Abstract
Backgrounds and Aims: Infection of sternotomy wound is a rare potentially fatal complication because of the risk for deep sternal infection. Current treatment comprises antibiotics, debridement, negative pressure wound therapy and sometimes transposition of muscle or omental flaps to fill the anterior mediastinal dead space.
Material and Methods: The management of 60 consecutive deep sternotomy wound infections is reviewed. The one stage treatment was mostly chosen. In 5 patients after debridement, negative pressure wound therapy was used before flap reconstruction. Fifty-seven patients were rewired and 3 patients had sternectomy. The choice of the flap was based mainly on anatomic location of a sternal wound defect and also on which grafts been used in cardiac operation.
Results: The unilateral turnover split pectoralis major flap was the choice for 50 patients. In 8 patients latissimus dorsi flap was used. Rectus abdominis was used as a standalone flap in 4 patients and in combination with pectoralis major in one. All patients survived after deep sternal wound infection. In only 33 patients the recovery was totally uneventful. In the remaining 27 patients there were one or more complications. Not a single flap was lost completely, but due to partial flap necrosis, a redo reconstruction was needed in 3 patients. Negative pressure wound therapy was used after flap reconstruction in eight patients with incomplete post-flap healing to prepare for wound revision and split thickness skin graft.
Conclusions: A structured approach including both cardiac and plastic surgery in case of deep sternal wound infection is recommended. A single stage surgery with the help of muscle flap reconstruction is our standard treatment. With our protocol, we have been able to keep the mortality low.
Keywords
INTRODUCTION
Deep sternal wound infection (DSWI) after sternotomy is a relatively rare complication with a reported global incidence in the range of 0.25–2.8% (1–3). However, this complication is associated with a significant mortality varying between 0–28% (4, 5).
The definition of poststernotomy mediastinitis, also commonly called deep sternal wound infection (DSWI), has been established by the Center for Disease Control and Prevention in the USA (6). According to these guidelines, diagnosis of mediastinitis requires at least one of the following:
an organism isolated from culture of mediastinal tissue or fluids
evidence of mediastinitis seen during the operation
one of the following conditions: chest pain, sternal instability, or fever (> 38° C), in combination with either purulent discharge from the mediastinum or an organism isolated from the blood culture or culture of mediastinal drainage.
The treatment of patients with DSWI is handled on a day-to-day basis by cardiac surgery departments, but as the reconstruction often involves flap surgery, the reconstruction itself is often performed as interdisciplinary teamwork between plastic and cardiac surgeons.
Today the treatment of DSWI comprises of the clearing of the sternal infection by wound debridement combined treatment with negative pressure wound therapy (NPWT) sometimes and subsequent reconstruction with final closure of the sternal defect with or without flap reconstruction. Early treatment strategies consisted primarily of open wound care. In the ensuing period, treatment changed to closed treatment with retrosternal and catheter based antibiotic irrigation. This treatment is still a part of today's regimen, although in an updated form. In 1980, Jurkiewicz et al. described the use of muscle flaps to reconstruct sternal defects. Since then, a variety of muscle flaps have been used to close mediastinum after sternal resection. The use of muscle flaps lowered the mortality even further and is today a well-integrated part of the treatment of DSWI (6).
Before the reconstruction of a sternal defect, the wound must be carefully debrided of infected and/or necrotic tissue. This is achieved by combining of the removal of sternal wires and sutures, surgical debridement of wound edges down to a healthy bleeding tissue. Debridement can be performed either as an independent procedure prior to a later separate reconstruction (a two-stage procedure), or as a single-stage operation where the wound debridement and flap reconstruction are done in the same operation. The single-stage approach is recommended as it yields shorter hospital stay and shorter postoperative periods of intensive care (6). Recently, there have been studies demonstrating the role of negative pressure wound therapy (NPWT) in the management of DSWI as well (1, 4).
During the last 50 years, there have been considerable improvements of both in survival and morbidity of these patients. This is due to the wide range of treatment options made available by advancements in medical and surgical science. However, there is a general lack of consensus regarding which treatment strategies and reconstructive techniques are best suited for this complication (7).
MATERIAL AND METHODS
The study population consists of 60 consecutive patients with deep sternal wound infection treated with a total of 63 flaps (Fig. 1) between January 2005 and May 2012. There were 50 patients (83.3%) undergoing coronary artery bypass grafting (CABG), four patients (6.7%) with valve repair, combined surgery in four patients (6.7%) and aortic reconstruction due to dissecation in two patients (3.3%) included in the analysis. The median age of the patients with deep sternal wound infection was 65 years (39–80 years).
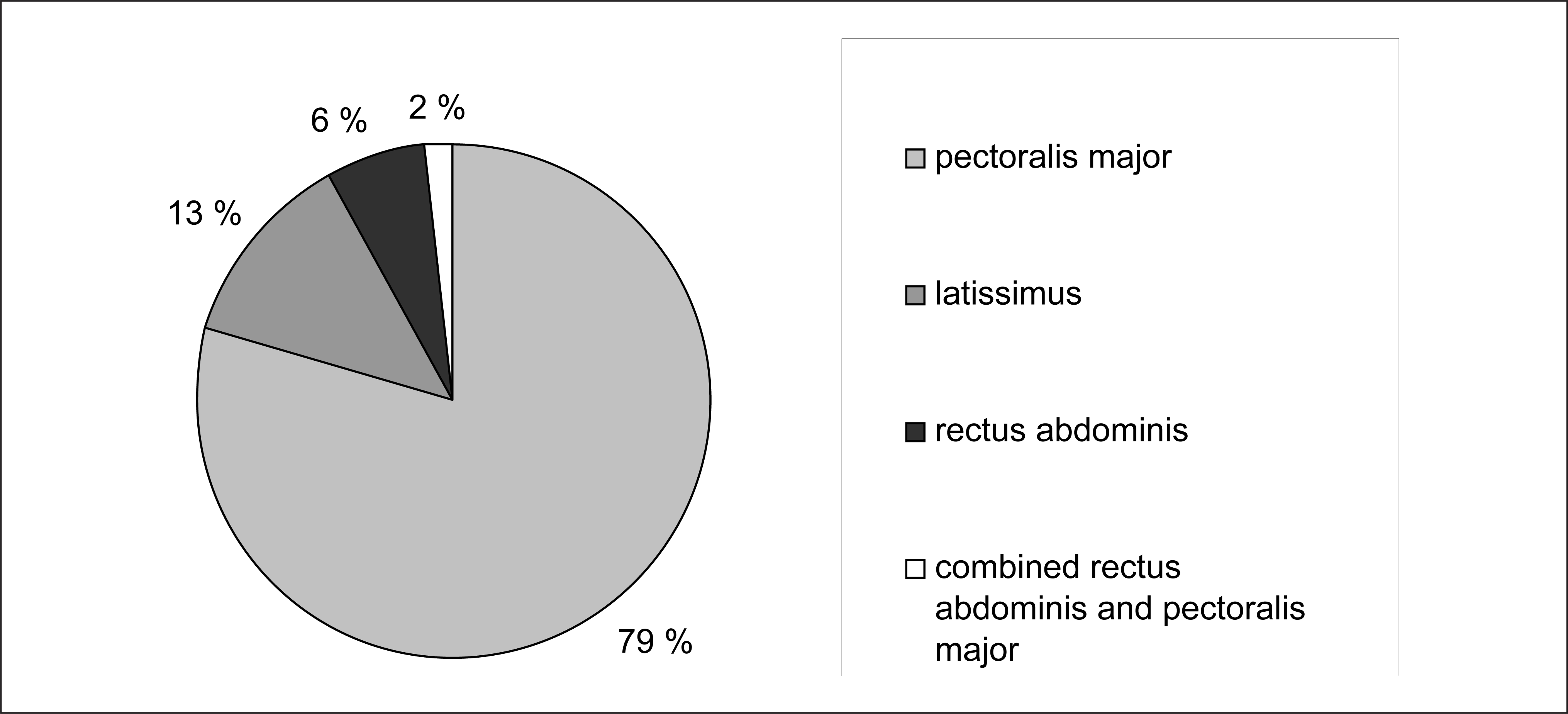
Flaps used for reconstruction
The decision for debridement and flap reconstruction was made, when diagnosis was clear based on laboratory and radiological findings and patients' clinical status. We did not postpone any debridement operation based on patients' unstable clinical condition or high CRP levels.
A prophylactic antibiotic regimen was used in all patients. The treatment followed the algorithm, which is described in Fig. 2. The one-stage approach was mostly used, where both debridement and flap reconstruction were done in the same operation. In five patients the debridement was done followed by negative pressure wound therapy and later on a flap reconstruction. Complete debridement of the wound following cardiac surgery was performed involving the removal of sternal wires, extension, necrosectomy of soft tissue and sternum. Bacterial samples from mediastinum and sternum were taken on all occasions. Also the fibrous adhesions of the right ventricle to the sternum were released to minimise the risk of laceration of the right ventricle. In 56 patients the remaining sternum was rewired and in 1 patient a plate reconstruction was used. Sternectomy was necessary in three patients. The choice of the flap used was based mainly on anatomic location of sternal wound defect and also which vessels have been used previously in the cardiac operation (Table 1) and if the sternotomy wound was heavily exudating, a continuous mediastinal irrigation was used for a couple of days postoperatively.
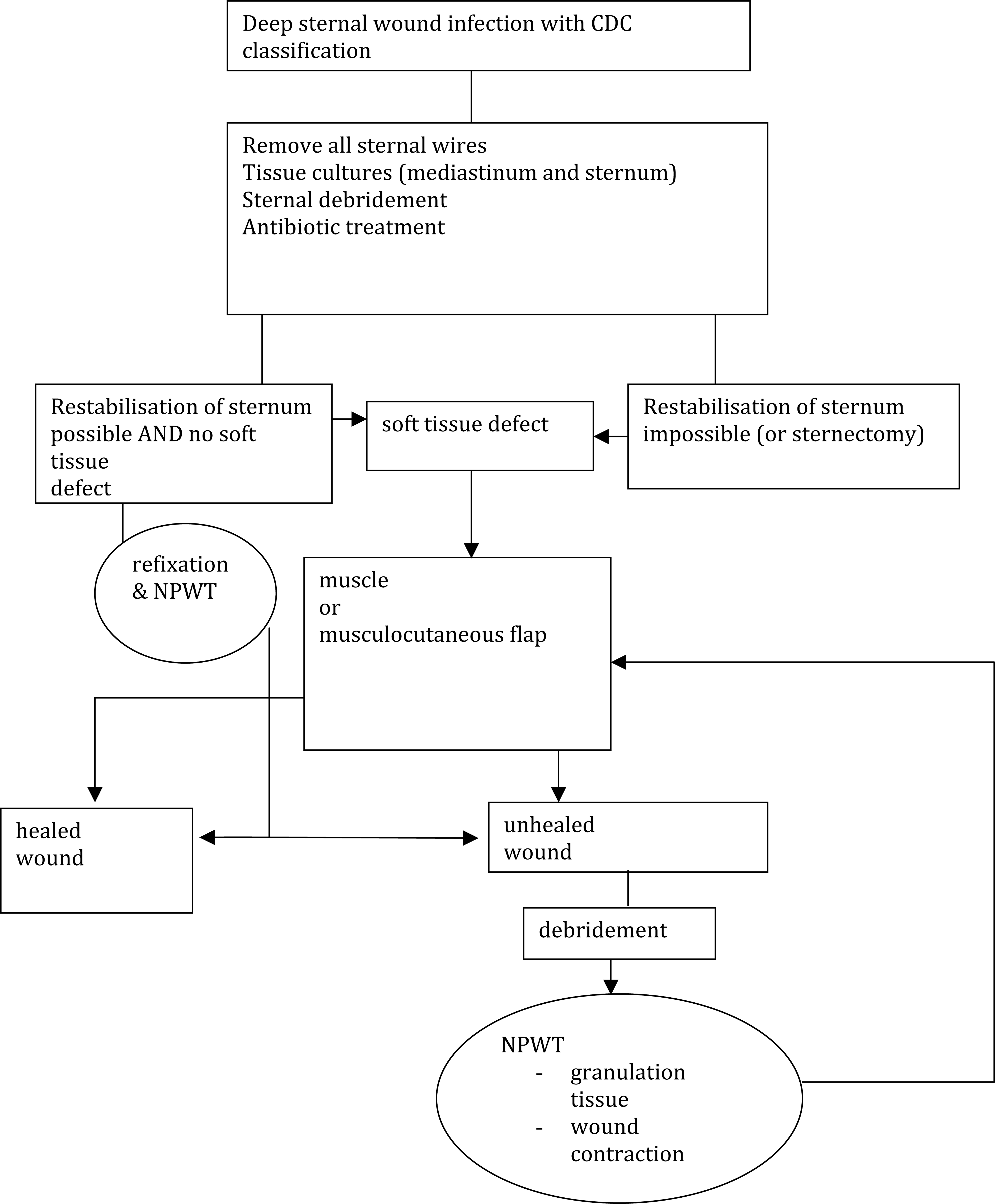
Treatment algorithm for DWSI patients
Flap selection based on both anatomical and previous use of different grafts for coronary artery bypass grafting.

Contralateral pectoralis major flap (depending on which IMA vessel had been harvested for CABG) was mainly used. It was raised from thoracic wall using diathermia beginning mobilization medially and continuing laterally. The muscle was detached from the humerus and then mobilized to the level of IMA perforators, which were indentified. The pectoralis muscle was then split according to perforators to use the pectoralis major flap divided into 2 to 3 different muscle portions to cover the whole defect (Fig. 3). If a skin paddle was needed, it was prepared with its' own IMA perforator (Fig. 3).
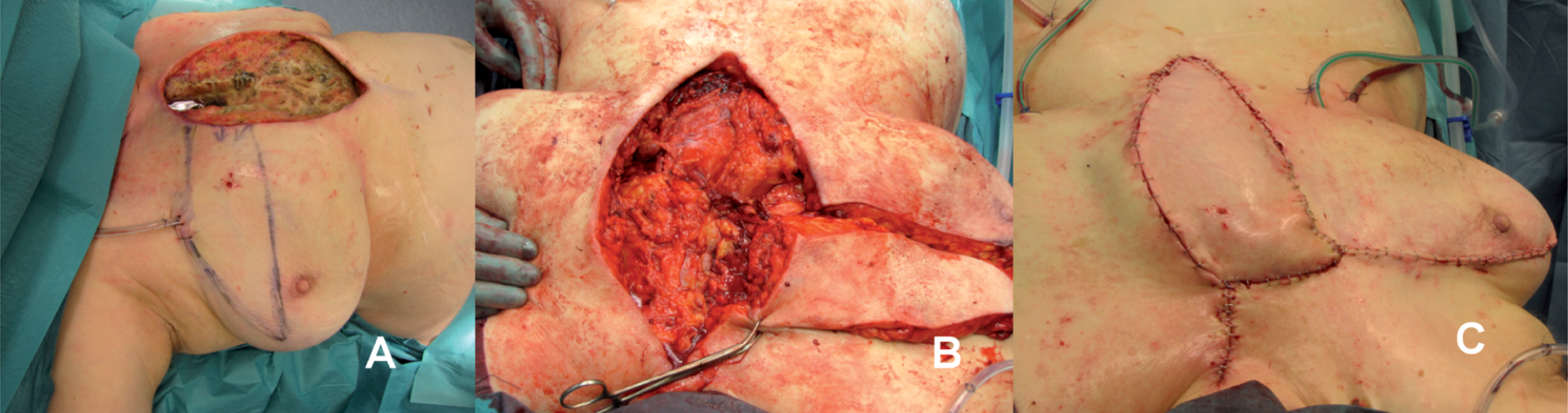
IMA perforator flap. A: IMA perforator flap design. B: Pectoralis major muscle portion sutured on sternal defect after debridement. C: IMA perforator flap sutured on skin defect of sternal skin defect.
For the latissimus dorsi reconstruction the patient's position was changed after debridement and the latissimus dorsi flap was drawn with a horizontally located skin island designed to fill the resultant cutaneous defect created by the sternal dehiscence and subsequent debridement. A longitudinal skin incision was made in the axilla parallel with the underlying muscle, and it was extended until it joined the skin island. Flap was elevated in the subcutaneous plane leaving an elliptical island of skin and subcutaneous fat tissue attached to the latissimus dorsi muscle. The sub-latissimus plane was entered and submuscular dissection was performed to undermine the entire latissimus dorsi muscle from the hemithorax. The segmental perforators on medial aspect of the muscle portion were clamped and divided. In the axilla, the thoracodorsal vessels were skeletonised up to axillary artery and vein continuing the incision to axillary fold, and then the flap was tunnelled in the subcutaneous tunnel to the midline defect to cover the whole sternal defect.
When placing the pectoralis major or latissimus dorsi flap into the sternal defect, the care was taken not to allow any dead space, and also the sternal wires were covered with muscle portion of the flap. Soft chest harness was used for external support postoperatively for all flap reconstructions.
RESULTS
There were all in all 5908 open cardiac operations performed during the study period. The overall incidence of DSWI was 1.0% within the study period. Postoperative mortality was 0% within 30 days after debridement and flap reconstruction. No postoperative sternal osteomyelitis necessitating a sternectomy was seen after debridement with flap reconstruction.
Consulting plastic surgeon evaluated reconstructive needs preoperatively and if simple split pectoralis major flap was chosen, then cardiac surgeon did both debridement and flap reconstruction (in 19 cases). If a modified pectoralis plasty or other flaps were needed, preoperatively, then the plastic surgeon made the reconstruction (31 cases). The pectoralis major was by far the most frequent flap (in 50 cases) used and it was only for very large sternal defects that the latissimus dorsi musculocutaneuos flap was chosen (in eight patients). For very distal located sternal defects, the rectus abdominis flap (in four patients) was used as a muscular or a musculocutaneous flap. In only one patient both pectoralis major and rectus abdominis were used. No microvascular flaps were needed.
Irrigation with flap reconstruction was used in 5 patients. In all of these patients their sternotomy wounds were heavily exudating pus preoperatively and 4 of these patients were septic before their debridement and flap reconstruction operation. The duration of irrigation varied from 2 to 5 days depending on the clinical situation.
The cultures from the mediastinum were positive in 56 of 60 cases (93.3%). Staphylococcus epidermidis was the most common pathogen (28 pts, 46,7%) and accounted for 28 of the infections corresponding to 46.7%. Other pathogens were Staphylococcus aureus (including one MRSA) (17 pts, 28.3%), Enterococcus faecalis in (3 pts, 3,3%), gram negative bacteria (Klebsiella, Morganella morganii, Pseudomonas aerunginosa, E. coli) in 4 patients (6.7%). In four patients no pathogens were detected (6.7%).
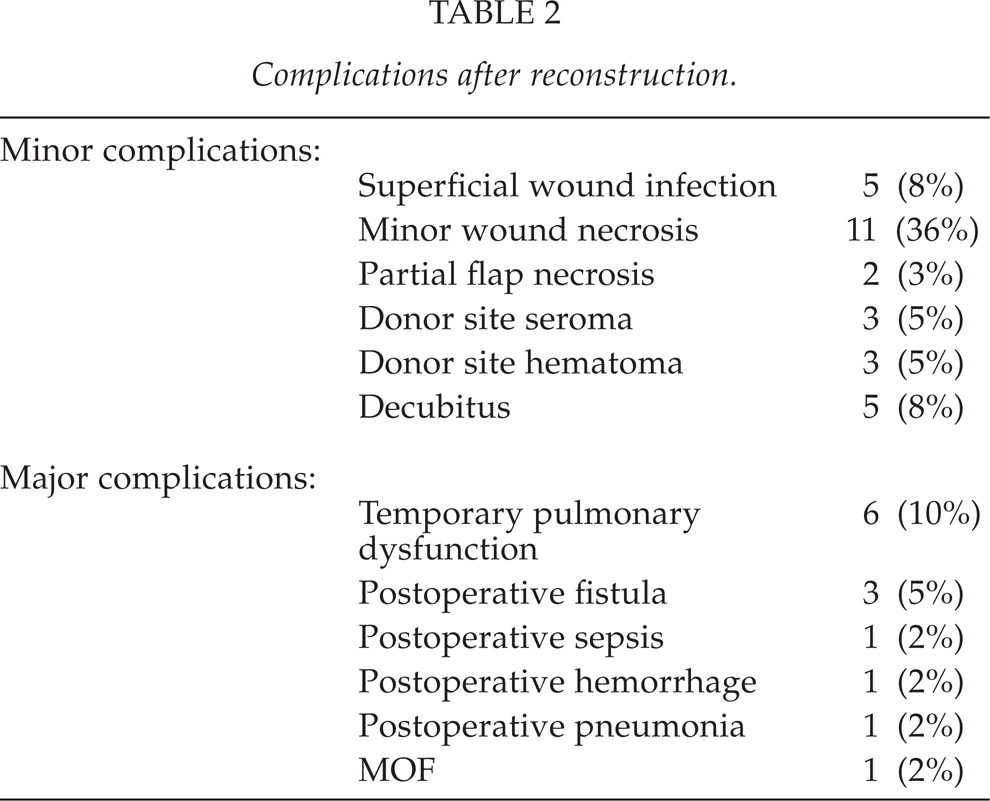
Complications are summarised in Table 2. Only in 33 patients (33/60 cases), treatment went without any complications. Even though there were many complications, these caused no mortality. Most of the complications were wound problems like superficial wound infection or minor wound necrosis. There were three cases with postoperative fistula formation, out of which two were treated with antibiotics and local wound care. In one patient with a fistula, a reoperation was necessary (soft tissue revision with perforator flap, which was modified from previous musculocutaneous latissimus dorsi flap). Reoperations were all in all needed in 12 cases (20%): for three partial pectoralis major flaps necrosis (three musculocutaneous latissimus dorsi flaps were used to repair these soft tissue defects), three reoperations for hemostasis at donor sites, five minor revisions with split thickness skin grafts. In one patient a revision with resuturation was done.
Complications after reconstruction.
Negative pressure wound therapy was used in five patients. In all of these patients, the wounds were finally closed with pectoralis major flaps. In one patient, an IMA perforator flap combined with a pectoralis major muscle was used. For postoperative wound problems, NPWT was used in eight patients to prepare the wound for final closure with split thickness skin grafts.
Seroma formation was seen in three patients with musculocutaneous latissimus dorsi flaps (3/9 patients). These seromas were treated conservatively. No flap necrosis was noticed in musculocutaneous latissimus dorsi flaps.
No right ventricular ruptures occurred during this period, but one vein graft ruptured during debridement and it was sutured. Afterwards this patient recovered uneventfully.
DISCUSSION
Deep sternal wound infection (DSWI) is an infrequent, but potentially devastating complication after cardiac surgery. The treatment is usually a prolonged one and may include multiple surgical procedures. The treatment has evolved from initial methods of wound debridement, primary sternal closure, and closed mediastinal catheter irrigation to the use of either muscle or omental flaps. Pedicled flaps do allow for an early closure of a large defect and help to reduce mortality (6, 8). Choice between muscle and omental flaps is made mainly by surgeon's preference, and is still a debatable issue.
Omentum provides a versatile tissue for wound coverage and can limit or even eliminate infection, but has a risk for peritoneal contamination or ventral hernia (9). Also it is contraindicated in patients who have intra-abdominal adhesion or are in a critical condition. There is also a risk for extensive visceral herniation, when omentum is brought up to the anterior mediastinum in front of diaphragm (10). To avoid these problems with the use of omentum, our choice was to use muscle flaps.
The primary limitation of our analysis is its retrospective nature and a quite small number of patients. As with any retrospective studies, the present series is susceptible to selection bias.
One important factor in successful outcome following DSWI is an early referral to a surgical centre with a structured approach and a well-known experience in wound-healing management. Delayed diagnosis and therapy will most likely lead to increased morbidity and mortality. During nearly the two past decades we have tried to build a practical and clear approach to the treatment of this patient group, combining the experience of both cardiac and plastic surgeons.
A well-performed debridement and restabilisation of the sternum and flap reconstruction for one-stage definitive treatment is a basis for a successful wound healing in DSWI patients. In our experience a restabilisation of sternum is necessary because of the possibility of the mechanical alteration of the instable sternum that may lead to injuries of the underlying tissues. However, if restabilisation is not possible, a well-performed wound care can lead to a firm connective tissue formation, which will act like a stable leaf making any further mechanical stabilisation unnecessary. At our unit restabilisation of sternum was mostly done with sternal wires, because it is a simple and inexpensive method and does not compromise the pectoralis flap reconstruction.
Negative pressure therapy (NPWT) was first described by Davydov et al (11) and has been successfully adapted for surgical wound management by Morykwas et al in 1997 (12). Several studies have reported the clinical benefits of NPWT in poststernotomy mediastinitis. In these studies NPWT has been successful in either as a single-line therapy, or as a procedure for providing optimal conditions for second-line treatment with delayed flap closure (13, 14, 15). NPWT is a valuable and effective adjunct to surgical management of mediastinitis, but when used for a prolonged period, it can lead to recurrent problems due to chronic infection of the deeper tissues including sternal osteomyelitis (16). Gdalevitch et al. have attempted to unveil predictors of NPWT failure and concluded that NPWT treatment failure is significantly associated with a positive blood culture, a high degree of bony exposure and sternal instability as well as wound depth of four centimetres or above (17). Also a couple of cases of fatal right ventricular rupture during NPWT therapy for DSWI have been reported (16, 18). We have tried to prevent these complications by judicious and limited use of NPWT.
In this retrospective analysis the unilateral pectoralis major flap was mainly used. It is a safe and technically very straightforward even with this modified method and also allows the use of skin paddle provided with its own perforator vessel. The pectoralis major muscles are close to the wound and easy to dissect, and thus the reconstructive method adds only a small amount of additional trauma to the patient with no risk of hernia formation. Rectus abdominis flap was not the first choice, but it was used in those four patients, in whom pectoralis muscle did not provide sufficient coverage for the sternal defect. Rectus abdominis can provide ample volume to fill the dead-space, because of its proximity to the sternal defect and it can be elevated as a musculocutaneous flap. Harvesting rectus abdominis flap requires extensive dissection with a risk for abdominal herniation and uneven contour of the abdominal wall. The latissimus dorsi flap is rarely used for sternal reconstruction after DWSI, but we have found it very useful in selected patients. The latissimus dorsi flap does not need a blood supply from the IMA and it can be elevated as a large musculocutaneous flap. The deployment of the latissimus dorsi does not entail a risk for hernia formation, neither does it result in a significant postoperative impairment of the shoulder strength or passive/active shoulder joint mobility or a significantly decreased postoperative FEV1 (19). We have not noticed any problems when using pedicled latissimus dorsi flap to cover also the caudal third of the sternum. However, donor site seroma is a frequent complication in our practice, too.
Based on these findings, the authors can recommend in the presence of DSWI a fast referral to a multidisciplinary centre. Aggressive treatment (radical debridement and a single stage flap reconstruction with muscle flaps) is recommended, whenever it is possible. NPWT is helpful for wound treatment in DSWI patients, but should not be a standard treatment for all DSWI patients.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
