Abstract
Most chest wall defects requiring reconstruction result from tumor resection. Bone and soft tissue sarcomas and recurrent mammary cancer are the most common tumors. Careful preoperative evaluation, meticulous surgical technique and active postoperative treatment are important. The selection of reconstruction is based on the nature, size and location of the defect as well as on the general health and prognosis of the patient. The goals of the reconstruction are adequate stability, water- and airtight closure of the chest cavity, and acceptable cosmetic appearance. The pedicled muscular or musculocutaneous flaps are usually the first choice for tissue coverage. These include flaps such as latissimus dorsi, vertical or transverse rectus abdominis and pectoralis. In certain cases also the breast flap or omental flap can be used. In selected cases, a free flap reconstruction is indicated if the local options for reconstruction have been used, or if they are unreliable due to earlier scars or radiotherapy. The free flaps to be used for chest wall can be harvested from the thigh (tensor fascia latae flap, anterolateral thigh flap), from the abdomen (transverse rectus abdominis flaps, deep epigastric perforator flaps) or from the chest wall (latissimus dorsi flap and other flaps based on the subscapular artery). Sometimes a fillet forearm can be used as a flap to cover a defect after extended forequarter amputation. Artificial meshes are commonly used to give stability in the defect and to give a platform for the flap. Methylmethacrylate embedded between the two layers of a mesh, or one or two rib grafts fixed to the mesh, can be used to give additional stability in extensive defects to prevent paradoxical movement.
INTRODUCTION
Modern surgical techniques of managing chest wall tumors developed in the 1960s and 1970s (1). The introduction of reliable, new methods of flap reconstruction, improvements in intensive care and rehabilitation have led to acceptable morbidity and mortality rates. Earlier reconstructions used random flaps, often resulting in flap necrosis and complications. Thanks to the evolution of flap techniques, the operations are nowadays easier and safer allowing more extensive resections and reconstructions.
The conventional options include the axial pattern muscle, myocutaneous and fasciocutaneous pedicled flaps, the breast flap, and the omental flap, which all have a reliable blood supply. These remain the mainstay of chest wall defect reconstruction following tumor resection, infection or radiation injuries. In different series, 75–90% of chest wall defects are covered with regional or pedicled myocutaneous flaps (2, 3).
The advent of microsurgery has made single-stage reconstruction possible also in extensive and more complex defects. The early reports of free flap reconstruction for chest wall defects used latissimus dorsi or tensor fasciae latae flaps. At present other flap options are also available. The use of microvascular flaps is reliable, and in selected cases better than regional or pedicled options. It has achieved an important role in the chest wall surgery in selected cases.
THE CHEST WALL DEFECTS
The chest wall defects are usually the result of a tumor resection, infection, radiation or trauma. They can be classified according to their anatomical location. They can be partial or of full thickness. Full thickness defects including all the tissue layers must usually be reconstructed reliably and immediately, in order to restore the stability of the chest wall, to protect and cover the exposed vital organs and to allow early recovery. In addition, postoperative oncological therapy often demands uneventful recovery.
The goals of the reconstruction are adequate stability in respiration (prevention of paradoxical movement), water- and airtight closure and acceptable cosmetic appearance.
The majority of defects requiring reconstruction results from tumor resections. Chest wall neoplasms are be classified as primary, locally invading, and metastatic. The most common primary malignancies in the chest wall are the soft tissue and bone sarcomas.
PRIMARY CHEST WALL TUMORS
Primary chest wall tumors account for 5% of all thoracic neoplasms and 1–2% of all primary tumors (4). Chest wall is the location for 6–7% of all sarcomas. About 45% of the primary malignant tumors arise from soft tissues and 55% appear in cartilaginous or bony structures (5). Chondrosarcoma is the most common primary chest wall malignancy. Chest wall is also a well-known site of radiation-induced sarcomas (6).
Both for soft tissue and bone sarcomas a wide local resection is the best treatment if the tumor is resectable (7). If the skin and subcutaneous tissues are adherent or infiltrated, a full-thickness chest wall resection is often indicated. Also the previous biopsy needle tracts and scars should be excised. It is essential to include wide radical lateral margins as well as a clear deep margin. This often necessitates a full thickness skeletal resection including the parietal pleura which itself provides a good oncological barrier.
LOCAL RECURRENCE
Local recurrence is one of the major problems in the treatment of breast cancer. Up to 20% of patients who have radical or modified mastectomy will sustain a local recurrence. Occasionally, small recurrences can be excised leaving only a superficial defect. However, more often extensive resection including bone and cartilage (full-thickness resection) is indicated demanding coverage with vascularized tissue.
Several conditions related to breast cancer can lead to an indication for chest wall resection: locally advanced primary cancer, local recurrent cancer, secondary tumors associated with radiotherapy (postradiation sarcoma, Stewart-Treves syndrome) or osteoradionecrosis.
Malignant neoplasms can also invade from adjacent tissues or organs. Breast cancer, lung cancer, mediastinal neoplasms or mesothelioma may locally invade the chest wall.
The chest wall may also be a site for distant metastasis of various malignancies. The nature and prognosis of these tumors will influence the extent of resection and the reconstruction.
Sometimes tumors of the shoulder girdle invade the brachial, axillary or subclavian vessels and nerves and also involve the thoracic wall. In these cases, a forequarter amputation and chest wall resection may be indicated (an extended forequarter amputation).
PREOPERATIVE EVALUATION
Extensive full-thickness chest wall resection combined with the reconstruction is one of the most challenging procedures in surgery with a high risk of morbidity and mortality. Individualized treatment and careful planning are indicated. Preoperative evaluation is important and, in particular, pulmonary, cardiac and nutritional problems should be indentified and treated. Ideally, smoking should be stopped 6–8 weeks prior to major surgery. Preoperative pulmonary function has to be evaluated, CT of the chest and trunk, and MRI of the tumor area and bone scan are indicated. Patients are usually treated postoperatively in the intensive care unit, where early extubation and active physiotherapy are mandatory. Epidural blockade is used to control the postoperative pain.
CURATIVE OR PALLIATIVE INTENTION
Some of the patients with extensive chest wall malignancies are not candidates for a curative resection. The tumour may already be disseminated which precludes the possibility of a curative approach. However, the patients may suffer from intense local pain, bleeding and discharge, continuing tumor growth and malodour. Another indication for palliative surgery may be an upper extremity which is swollen, paralytic, painful or infected. This kind of limb can develop a secondary malignancy after chronic lymphoedema or radiation therapy. It is important to provide a safe and quick recovery.
SOFT TISSUE RECONSTRUCTION
Soft tissue coverage can be achieved with the use of a direct closure, skin grafts, local skin advancement, the breast flap, the omentum flap, pedicled myocutaneous flaps, perforator flaps and free flaps. These different methods can also be used in various combinations. The choice of a reconstruction method is based on the location and size of the defect, availability of the local and pedicled options, previous operations or radiotherapy and the general condition and prognosis of the patient.
Direct closure is possible in small resections, or in tumors in which skin and subcutaneous tissue can be preserved with a good blood flow so that, for example, a mesh can be safely covered.
Skin grafts can be used only if the surface is suitable (muscle, fascia, omentum) but not on bone, cartilage or overlying a mesh. Previous radiation therapy may prevent the healing of skin grafts.
LOCAL FLAPS
Breast flap
The breast can be used as a flap to cover defects located mostly in the midline. This method can be used in elderly, morbid patients. In these cases a short operation time is preferable and they can accept the aesthetic deficiency. The incision is placed in the inframammary crease, it can be extended laterally up to the anterior axillary line and the breast is advanced to cover the defect. The procedure can be performed in one position, the circulation of the breast is usually good and the operative risks are moderate.
PEDICLED FLAPS
The regional pedicled muscular or musculocutaneous flaps have been the first choice for soft tissue coverage of the chest wall defects. Flaps such as the latissimus dorsi, vertical rectus abdominis (VRAM), or transverse rectus abdominis (TRAM), and pectoralis, serratus and external obliquae muscles have been commonly employed (8, 9, 10). Also multiple local and pedicled flaps can be used in combinations.
The latissimus dorsi muscle or musculocutaneous flaps are easy to elevate, their arc of rotation is wide and they are usually large and can be tailored to the defect. Serratus anterior muscle slips or the parascapular flap or scapular flap can also be included in the flap. Latissimus dorsi flap covers anterior, lateral and posterior regions of the chest, and also defects of the upper half of the sternum.
The pectoralis major muscle can be based on the thoracoacromial or internal mammary vessels. It is the workhorse flap for reconstruction of the upper or middle sternal and parasternal region. The pedicled rectus abdominis muscle, or musculocutaneous flaps, VRAM or TRAM, are used to cover the epigastric and sternal area and the anterolateral chest wall. Previous surgery or radiation injury to the internal mammary vessels may preclude the use of the flaps based on these vessels. The trapezius muscle or musculocutaneous flap can provide coverage over the posterior chest wall area. These flaps are based on the dorsal branch of the thyreocervical trunk.
Also perforator flaps such as thoracodorsal artery perforator (TAP) flap or intercostal perforator flaps have been used in reconstruction. They can cover small and moderate sized defects on the anterior and lateral aspects and can be used in combination with other flaps.
The omental flap
The omentum can be used to cover anterior and lateral defects (11), but it will not reach the posterior area of the chest wall. The blood flow is based on the right, left or both gastroepiploic vessels. Usually this flap is very soft and pliable, and therefore, in wide defects a mesh is often used to give a necessary platform for the flap and stability. The omental flap is usually covered with skin grafts. The harvesting of the omentum may affect respiration postoperatively. Infection or dehiscence in the abdominal wound were the commonest complications, and later there was a risk of a ventral hernia. The flap can be safely divided 6–12 months after the reconstruction and the abdominal fascia layer closed.
FREE FLAP RECONSTRUCTIONS
In selected cases, free flap reconstruction is indicated. The local options for reconstruction may have been already used, or they may be unreliable due to earlier scars or radiotherapy. The pedicle of the flap may have been damaged by previous operations or radiation therapy, or the flap size may not be adequate to cover the whole defect. In particular, large anterior midline defects extending to the epigastrium and large thoracoabdominal defects may be difficult to reconstruct with pedicled options. Also repeated lateral thoracotomies often damage the thoracodorsal vessels, so that latissimus or serratus muscles are not available to cover a defect on the lateral chest wall, or sternal area, or inside the chest cavity (12, 13, 14). In addition, the general condition and prognosis of the patient must be taken into account. The advantages of microvascular reconstruction should outweigh the disadvantages such as longer operative time and potential anastomotic problems.
Microvascular tensor fascia latae flap (TFL) is one of the oldest flaps to cover extensive defects. The TFL flap is easy and quick to harvest both in the lateral and supine positions, and the flap can be large (20×35 cm) with a constant and reliable pedicle. It allows a two-team approach (15). The flap contains a firm thick fascia, and also rectus femoris muscle can be included in the flap if extra bulk is needed for example to fill the chest cavity. The donor site in the thigh has no influence on respiratory function.
The rectus abdominis myocutaneous (TRAM) flap has many advantages and it has also been frequently used for chest wall defects. The TRAM flap is a reliable and large flap which can be used to cover chest wall defects (16), and if indicated, give breast contour at the same time. As a bulky flap, it can also be used as a filler in large intrathoracic defects. In many patients the TRAM flap can provide large surface area, but in thin patients the size is not as large. In addition the anterior chest wall defect may increase, when TRAM donor site is closed. The tight abdominal donor site may postoperatively adversely affect respiratory function in thin, morbid patients.
Fillet forearm flap. After forequarter amputation the wound can be usually directly closed using the standard posterior flap. However, in some cases the extent of the tumor demands resection of soft tissues or ribs so that closure without a reconstruction is impossible. In these cases the amputated extremity can provide a free flap (17, 18). If the forearm and distal upper arm are not affected, a fillet forearm flap (with a pedicle up to the distal brachial vessels) can be dissected. Usually the radius is excised, and the ulna can be preserved in the flap, and after osteotomy and plate fixation, it can give stability and provide contour of the shoulder.
CHEST WALL STABILITY
The flaps used for reconstruction give some stability on their own depending on their composition and thickness. Tensor fascia latae with a strong fascia is rather stable, and it can be sutured like a mesh to the defect. Also a thick musculocutaneous flap (latissimus dorsi flap, VRAM, TRAM) is quite stable alone in smaller defects. However, the omentum flap or a thin muscle flap are often too soft and pliable. They are commonly used with a mesh to give platform and stability for the flap.
In particular, the following defect locations are at the risk of herniation or paradoxical breathing: total or subtotal sternal resection including several ribs bilaterally, high lateral resection (more than 3–4 ribs), or extended forequarter amputation with resection of 3 or more ribs. In these cases, the patients are prone to have respiratory problems, if the stability is not adequate as early as during the immediate postoperative period (15). Instability or even slight paradoxical movement is better tolerated, when the defect is located in the lower lateral area or both in the chest and the abdominal wall.
Various autologous materials such as free dermis flaps, fascia latae, fibula or rib grafts have been used to stabilize the chest wall (19). They are usually well tolerated but may alone be either too flaccid or resolve during time. Currently, polypropylene or Marlex mesh, polytetrafluoroethylene (Goretex) are used for stabilization and prevention of herniation (20, 21). Forty patients undergoing a chest wall reconstruction with mesh and flap recovered significantly better (shorter time on respiratory support and shorter hospital stay) compared to those having only flaps reconstruction (20).
To get extra stability, rib grafts have been used, too. With one or two rib grafts superimposed a large defect covered with mesh can be divided into smaller defects with adequate stability and movement. Many centers have used methylmethacrylate embedded between two layers of mesh (23). The rigid area should be about 1–2 cm smaller in diameter compared with the defect size. This allows the sandwiched mesh to be securely fixed to the defect wall leaving the hard methylmethacrylate portion about 1 cm away from the bony defect and still provide proper convexity and rigidity with slight movement (Fig. 1 A–D).
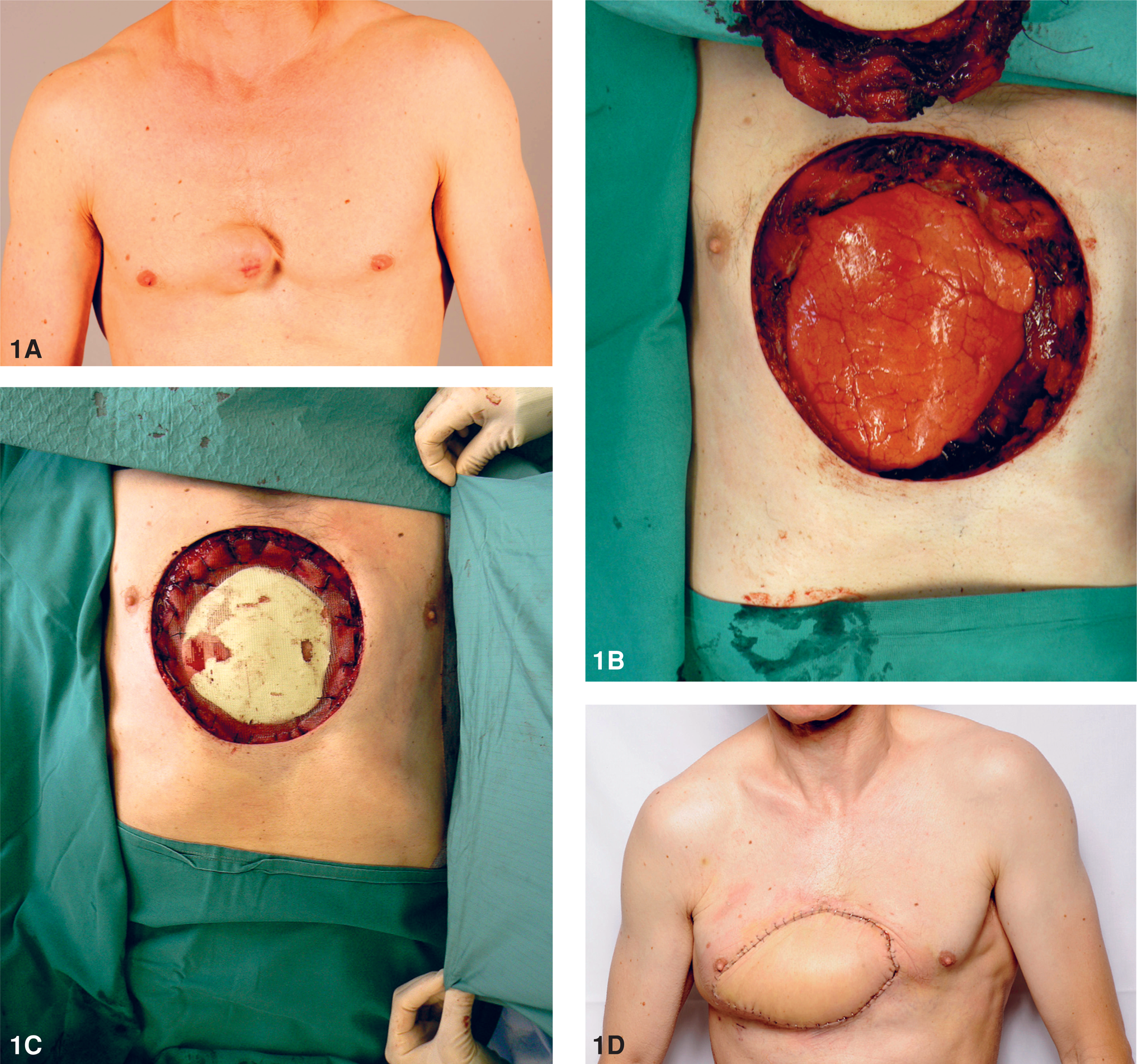
Reconstruction with a mesh, methylacrylate and microvascular TFL flap.
In the thoraco-abdominal cases, the infero-lateral diaphragm often forms a barrier and should be resected along with the tumor. It is important to reconstruct the missing portion of the diaphragm with a similar-sized mesh and suture it to the original position of the diaphragmatic muscular insertion in order to maintain proper pleural cavity size and adequate respiratory physiology (15).
PROGNOSIS
The prognosis of chest wall tumors has become better during the last decades. For the primary chest wall sarcomas the five-year overall and disease-free survivals were 66% and 47%, respectively (24). Favorable prognostic variables for survival included age below 50 years, bony tumor, chondrosarcoma, and low grade soft tissue sarcoma (7). Microscopically clear margins could be achieved in 80% of the primary sarcomas and 64% of the recurrences. The chances for local control were lower after surgical treatment of recurrent sarcoma compared to primary sarcomas (25). Also in locally recurrent mammary cancer the chest wall resection and reconstruction has reasonable long term survival and it has proven to be safe and effective procedure with acceptable morbidity (22, 26)
