texture due to fibrosis and lower fat content has not been clarified in the literature. This study showed that a single shot of octreotide did not lead to histological alterations in pancreas. Another issue is the duration of octreotide action, which was about an hour in the present study. The pharmacokinetics of octreotide suppose that it reaches its maximal activity within minutes after application, irrespective of its route of administration (19). Konstadoulakis et al. (13) observed a hardening effect within minutes, too. However, Foitzik et al. (14) showed that the longer the treatment, the harder the pancreas. Furthermore, it is hard to expect any permanent tissue hardening after a single bolus application of the drug, especially at a relatively low dose of 500 µg. The number of patients was adequately calculated to clear the primary end point of the study; however, it was too small to reveal any clinically relevant consequences in terms of morbidity and mortality rates. These appeared to be the same in both groups, which is not a surprise having in mind that both groups were identical with respect to patient characteristics, pathological findings, and surgical procedures. The rate of POPF was high, which reflects the selected homogenous group of high-risk patients, all of them with soft pancreas and low histological score.
In conclusion, based on the findings of this trial, one cannot expect any immediate, long-lasting, and clinically relevant tissue hardening after intraoperative application of octreotide in patients with soft pancreas. Future studies on the hardening effect of octreotide should focus on the possible mechanisms of this effect and employ repeated or continuous administration of this drug.
Footnotes
Declaration of Conflicting Interests
There are no known conflicts of interest associated with this publication.
Funding
This study was partially funded by a FoRUM grant of the Ruhr University of Bochum, AZ F694-2010, Nr. 842920.
