Abstract
The use of free vascularised bone grafts is an infrequently performed surgical technique for the reconstruction of spinal defects. This field of surgery brings many challenges concerning the choice of free vascularised bone graft, planning of the operative procedure and selection of recipient vessels. This study aims to report our experience with free vascularised bone grafts, with special emphasis on the surgical approach and the selection of recipient vessels.
Over a period of 17 years (1994–2011), we used these grafts for anterior spinal reconstruction in 30 patients. In 28 patients, a free vascularised fibular graft was used, and in two cases a free vascularised iliac crest graft was used. The spinal segments reconstructed involved the cervical or cervicothoracic spine (6 cases), the thoracic spine (11 cases) and the thoracolumbar and lumbosacral spine (13 cases). Revascularisation of the free vascularised bone graft proved to be technically feasible in 30 patients, but failed in one fibular graft due to difficulties with recipient vessels in the lumbar region. Technical challenges were met with respect to the choice of the recipient vessel at various anatomical sites. Availability of acceptor vessels was highly dependant of the type of surgery (resection or stabilisation) and the selected surgical approach. Based on these findings, a preferred approach is given for each region.
The use of free vascularised bone grafts is a valuable technique for the reconstruction of complex spinal disorders. Successful execution requires microvascular expertise with respect to graft harvesting and appropriate choice of recipient vessels. Adequate preoperative planning in a multidisciplinary setting and adherence to the basic principles for spinal reconstruction are required.
Keywords
INTRODUCTION
The use of free vascularised bone grafts (FVBGs) was introduced by Taylor in 1975 (22), and is currently a well-accepted and frequently used surgical technique in the reconstruction of large skeletal defects (15, 29). The advantages of vascularised over non-vascularised bone grafts include rapid consolidation, resistance to infection, hypertrophic reaction guided by mechanical load and tolerance of therapeutic levels of radiation (6, 12, 21, 30).
FVBGs maintain their cellular viability as a result of restoration of endosteal and periosteal blood flow. Therefore, revascularised bone grafts participate actively in bony union and provide fast and reliable periosteal growth and healing, comparable to fracture healing under physiological conditions. In spinal surgery, FVBG are used for the reconstruction of complex primary or secondary defects and/or loss of structural integrity of one or more spinal segments that require adequate, long-term, mechanical stability. The indications include progressive symptomatic spinal deformities, trauma, (chronic) infections and vertebral corporectomies for primary or metastatic spinal tumours (10, 19, 31). Additional anterior and/or posterior instrumentation is necessary to ensure initial stability.
In spite of the superior quality of vascularised bone in the reconstruction of skeletal bony defects, reports on use of FVBGs for spinal reconstructive surgery are limited (1, 11, 19, 24). This is not surprising, since there are many difficulties relating to the preoperative planning and the surgical technique of the use of FVBGs in spinal reconstructions.
For the reconstructive surgeon, this field of surgery brings many challenges concerning choice of FVBG, planning of the operative procedure, technical aspects of inserting and securing the graft and specially selection and dissection of recipient vessels and revascularisation. This study aims to report our experience with FVBGs in spinal reconstruction in 30 patients over a period of 17 years, with special emphasis on the selection of acceptor vessels.
The techniques used for instumentation and fixation of the spine do not differ significantly from other forms of spinal surgery and are therefore not included in this paper.
MATERIALS AND METHODS
Thirty patients underwent spinal reconstruction with the aid of FVBGs over a period of 17 years (1994–2012). All FVBG transfers and revascularisations were performed by one of the authors (HW) in close collaboration with spine surgeons and general surgeons. Follow-up data were obtained from the patients' medical files.
PLANNING AND SURGICAL TECHNIQUE
In the first 2 cases, an iliac crest FVBG was used. Thereafter, the fibula was used as FVBG because of its superiority in terms of mechanical quality, quantity and donor-site morbidity (28). To establish the suitability of the fibula for vascularised transfer, a preoperative vascular analysis of the peroneal vessels was performed in all cases by angiography or duplex ultrasonography. Surgical procedures included anterior, anterolateral, lateral or posterior spinal approaches of the cervical, thoracic, thoracolumbar or lumbosacral spine. The reconstructive surgeon participates in the spinal approach to the extent that the recipient vessels are selected and dissected as early in the operation as possible. Both arterial and venous recipient vessels are selected and, if necessary, dissected carefully. These vessels are then guarded throughout the duration of the surgical procedure. This goes in particular when intercostal or lumbar vessels are selected as acceptor vessels. During subsequent resection of the affected vertebral bodies, the free vascularised iliac crest graft or the free vascularised fibular graft is prepared and harvested simultaneously from the iliac crest or lower leg. The FVBG is left attached to its vascular pedicle until the moment of actual transfer (29). The length of the FVBG is accurately measured depending on the size of the spinal defect. When necessary, the fibula is osteotomised and folded in a double-, triple- or quadruple-barrel construct to increase the graft strength (Fig. 1). When osteotomising and folding the fibula, sometimes a small (1 cm) segment of fibula needs to be removed to facilitate a smooth bend in the vascular pedicle. Depending on the selected recipient vessels, the fibular graft will be positioned with the vascular pedicle ending in the direction of the recipient vessels. Additional anterior spinal instrumentation is applied prior to performing the vascular anastomoses in order to eliminate the risk of kinking or compression of the vascular pedicle during instrumentation. Either end-to-end or end-to-side arterial and venous anastomoses are performed using an operation microscope or 4.5× ocular loupes.
In most cases – independent of the level – additional stability is created with instrumentation through a separate dorsal approach (Fig. 2). This can be done at the same time as the resection and reconstruction, or at an earlier stage. The two-step method has multiple advantages, but is not always possible. Technique and timing of the dorsal fixation is beyond the scope of this paper as we aim to focus on the microsurgical reconstructive procedure, with special emphasis on surgical approach and acceptor vessels. In contrast to the FVBG technique for reconstruction of bone defects in the extremities and the head and neck region, FVBG in spinal reconstruction encompasses additional technical difficulties in relation to selection of recipient vessels at different anatomical locations of the cervical, thoracic and lumbo-sacral spine.
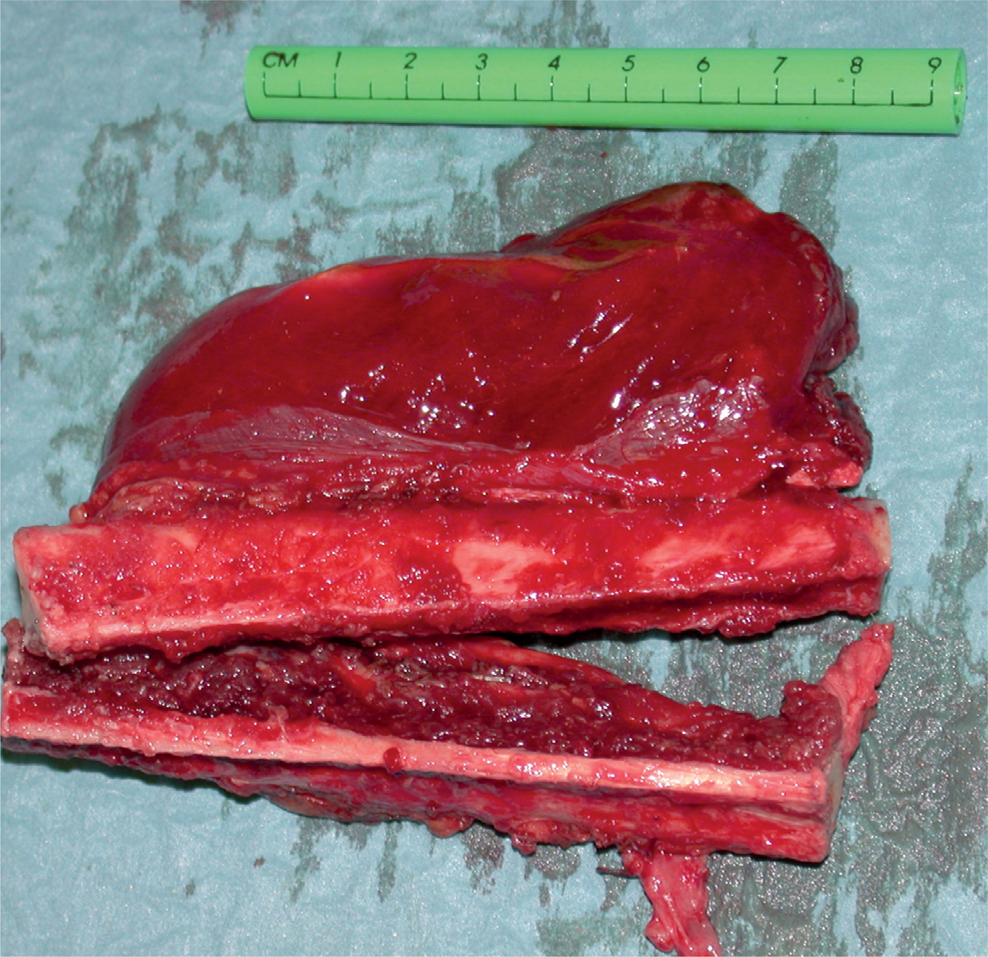
Double barrel fibula strut
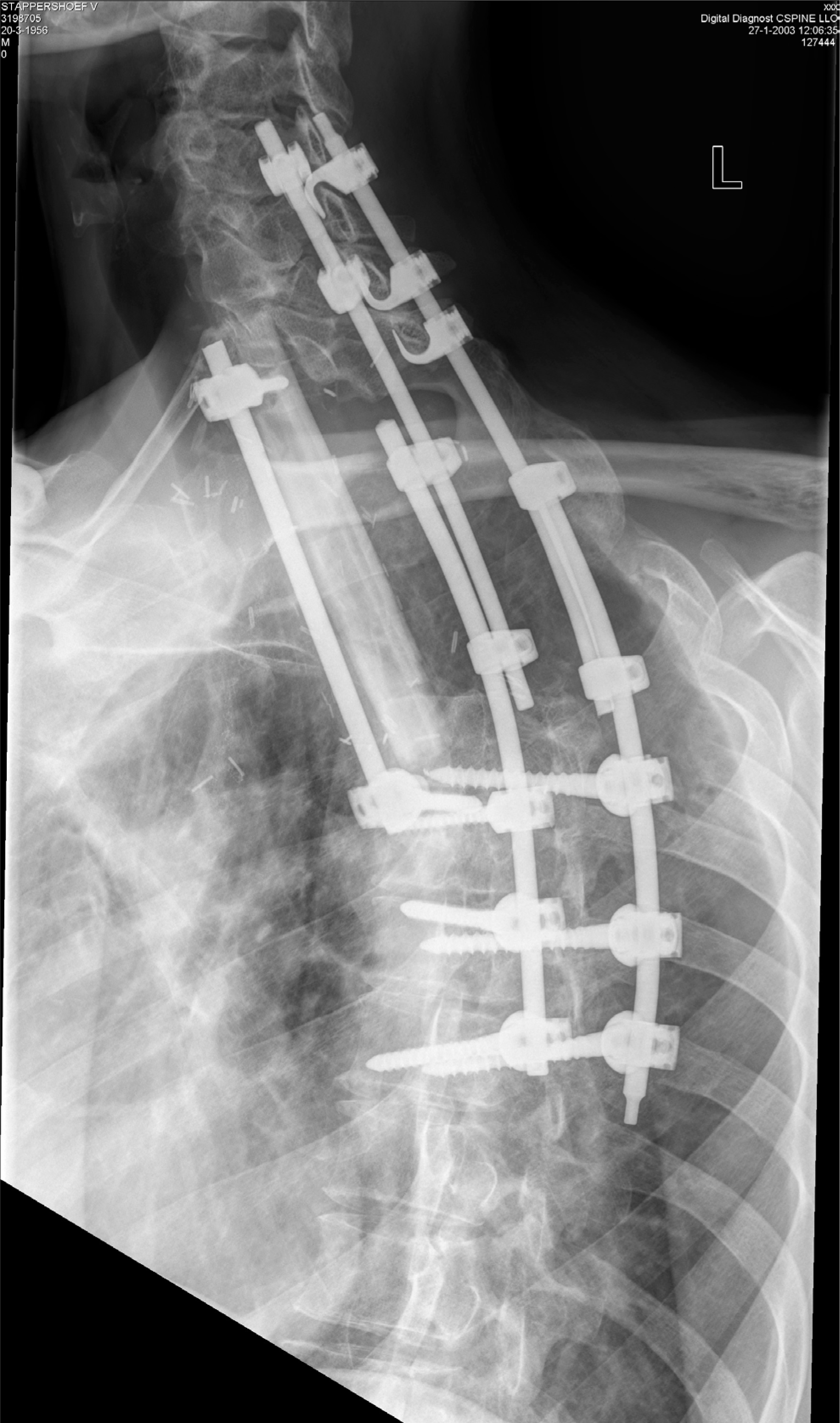
Double barrel fibula, bridging a large spinal defect. Additional fixation is clearly visible
Cervical spine
Because of the relative proximity and good quality of the recipient vessels in the cervical area, microsurgical revascularisation of FVBG rarely presents any difficulties. In this region the exposure of the vertebral corpora is the bigger problem. An anterolateral approach is favoured to reach the acceptor vessels and vertebrae from C3 down. For high defects (C1, C2) a combined dorsal and lateral approach is often preferred, if necessary combined with a trans-oral approach (Fig. 3). In these cases, the anastomoses are performed through a separate incision (submandibular).
Thoracic spine
The thoracic area, on the other hand, is a difficult and challenging area for spinal reconstruction using microsurgical FVBG. Selection of recipient vessels greatly depends on the approach that is chosen and should be carefully planned prior to surgery. To gain access to the thoracic vertebrae by a posterior approach only, (23) the onsets of two to three ribs have to be removed. The reconstructive surgeon should partake in the posterior surgical approach from the beginning, in order to dissect and preserve the intercostal vessels that are selected as recipient vessels. Size and quality of intercostal vessels can be inadequate, especially if a long pedicle is needed. The vein can be thin-walled and adherent to the rib, making it difficult to dissect. The complete resection and reconstruction will have to be performed manoeuvring around the myelum, making this approach extremely difficult. We have performed this technique in one case, but do not advocate the use of this approach in combination with microsurgical FVBG. An intercostal thoracotomy provides reasonable access to the spine, but the reconstructive surgeon is confronted with a relatively deep cavity through a relatively narrow opening. This can make it difficult to reach the target recipient vessels with standard microsurgical instruments. Long, 21-cm micro-needle-holders and microforceps are of great value in this type of surgery. In some cases, we were not able to use the operating microscope due to the limited accessibility, necessitating the use of high-power (4.5×) ocular loupes to perform the vascular anastomoses. Here (partial) removal of one or two ribs can greatly enhance the accessibility. Recipient vessels will most likely be intercostal arteries and veins, or azygos/hemiazygos veins. In cases of spinal or paraspinal tumours, the azygos and/or hemiazygos veins can be displaced by, or included in the tumour, and therefore, may not be usable for microvascular reconstruction, making it extremely difficult to find suitable receptor vessels. We have demonstrated that the thoracic duct can be a valuable alternative for the venous recipient in these cases (25). Despite the successful use of the thoracic duct in this one case, we do not advocate its use as a standard venous receptor vessel.
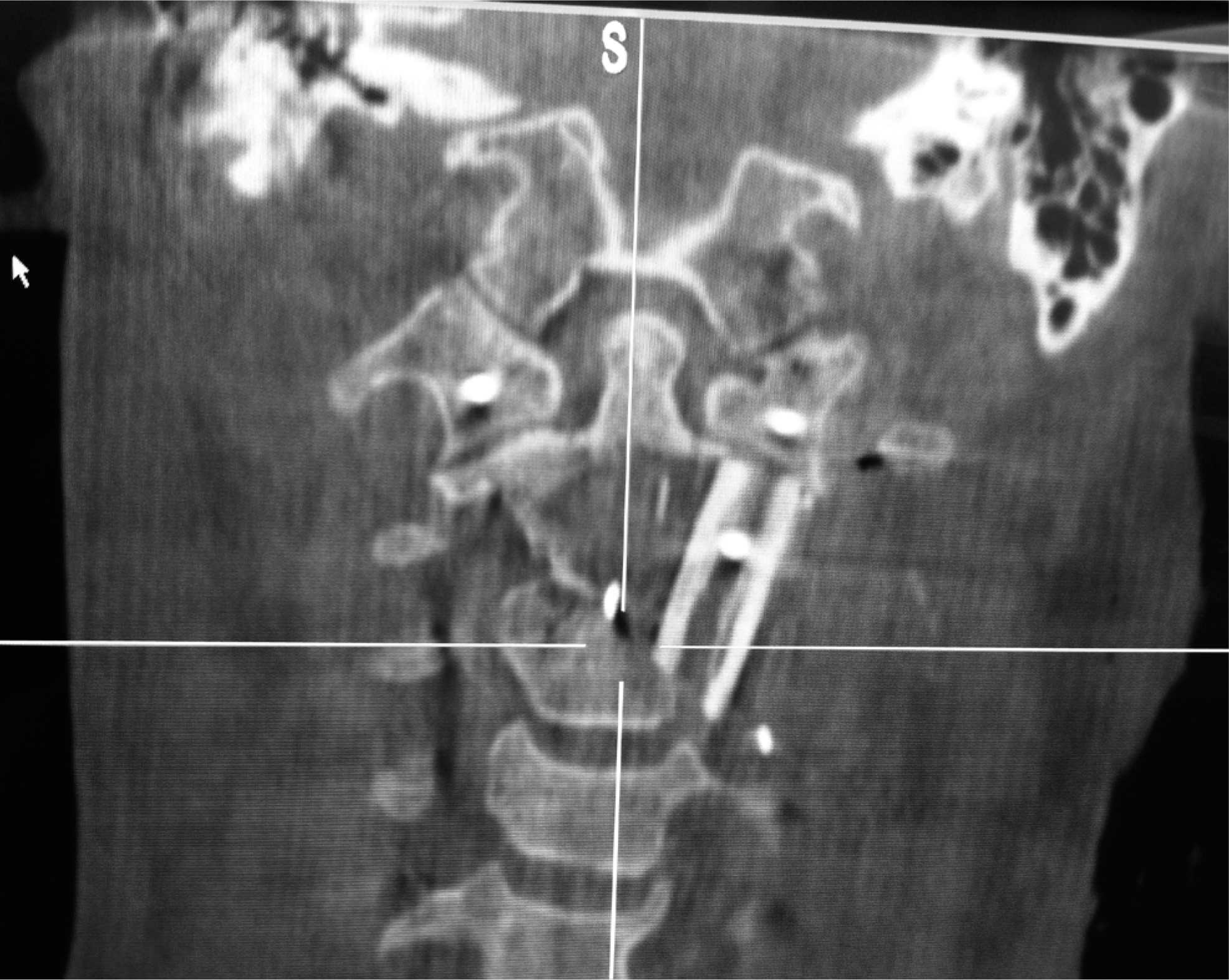
Single fibula strut for a C1–C3 defect
The anterior approach via a midline sternotomy proved to be a much better approach of the high thoracic and low cervical vertebrae for FVBG. The spine is reached through the mediastinum. The distance between sternum and spine is short, thus giving an excellent accessibility. In addition, the internal thoracic artery and vein provide good-quality recipient vessels (8, 14, 18). They are easy to dissect and provide adequate length to reach any thoracic defect. Furthermore, they are well away from the area of the ablative surgery, lessening the risk of intra-operative damage. We used the transsternal anterior approach in 4 cases, and we consider this approach a major improvement for treatment of anterior spinal disorders of the thoracic region (3). In these cases there is no need to dissect the acceptor vessels in an early stage of the operation, albeit dissection before the actual flap transfer is strongly advised. In cases where the cervicothoracic spine is involved, the Shaw-Paulsen approach is a good alternative (31). The patient is placed in a lateral position, the scapular muscles (trapezius and rhomboid) are detached from the ribs, starting at the spine, and the arm, including the scapula and all of its dorsal muscles, is rotated forward. The first and second rib are then removed, creating good access to the cervicothoracic and high thoracic spine. The vascular pedicle can be anastomosed end-to-side with the subclavian artery and vein, as these vessels are easily accessible and close to the spine. We have used this approach in two cases, and it proved to be very useful.
Thoracolumbar spine
Approaches to the lower thoracic and upper lumbar vertebrae are through a thoracophrenico-laparotomy. This is likely to be the most difficult region for microvascular anastomosis. Intercostal or lumbar vessels are the first choice as acceptor vessels, but are often difficult to reach and easily damaged. In this region creativity and ingenuity are called for. The aorta and vena cava are difficult to expose both at the same time. Through a right-sided approach, the vena cava can be reached and used in an end-to-side fashion, but the aorta will be virtually too deep to properly reach. For a left-sided approach this is the other way around. The superior and inferior mesenteric vessels can be used for end-to-side anastomosis, or minor branches can be used for end-to-end anastomosis. However, even though we did not encounter this in our series, there is a risk of inducing intestinal ischaemia and necrosis. In one patient we performed an end to side anastomosis to the right renal artery and used an end-to-side anastomosis to the vena cava for venous outflow. If no local acceptor vessels can be found, there is a possibility to construct an arteriovenous loop from the greater saphenous vein. This is dissected from the leg, to as low as the ankle and then anastomosed to the femoral artery in an end-to-side fashion. This loop can be brought up to the desired level and, after insetting the graft, divided and used for revascularisation of the graft. We have used this technique several times, albeit not for spinal reconstruction.
Lumbar spine
For lower lumbar vertebrae, up to S1, the anterior approach according to Brau (32) is an excellent way to create good exposure of the anterior spine and of the common iliac vessels (Fig. 4). These vessels are well situated for revascularisation of the FBVG by end-to-side anastomosis. In this approach the skin is incised transversely over the rectus abdominis muscle belly, and the rectus muscle is mobilised to facilitate the dissection. As we normally need a wider exposure, we adapted this technique in the way that a paramedian longitudinal skin incision is made. The dissection continues through the rectus sheath, mobilising the lower part of the rectus and finally keeping it retracted to the lateral side, as described in the original article by Brau (32). Because the dissection stays completely extraperitoneal, fluid loss and the risk of abdominal complications remain low.
RESULTS
Our experience with the use of free vascularised bone grafts for anterior spinal reconstruction, to date, consists of 30 cases, 8 of whom have been reported in an earlier article in 1999, and 23 which have been reported in 2008 (31, 33).
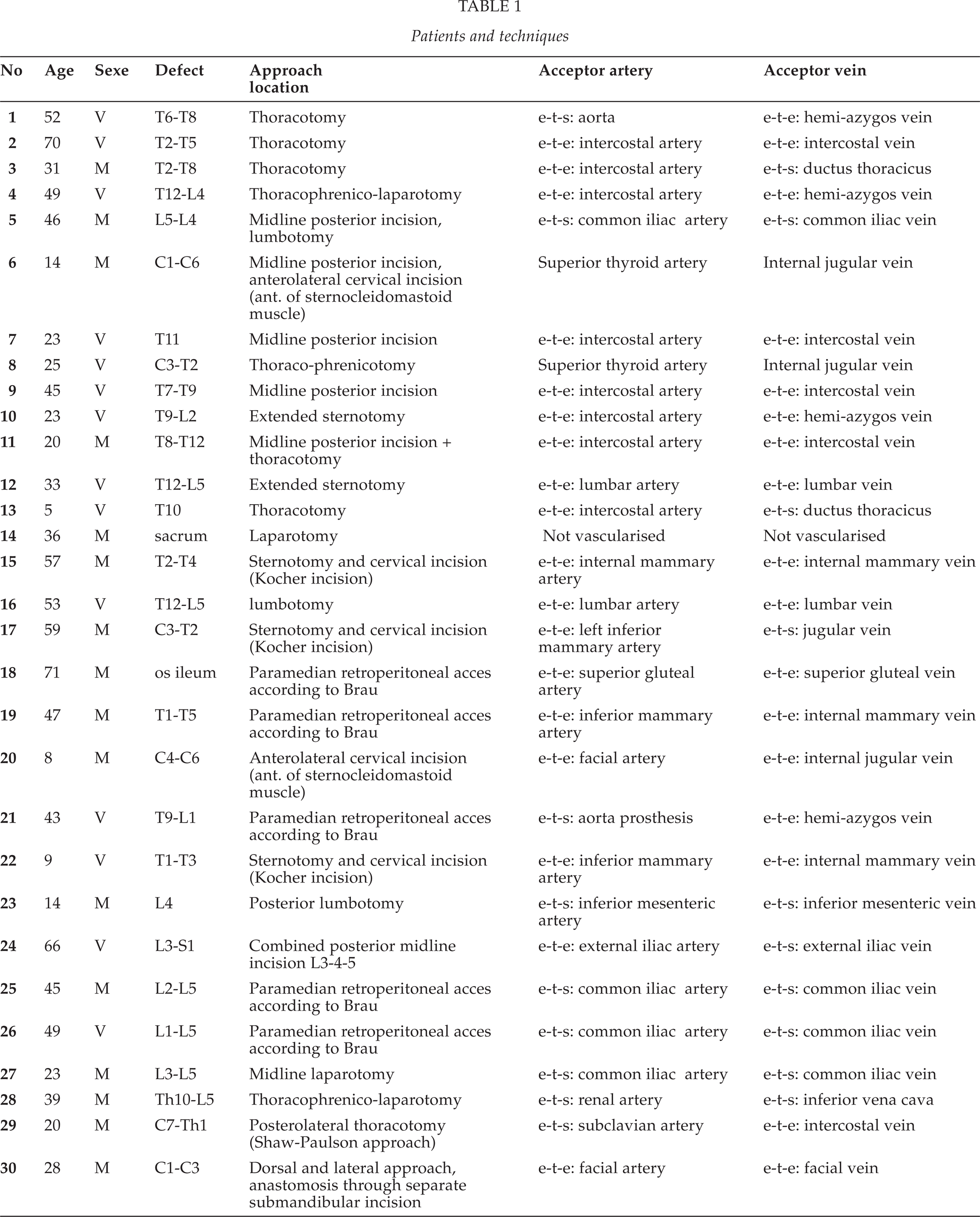
A free vascularised fibular graft was used in 28 patients and a free vascularised iliac crest graft in 2 patients. In one case treated with a free vascularised fibular graft, microvascular anastomosis proved to be impossible due to peroperative loss of all recipient vessels (case 14). Here, the fibula was used as an unvascularised graft finally. Table 1 shows the level of reconstruction, the chosen approach and the acceptor vessels used in each patient.
DISCUSSION
No matter how dedicated the reconstructive surgeon may be, successful revascularisation will not always be possible. Despite our great effort to preserve the initially well prepared recipient vessels during the multidisciplinary surgical procedure in case no. 14, we lost all usable recipient vessels at one stage in the case of a complete resection of the sacrum. Due to the condition of the patient, there was no time to prepare an arteriovenous loop as alternative recipient vessel. Therefore, the microvascular part of the reconstruction procedure had to be aborted and the reconstruction was performed using the already prepared fibula as a non-vascularised graft.
The use of a pedicled, vascularised iliac crest graft, based on the iliac branches of the iliolumbar artery, is a valuable alternative technique for reconstructive procedures in the lumbosacral area (26, 27).
For anterior spinal reconstruction, a substantial volume and/or length of bone is needed in most cases. High compression strength is also desired. The fibula is superior in axial strength; provides up to 30 cm of bone; is easily accessible in the prone, supine and lateral positions; has a low donor-site morbidity and can easily be harvested simultaneously with the spinal surgery (2, 7, 13, 16, 17, 20). Therefore, after starting out with vascularised iliac crest grafts in the first two cases, we changed to the fibula as our FVBG of choice. In most cases, we used double-, triple- or quadruple-barrel fibular grafts to improve axial strength of the graft, the double barrel being the most common. However, by using multi-barrel fibular grafts, there is a risk of pushing the most posterior fibular strut into the spinal cord. For stabilizing procedures, where no vertebrae have been completely resected, this risk is absent.
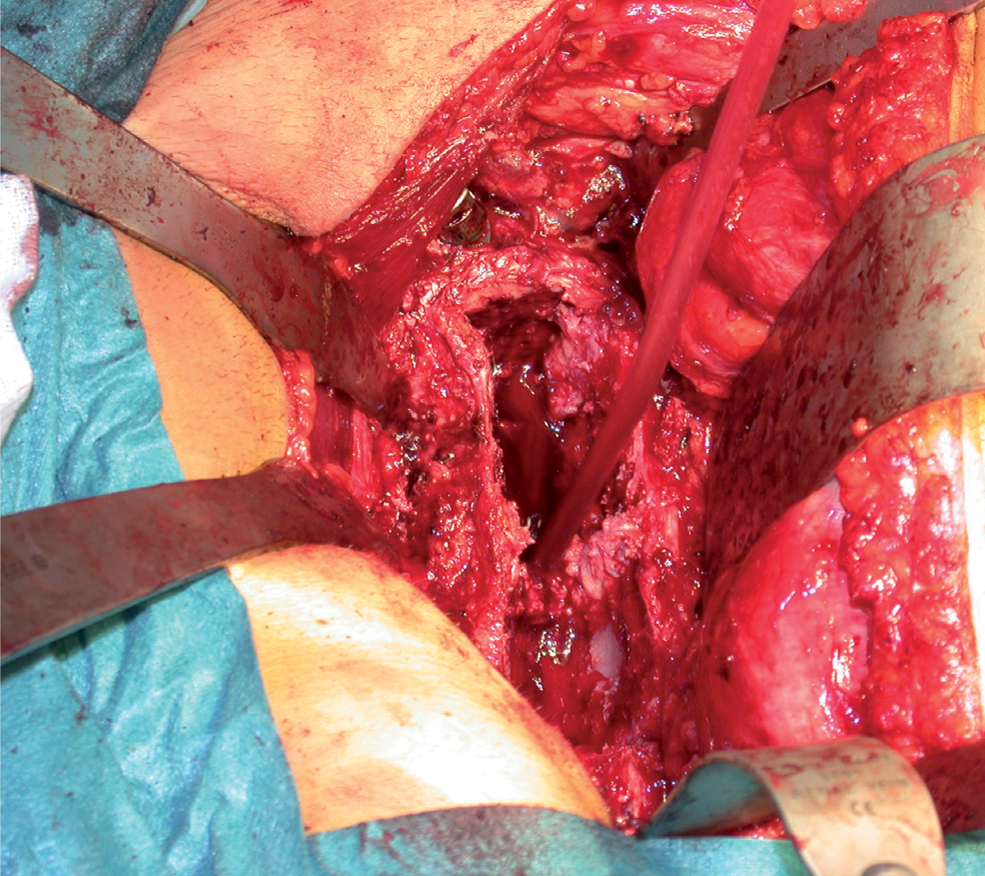
Paramedian retroperitoneal access according to Brau: excellent exposure of lumbar spinal corpora
Patients and techniques
Complex spinal surgery often implies wide surgical exposure and long-lasting procedures with extensive blood loss. As a result, these patients are susceptible to hypothermia and poor peripheral circulation, with an increased risk of deep venous thrombosis (5, 9). We found venous thrombosis in the deep peroneal veins of the already-raised fibular flap in three patients. In all these three patients, there had been an extensive period of time (3–8 h) between the dissection of the fibular flap and the actual transfer, leaving the flap attached to the lower leg by its vascular pedicle only. We have never experienced deep venous thrombosis in more than 200 fibular flaps used for mandibular reconstruction in head and neck surgery, albeit, in many of those cases, there has also been a period of many hours between dissection and transfer of the flap. Even though there is no proof of the efficacy of these measures, we try to minimise the risk of deep venous thrombosis by keeping the donor site covered and warm and by raising the flap as close to the moment of actual transfer as possible. Postoperative monitoring is important to prove the viability of the flap after revascularisation. Immediate postoperative revascularisation can be demonstrated by selective angiography, gadolinium-enhanced magnetic resonance images (MRIs), three-phase technetium-99 m bone scintigraphy, positron emission tomography (PET) studies, single-photon emission computerised tomography (SPECT), or the use of a skin paddle. From these techniques, only the use of a skin paddle has been proven to be a reliable indicator of the viability of the postoperative FVBG (4). Monitoring the postoperative vascularisation of FVBG in anterior spinal reconstruction with the use of a skin paddle is simply not possible in most cases. For reconstructions of the cervical spine, it may be possible to include a small skin paddle that can be sutured into the neck for the purpose of monitoring, but for anterior reconstructions at the thoracic or lumbar spine, the use of a skin paddle is impossible. We have performed bone scans and SPECT scans in several patients early in the series, but, apart from giving only information about one moment in time, they proved to be difficult to interpret in the spinal region, rendering us uncertain in most cases. As these patients are always in the intensive care unit (ICU) in their early postoperative days, performing these scans was quite a burden for patient and personnel. Therefore, we decided to stop these scans. In recent years, implantable doppler probes have proven their value in postoperative flap monitoring. In this series they were not available to us. We feel that monitoring is only useful when reoperations are performed in case of obstructed or absent blood flow. However, in cases of early flap failure, it is highly questionable if these patients – after just having had major surgery for up to 2 days – can safely be reoperated. Therefore, the clinical consequences of monitoring may be very limited in these patients. The main reason for monitoring in these patients will be to prove the superiority of vascularised reconstructions over other reconstructive methods. Our rationale for using vascularised bone in spinal reconstruction comes from the well-known facts and figures for bone reconstruction in the extremities and head and neck region, and from the assumption that these will be the same for reconstruction of the spine. Even though this is very likely, it still has to be proven. Unfortunately, due to the lack of adequate monitoring, our series will not provide this proof.
CONCLUSION
The technique of FVBG can be applied for the reconstruction of complex spinal disorders. The free vascularised fibular graft is considered as the first choice; however, a free vascularised iliac crest graft can be used in cases where the fibula is not available for vascularised transfer. Free vascularised fibular grafts can be used in a double-, triple- or quadruple-barrel construct to increase the graft strength. Selection and dissection of recipient vessels at different anatomical locations of the anterior spinal column is case- and approach-dependent and can be difficult, the cervical region providing the least problems in terms of acceptor vessels and the thoracolumbar region being the most difficult in this aspect. A trans-sternal approach for mid-thoracic reconstructions and an anterior, retroperitoneal approach for lumbosacral reconstructions are preferred over the traditional thoracotomy and lumbotomy.
Internal thoracic vessels, mesenteric vessels, common iliac vessels and even renal vessels can be used as recipient vessels in stead of the more obvious intercostal and lumbar vessels. Successful execution requires microvascular expertise and adherence to basic principles of spinal reconstruction. The superiority of spinal reconstruction using FVBG has still to be proven.
