Abstract
The occurrence of an enteric fistula in the middle of an open abdomen is called an enteroatmospheric fistula, which is the most challenging and feared complication for a surgeon to deal with. It is in fact not a true fistula because it neither has a fistula tract nor is covered by a well-vascularized tissue. The mortality of enteroatmospheric fistulae was as high as 70% in past decades but is currently approximately 40% due to advanced modern intensive care and improved surgical techniques. Management of patients with an open abdomen and an enteroatmospheric fistula is very challenging. Intensive care support of organs and systems is vital in order to manage the severely septic patient and the associated multiple organ failure syndrome. Many of the principles applied to classic enterocutaneous fistulae are used as well. Control of enteric spillage, attempts to seal the fistula, and techniques of peritoneal access for excision of the involved loop are reviewed in this report. Additionally, we describe our recent proposal of a lateral surgical approach via the circumference of the open abdomen in order to avoid the hostile and granulated surface of the abdominal trauma, which is adhered to the intraperitoneal organs.
Keywords
Introduction—Definition
Open abdomen (OA), initially used by Ogilvie (1) in 1940, is currently applied in the context of three broad clinical conditions: (a) damage control surgery for severe abdominal trauma, (b) damage control surgery for severe abdominal sepsis (source control–temporary drainage), and (c) management of abdominal compartment syndrome. A very challenging task for the surgeon dealing with an OA is to know in detail the available and effective techniques for temporary abdominal closure in order to manage the OA during the first weeks. Additionally, the patient has to be treated in a surgical intensive care unit (SICU) with relative experience in managing specific problems and complications related to patients with an OA, namely, severe dehydration due to excessive fluid losses from the exposed surface of the OA, hypercatabolism associated with the underlying disease, electrolyte and acid–base disturbances, septic complications, and the most dreaded complication, the development of an enteroatmospheric fistula (EAF).
The occurrence of an enteric fistula in the middle of an OA is called an EAF, which is the most challenging and feared complication for a surgeon to deal with. It is in fact not a true fistula because it neither has a fistula tract nor is covered by a well-vascularized tissue (Levy et al. (2) coined the term “exposed fistula”), as are the majority of EAFs. For these reasons, spontaneous closure or sealing of EAFs is almost impossible.
Etiology—Classification—Clinical Features
EAFs usually develop as a consequence of one or more of the following factors: postoperative anastomotic disrupture, deserosalizations occurring during laparotomy, exposure of the dehydrated and desiccated bowel to several materials used for temporary abdominal closure, adhesions between the edematous bowel and the anterior abdominal wall, severe wound infections, burst abdomen, severe trauma, sepsis with known precipitating factors (low serum albumin, decreased collagen synthesis, etc.), and finally, preceding bowel ischemia. Although negative pressure wound therapy is currently used for EAF management (i.e. the widely applied VAC® system (vacuum-assisted closure)), it is reported to be implicated in the development of new EAFs on the OA in a small proportion of these patients (5%). However, recent experimental data show that negative pressure is not transmitted on the bowel surface (3).
The incidence of EAFs depends on the underlying abdominal pathology and varies between 2% and 25% for trauma patients, 20% and 25% for abdominal sepsis, and up to 50% for infected pancreatic necrosis (4). Clinically, EAFs are usually seen after the first week of OA, but they can occur at anytime in the hospitalization especially of patients requiring many months of OA. EAFs can be classified according to their (a) anatomic location (proximal/distal), (b) fistula volume output (low < 200 mL/moderate 200–500 mL/high > 500 mL), and (c) location in the OA (deep/superficial) (5, 6).
Location and fistula output are factors crucially affecting the prognosis and spontaneous closure of the EAF. More specifically, distal and low-output EAFs have a high spontaneous closure rate (which cannot be accurately determined because outcome depends on several other factors as well) in contrast to more proximal and high-output EAFs. Additionally, the rare location of the EAF deep in the peritoneal cavity is considered a surgical emergency due to ongoing peritonitis and should therefore be managed immediately. On the contrary, superficial EAFs occurring on the granulated surface of the OA are more frequently encountered in clinical practice but are more difficult to manipulate due to the inability to effectively control enteric spillage on the OA surface, which triggers ongoing sepsis (5).
Fistula location, demonstration of any other intra-abdominal abscesses or associated collections, and exclusion of any distal gastrointestinal (GI) obstruction can be demonstrated with a wide variety of imaging diagnostic methods, that is, methylene blue test, upper and lower GI series with water soluble contrasts, fistulography, computed tomography, and magnetic resonance imaging.
Morbidity rates of EAFs are high and are directly related to their pathophysiological consequences, which include severe fluid and electrolyte losses, acid–base homeostasis derangement, hypercatabolism (hypoalbuminemia and hypoproteinemia), vitamin and trace element deficiencies, and septic wound complications due to spillage of enteric effluents on the open abdominal surface. The mortality rates of EAFs were as high as 70% in the past decades but currently are lowered to 42% due to advanced modern ICU and improved surgical techniques (4). However, after a fistula is closed, recurrence is always a probability and involves 9%–33% of spontaneously sealed EAF patients. The severity in means of morbidity and mortality of the EAF is reflected in Bjorck’s (7) classification of the OA reported in 2009, where an OA with an EAF is considered as grade 3, a step just before the frozen and inoperable OA (grade 4).
Principles of Management
Management of patients with an OA and an EAF is very challenging. These patients are usually critically ill and hypercatabolic and deteriorate rapidly if complications occur during their hospitalization in the SICU. Unfortunately, proximal diversion of enteric contents is almost technically impossible to achieve, due to the thick and shortened mesentery, the edematous bowel, the noncompliant abdominal wall and the rather hostile environment of the OA. A multidisciplinary team consisting of experienced senior surgeons, dedicated medical and nurse ICU personnel, experienced stomal therapist, and a plastic surgeon experienced in abdominal wall reconstruction is necessary in order to accomplish this difficult task.
General Support of the Patient
Basic principles of the nonoperative support of the critically ill patient with an EAF are the following:
Early recognition and management of sepsis. Progressive organ dysfunction or failure should be promptly managed according to currently used guidelines for severe sepsis (8, 9).
Source control. Radiologically guided drainage of abscesses or infected collections is crucial for resolution of sepsis and can alter the antibiotic regimen based on cultures results.
Reducing fistula output. Nil per os (NPO), effective drainage of the stomach via a nasogastric tube (Levin) as well as an attempt to reduce GI secretions by administering proton pump inhibitors and to reduce enteric and pancreatic secretions by means of somatostatin or analogs (octreotide) are general measures for reducing the fistula output.
Nutritional support. The patient with an OA is considered hypercatabolic, while the presence of an EAF considerably augments catabolism. Thus, nutritional support should be based upon three main parameters: (a) increased caloric requirements, usually calculated by 30–35 kcal/kg/day, (b) increased protein depriving, calculated by adding 1.5 g protein/kg/day and 2 g protein losses for each liter of fluid collected from the raw surface of the OA, and (c) deficiencies of vitamins and trace elements. Adequate nutritional support based on patient nutritional status, a positive nitrogen balance, adequate trace minerals, and vitamin replacement along with glycemic control may allow the surgeon to proceed to surgical treatment of the fistula. Additionally, several known parameters, such as weight, prealbumin, albumin and transferrin, are correlated with postoperative mortality and morbidity and spontaneous fistula closure rates (10).
Isolation of the Enteric Effluent
Spillage of enteric contents of an EAF on the adjacent OA surface serves as a factor of continuous impairment of the healing process, which aggravates local wound sepsis and is considered a source of major morbidity. Thus, methods of isolation of bowel effluent have been reported and are under study. These are important in order to protect the OA, prevent ongoing sepsis, estimate fluid losses volume and consistency, and facilitate nursing of the patient. These methods are described collectively in a chronological order as follows:
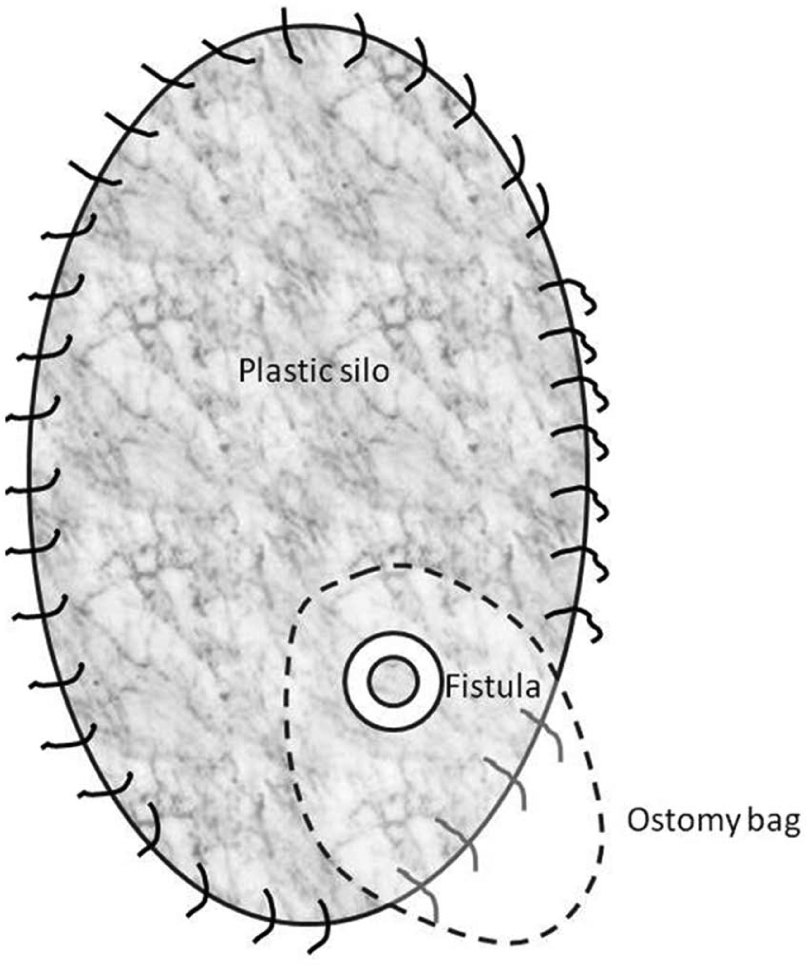
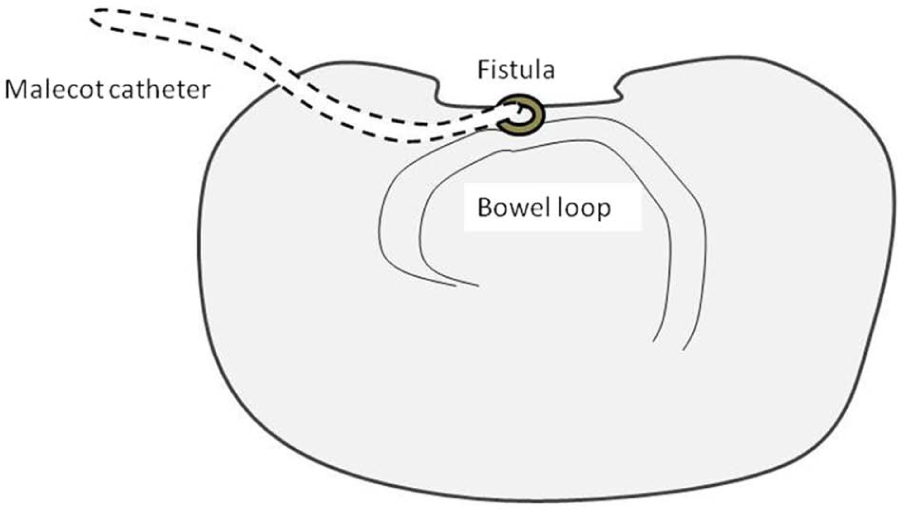
The “floating stoma” (schematic drawing).
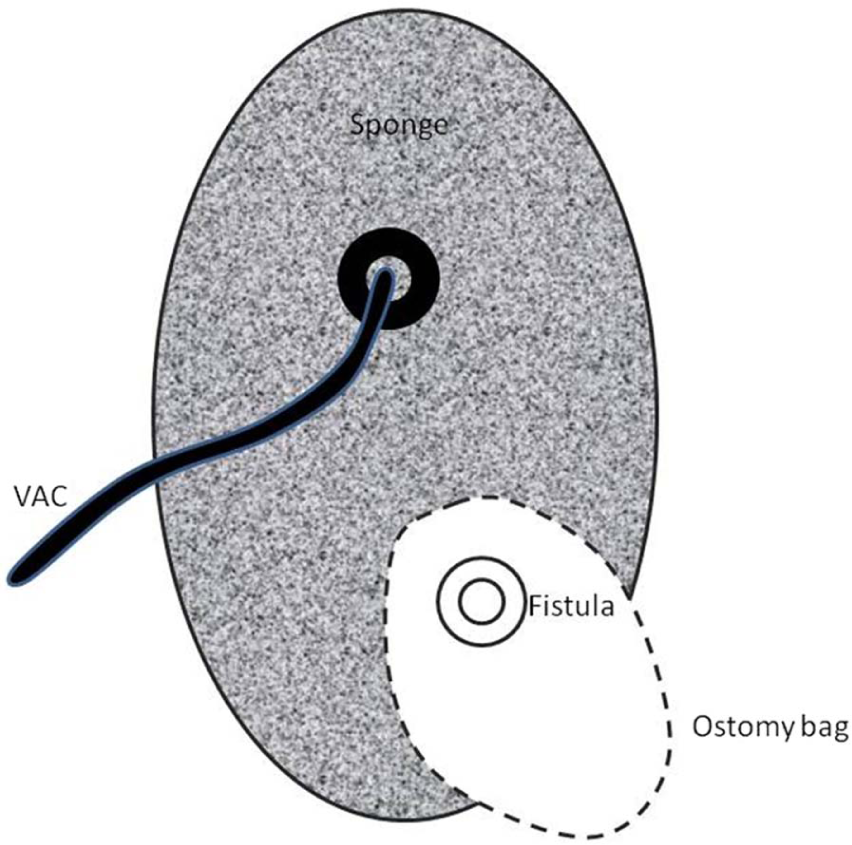
The “fistula VAC” (schematic drawing).
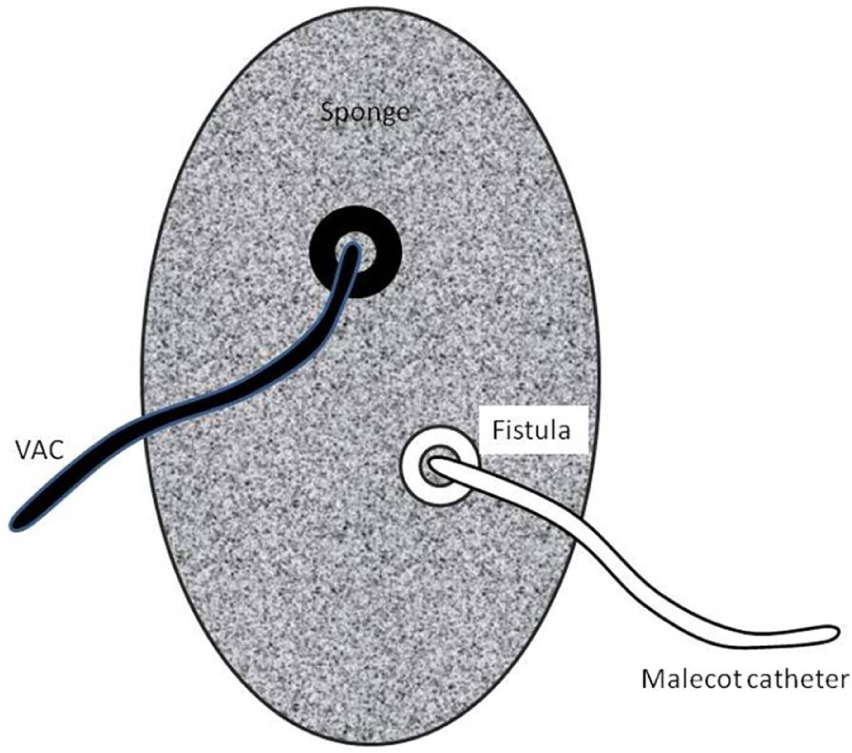
The “tube VAC” (schematic drawing).
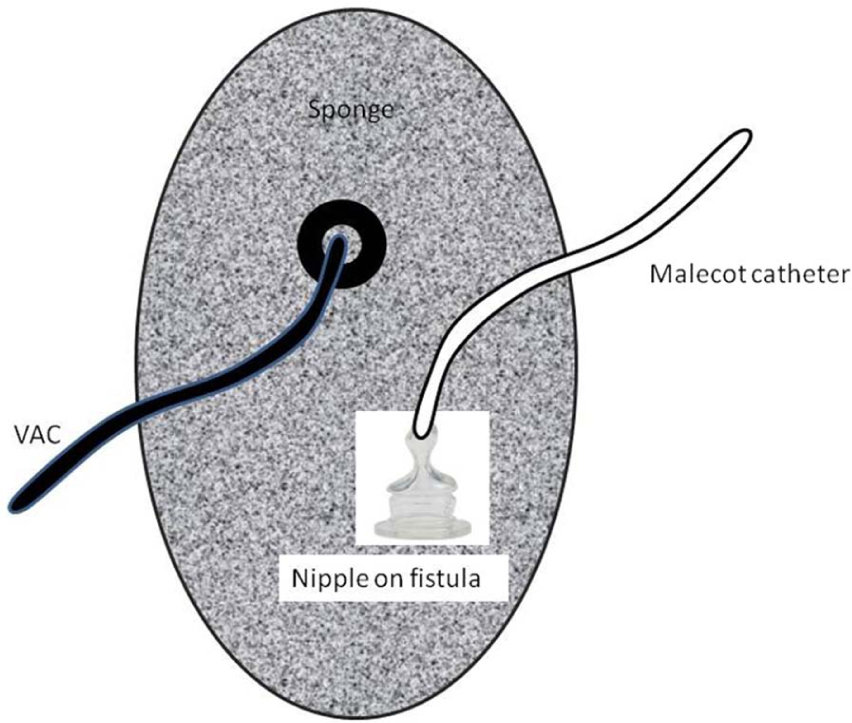
The “nipple VAC” (schematic drawing).
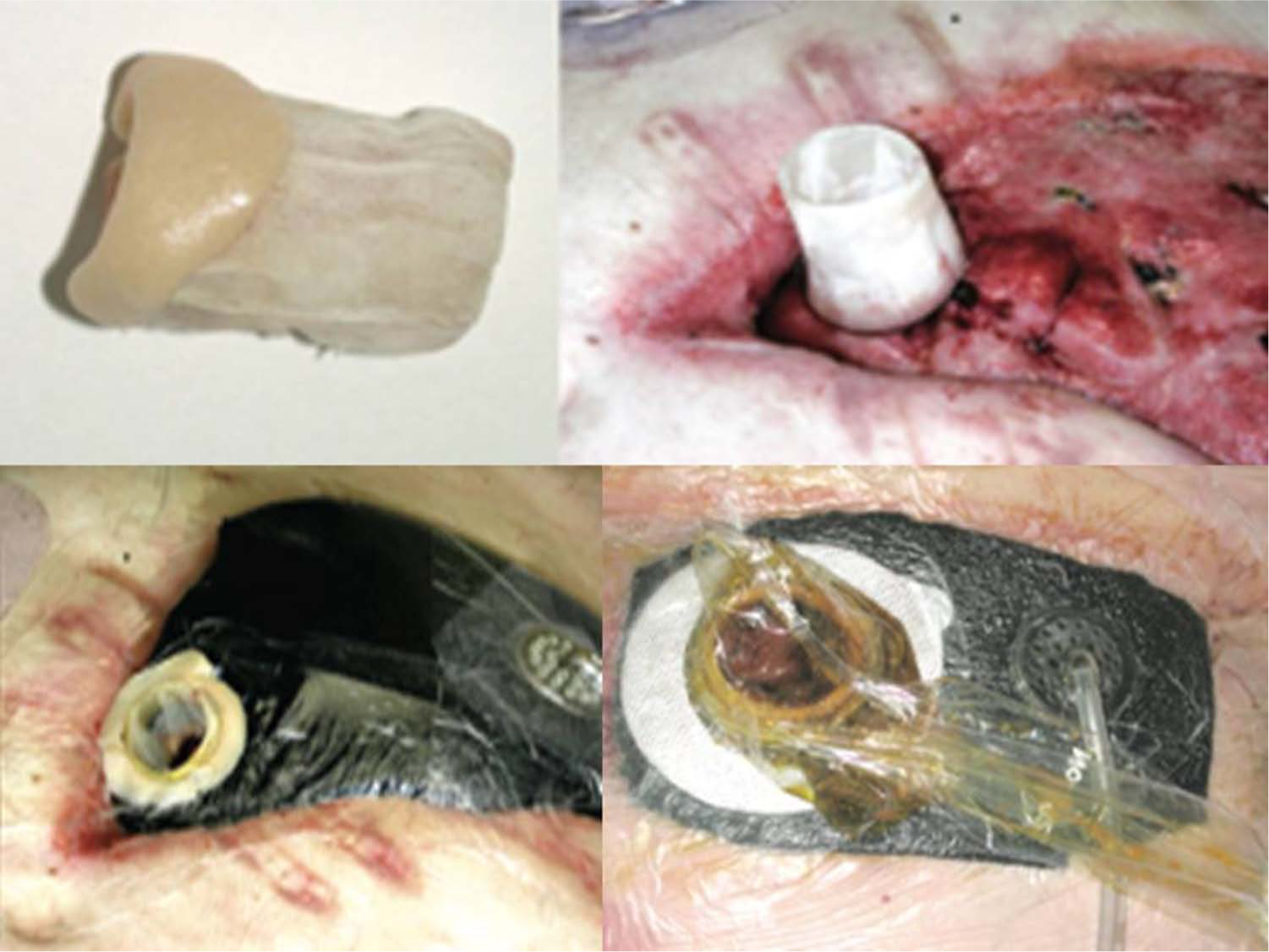
The “silo” method.
Fistula intubation and conversion to an enterocutaneous fistula (schematic drawing).
Techniques To Seal Eafs
After accomplishing the goal of effective diversion of enteric effluents from the granulated surface of the OA, several reports have been published related to efforts made to seal the fistula opening and eventually close it. The use of several biologic dressings such as acellular dermal matrix (Alloderm), STSG, and pedicled (skin or musculofascial) flaps seems to be promising for this goal. These reports are rare and include mainly the following:
The first report was from Girard et al. (20) in 2002, which proposed and applied Alloderm and fibrin glue successfully in three patients.
A preliminary report from Jamshidi and Schecter (21) in 2007 using two components: (a) human acellular dermal matrix applied via fibrin glue on the fistula opening and (b) cadaveric STSG and VAC or pedicled flaps to cover the OA around the fistula; this technique had a 71% success rate.
Resection Of The Involved Enteric Area (Aggressive Intervention)
The resection of the involved enteric loop is actually the most definite way of treating an EAF. However, the surgeon should choose this therapeutic option in clinically stable patients, which should be in good general status and free of infection (4, 5). These conditions are possible to attain in some patients as early as 1–2 months (4, 22), while others accomplish this target after 6–12 months or even after a 1-year period (21). Regarding the operative strategy, the main goal is to approach the peritoneal cavity not directly through the OA but rather with laterally placed incisions away from the granulating surface of the OA. Several reports have been published describing various techniques for approaching and finally resecting the involved bowel loop:
One of the first reports was by Prof. Demetriades (23) in 2003, approaching the abdominal cavity via long vertical incisions approximately 8–10 cm lateral to the open abdominal wound, mobilizing the bowel under direct vision toward the midline, and finally, resecting en masse the involved loop and reestablishing GI continuity with an entero-enteric anastomosis. The abdominal defect was covered by a Marlex mesh and the skin and subcutaneous tissue closed over the mesh.
In 2006, Sriussadaporn et al. (24) reported a case series (eight patients) entering the abdomen with an incision around the granulating tissue of the OA. Once the involved enteric loop was identified and resected (with an anastomosis performed), they closed the open abdominal wound defect with an absorbable mesh (Dexon), which was subsequently covered with bilateral bipedicled anterior abdominal skin flaps (Fig. 7).
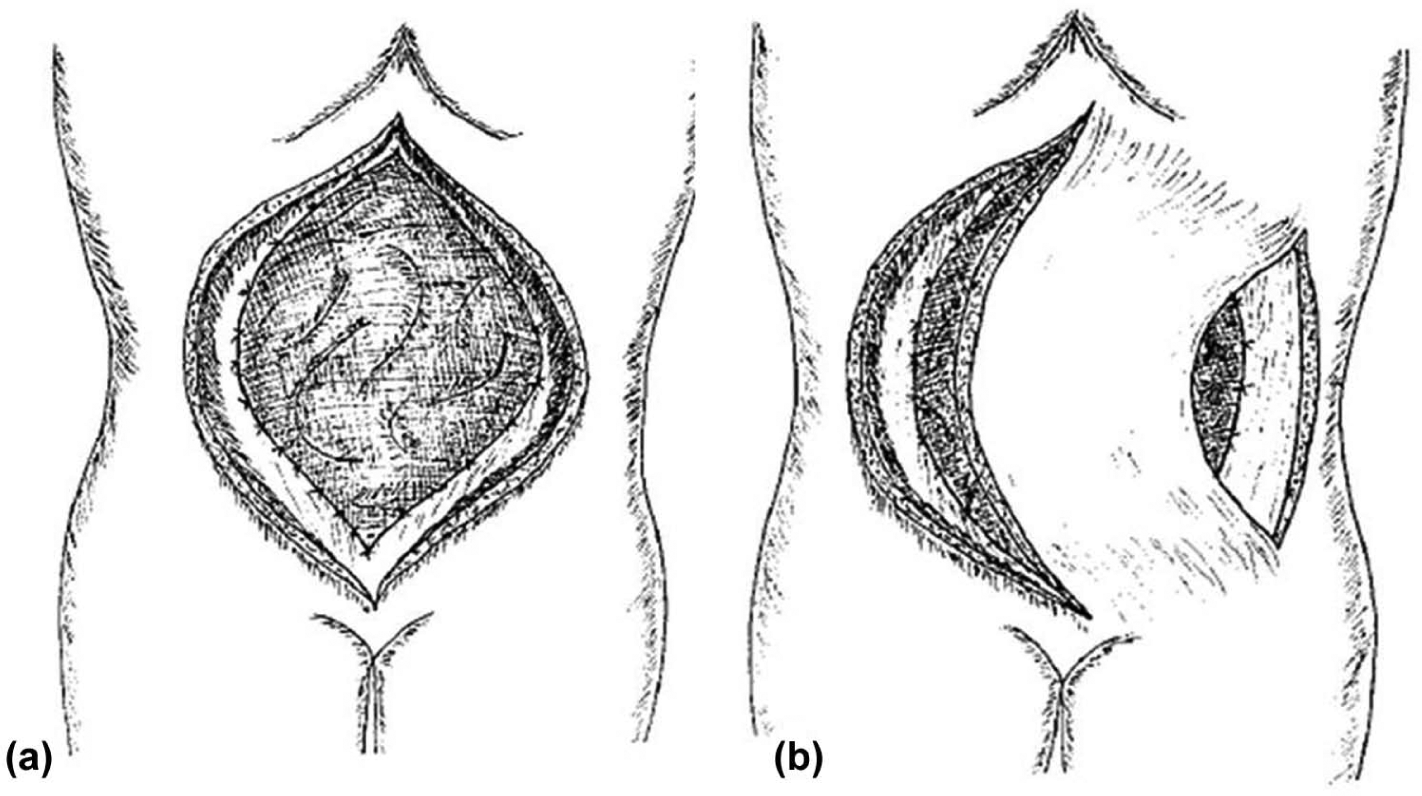
A) Polyglycolic acid (Dexon) mesh sutured to the rectus abdominis muscle. B) Bipedicle anterior abdominal skin flap and relaxing incision at the anterior axillary line.
Our Experience And Proposed Surgical Technique
In our department, we have experienced several cases with EAFs and have proposed a relatively early intervention of accessing the hostile granulated OA (22). Thus, we use a lateral surgical approach via the circumference of the OA in order to avoid the hostile and granulated surface of the abdominal trauma, which is densely adhesed with the intraperitoneal organs (Fig. 8). Immediately after entering the abdomen, the separation of the involved loops is performed relatively easy. We have used this technique successfully in three of our patients (Table 1). After entering into the abdominal cavity, an enterectomy of the associated enteric loop was performed, taking down the EAF. The abdominal defect was closed by an absorbable mesh, which was either left free to granulate as was the case in two patients or a VAC device was applied in one patient. Finally, an STSG covered efficiently the granulated tissue. In the fourth patient (Table 1), an EAF occurring in the lateral edge of the abdomen was managed with topical measures (suture and biological glue), with cessation of the EAF and finally, coverage with an STSG. All patients had an eventful outcome.
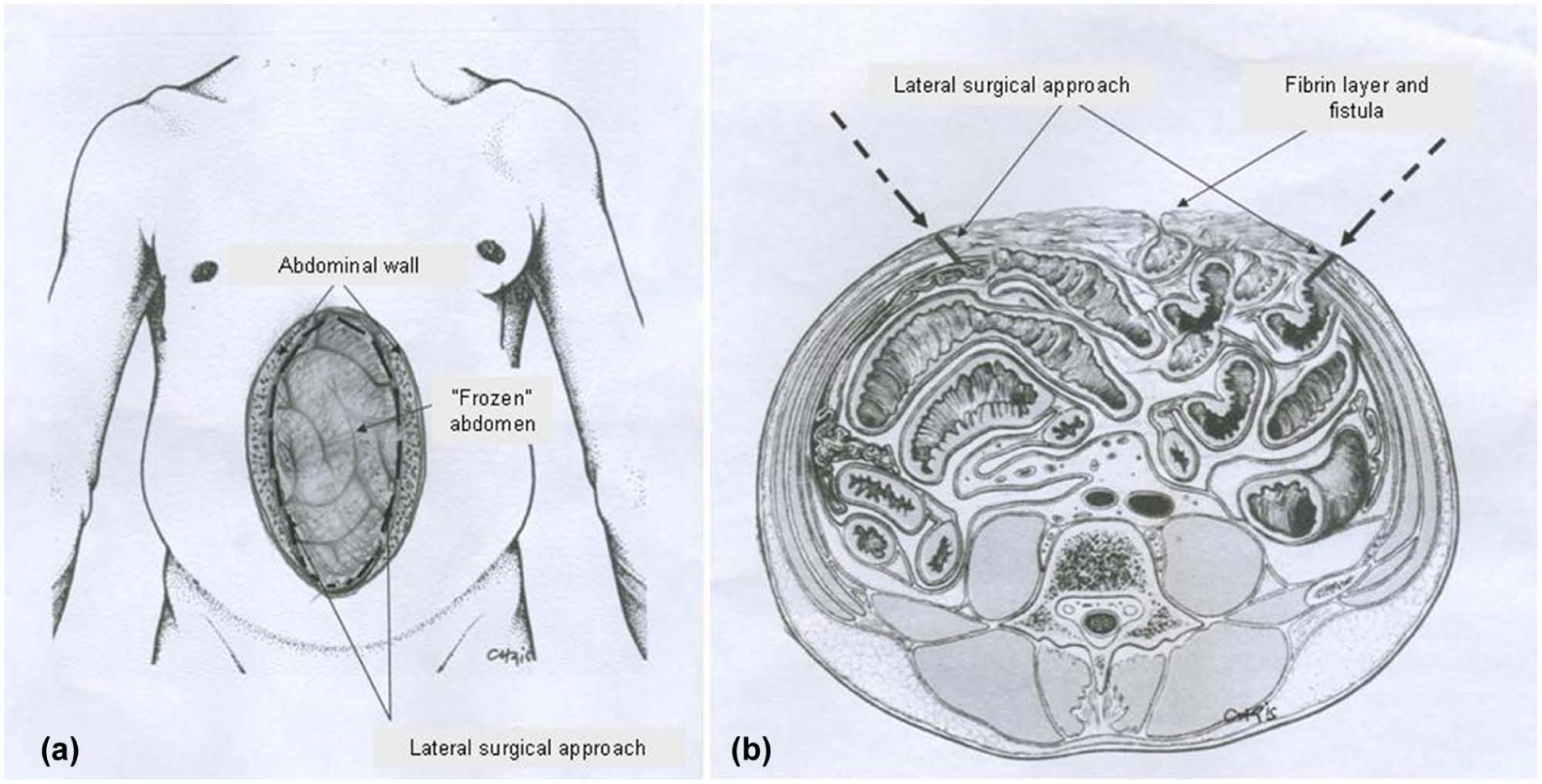
A) Safe dissection via circumference of abdominal wound. B) Lateral surgical approach.
Epidemiologic, clinical data, etiology, treatment, and LOS of our patients with EAF.

LOS: length of stay; EAF: enteroatmospheric fistulae; STSG: split-thickness skin graft; VAC: vacuum-assisted closure.
Regarding the extent of bowel resection, the surgeon aims to resect as less bowel as possible, keeping in mind that this operation is mainly a life-saving procedure. The possibility of developing a short bowel syndrome and its associated nutrient malabsorption is a problem of secondary priority, which can be managed today with specialized methods of nutritional support (low fiber and elementary diets, etc.) (10).
The incidence of developing a new EAF when VAC is applied reaches an average of 5.7% (ranging from 0% to 15%) (13, 25, 26). If VAC is directly applied upon the exposed bowel, a new EAF will most probably develop, further leading to increased mortality (27); if VAC is directly applied upon the EAF, it will probably not close. But if the fistulous tract is indirect, meaning that it is deeper seated within the OA, the application of VAC will possibly lead to closure of the fistula (22). Thus, after resecting the involved bowel, the anastomotic line should preferably be placed deep in the OA, in order to apply the VAC on the free surface. The use of a stapled anastomosis is generally avoided in order to gain bowel length (28) and could raise some concern regarding the final stapling line with the TA™ used to close the entrance openings on the bowel for the GIA™ stapler. The TA staple line is everted and may be prone to higher leakage in the case of negative pressure application, in contrast to the inverted hand-sewn anastomosis. However, placement of the final anastomosis far away from the surface where negative pressure is directly applied will avoid anastomotic leakage and subsequent development of a new fistula.
Prevention of Eafs
The high morbidity and mortality associated with the EAFs dictate the need to prevent the development of this very hazardous complication. Several principles are considered as cornerstones of prevention (5, 29, 30):
Careful manipulation of the bowel during initial operation is very important to avoid deserosalizations, which could possibly burst postoperatively, leading to the development of an enteric fistula.
The intestine in patients with OA should be meticulously protected and covered by means of early temporary abdominal closure techniques.
Access to the OA should be permitted only to one or two experienced senior surgeons who know the wound and are familiar with the healing process. Free access to the wound by all members of the medical and nursing staff almost guarantees the development of an EAF.
Conclusions
The EAFs develop in the middle of the OA and are associated with high morbidity and mortality. The patient should be managed by a multidisciplinary team usually in a surgical ICU where the goal of optimization of patient’s general condition should be strongly considered. Thorough knowledge of manipulating the fistula, namely, diversion of enteric effluents, sealing the fistula, and resection of the involved enteric loop, is critical for the outcome of these patients. Usually, small and low-output EAFs are covered by biologic dressings in order to spontaneously heal, while larger and high-output EAFs are managed by using diversion techniques in combination with VAC therapy (“fistula or tube or nipple VAC”). Finally, non-responsive EAFs should be resected when the patient is in a stable clinical condition and is free of infection. The comprehension of the recently accepted total OA management (6) principle of “open early and close early” has led to a significant reduction of complications such as EAFs and a higher rate of early primary closure.
Footnotes
Acknowledgements
The authors would like to thank the editors who provided permission to use figures for this article: (a) Prof. Po-Huang Lee, Editor-in-Chief, Asian Journal of Surgery, (2) Michael McGovern, Managing Editor, WOUNDS, and (![]() ) Jason B. Schappert, Reprint Coordinator, Mary Ann Liebert Inc.
) Jason B. Schappert, Reprint Coordinator, Mary Ann Liebert Inc.
