Abstract
Patients with critical limb ischaemia (CLI) are usually elderly and suffer from several co-morbidities. The goal of surveillance after both endovascular and surgical revascularization for CLI is not only the protection of re-established distal perfusion and sustained ambulation but also the reduction of systemic atherothrombotic risk and mortality by ensuring continued best medical care. However, preferred format and rhythm of structured follow-up programs have remained controversial, mainly because of lack of compelling evidence. This review aims to summarize and to appraise available information critically. Thereby, it underlines the importance of systematic surveillance after both surgical and endovascular revascularization for CLI. Recent European guidelines are considered and areas of uncertainty are highlighted and discussed. According to currently available literature and recent guidelines, the early duplex scan is justified in all patients undergoing endovascular or surgical distal revascularization for CLI. There is no best level evidence supporting continued long term duplex surveillance of revascularizations with normal findings at early duplex scan, whereas those patients with abnormal early duplex scan or high risk revacularization are likely to benefit from continued duplex surveillance. Regular clinical follow-up is suggested and clinical deterioration should trigger duplex scanning to ensure revascularization patency.
INTRODUCTION
Follow-up after revascularization for chronic critical limb ischemia (CLI) aims at sustained local treatment success and continued best patient care. As most CLI patients are elderly and frail, sustained resolution of CLI and preserved ambulatory capacity may decide over independent living and overall prognosis. However, the widespread assumption that, in the context of CLI, efficacy of initial revascularization will determine prognosis during the short remaining life expectancy has all but discouraged focused and prospective research into preferred type and structure of follow-up. Therefore, apart from a few exceptions, available evidence is retrospective and uses inconsistent reporting standards, and hardly any study has been stratified for presence of CLI (1). This narrative review attempts to summarize currently available data concerning post-interventional surveillance and aims to appraise available evidence for best clinical practice critically including anti-platelet and antithrombotic therapy, clinical surveillance, use of duplex ultrasound, and indications for and preferred type of repeat interventions for failing and failed reconstructions.
GENERAL CONSIDERATIONS
Approximately 10 to 20% of all vein grafts fail within the first year after bypass, and up to 25% within 3 years (2). Depending on its timing, vein graft failure can be divided into three phases (3). Early (< 30 days) failures are usually due to technical mistakes, such as poorly constructed anastomosis, insufficient in- or outflow, retained venous valves, or poor-quality vein. Any of these reasons can reduce graft flow below the thrombotic threshold velocity and lead to graft occlusion. Hypercoaguable states may also cause early vein graft failure (4). Intermediate (30 days-2 years) vein graft failure is mainly due to intimal hyperplasia. As a response to the arterialization of the vein graft and surgical trauma, vascular smooth muscle cells (SMSs) migrate from the media to the intima and switch phenotype to a more proliferative form which leads to a production of excessive amounts of extracellular matrix and subsequent intimal hyperplasia (5). Although all vein grafts probably undergo some degree of intimal hyperplasia, it remains unclear why some are affected more than others. Finally, late (> 2 years) vein graft failure is commonly attributed to progression of the underlying atherosclerotic disease with critically compromised in- or outflow (5). Similar mechanisms may affect patency of endovascular revascularizations. Technical failures include over- or undersizing of balloon angioplasty, bulky recoil of the dilated segment, local trauma such as wall dissection or perforation, and inadequate in- or outflow. Analogously, the intimal hyperplasia reaction is triggered by the arterial response to local trauma and continued mechanical irritation by a stent. However, in contrast to surgical bypass, the preserved collateral vessels after endovascular revascularization may attenuate the immediate clinical impact of local restenosis or reocclusion (1).
COLOR-DUPLEX SURVEILLANCE AFTER BYPASS
Graft stenoses are usually asymptomatic at first but, if not corrected, may limit the graft flow critically and lead to subsequent graft occlusion. As graft salvage after occlusion is poor (2-year patency rates are as low as 19%–31%) (6), it is crucial to identify and revise significant stenoses prior to graft occlusion. The positive predictive value of ankle-brachial index (ABI) alone in detecting a failing vein graft has been estimated as low as 12%–34% (7). In contrast, stenoses can be detected reliably with colour-duplex (CD) ultrasound. However, optimal intensity and target population of cost-effective vein graft CD surveillance have remained controversial, since graft failure does not necessarily imply limb loss, which is the clinically relevant outcome. It has been estimated that a systematic surveillance programme has to improve the limb salvage rate by at least 5% to justify associated costs (8). Four randomised controlled trials (RCTs) assessed the efficacy of systematic CD surveillance. Lundell et al. (9) studied both vein graft and prosthetic bypasses and reported a better identification of failing vein grafts by intensive duplex surveillance, which led to significantly better assisted primary and secondary patency rates when compared to clinical surveillance alone. However, leg salvage rates did not differ between the groups. Ihlberg et al. (10, 11) on the other hand, failed to demonstrate any difference in assisted primary patency, secondary patency or limb salvage rates between the duplex and the clinical surveillance group. Lastly, the largest RCT published by Davies et al. (3) randomised 594 patients either to clinical (n = 290) or to duplex (n = 304) surveillance. More graft stenoses were missed in the clinical surveillance group, but primary, assisted primary and secondary patency rates did not differ between the groups. Table 1 summarizes available studies assessing postoperative duplex surveillance. A systematic review of 6649 vein grafts concluded that CD surveillance significantly reduced the number graft occlusions, but failed to improve limb salvage (12). Thus, the second TASC consensus document recommends clinical surveillance of vein grafts but does not recommend routine duplex scanning (13).
Studies assessing duplex surveillance after distal bypass for CLI. Modified from Dick et al 2011 (1)
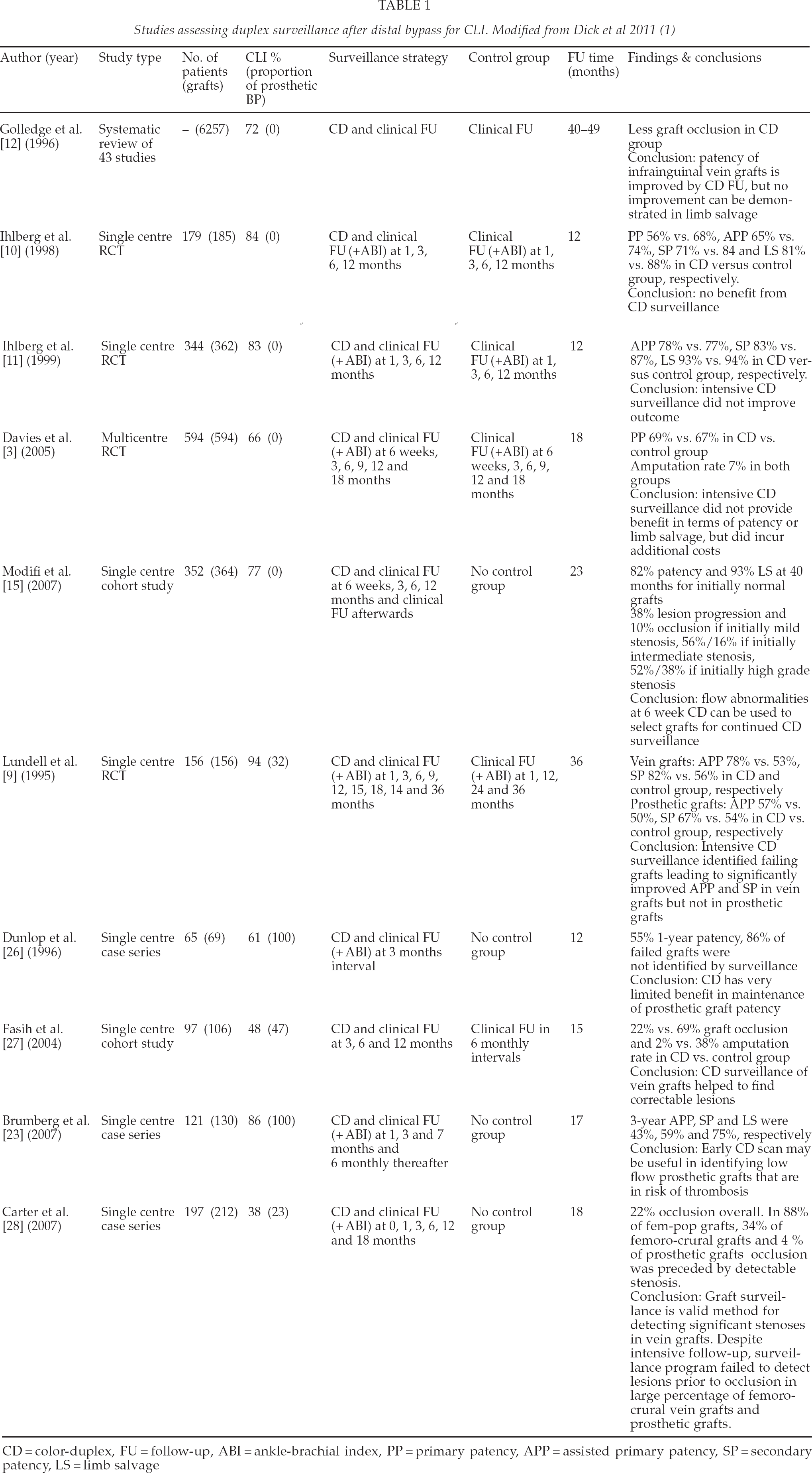
CD = color-duplex, FU = follow-up, ABI = ankle-brachial index, PP = primary patency, APP = assisted primary patency, SP = secondary patency, LS = limb salvage
However, CD surveillance may still be (cost-) effective if limited to a suitable subset of patients at risk. Several studies have assessed characteristics of failing grafts to identify those patients who would benefit most from intensive duplex surveillance (1). On an early duplex scan approximately 25% of all patients present with abnormal findings (i.e. peak systolic velocity (PSV) < 45 cm/s; PSV > 300 cm/s or PSV ratio > 3) (3). Half of them will need repeat interventions later on to maintain graft patency and limb salvage (14). In contrast, patients with a normal duplex scan at 6 weeks to 6 months have a low risk of subsequent graft occlusion if clinical surveillance (ABI, limb status) remains unchanged (15, 16). Other predictors of vein graft failure are decreasing serial ABI, small diameter (< 3 mm) grafts, non-single segment or long (> 50 cm) great saphenous vein grafts, and redo bypass (17, 19). Arm vein grafts are particularly prone to stenosis (20, 21). Therefore, an early duplex screening following vein bypass seems justified for all CLI patients, but only those with an abnormal early scan or specific patient- or procedure related risk factors will benefit from continued duplex surveillance (1).
For prosthetic grafts, there is currently no clear evidence to support any form of duplex surveillance (9, 22). The mechanism of prosthetic graft failure is believed to relate mainly to low graft flow. Although theoretically, anastomotic stenoses predisposing to low flow can easily be detected with colour duplex, evidence of its cost-effectiveness is still lacking. According to Brumberg et al. (23), however, an early duplex scan of below knee prosthetic grafts may still be reasonable – not for triggering revision of stenotic anastomoses but for identifying low-flow grafts in order to select patients for anticoagulation.
COLOUR-DUPLEX SURVEILLANCE OF ENDOVASCULAR REVASCULARIZATIONS
Imaging surveillance of endovascular revascularizations is mainly confronted with the challenge of localizing the treated arterial segment again. Local treatment failure such as restenosis or occlusion is easily confounded with progressive atherosclerotic disease aside. Moreover, established collaterals may attenuate the clinical impact of local restenosis or occlusion suggesting that reinterventions should be driven by clinical need rather than prophylactically. Therefore, usefulness of duplex surveillance in addition to close clinical follow-up might be limited in an endovascular context theoretically. So far, no RCT has investigated the efficacy of routine CD after endovascular interventions. However, as with surgical bypass, early CD screening could be beneficial to detect residual stenoses which identify patients at risk. Such stenoses are missed on completion angiography in up to half of patients, and they are known to be associated with a higher amputation rate when compared to normal early duplex (20% vs. 5%) (1).
BEST MEDICAL TREATMENT
Best medical treatment and smoking cessation advice certainly represent the mainstay of care for all vascular patients, even though this has not been explored specifically in the context of revascularized CLI. However, to reduce the increased risk of cardiovascular death, all recommendations regarding risk reduction therapy and secondary prophylaxis apply to these patients, and monitoring of patient compliance may be particularly important after (successful) revascularization. In addition, the best medical therapy including platelet inhibitors, antithrombotic agents and statins positively affect patency of both surgical bypass and endovascular reconstructions (1).
Of note, available evidence is based on heterogeneous studies involving various proportions of patients with CLI and different types of reconstructions (1). In these, platelet inhibitors consistently improved graft patency rates as compared to placebo. Patients with a prosthetic graft appear to benefit more from platelet inhibitors than those with a venous graft, whereas patients with a venous bypass appear to benefit more from vitamin K antagonists, particularly following below-knee bypass (24). After endovascular revascularizations for CLI, there is no consistent evidence regarding the optimum antithrombotic strategy. However, long-term platelet inhibition (50 to 300 mg ASA daily) appears to be preferred to vitamin K antagonists and should commence prior to intervention. Evidence from coronary interventions suggests that more potent platelet inhibitors such as thienopyridines (clopidogrel) or dual antiplatelet therapy might confer additional benefits (1). However, specific data for dual antiplatelet therapies for CLI revascularizations is lacking. The CASPAR trial (25), for example, included 851 patients, but one third of patients were claudicants.
REPEAT INTERVENTIONS
Approximately 40% of CLI patients undergoing vein bypass will need a secondary intervention during follow-up, and in 35% of patients a repeat intervention is needed within one year after endovascular reconstruction (1). Success rates of such secondary procedures are generally high, as long as the bypass or the angioplasty segments were not occluded. In contrast, once a graft has failed long-term secondary patency and limb salvage rates are markedly impaired. Stenotic vein grafts can be salvaged by endovascular and surgical techniques with similar overall success, even though surgical revision necessitates less subsequent repeat interventions. Overall revision failure is in the range of 30% and similar between surgical and endovascular approaches. Therefore, the choice of the preferred technique depends on anatomic characteristics and the accessibility of the lesion. Outcome of recurrent stenosis or lesions within anastomoses is markedly inferior after repeat endovascular angioplasty; therefore, such lesions probably benefit from surgical revision. In contrast, endovascular revision is probably suited best for short (< 2 cm) and late-appearing (> 3 to 4 months) lesions involving the mid-graft (1).
Bypass occlusion, on the other hand, is a critical event and should undergo urgent revision if the bypass is to be maintained. Nonetheless, salvage attempts fail in up to 65% of patients, and about every second occlusion will eventually lead to amputation. Surgical revision including placement of a new bypass is preferred for occluded vein grafts, whereas for occluded prosthetic bypasses, graft salvage attempts lead to acceptable results only for above knee and extra-anatomic grafts. In contrast, prosthetic below knee grafts should be replaced using an autologous vein conduit to a new outflow artery (1).
DISCUSSION
It has been estimated that on average the cost for 5-year surveillance including CD is equal to the cost of an initial bypass procedure, whereas the cost of a bypass procedure plus the cost of surveillance during 5 years approach the overall cost of a primary amputation. Moreover, the revision of a stenotic but patent graft (as identified by CD) is significantly less expensive than the revision of an occluded graft, which also more often leads to major amputation (33% vs. 2%) (1). Therefore, limb salvage-related expenses including CD surveillance seem economically reasonable in patients with CLI as long as amputation rates are reduced by at least 5% (8).
The main reason for the lacking level 1 evidence for routine CD surveillance of CLI revascularizations is that none of the published RCTs (3, 9 –11)was sufficiently powered and stratified for CLI, and that the available RCTs only included vein grafts that were still patent at 4–6 weeks after surgery. However, as elaborated in recent guidelines (1), CD screening still has an accepted role to identify grafts at risk including those with early flow disturbances. Moreover, there are defined subgroups of patients that benefit from intensive surveillance as opposed to others. For instance, arm or spliced vein grafts are often the last autologous option for bypass, but they are prone to focal stenoses. Hence, every effort should be made to monitor and maintain such “high-risk grafts”.
Another important issue that cannot be based on appropriate evidence is the preferred duration of CD surveillance. The obvious targets of focused surveillance are developing stenoses within the arterial reconstruction. Therefore, CD surveillance efforts could be limited to early and intermediate revascularization failure, whereas late revascularization failure due to progressive disease is conceptually addressed by best medical care including statin and antithrombotic therapy and smoking cessation. As early and intermediate failures most often occur within the first two years after revascularization, this period might represent an appropriate duration of CD-surveillance, after which clinical follow-up should be sufficient to ensure best medical care.
However, as long as well-designed studies with a prospective focus on surveillance of CLI patients after arterial reconstruction are lacking, the optimal duration and combination of CD and clinical surveillance will remain uncertain. Similarly, exact role and preferred duration of CD surveillance after endovascular interventions need to be established. Moreover, studies should focus on differential impact of dedicated endovascular adjuncts such as bare or drug eluting stents, drug eluting balloons or atherectomy devices. Last but not least, cost-effectiveness analyses will be indispensable to establish a generally accepted surveillance algorithm after both bypass and endovascular revacularization procedures for CLI patients.
In conclusion, critical appraisal of currently available evidence including recent guidelines suggests that a single early duplex scan is justified in all patients undergoing any type of arterial reconstruction for CLI. The vast majority presents with normal findings at 1 to 6 months after surgical bypass or endovascular revascularization and will be sufficiently monitored by regular clinical examinations thereafter. Thus, in these patients there is no best level evidence to support routine long term duplex surveillance. Importantly, however, clinical deterioration and decreasing ABI should trigger repeat duplex scanning of the reconstruction. In contrast, those patients with abnormal early duplex scans or arterial reconstructions at risk as identified by graft-, flow- and patient-related factors are likely to benefit from continued duplex surveillance. The best medical care should be continued and patient compliance monitored after every revascularization, as it effectively reduces cardiovascular mortality whereas patency of reconstruction and limb salvage are improved. Finally, repeat interventions need to be tailored carefully to the underlying cause of graft failure, and should involve both endovascular and open surgical salvage procedures.
