Abstract
Background and Aims:
We report the results for the first consecutive 360 Roux-en-Ygastric bypass (RYGB) operations performed in a district hospital in Finland.
Material and Methods:
Demographic data, perioperative characteristics, and follow-up data were entered prospectively in the hospital's database for bariatric patients.
Results:
We performed 325 primary laparoscopic Roux-en-Ygastric bypass (LRYGB) operations, one open RYGB, and 34 revisions. Mean BMI before the operations was 47.5 ± 7.8 kg/m2 (31.5–91.0 kg/m2). The percentage of patients with type 2 diabetes mellitus (T2DM) was high; 52.3% (170 patients). The mean operative time decreased from 110 minutes during the first 108 operations to 82 minutes for the second 108 operations, and to 74 minutes for the last 109 operations (P < 0.001). Postoperative hospital stay was significantly reduced (P < 0.001) for the last 109 patients compared to the first and second group of patients. Overall morbidity averaged 19.1% (62/325). Severe complications were detected in 15 patients (4.6%). There was a tendency to more serious complications in the first group of patients. There was one death (mortality 0.3%). With a follow-up rate of 97% at 3 months T2DM had resolved in 48.2%, and had resolved or improved in a minimum of 92.9% of the diabetic patients. Weight loss (WL) and excess weight loss (EWL) averaged 20% and 46% respectively.
Conclusions:
Operative time and postoperative hospital stay decrease significantly with increasing experience. The first one hundred patients may be at higher risk for complications. LRYGB operations may have an important impact on the resolution of T2DM in the operated population.
Keywords
INTRODUCTION
The worldwide rapid increase in the prevalence of morbid obesity and “diabesity” has lead to increasing demands for effective surgical treatment options. As a consequence, many surgical centers without previous experience in this field, are adding bariatric surgery to their practice. This needs to be done as safely as possible.
According to the International Diabetes Federation (IDF) statement T2DM now affects more than 280 million people worldwide, and the number is expected to nearly double by the year 2030 (1). The IDF statement calls for increasing interventional procedures for obese patients with T2DM even in the BMI (kg/m2) range of 30 to 35 if the disease is poorly controlled despite medical therapy (1). T2DM is strongly associated with obesity as the more and more commonly used term “diabesity” also implies (2, 3). Less than 15% of T2DM patients have normal body weight (1). With increasing evidence LRYGB can be regarded as a metabolic operation, leading to long-term remission of T2DM, and dramatic improvement of other metabolic disorders, such as hyperlipidemia and hypertension in the majority of patients (4 –8). Weight loss is a secondary endpoint. Many previous studies have demonstrated that these metabolic effects are not simply a consequence of drastic weight loss and reduced caloric intake, but also of endocrine changes in the gut hormone profiles that result from surgical manipulation of the gastrointestinal tract (6). In this regard the LRYGB operation differs from purely restrictive operations, such as the gastric banding operation.
We initiated bariatric surgery with mainly gastric banding operations at our institution among the first hospitals in Finland in 1996. We have shown earlier that the long term outcome following gastric banding operations is not always convincing (9). Furthermore, the follow-up of banding patients requires a lot of resources, and we have gradually shifted towards LRYGB operations.
As the prevalence of T2DM in Finland is high, and 52% of our patients suffered from diabetes, our primary goal with bariatric surgery is resolution of T2DM, and better control of associated other metabolic disorders. Keeping this in mind, the LRYGB serves us better. However, compared to gastric banding it is a more complex procedure, and associated with a significant risk of major complications and mortality. According to a retrospective review of 3828 gastric bypass procedures anastomotic leaks occurred in 5.2% of cases, and overall leak-related mortality was 0.6% (22/3828) (10). Especially jejunojejunostomy (JJ) leak is dangerous since it is more difficult to diagnose, have a longer diagnostic delay compared to gastrojejunostomy (GJ) leaks, and is associated with 40% mortality rate (10). Overall 30-day mortality following LRYGB is reported to be less than 0.5% in most series (5,11).
The impact of the learning curve on the primary outcome of LRYGB has been reported by different authors (12 –15). Many of these reports come from larger academic institutions, and there is no previous data available from Finland. We report our experience of the introduction of LRYGB in a small district hospital in the West of Finland. We looked in more detail at operative time, postoperative hospital stay, major complications, mortality, short-term weight loss at 3, 12, and 24 months, and the resolution of T2DM at three months postoperatively.
PATIENTS AND METHODS
Bariatric surgery – mainly gastric banding operations – was initiated in 1996 at our hospital, and we introduced laparoscopic gastric bypass operations (LRYGB) in 2006. From May 2006 to March 2011, a total of 525 bariatric procedures for morbid obesity were performed. The procedures included gastric bypasses (n = 360), gastric bandings (n = 91), sleeve gastrectomies (n = 64), biliopancreatic diversions (Scopinaro, n = 2), duodenal switches (n = 2), intragastric balloons (n = 4), and distal gastric bypasses (n = 2). Of the 360 bypasses, 325 were primary laparoscopic gastric bypass operations, and one operation was performed by open technique. Thirty-four patients (9 open, 25 laparoscopic) had revisional surgery for failed previous restrictive procedures, or they had had other upper GI-surgery earlier (two fundoplications and one Heller's myotomy). These patients were analysed separately. Demographic data, perioperative characteristics, and follow-up data were entered prospectively in the hospital's database for bariatric patients. The first 30 operations were performed with circular stapling technique in the GJ, and the next 295 operations with the antecolic double loop linear stapling technique with a 150 cm alimentary limb and a 75 cm biliopancreatic limb. Both techniques have been described in detail earlier (16). The authors performed all operations. The first author performed the majority (n = 231) of the operations, and assisted the second (n = 76) and third author (n = 18) in the rest of the operations. All revisional operations (n = 34) were performed by the first author. The mesenteric defects were closed routinely with running sutures from patient number 178 onward. Patients were encouraged to follow a very low calorie (VLC) diet for 2–4 weeks preoperatively, aiming at approximately 8% weight loss before surgery.
DEMOGRAPHIC DATA
Mean age of the patients was 45.0 ± 10.8 years (range 19–67 years), and 63.7% were females. Mean preoperative weight of the patients was 138.1 ± 28.0 kg (84–285 kg), and mean excess weight 65.5 ± 24.6 kg (18.9–206.7 kg), which added up to 90.1 ± 31.2% (25.9–263.9%) in percentage. Mean BMI before the operation was 47.5 ± 7.8 (31.5–91.0). Of all patients, 308 (94.8%) suffered from at least one of the most typical co-morbidities associated with morbid obesity (such as hypertension, type 2 diabetes mellitus, arthritis, sleep apnoea, dyslipidemia, hyperuricemia and/or asthma). The number of type 2 diabetics was high, 170 patients (52.3%).
Major complications were defined as potential treats to life as shown in Table 2. All other complicatrions were considered minor.
STATISTICS
Microsoft Excel was used for data management. Statistical analyses were performed with SPSS statistical software (SPSS Inc., Chicago, IL, USA). Data are expressed as mean (range) ± standard deviation (SD). The chi-square test or Fisher's exact test was used for categorical data. The unpaired t test was used for parametric data. P < 0.05 was considered significant.
RESULTS
WEIGHT LOSS
Preoperative weight loss averaged 8.6% ± 5.3% (range 0.0–35.9%). Weight parameters before operations and at 3, 12, and 24 months are outlined in Table 1.
Weight parameters before and after 325 primary laparoscopic gastric bypass operations.
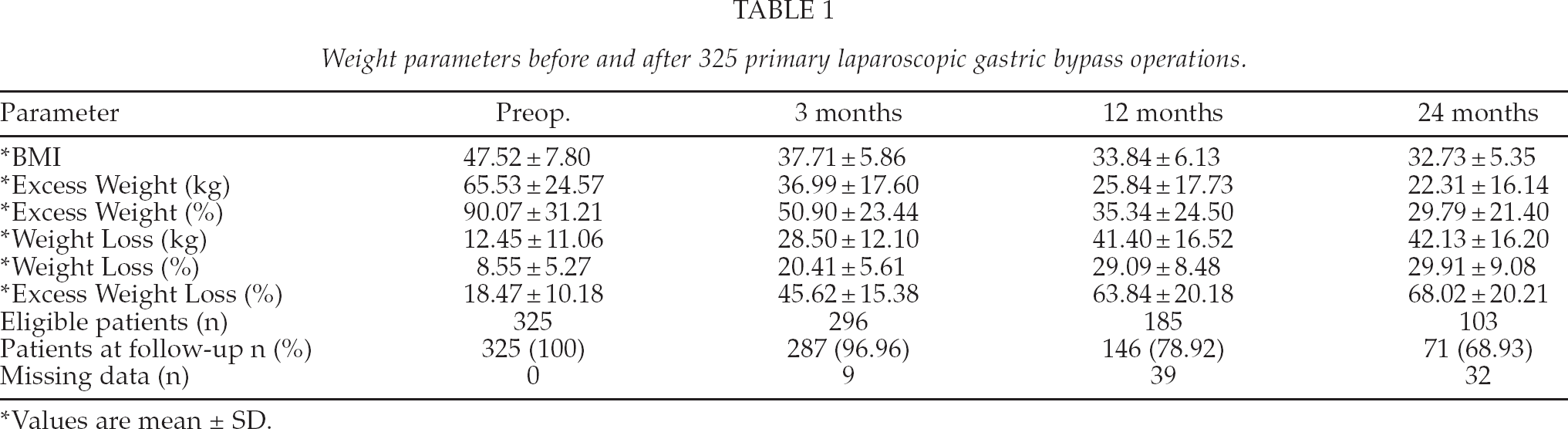
Values are mean ± SD.
RESOLUTION OF TYPE 2 DIABETES
Of the 170 diabetic patients 82 (48.2%) had discontinued all their diabetes medication by the time of their first postoperative visit at three months, and their glycosylated haemoglobin (HbA1C) levels were confirmed normal (< 6.0 % in all cases). The rest of the eligible patients (n = 76, 44.7%) had been able to reduce their medication, and had better glycemic control. Twelve patients had not yet visited the outpatient department. In conclusion, three months after the surgery type 2 diabetes had resolved or improved in a minimum of 92.9% of the diabetic patients. More long term data are not yet available.
MORBIDITY AND MORTALITY
Early major morbidity (less than30 days) is listed in Table 2, and late morbidity (more than 30 days) requiring reoperations in Table 3. Overall morbidity averaged 19.1% (62/325). Severe complications were detected in 15 patients (4.6%, Table 2). There were four leaks (1.2%), one in the JJ, two in the GJ, and one in the staple line of the remnant stomach (Table 2). Twenty-one patients (6.5%) needed 25 reoperations (7.7%, Tables 2 and 3). Two operations were converted to an open procedure (conversion rate 0.6%). One patient died (mortality 0.3%) three weeks after the operation from septic complications following a rupture of the staple line of the excluded stomach (blow-out) on the third postoperative day. She did not recover from a deep septic shock despite immediate reoperation. The cause of this fatal complication was a blood clot in and around the JJ causing twisting and obstruction of the biliopancreatic limb.
BMI, operative time, postoperative hospital stay, major complications (less than 30 days), and reoperations in the first, second and third group of patients in a total of 325 consecutive primary laparoscopic gastric bypass operations.
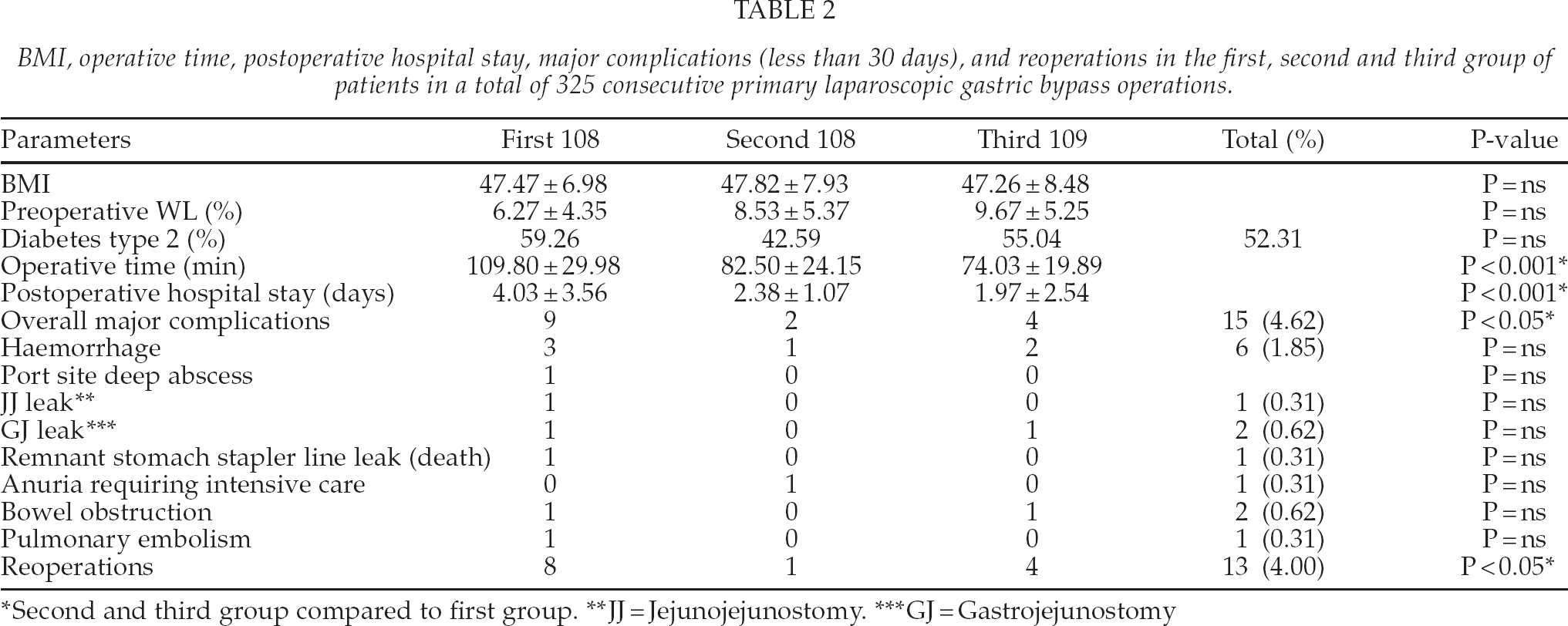
Second and third group compared to first group.
JJ = Jejunojejunostomy.
GJ = Gastrojejunostomy
All the six internal hernias in our series have been diagnosed among the first 177 patients (6/177 = 3.4 %). During their operations the mesenteric defects were not closed on a routine basis (Table 3). Until today, no internal hernias have been diagnosed in the patients operated since closure became a routine.
Complications requiring late (more than 30 days) reoperations during first, second and third group of patients in a total of 325 consecutive primary laparoscopic gastric bypass operations.
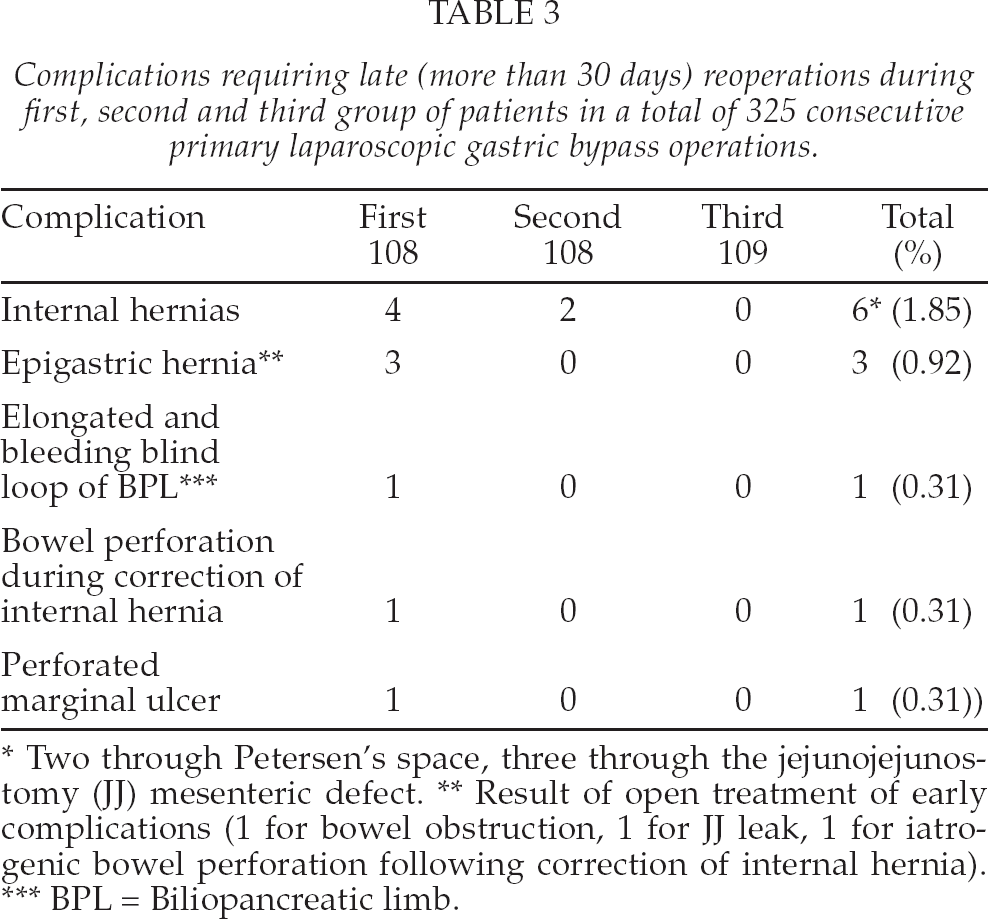
Two through Petersen's space, three through the jejunojejunostomy (JJ) mesenteric defect.
Result of open treatment of early complications (1 for bowel obstruction, 1 for JJ leak, 1 for iatrogenic bowel perforation following correction of internal hernia).
BPL = Biliopancreatic limb.
IMPACT OF THE LEARNING CURVE
Overall mean operative time was 87.4 ± 30.0 minutes (range 45.0–238.0 minutes), and mean postoperative hospital stay was 2.8 ± 2.7 days (1–30 days). Operative time, prevalence of type 2 diabetes, BMI, postoperative hospital stay, major complications, and reoperations (less than 30 days) during the first, second and third group are listed in Table 2.
REVISIONAL SURGERY
Thirty-four patients had revisional surgery, and are listed in Table 4. The most common reason was a failed gastric banding operation (Table 4). Mean operative time in this group was 113.2 ± 34.0 minutes (range 54–184 minutes). Nine patients (26.5%) suffered from postoperative complications, and three of these (8.8%) were considered serious; two leaks in the GJ (5.9%), and one pulmonary embolism (2.9%). In addition, one pneumonia, one small subphrenical abscess, 2 bleedings, and 2 unclear infections were managed conservatively. Excess weight loss at three months in this group did not differ from the group that had had no previous bariatric surgery, and averaged 44.0% (vs 45.6%, Table 1).
Conversions to laparoscopic Roux-en-Y gastric bypass (LRYGB)in 34 patients.

Seven operations by laparoscopic band removal and conversion to LRYGB during the same operation.
DISCUSSION
The increase in bariatric procedures in Finland has been slower compared to other Scandinavian countries. The total annual number of bariatric operations in Finland in 2010 was 766 operations, compared to more than 8000 operations in Sweden, and more than 5000 operations in Denmark. However, LRYGB procedures for morbid obesity are currently increasing in Finland with many new centers starting up this practice.
This is the first Finnish report on the primary outcome of LRYGB, and represents a series of 360 consecutive cases performed in a district hospital. We have shown that both operative time and postoperative hospital stay decrease with increasing experience. Similar effects of the learning curve have been reported earlier (13, 14). Although our major complication rates are similar to those of other published series (4, 5, 13,17), their number diminished after the first third of the operations, showing the effect of the learning curve. In other words, the first 108 patients were at higher risk. Type 2 diabetes may increase operative risk, but the number of diabetics was not significantly different in the first 108 patients compared to the later groups of patients (Table 2). Setting up a new practice includes high risks, but those risks should be eliminated with proper planning, training, and expert assistance. Our results show that we have failed in this regard. The effect of the learning curve could not be entirely avoided.
As the giants of bariatric surgery, Henry Buchwald and Nicola Scopinaro, point out in a leading article (18), there should not be any learning curve. The initial higher risks can, and should be eliminated with proper training, by visiting expert centres, consulting expert assistance, and avoiding high-risk patients during the first operations. We took all these steps of caution. We visited several high volume centres in Belgium, Spain, Sweden, Norway and Denmark. We trained in animal laboratories. An outside experienced expert assisted in the first two operations. In addition, we were not unfamiliar with advanced laparoscopic surgery or other bariatric, restrictive procedures. Laparoscopic surgery, intra-corporeal suturing and anastomotic techniques had been practised during a decade of fundoplications, re-do fundoplications, gastric bandings, laparoscopic hemicolectomies and subtotal gastrectomies. Despite all this we encountered more major complications in the first group of patients than in the later groups, and we lost one patient (37th in the series).
On the other hand, our results are acceptable when comparing major complication rates (4.6%), leaks (1.2%), haemorrhage (1.9%), reoperations in less than 30 days (4.0%), and mortality (0.3%) with the rates in the meta-analysis by Maggard et al. (19), or the multicentre study by Flum et al. (17), showing major complications in 4.3%, leaks in 2.2%, bleedings in 2.0%, and mortality in less than 0.5% of the operations.
Revision surgery is associated with prolonged operative time and higher risks (20). It is therefore justified to analyse these operations separately. Serious complications and leaks were more common in this group compared to those with a primary operation. There was no mortality in this group. According to a retrospective review of 3828 gastric bypass procedures, anastomotic leaks following revisions occurred in 8% of cases (10). We encountered two leaks (5.9%) in this group (overall rate 6/360 = 1. 7%), both of them in the GJ. The first one was detected in a patient with a previous open vertical banded gastroplasty performed more than 15 years earlier. She had a conversion to a LRYGB with some technical difficulties and developed a small leakage in the posterior stapled corner of the GJ. The other patient with leakage had had an obviously successful laparoscopic operation for achalasia 10 years earlier (by the first author) by Heller's myotomy and a Toupet fundoplication. There was dense scarring, and some tension in the anastomotic staple line could not be entirely avoided. However, both patients were managed with immediate drainage, repeated endoscopic tissue gluing, and both fistulas had sealed completely after one month. Seven of the 28 conversions to RYGB following failed gastric bandings (Table 4) were performed by laparoscopic removal of the band and conversion during the same operation. The rest of these operations were performed in two steps; the band was removed during the primary operation, and a RYGB was performed 4–6 months later. This may be considered a safer way to accomplish conversion from gastric banding to LRYGB (21).
Whether the mesenteric defects should be closed or not is still disputed, at least in Scandinavia. The incidence of internal hernias following LRYGB without closure has been reported to be in the range of 1–5% (19, 22, 23), although the true incidence is not known, and is probably higher. Many patients complain about abdominal pain after bypass surgery, and some of these patients may suffer from undiagnosed intermittent internal hernia. Closing the defects may not entirely eliminate the risk, which is one of the arguments against closing them. The overall incidence in our study was 1.7% (6/360), but it may rise with longer follow-up. One of the reasons for internal hernia formation following LRYGB can be the diminished intra-abdominal adhesion formation (compared to open surgery) associated with laparoscopic surgery in general. During the era of open RYGB operations the problem of internal hernias was less heard of. The symptoms can be very dramatic for the patient, and the condition potentially fatal. The diagnosis is difficult, especially if the patient is seeking advice in a center not familiar with bariatric surgery. We did not close the defects in the beginning (the first 177 patients), and until today we have encountered 6 internal hernias (3.4%). We discovered how to close both mesenteric defects – the space between the antecolic alimentary limb and the transverse mesocolon (Petersen's space), and the defect created by forming the jejunojejunostomy – in a safe and rapid manner. It prolonged our operative time approximately 10 minutes, but despite this the mean operative time decreased, and is still decreasing (data not shown). After we started closing the mesenteric defects in a routine manner, no internal hernias have been diagnosed.
Our short-term outcome, with a mean excess weight loss of 68% at two years is consistent with the commonly reported range of 60–70 % (4, 5, 19). More interesting is the fact that almost half of our T2DM patients had a complete remission of the disease, and the other half had been able to reduce their medication already at the time of the first postoperative visit at 3 months. Excess weight loss at this time averaged 46%. The number of T2DM remissions at 3 months is probably underestimated since many patients were still using medical therapy despite normal HbA1C levels. It is difficult to conclude whether those normal levels were the result of successful medical therapy or if the medication had become unnecessary. According to our follow-up programme the diabetologists will make more permanent conclusions in the future, and the follow-up in this regard is still incomplete. The percentage of remissions is likely to rise with further follow-up. In the meta-analysis by Buchwald et al. (4), complete remission of T2DM following LRYGB averaged 80% after various follow-up periods. However, according to the SOS study long-term (more than 10 years) remission rate was only 36 % (8). The majority of the operations in the SOS study were restrictive operations, which are less effective in treating metabolic disorders than RYGB.
The incidence of T2DM is rising rapidly worldwide, and T2DM has been described as the “global health crisis of the next generation” (24). The prevalence of T2DM in Finland is high, and 52% of our patients suffered from this disease. The number is much higher than the usual 10–20% T2DM rate reported in most series (4,5,19). Bariatric surgery, especially LRYGB will be a valuable treatment option in the fight against this chronic disease and its many complications. It is of great importance that the IDF now acknowledged this fact in their recent statement at the Second World Congress on Interventional Therapies for T2DM (1). Surgeons and internists should work together. There seems to be enough evidence for offering surgical interventions for even less overweight T2DM patients, in the BMI range 30–35, when conventional treatments are insufficient or have failed (1).
In conclusion, we have showed that LRYGB can be introduced relatively safely on a larger scale in a small hospital with acceptable complication and mortality rates, with good short-term weight loss, and a valuable impact on T2DM. However, the effects of the learning curve could not be entirely avoided.
Footnotes
ACKNOWLEDGEMENT
We thank MD, MA Jaana Elberkennou for her kind revision of the English language.
Drs Mikael Victorzon, Pipsa Peromaa, and Pekka Tolonen have no conflicts of interests or financial ties to disclose.
