Abstract
Background and Aims:
This study was conducted to clarify the effects of age on human liver regeneration.
Patients and Methods:
Thirty major hepatectomies, equal to or more than two segmentectomies for hepatocellular carcinoma, were performed. Ages ranged from 37 to 85 years and five octogenarians were included. The early regenerative index was defined: (liver volume after 7 days after hepatectomy − estimated remnant liver volume before hepatectomy)/estimated remnant liver volume, using three-dimensional computed tomographic volumetry. Farnesoid X receptor and forkhead box m1 expression in the liver, which has been reported to age-related decrease of liver regeneration in animal model, were examined using real-time polymerase chain reaction. The patients were divided into two groups: low early regenerative index (n = 15), early regenerative index less than 55% and high early regenerative index (n = 15), early regenerative index equal to or more than 55%.
Results:
The mean early regenerative index was 57%. Age (R2 = 0.274, P = 0.003) and estimated blood loss (R2 = 0.134, P = 0.0466) were inversely correlated with the early regenerative index, and the expression of farnesoid X receptor and forkhead box m1 was not. The incidence of posthepatectomy liver failure in the low early regenerative index group was higher than that in the high early regenerative index group (P = 0.0421).
Conclusions:
Age and intraoperative blood loss are inversely correlated with early liver regeneration in humans. In elderly patients, massive blood loss should be avoided in view of liver regeneration.
Introduction
The liver is a unique organ that is able to regenerate itself after surgical resections (1–3). The loss of regenerative capacity is the most dramatic age-associated alteration in the liver. The regenerative capacity of the liver has been widely proven in experimental animal models. Although this phenomenon in experimental animal models was reported over 40 years ago (4), aging and liver regeneration in humans are not fully understood. The effects of liver cirrhosis (5) and portal pressure (6) have been reported, but the effect of age has not been well studied. This may be because patients who underwent hepatic resection were not very old in previous studies.
In developed countries, the number of elderly patients with hepatobiliary malignancies has increased due to life expectancy being prolonged (7). In hepatocellular carcinoma (HCC), Wu et al. (8) reported zero mortality in 21 patients over 80 years of age who underwent hepatic resection. The authors also showed that more than 10% of the patients who underwent hepatic resection were octogenarians, and they demonstrated the feasibility of hepatic resection in patients over 80 years of age (9). Nevertheless, the number of major hepatectomies in these patients is limited, and liver regeneration in elderly patients remains unclear.
The mechanism of reduced cellular proliferation during aging remains unclear, even in animal models. Reduced cellular proliferation during aging has been reported to be associated with a progressive decline in both growth hormone secretion and forkhead box m1 (Fox M1B) expressions (10). Thus, one of the causes of decreased liver regeneration in elderly animals is reported to be decline in Fox M1B expression. Recently, Chen et al. (11) showed that Fox M1B is a target gene of the farnesoid X receptor (FXR). Taken together, these findings suggest that both FXR and Fox M1B may be related to deterioration of liver regeneration in elderly patients.
A retrospective study was carried out at our institution to investigate the correlation between liver regeneration and aging. Furthermore, the expression of FXR and Fox M1B was examined in these patients.
Patients and Methods
Patients
Between October 2005 and March 2011, 30 patients who underwent major hepatectomies for HCC at Kyushu University Hospital were enrolled into this study. The inclusion criteria were (1) major hepatectomies, which were equal to or more than two segmentectomies for HCC and (2) frozen tissue for reverse transcription polymerase chain reaction (RT-PCR) was available. The patient backgrounds are shown in Table 1. The mean age was 68 years, and it ranged from 37 to 85 years.
Clinicopathological background.
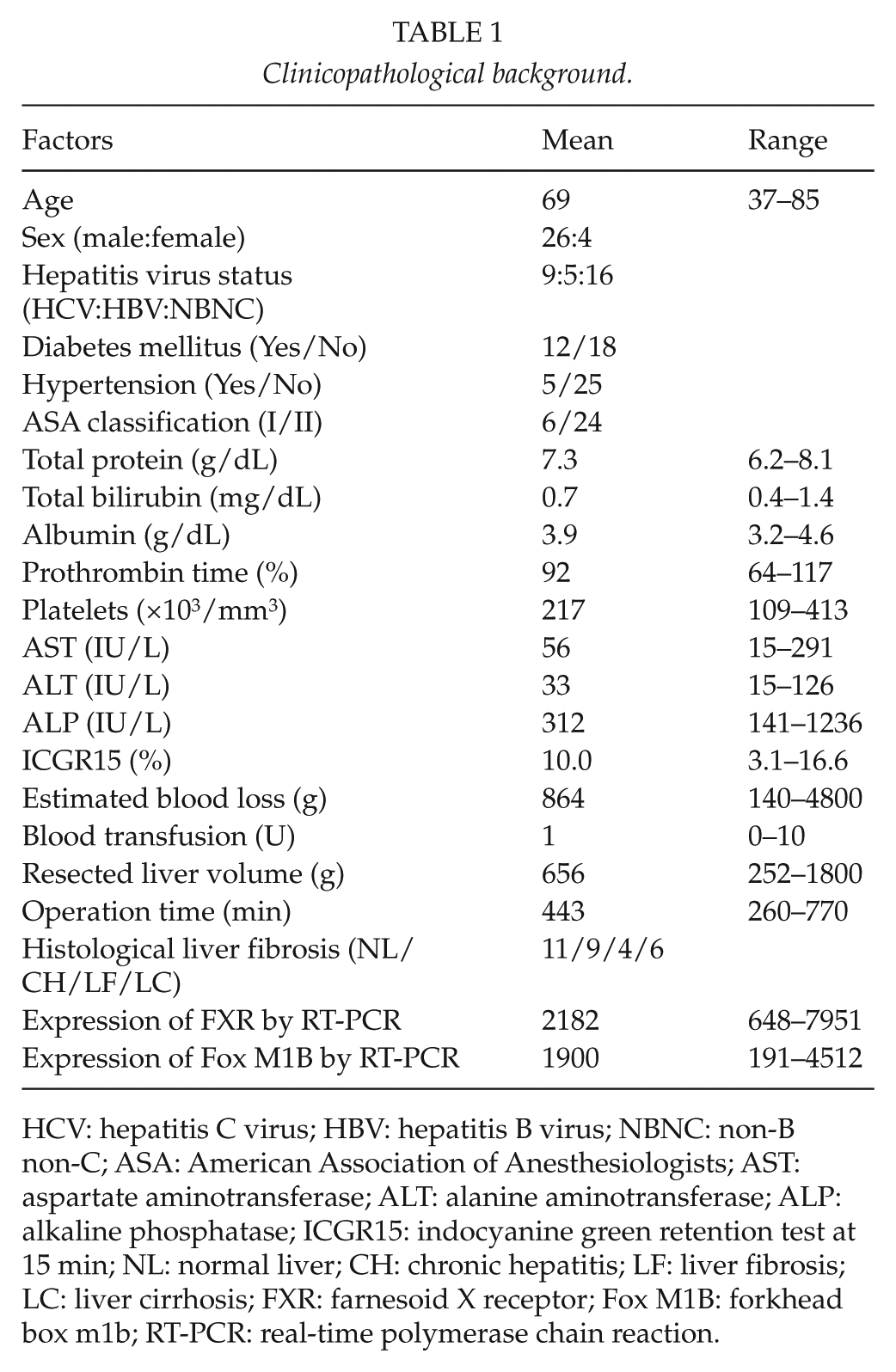
HCV: hepatitis C virus; HBV: hepatitis B virus; NBNC: non-B non-C; ASA: American Association of Anesthesiologists; AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase; ICGR15: indocyanine green retention test at 15 min; NL: normal liver; CH: chronic hepatitis; LF: liver fibrosis; LC: liver cirrhosis; FXR: farnesoid X receptor; Fox M1B: forkhead box m1b; RT-PCR: real-time polymerase chain reaction.
Methods
Criteria for hepatic resection
The criteria for hepatic resection for HCC were as previously described (12). The surgical procedure was selected according to the following criteria: trisegmentectomy for indocyanine green retention test at 15 min (ICGR15) < 15% and bisegmentectomy for ICGR15 < 25%.
Early regenerative index
Volumetric measurements were performed as previously reported (13). Total volumes and segmental volumes were measured for total liver, future liver remnant (FLR), and liver remnant (LR). The total and segmental early regeneration indexes, defined as [(VLR − VFLR)/VFLR)] × 100, where VLR is the volume of the LR and VFLR is the volume of the FLR, were calculated. Preoperative multidetector helical computed tomography (MDCT) images were performed with 2-mm-thick slices represented on computed tomography (CT) machines, as previously reported (14). Enhancement was achieved by an intravenous bolus of a contrast nonionic medium (Iopamion, Schering, Erlangen, Germany) at a speed of 5 mL/s. Three-dimensional (3D) reconstruction of the liver and graft was obtained from the MDCT data with the Zio M900 (Zio Software, Inc., Tokyo, Japan).
Postoperative course
Posthepatectomy liver failure was defined as serum levels of total bilirubin 3.0 mg/dL or a prothrombin time less than 50% on the fifth operative day, according to Figueras et al. (15). Postoperative complications included surgical site infection, bile leakage, and prolonged hyperbilirubinemia.
To examine the effects of liver regeneration on the postoperative course, the patients were divided into two groups, according to the median value of early regenerative index (ERI). The low ERI (n = 15) group included patients with an ERI less than 55% and the high ERI (n = 15) group included those with an ERI equal to or more than 55%. The postoperative clinical course was compared between the two groups.
Quantification of messenger RNA of liver tissues using real-time quantitative RT-PCR
Total RNA was isolated from noncancerous tissues of HCC patients using an ISOGEN (Nippon Gene, Tokyo, Japan) according to the manufacturer’s instructions. The complementary DNA (cDNA) was synthesized with random hexamer primers and Superscript III reverse transcriptase according to the manufacturer’s instructions, and the product was used for further analysis. Fox M1B and FXR transcriptions were quantified using the LightCycler (Roche Molecular Biochemicals, Mannheim, Germany) polymerase chain reaction (PCR) protocol, in which fluorescence emission is attributable to binding of SYBR Green I dye to amplified products and it can be detected and measured. The relative quantization value is expressed as 2-Ct, where Ct is the difference between the mean cycle threshold (Ct) value of triplicates of the sample and of the endogenous β-actin control (16). The primer sequences for real-time RT-PCR were as follows: Fox M1B, 5′-CGTGGATTGAGGACCACTTT-3′ (forward) and 5′-TCTGCTGTGATTCCAAGTGC-3′ (reverse); FXR, 5′-ATCAAAGGGGATGAGCTGTG-3′ (forward) and 5′-AAGCATTCAGCCAACATTCC-3′ (reverse); and β-actin, 5′-CTGGCACCACACCTTCTACAATG-3′ (forward) and 5′-GGCGTACAGGGATAGCACAGG-3′ (reverse).
Statistical analysis
Values are presented as means and standard deviations. A regression test was used to examine the relation between ERI and other clinicopathological factors. Continuous data were analyzed with the Student’s t test, and categorical data were analyzed with the chi-square test. Any variable identified as P < 0.05 on univariate analysis was considered a candidate for multivariate analysis. StatView software (version 4.11; Abacus Concepts Inc., Berkeley, CA) running on a Macintosh computer was used for the adjustment of all covariates and stepwise regression analyses. The values of P < 0.05 were considered statistically significant.
Results
The clinicopathological background is shown in Table 1. A total of 31 patients underwent hepatic resections for HCC. In all 31 patients, the mean ERI was 57.4%, and it ranged from 28.6% to 157.3%. Regression tests showed correlations between ERI and factors related to ERI (Table 2). Age was inversely correlated with ERI (Fig. 1A) (R2 = 0.274, P = 0.0030). The estimated blood loss was inversely correlated with ERI (Fig. 1B) (R2 = 0.134, P = 0.0446). There were no statistically significant factors among other clinicopathological factors.
Clinicopathological factors linked to the ERI.
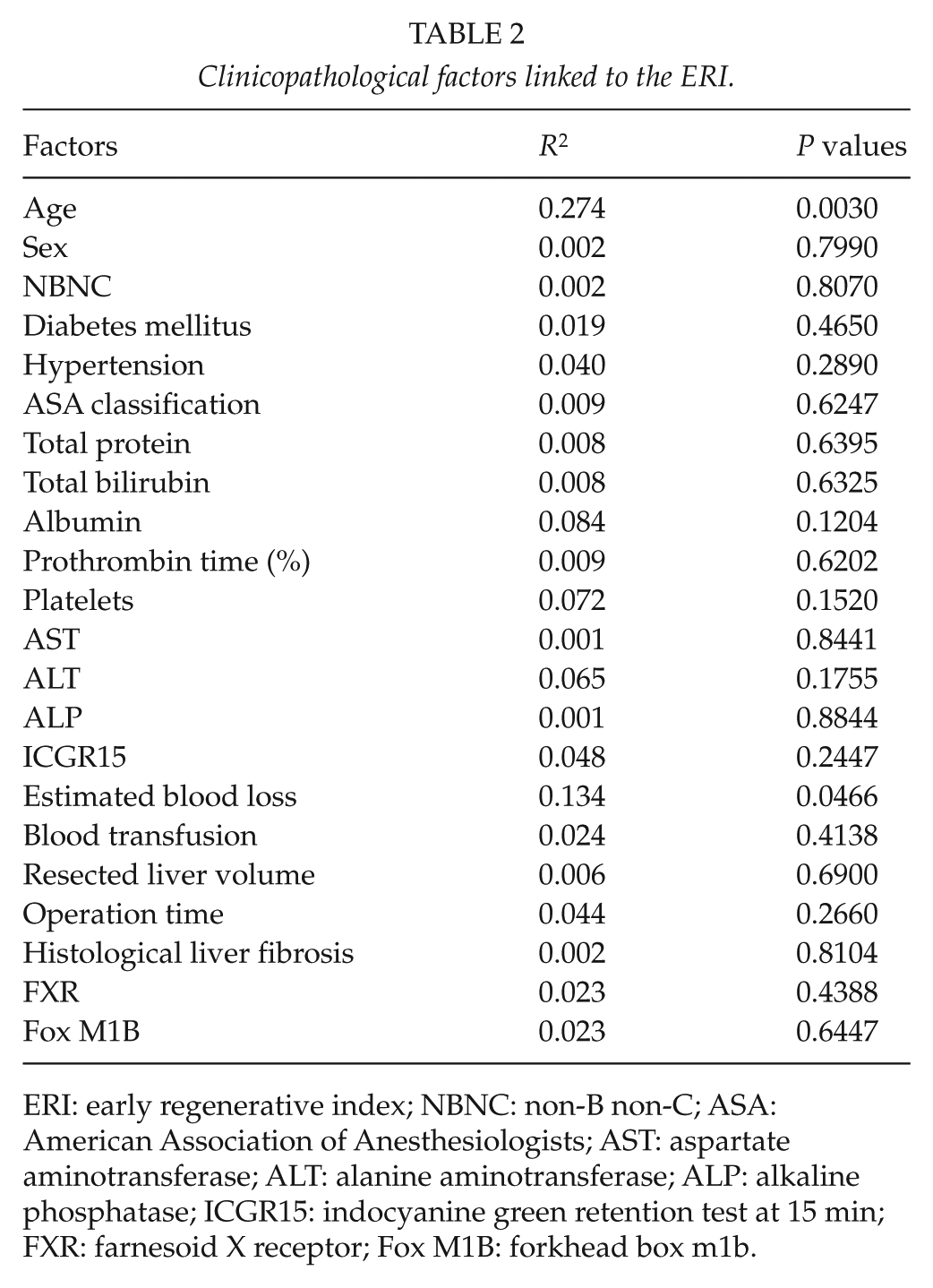
ERI: early regenerative index; NBNC: non-B non-C; ASA: American Association of Anesthesiologists; AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase; ICGR15: indocyanine green retention test at 15 min; FXR: farnesoid X receptor; Fox M1B: forkhead box m1b.
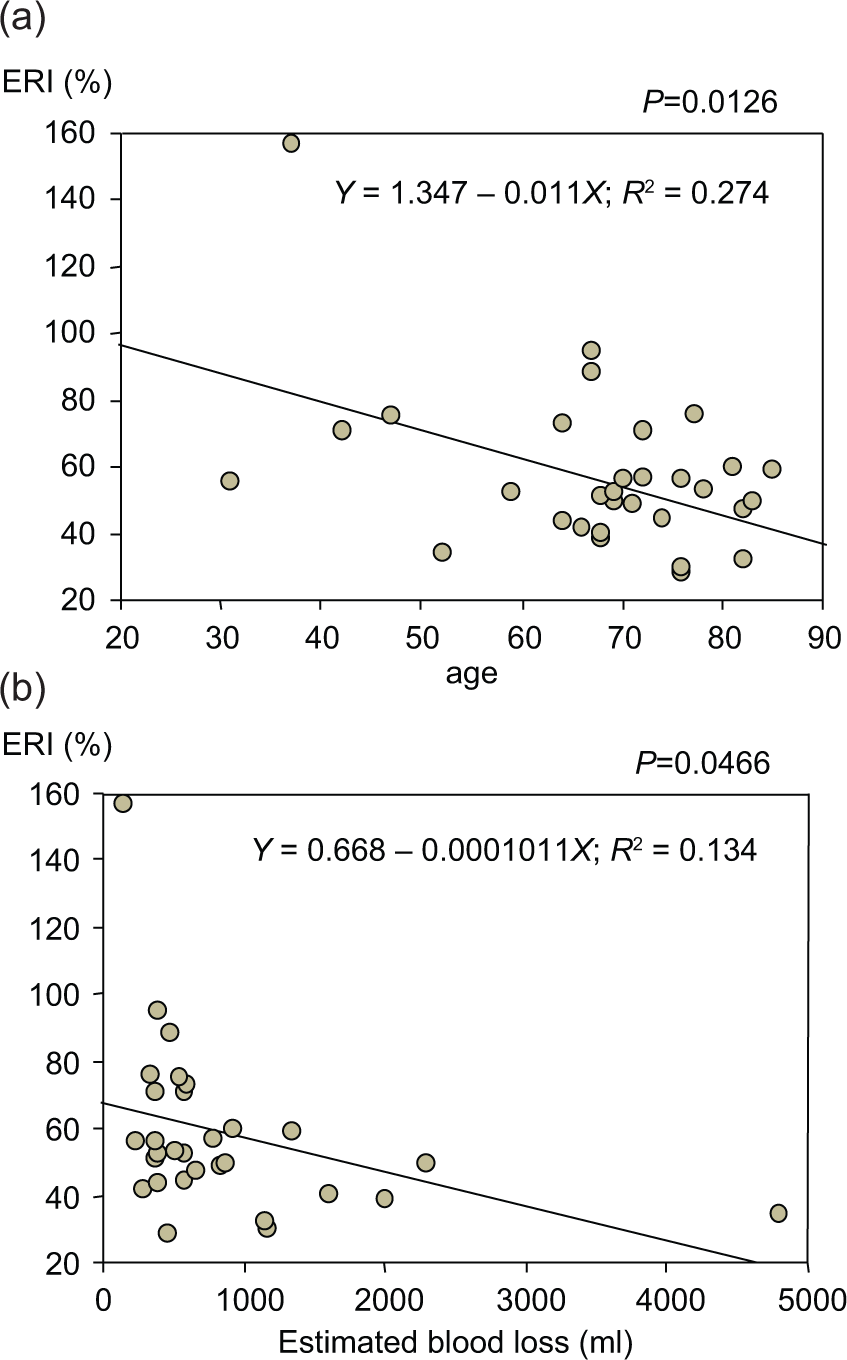
A) Age was inversely correlated with ERI (R2 = 0.274, P = 0.0030). B) the estimated blood loss was inversely correlated with ERI (R2 = 0.134, P = 0.0446).
The correlation between FXR and Fox M1B messenger RNA (mRNA) expressions and that between ERI and FXR and Fox M1B mRNA, and between age and FXR and Fox M1B mRNA was examined. The mRNA values of FXR were not correlated with Fox M1B (data not shown). Either the FXR mRNA values or the Fox M1B mRNA values were not correlated with ERI (Table 2). Furthermore, either the FXR mRNA values or the Fox M1B mRNA values were not correlated with age (data not shown).
Comparison of the postoperative clinical course between the low and high ERI groups is shown in Table 3. Serum total bilirubin levels at day 7 after hepatectomy tended to be higher in the low ERI group than those in the high ERI group (P = 0.0943), and prothrombin time was significantly lower in the low ERI group than that in the high ERI group (P = 0.0389). The incidence of posthepatectomy liver failure was compared between the low and high ERI groups. The incidence of liver failure was significantly higher (5 of 15, 33%) in the low ERI group than in the high ERI group (0 of 15, P = 0.0421).
Outcome after hepatectomy and ERI.
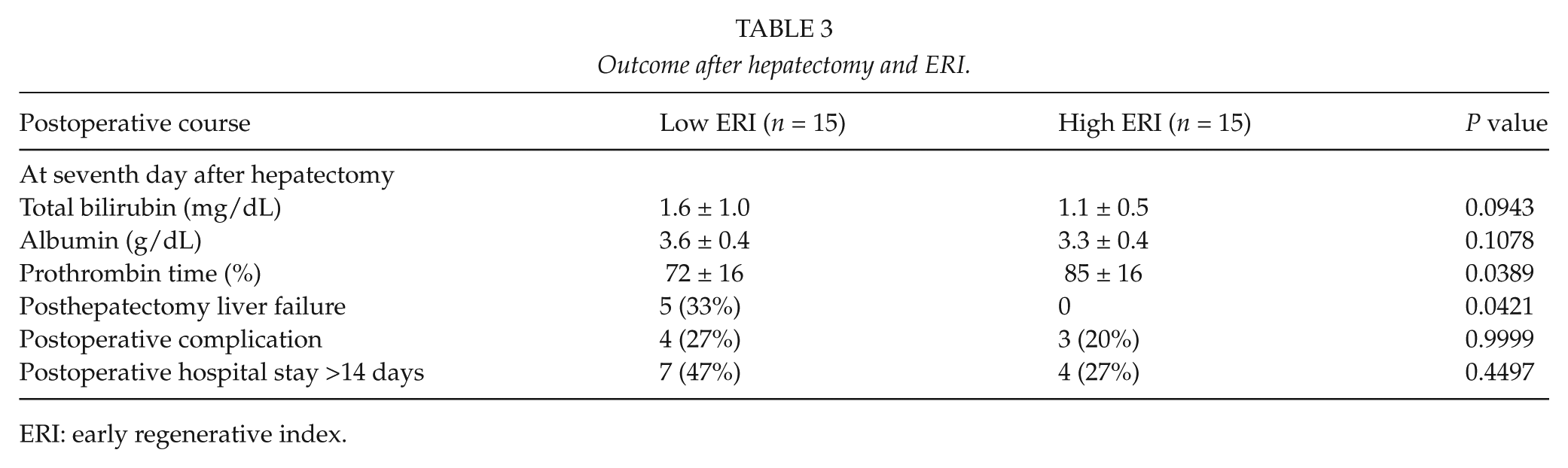
ERI: early regenerative index.
Discussion
In this study, we evaluated liver regeneration 7 days after hepatectomy. Most previous studies (5, 17, 18) evaluated regenerative liver volume at 1, 3, or 6 months, and Akamatsu et al. (19) showed that changes in liver volume between 1 and 3 months are minimal, indicating that liver regeneration occurs within 1 month. Pomfret et al. (20) showed that most liver regeneration occurs within 1 week, and several studies showed that the ERI at 7 days after hepatectomy is critical for liver regeneration. Furthermore, most postoperative liver failure occurs within 1 month (21). In our study, the incidence of delayed liver dysfunction was significantly higher in the low ERI group compared with that in the high ERI group. This result clearly shows that ERI is a good predictor for clinical course in hepatectomized patients.
Mean ERI was 57.3% in the current study. This result is similar to that in the previous studies, which showed 64% (20) and 59.8% in females and 61.5% in males (13). In our study, 3D CT volumetry was used for evaluation of the ERI. This method has been reported to be good for estimating volumetry.
There are many studies regarding aging and liver regeneration in the animal experimental setting. Nevertheless, there are only a few studies in the clinical setting. Shimada et al. (17) showed that age was inversely associated with liver regeneration in patients who underwent right lobectomy, and Yamamoto et al. (18) showed no association between age and liver regeneration in elderly patients. Therefore, liver regeneration and aging are still controversial in the clinical setting. In our study, liver regeneration gradually and significantly decreased with age, and this may be because our study included very high-age patients who were octogenarians.
The mechanism of aging deteriorating liver regeneration remains unclear. Forkhead box transcription factors are an extensive family of transcription factors, consisting of more than 50 mammalian proteins (22), which share homology in the winged helix DNA-binding domains (23). Previous liver regeneration studies (10, 11, 16) have shown that increased hepatocyte expression of Fox M1B alone in elderly mice was sufficient to stimulate regenerating hepatocyte DNA replication and mitosis to levels found in young regenerating mouse liver. Fox M1B is reported to be a target gene for FXR. Furthermore, growth hormone stimulates proliferation of old-aged regenerating liver through Fox M1B (23). Nevertheless, there was no significant correlation between preoperative FXR and Fox M1B gene expression and liver regeneration in this study. Fox M1B expression is induced by greater than 40-fold during cellular proliferation (24, 25). Therefore, activation of these genes during liver regeneration, but not preoperative levels of these genes before hepatectomy, may be important. Further studies are required to determine this issue.
Another important factor related to liver regeneration was estimated blood loss during hepatic resection in this study. Previous studies have shown that cytokines and humoral factors, such as interleukin 6, hepatocyte growth factor, and growth hormone, are important in the liver regeneration (26). Reduced humoral factors after a significant amount of blood loss may be related to decreased liver regeneration. Nevertheless, Ikegami et al. (27) demonstrated that liver regeneration of liver grafts from elderly donors was decreased in living-related donor liver transplantation compared with liver regeneration in liver grafts in young donors. This result suggests that the humoral factors may not be important in liver regeneration.
Nagasue et al. (5) showed that decreased liver regeneration is related to liver cirrhosis. Shimada et al. (17) demonstrated that intraoperative portal pressure is significantly correlated with liver regeneration. However, in our study, histological fibrosis was not related to liver regeneration. Under strict patient selection, histological fibrosis may not have an effect on liver regeneration.
In conclusion, liver regeneration occurs early, already during the first week after surgery. The independent factors related to liver regeneration were aging and estimated blood loss during hepatic resection. Massive blood loss should be avoided in elderly patients because liver regeneration might be suppressed.
Footnotes
Declaration of Conflicting Interests
Ken Shirabe and other coauthors have no conflict of interest.
Funding
This study was supported in part by 2010 Research Award of Japanese Society for Advancement of Surgical Techniques.
