Abstract
Background and Aims:
Functional gastric outlet obstruction is a common problem after esophagectomy. The aim of this study was to evaluate the safety and efficacy of treating this group of patients with pneumatic dilatation of the pyloric sphincter region using a large-diameter (30–35 mm) balloon.
Material and Methods:
A review of all patients who had undergone pneumatic dilatation of the pylorus sphincter because of gastric outlet obstruction symptoms after esophagectomy at the Karolinska University Hospital from 2006–2011 was completed. Main outcomes were recordings of nausea, regurgitation and bloating.
Results:
A total of 13 patients received pneumatic dilatation after an esophagectomy. The median time between esophagectomy and the first dilatation was 100 days, and the patients underwent a total of 21 dilatations (1–3 per patient) to a final median diameter of 30 mm. No procedure-related complications occurred. The median follow-up time was 205 days, and nausea and regurgitation improved significantly (p < 0.001, Fisher’s test).
Conclusions:
Pneumatic dilatation of the pylorus using a large-diameter pneumatic balloon seems to be a safe and effective method for treating symptoms suggestive of gastric outlet obstruction after esophagectomy. To document its true effectiveness, a randomized and sham-controlled study is needed.
Keywords
Introduction
Patients undergoing esophagectomy are at risk of developing impaired gastric emptying, which consists of symptoms such as nausea, regurgitation, bloating and postprandial early satiety and fullness. This is referred to as gastric outlet obstruction (GOO) or delayed gastric emptying. Randomized trials have shown that drainage of the pylorus (pyloromyotomy or pyloroplasty) during esophagectomy may reduce GOO in the early postoperative phase (1, 2), whereas there does not seem to be any significant effect of this intervention at long-term follow-up (3). This means that GOO may recur even after a drainage procedure has been performed. GOO after esophagectomy has been reported to occur in around one out of four patients (4, 5) . The diagnosis in these patients is more or less always primarily based on clinical symptoms. Scintigraphic imaging may be useful for the establishment of diagnosis, but its correlation to symptoms is not complete (6). Patients who develop GOO early, while still in hospital, are typically treated conservatively with nasogastric decompression and prokinetic agents. If these fail, or if the symptoms develop at a later stage after discharge, other measures may be required. The most widely used treatment is endoscopic dilatation of the pylorus. Previous uncontrolled studies of endoscopic dilatation have reported positive effects of balloons with a final median diameter of 17–20 mm (5, 6) . Applying a conceptual view on the underlying mechanism for GOO as being an incomplete relaxation of the pyloric sphincter, very similar to the lower esophageal sphincter in achalasia, it would be logical to use a more aggressive dilatation strategy. In this article, we report a series of patients, who presented with GOO symptoms after esophageal resection, where pneumatic balloons with a diameter of 30 mm or more were used for dilatation of the pylorus.
Materials and Methods
Study Design
We completed a retrospective review of computerized medical records on all patients who had undergone esophagectomy (n = 195) from September 2006 to October 2011 at the Karolinska University Hospital. The vast majority of these patients had a pyloric drainage procedure added; however, this part is not mandated in the standard operative procedure protocol. Our routine is to perform a pyloromyotomy, which is converted to a pyloroplasty, in case of perforation of the mucosa. The search criteria included endoscopic dilatation of the stomach/pylorus/duodenum or gastric anastomosis. Thirteen patients who had pneumatic dilatation of the pylorus after esophagectomy were identified.
Dilatation Procedure
All dilatations were done under general anesthesia. A 10-mm endoscope was used to identify the pylorus, which was passed with a Savary-type guidewire. Dilatations were performed only after exclusion of anastomotic stricture by endoscopy, and there were no reports of scarring or fibrotic tissue in the pylorus. The final dilatation in each patient was done with a 30- to 35-mm pneumatic balloon intended for treatment of achalasia (Rigiflex®, Boston Scientific), which was placed to straddle the pylorus over the guidewire under fluoroscopic control. The balloon was then inflated for 1–2 min. During inflation of the balloon, the disappearance of a waist on the mid part of the balloon was strived for, but this was not always documented. After dilatation, the mucosa of the dilated pylorus was reexamined to exclude perforation. In four cases, a planned two-stage procedure was performed with a 5-mm increase in balloon diameter during the second procedure, which was usually performed 1–2 weeks after the first procedure. There was no suspicion of recurrent tumor among patients who underwent dilatation.
Exposure Data and Outcome
During review, information on bloating, nausea and regurgitation before treatment was collected from the patient records. Data on all symptoms were not always available. At follow-up, the patients were asked whether the symptoms had improved. In cases where there was no effect of dilatation or if symptoms recurred, redilatation was usually attempted. Results after the final dilatation are presented herein.
Statistical Analysis
The data are presented as median values and range. Fisher’s exact test was used for determining statistically significant differences (p < 0.05). Sigma Stat software (Jandel Corp., Sausalito, CA) was used for calculations.
Ethics
This research was conducted in accordance with the Helsinki declaration.
Results
Patient Data
All 13 patients (Table 1) had undergone esophagectomy with a narrow gastric tube (<4 cm). The indication or esophagectomy was cancer in 12/13 cases and iatrogenic perforation in one case. In 12/13 cases, a transthoracic esophagectomy was performed, and one patient had a transhiatal operation. A Kocher mobilization and a pylorus drainage procedure were done in all except one case (patient 5, transthoracic). Patients 4, 8 and 10 had anastomotic leaks (Table 1). Patient 4 underwent four dilatations of the anastomosis because of stricture before the first dilatation of the pylorus was performed. Six patients had metoclopramide or ondansetron against nausea administered on demand both before dilatation and at follow-up. All patients used proton-pump inhibitors both before and after the intervention. Four patients were on opiates before dilatation (one still in hospital after esophagectomy). At follow-up, two of these patients had opiates discontinued, while two other patients had started treatment with opiates during the follow-up. None of the patients required intermittent nasogastric tube insertion to decompress the gastric tube prior to the endoscopic intervention. Six of the patients were examined with barium swallow examination, and in three of these, there were signs of delayed emptying of the gastric conduit. One patient was examined with a radio-labeled meal that showed signs of delayed emptying. Two cases had signs of retained food during diagnostic endoscopy.
Summary of pneumatic dilatations of the pylorus in 13 patients with gastric outlet obstruction (GOO) symptoms after esophagectomy.
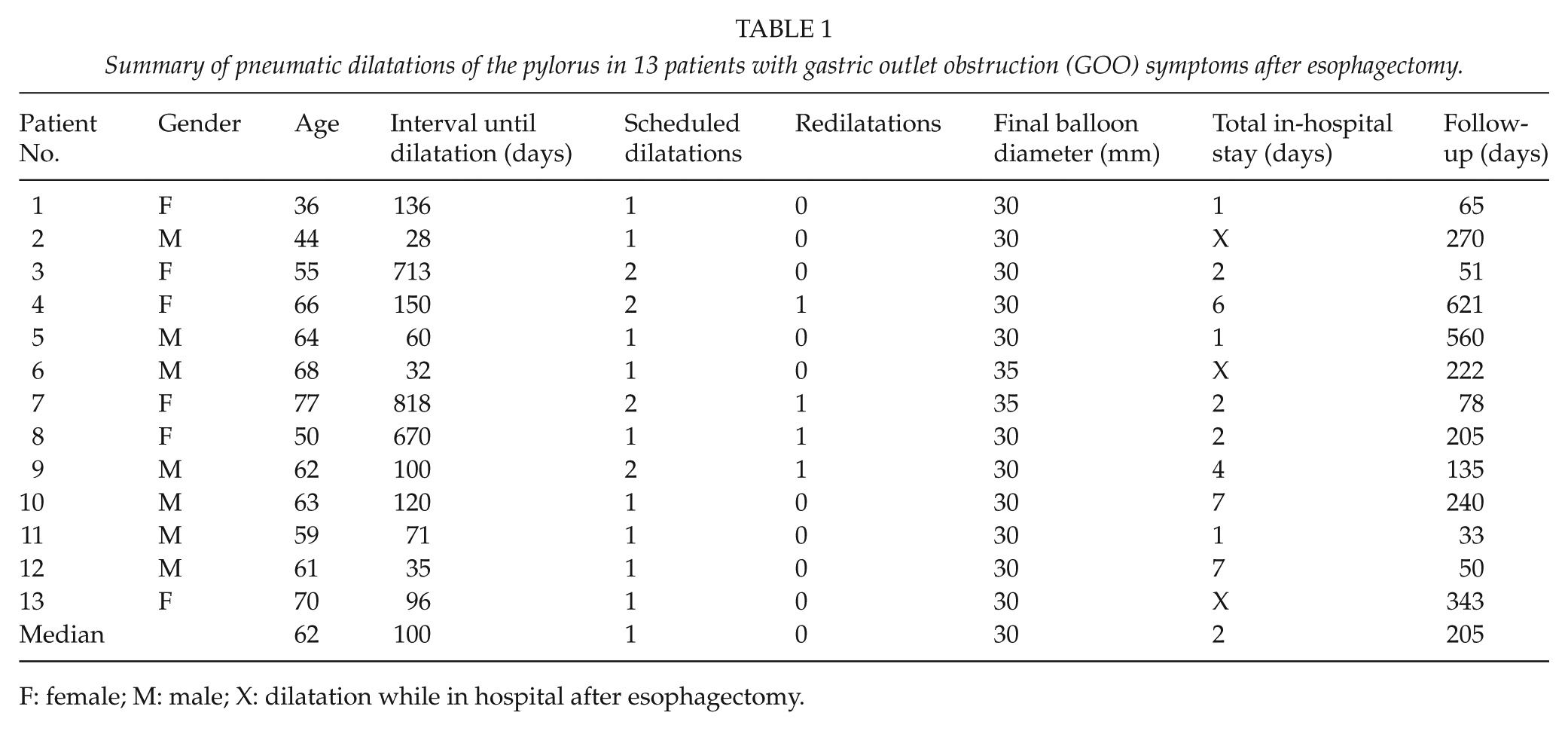
F: female; M: male; X: dilatation while in hospital after esophagectomy.
Effects of Pyloric Dilatation
The 13 patients included herein underwent a total of 21 dilatations (Table 1). Four of the patients had unscheduled redilatations because of persisting or recurrent symptoms. There were no complications after intervention. Patients 4, 10 and 12 had a total length of stay of 6–7 days. Patient 4 was from another city and was in hospital over a weekend waiting for a scheduled second dilatation, which however was postponed until 1 week later. Patient 10 was under treatment for gallstone pancreatitis and received dilatation during hospitalization, and Patient 12 was hospitalized because of abdominal pain and increased inflammatory parameters due to an intra-abdominal hematoma. Dilatation resulted in a significant improvement of nausea and regurgitation (p < 0.001 for both symptoms). Bloating was documented in only four of these patients and did not improve significantly. After initial improvement of symptoms, three patients reported new or recurrent symptoms. Patient 2 reported postprandial abdominal pain, Patient 4 experienced early satiety, and Patient 9 reported possible recurrence of nausea and regurgitation.
Discussion
The present retrospective and uncontrolled study could best be described as being a hypothesis-generating feasibility and safety study. Two symptoms suggestive of GOO, nausea and regurgitation, were relieved in most patients by forceful pneumatic dilatation of the pyloric ring after an esophagectomy. Two patients had concurrent intra-abdominal conditions that may have contributed to the GOO symptoms. The response to treatment was variable since some patients needed redilatation. Although the functional consequences of vagal denervation on the pyloric sphincter function after esophagectomy are not fully elucidated in humans, an achalasia-like situation may reside in the gastroduodenal junction in these situations because of incomplete relaxation of the pylorus and impaired motility of the gastric tube. Accordingly, the logical step would be to apply a similar therapeutic strategy in the gastroduodenal junction as proved to be effective in the gastroesophageal junction area in achalasia. Forceful pneumatic dilatation would therefore be required with a larger balloon than in previous publications about this technique (5, 6) . Such an intervention cannot be performed in unsedated patients, since significant pain is generated. Therefore, it is possible to assume that the previous observations on the effect after balloon dilatation in the 10- to 20-mm range may have been a placebo effect to a greater extent than dilatation with 30- to 35-mm balloons presented herein. The diagnosis and assessment of the severity of symptoms of GOO are difficult and have merely to be based on a comprehensive symptom analysis. Objective measures have been tested, such as gastric and/or pyloric motor function tests, with or without the addition of emptying characteristics of a radio-labeled meal. The problem is that the relation between objective parameters and symptoms is incomplete (6). Therefore, clinical assessment and symptom analyses are critical in these patients according to whose requirements structured research questionnaires can be amended, whereas the value of radiological imaging is limited. Another question that emerges is why forceful dilatation of the pylorus should be effective if pyloroplasty is devoid of a clear preventive effect on long-term GOO symptoms after esophagectomy (1). The early beneficial effects of pyloric drainage are obvious, whereas the durability is questionable (3). It is also possible that the effect of pneumatic dilatation is accordingly only temporary, although our preliminary results suggest a durable clinical effect. Again, these and other related issues have to be addressed within the framework of a randomized clinical trial framework. From a methodological perspective, it is crucial that a corresponding trial should have the design of being sham-procedure controlled.
