Abstract
Background and Aims:
The unique anatomical and physiological function of the perineum region makes it difficult to be repaired after tumor resection. We aim to evaluate the efficacy of PSC divisional reconstruction strategy in the reconstruction of perineal skin defect.
Materials and Methods:
This study includes patients undergoing perineal skin defect reconstruction with PSC strategy—P (penis), S (scrotum), C (circum-penoscrotal skin) divisional reconstruction strategy.
Results:
From August 2013 to August 2018, 47 patients were enrolled in the surgical procedure. The defect area after resection measured 2 cm × 2.5 cm, minimum, and 12 cm × 18 cm, maximum. Among them, the cases involved one, two, and three zones are 12, 10, and 25, respectively. The skin defects were divisionally repaired. All flaps were well survived without complications or scar contracture. No tumor recurrence happened.
Conclusion:
The application of PSC divisional reconstruction strategy is a promising way to repair wounds in circum-penoscrotal skin area. Moreover, this strategy is easy to process and shows no significant complications during follow-up period.
Introduction
The common skin malignant tumors in perineum include squamous cell carcinoma (SCC), Paget’s disease, basal cell carcinoma, Bowen’s disease, malignant melanoma, and sarcoma (1). Due to the unique anatomical structure and the physiological function of the perineum, the residual wounds are usually very complex to be constructed after the removal of the perineal skin tumor. In addition, urinary, sexual, and gastrointestinal functions should be considered in the reconstruction process. Reconstruction based on the anatomical division is one of the basic principles in reconstructive surgery. Our team designed PSC strategy—P (penile), S (scrotum), and C (pericarpal skin) regional reconstruction strategy for wound reconstruction after perineal tumor resection to achieve better therapeutic, cosmetic, and prognostic outcomes. This method is reported as follows (Fig. 1).
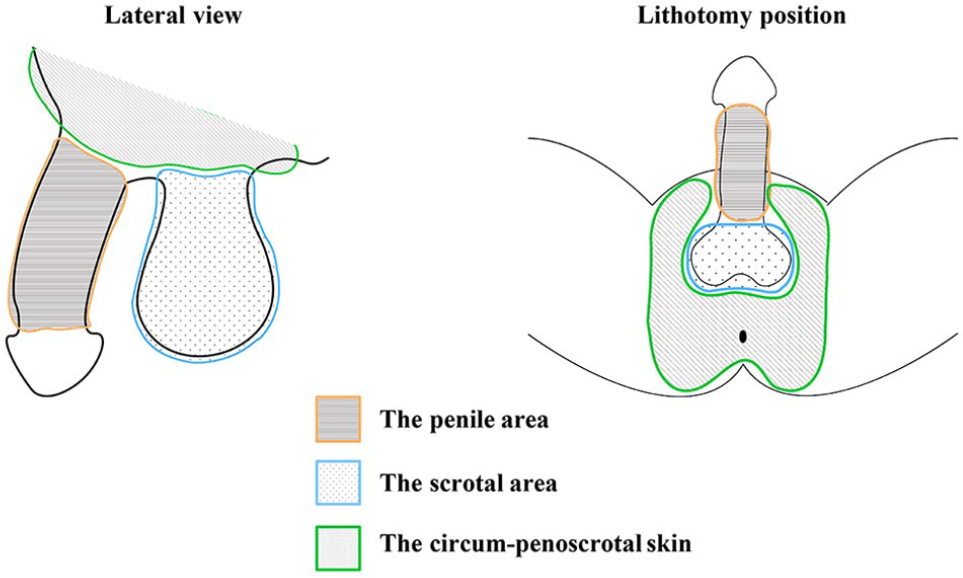
Schematic illustration of the anatomical divisions of the perineum region.
Materials and Methods
Clinical Materials
From August 2013 to August 2018, 47 cases admitted in plastic surgery from Changhai Hospital of Shanghai Second Military Medical University were enrolled in this study. The patient’s consent was obtained. Standardized data collection templates are used for statistical variables, including patient demographics, tumor types, defect size, and follow-up period during hospitalization and reexamination.
Surgical Technique
Tumor resection
Preoperative ultrasonography for inguinal lymph nodes and biopsies for enlarged lymph nodes were performed. All surgical procedures were performed under general anesthesia. The excision was designed according to the guidelines for each type of tumor, with histologically clear margins and bases confirmed by intraoperative frozen pathologic section. Generally, the incision was designed 1 cm away from the tumor and the tumor was resected above the layer of deep fascia. For Paget’s disease, the incision was made 2 cm away from the tumor border, another 2-cm incision will be made if the frozen pathology showed a positive surgical margin. For specimen smaller than 15 cm2, 6 points were examined in frozen pathology, while 12 points were examined for those larger than 15 cm2. Inguinal lymph nodes dissection was conducted if lymphadenopathy was detected before surgery.
Flap design and divisional reconstruction
The PSC reconstruction strategy was performed to design the flap, according to the location and size of the wound. Most of the tumors are located at the base of penis, so the skin defect is difficult to reconstruct. The location of the tumors made flap reconstruction rather difficult. The excellent ductility makes scrotal skin flap the best choice during divisional flap repair. Furthermore, the scrotal septal flap is preferred due to its constant blood supply and abundance tissue reserve. The defect in the perineum could be repaired by perforator flap of this area. Either direct suture or flap coverage was performed in the donor sites depending on the flap size and mobility of surrounding tissues.
Scrotal septal flap is an ideal choice to repair penile skin defect. After marking the flap area, incisions and undermining were made down to the superficial scrotal fascia, and the vertical vessels in the scrotal septum area were carefully preserved during dissection. The penis defect could be repaired by scrotum flap through subcutaneous tunnel. The subcutaneous anchor attachment was carried out at the root of the penis to maintain the penoscrotal angle.
For skin defects at the circum-penoscrotal skin area, it is optimal to choose superficial circumflex iliac artery perforator flap. The area of the perforator flap should be relatively larger than the defect area. The center of blood supply and the axis of the flap located at 2- and 1-cm below inguinal ligament, respectively. The pedicle of the flap should be as wide as possible when the defect area is relatively small. The dissection of blood vessels can be conducted to increase the mobility and covering area of the flap if the defect is large. For skin defects in scrotum area, local scrotum flap is an ideal choice in the reconstruction strategy (Fig. 2).
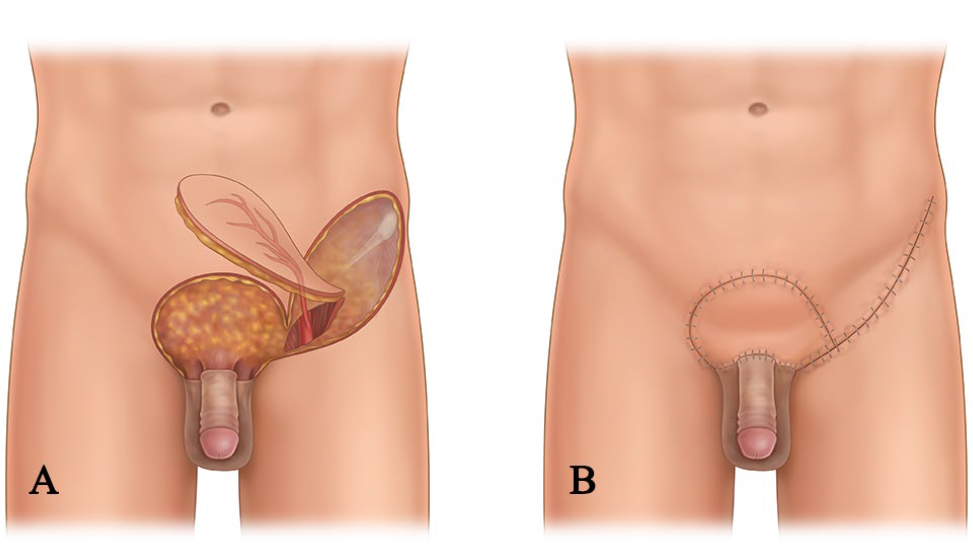
Schematic illustration of the design of superficial circumflex iliac perforator flap. A) The skin defect at the circum-penoscrotal skin area design of superficial circumflex iliac perforator flap. B) The reconstructive outcome after flap covering.
Results
A total of 47 patients aged 45–75 years old were included in this study. The surgical indications for all patients were malignant tumors, including Paget’s disease (25 cases), SCC (10 cases), and basal cell carcinoma (12 case). Among those cases, defects in P, PSC, SC, and PC region were 12, 25, 7, and 3 cases, respectively. Histologically clear margins and bases were confirmed by intraoperative frozen pathologic section except for one patient with SCC. The extended resection of another centimeter was conducted and was confirmed to be clear of malignant cells. Right inguinal lymph node dissection was performed in one case of SCC with lymphatic metastasis identified by biopsy before surgery.
Among all the cases, the maximum wound area was 12 × 18 cm2 and the minimum resection area was 2 × 2.5 cm2. There were no intraoperative or postoperative complications. The flap at the recipient sites shows favorable texture and color. The outcomes were satisfactory from both functional and aesthetic point. Furthermore, no tumor recurrence at the transplanted sites occurred during the follow-up period of 6–20 months.
Typical Cases
Case 1—P, S, C Areas Skin Defect
A 56-year-old male presented a lump in the pubic area which was identified 4 years ago and enlarged rapidly in the past 2 months. The pathological part involved the root and skin of penis, the scrotum, and the pubic region. The extent of resection area was 1 cm beyond the tumor boundary, and the excision depth was above the deep fascia. Histologically clear margins and bases were confirmed by intraoperative frozen pathologic section. The wound size after resection was 9 cm × 15 cm. Divisional reconstruction was performed using a scrotal local flap (measured 3 cm × 7 cm) designed to cover penis skin defect and the superficial circumflex iliac artery perforator flap (measured 7 cm × 14 cm) to cover the remnant defect in a manner of 70° and 120° rotation, respectively. All of the defects and donor sites achieved tension-free closure. All flaps survived without partial or total defects or donor complications. There was no tumor recurrence during the 3-month follow-up (Fig. 3).
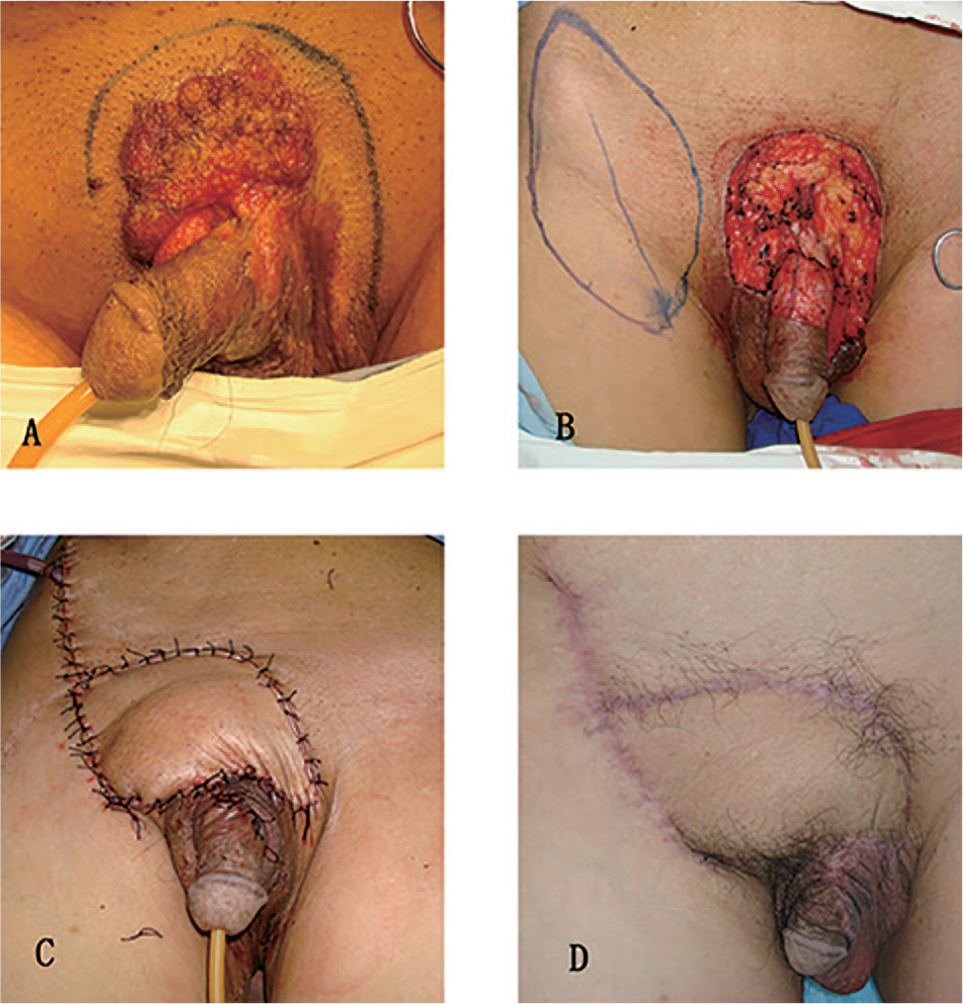
Perineal reconstruction after squamous cell carcinoma resection. A) Preoperative view of the tumor and design of incision. B) Immediate after tumor resection. Perineal defect of 9 cm × 15 cm including the skin of penis, pubic area, and scrotum. The design of scrotal flap and perforator flap. C) The repair of penis and scrotal skin wound with scrotal flap and perforator flap to cover the donor site defect. D) Three months postoperative view.
Case 2—P Area Skin Defect
The second patient is a 45-year-old male with a lump at the base of the ventral side of his penis. This lesion was detected more than a year ago and grew rapidly. The tumor was resected 1 cm away from the tumor boundary above the deep fascia, causing a 2.5 cm × 4 cm skin defect. Notably, the margins were clear based on intraoperative frozen pathologic section, which also showed this lesion to be SCC. Preoperative ultrasonography revealed that the right inguinal lymph nodes were enlarged, and the biopsy confirmed it to be lymph node metastasis. Therefore, right inguinal lymph node dissection was performed. A scrotal septal vascular pedicle flap was designed to cover the penile skin defect. The flap was advanced about 7 cm. The flaps showed no congestion, wound dehiscence, or other severe complications during the follow-up period (Fig. 4).
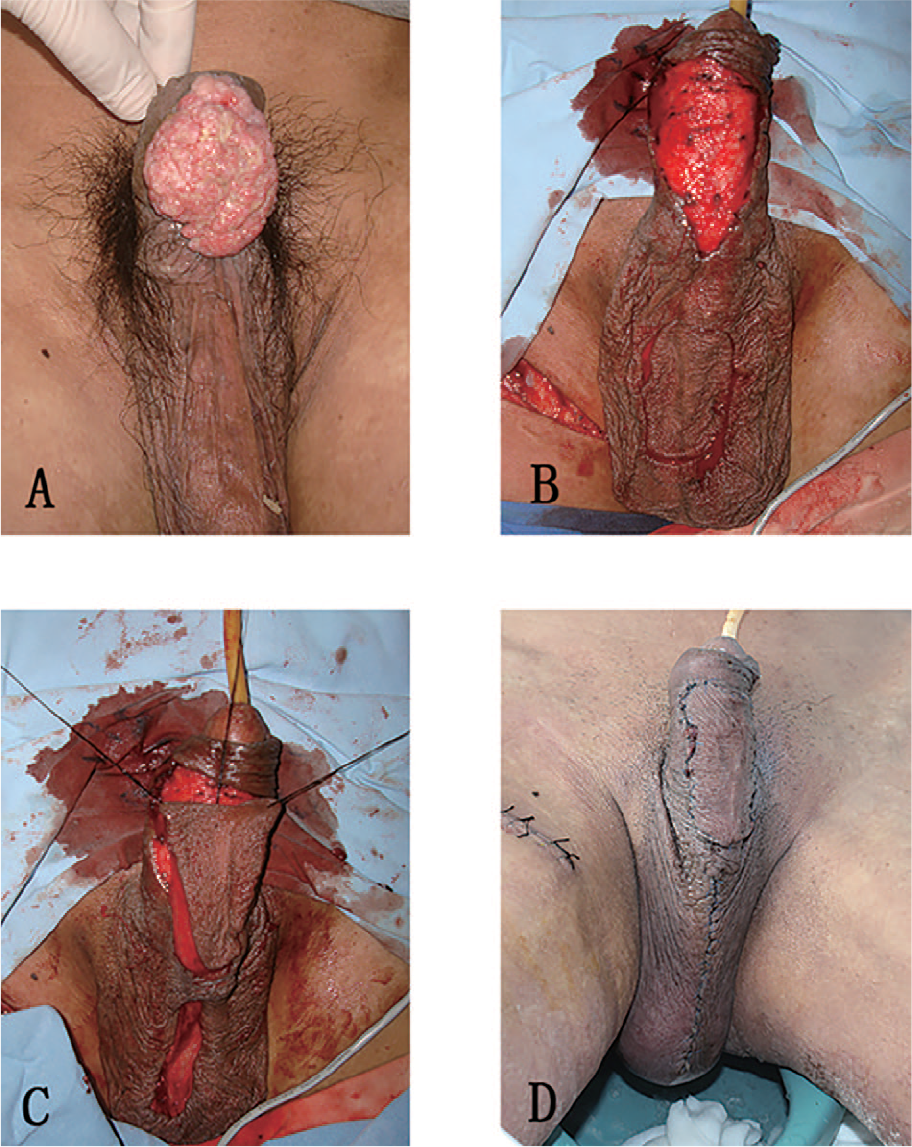
Perineal reconstruction after penis squamous cell carcinoma resection. A) Preoperative image of the tumor. B) Scrotal septal skin flap design after penis squamous cell carcinoma resection. C) Scrotal septal skin flap was harvested and the wound was covered. D) One week post-operation.
Case 3—P, S, C Areas Skin Defect
The third patient was a 45-year-old male diagnosed with extramammary Paget’s disease. The scrotum ulceration lasted for more than 10 years and was treated with external ointment. The ulceration enlarged to 5 cm × 5 cm within a year with partial hair loss. The lesion was removed, and the margins were clear according to intraoperative frozen pathologic section. The skin defect measured 8 cm × 6 cm. Divisional reconstruction was performed using scrotal septal flap (measured 4 cm × 3 cm) to cover the defect and the inguinal flap (measured 7 cm × 4 cm) was designed to cover the donor site in a manner of 40° and 60° rotation, respectively (Fig. 5).
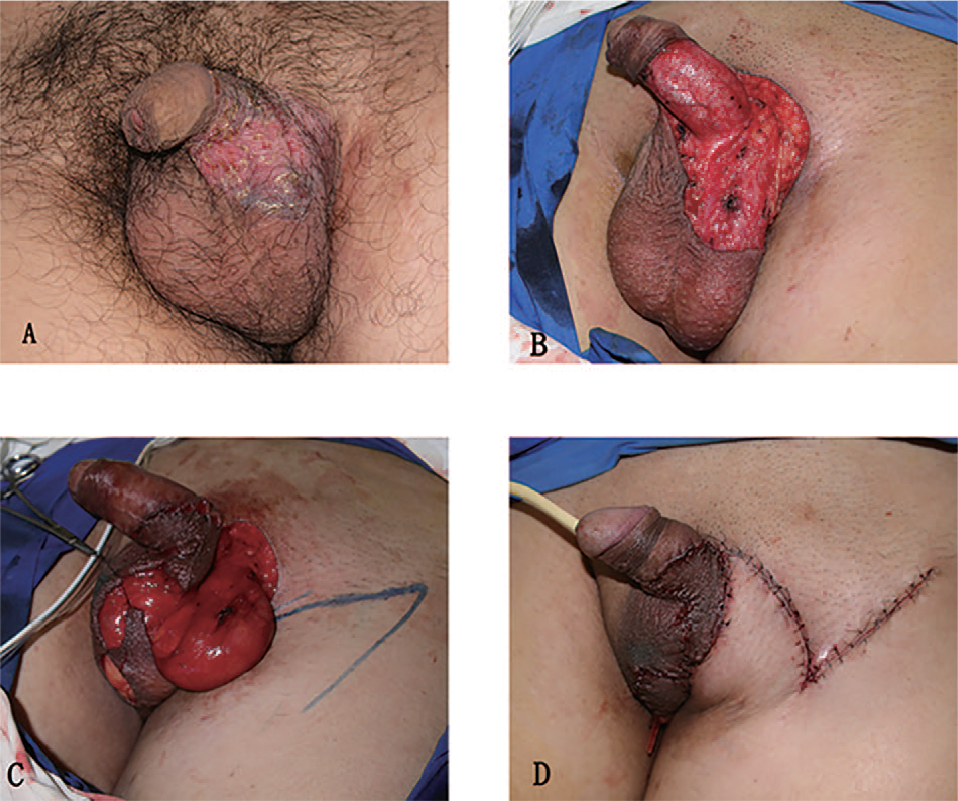
Perineal reconstruction after extramammary Paget’s disease resection. A) Preoperative image of the tumor. B) Immediate after tumor resection. C) Scrotal flap transferring and the design of perforator flap. D) Immediate after reconstruction.
Discussion
The unique anatomical features and functions of the perineal area make the reconstruction of this region a considerable challenge to the surgeons. The skin color, texture, thickness of soft tissue, and degree of flexibility in the perineum region are distinct from other anatomical regions. In addition, the local soft tissue in the perineal area has certain tension and shear force changes during walking or running. Therefore, the specific function in this area should be emphasized during the reconstruction procedure.
The anatomical structure and function are closely related, and as such, reconstruction based on anatomical division could achieve better clinical outcomes. It has been reported that better clinical outcomes could be achieved if divisional repair based on aesthetic unit is applied in facial defect reconstruction (2). In recent years, this concept has also been emphasized in the repair of foot wounds (3). According to anatomical features and partitioning methods proposed by Ahn et al. (4), we divided perineum region into three functional and aesthetic areas for reconstruction: the penile, scrotal, and perineal skin area (Fig. 1). We believed that the structure and function of the penile area is more vital than the other two parts. The function of whole perineal area will be ensured if ideal reconstructive effect of the penis is achieved.
Notably, there are numerous kinds of reconstruction strategies available after surgical resection of tumors in the perineum (5, 6), including skin grafts, local skin flaps, myocutaneous flaps, and free flaps. Skin graft in the perineum region hardly achieves satisfactory clinical effects due to its proximity to the anus and difficulty in bandage (3). Although vacuum sealing drainage system with more powerful and stable fixation has been suggested, the reconstruction effect was still limited by postoperative scar contracture (7).
Thus, flap reconstruction is usually applied in C region. Local flap and myocutaneous flap are the most commonly used reconstructive approaches. The myocutaneous flaps including transverse rectus abdominis myocutaneous (TRAM) flap (8), gracilis muscle flap (9), Singapore flap (perineal flap) (10), as well as various gluteus flap (11) and thigh flap (12) are advantageous due to its potent anti-infection and volume-filling capability, and are widely used in perineum reconstruction especially for those contaminated wound with volume defect. With the development of microsurgery, free flap has been an important choice for skin defects in C region, which usually demonstrate the characteristics of large area, inappropriate for skin transplant, or the exposure of deeper structures. However, free flap surgery can be technically difficult and the risk of failure, increased donor-site morbidity, and the matching degree with less ideal skin is relatively high.
Perforator flap was first described by Koshima and Soeda (8) when they reported the inferior epigastric artery perforator flap. In 1993, they described the application of anterolateral thigh perforator flap (13). The application of perforator flap in the perineum was explored and widely used since then (14–18). The blood supply of perforator flap comes from small and perforating branch vessels which go through the deep fascia. Perforating branches originate from multiple arteries are abundant in perineal, facial, peri-umbilicus, and inguinal region (16). There are four perforating branches in the perineum region, namely, inguinal and perineum perforating branch of external pudendal artery in the perineum region, scrotum (labium) posterolateral artery perforator, and cutaneous branch of the obturator artery perforator. The locations of these perforate arteries were relatively consistent, and these arteries connect with each other extensively.
Based on the recommendations by Dancey and Blondeel (19), this simplified management of skin flap could significantly reduce the operating time compared to other approaches. In addition, perforator vessels are often accompanied by cutaneous nerves in the perineum region, which could maintain sensory function of skin flap after flap transplantation. Kim et al. (16) noted that perforator flap reconstruction is an ideal procedure for the repair and reconstruction of the perineum. Furthermore, since skin in scrotal area is similar to the penis in texture, thickness, and ductility, it plays a particular role in the penis skin defect reconstruction (20–22). Based on the features, we used the scrotal skin flap to repair the penile area while the perforator flap to repair circum-penoscrotal skin area, and achieved ideal outcome. Although divisional reconstruction by combining scrotum flap and perforating flap is relatively complicated, we observed a significant increase in the degree of satisfaction using this approach.
Conclusion
The results showed that the application of PSC strategy in the repair of perineal skin defects after tumor resection can provide reliable blood vessels and sufficient skin supply, which is an ideal way for surrounding skin to cover a wide range of perineal defects and achieve tension-free closure. We recommend this as the best choice for perineum reconstruction.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
