Abstract
Neuropathy, peripheral arterial occlusive disease and microvascular disturbances are important factors contributing to foot problems in diabetic patients. In the diabetic foot with ischemia, the alterations in skin microvascular function are pronounced including severely reduced capillary circulation and abolished hyperaemic responses. These microvascular disturbances, which are superimposed on the already existing structural diabetic microangiopathy, are compatible with a state of “chronic capillary ischemia” and an increased shunting of blood through arteriovenous channels. This maldistribution of blood in skin microcirculation is not detected by measurement of peripheral blood pressure (systolic ankle blood pressure, systolic toe blood pressure). As indicated in several studies toe blood pressure is a poor predictor of local tissue perfusion, tissue survival and healing of chronic foot ulcers. Consequently, the disturbances in peripheral tissue perfusion of the diabetic foot may be underestimated leading to delayed vascular interventions and/or medical treatment. Thus, measurements of peripheral blood pressure, e.g. toe blood pressure, should be combined with investigations of local tissue perfusion in order to get an adequate estimation of peripheral tissue perfusion in diabetic patients. For this purpose local skin microcirculation can be investigated by transcutaneous oxygen tension of the forefoot. Also, due to these reasons, the threshold for revascularization should be lower in diabetic patients with foot ulcer.
Keywords
PERIPHERAL ARTERIAL OCCLUSIVE DISEASE
Peripheral arterial occlusive disease (PAOD) is 2–4 times more common in patients with diabetes than in patients without diabetes (1). It also developes at a younger age, and women are equally affected as men. In a group of patients with type 1 diabetes and a mean age of 28 years, 12% had developed asymptomatic PAOD when reinvestigated six years later (2). Glycemic control is important for decreasing the risk of PAOD (2). In United Kingdom Prospective Diabetes Study (UKPDS), 11% had developed PAOD six years after diagnosis of type 2 diabetes and for each percent increase of glycated haemoglobin (HbA1c) the relative risk of PAOD increased by 25–28% (3).
The vascular changes in diabetic patients are more diffuse and distally located, commonly at an infrapopliteal level (1). Stiff arteries (i.e. mediasclerosis or Mönckeberg's sclerosis), is more common in patients with diabetes, neuropathy and/or kidney failure (4). Mediasclerosis is characterized by calcification of the smooth muscle cells in the arteries sometimes causing incompressible arteries and falsely elevated blood pressure (pseudohypertention) especially at the ankle level. Since mediasclerosis is almost absent in the digital arteries, measuring toe blood pressure is better for evaluating the arterial circulation of the legs (5). It should also be stressed that decreasing the central blood pressure by antihypertensive medication may be deleterious for the peripheral blood supply in patients with PAOD, especially so in patients with critical limb ischemia (CLI).
Smoking is deleterious for patients with PAOD, and a clear dose-response relationship has been shown in these patients (6). The progress of PAOD is more aggressive in smokers and vascular interventions are less successful. Cessation of smoking reduces both the risk of severe symptoms and the risk of death in vascular complications.
CRITICAL LIMB ISCHEMIA
CLI represents an advanced stage of PAOD where the arterial blood supply to the limb and foot is severely reduced and the viability of the tissue jeopardized. The atherosclerotic changes of the large arteries lead to an increased resistance to blood flow with a critical reduction in perfusion pressure and nutritive blood supply to the tissues. The reduced blood supply may lead to tissue ischemia causing rest pain, tissue necrosis, non-healing foot ulcers and gangrene. CLI is a severe condition associated with high morbidity, poor prognosis, high medical costs and severe suffering for the patients. Patients are also rather old and with other disabling conditions such as renal insufficiency and cardiovascular disorders.
A typical symptom of CLI is pain in the supine position. In order to reduce the pain the patient has to take a sitting, or standing position. The reason for this improvement is that the local hydrostatic blood pressure increases to the limb and foot with an improved nutritional blood supply to the ischemic region. If no treatment is performed or is possible in this situation, the poor blood perfusion may lead to tissue necrosis and risk of amputation. Notably, the pain in diabetic patients is often less dramatic due to a concomitant peripheral neuropathy. There are also other factors which may disguise the clinical suspicion of ischemic ulcers in diabetic patients, e.g falsely high ankle blood pressure due to mediasclerosis and reduced inflammatory responses to tissue injury. Consequently, medical investigation and treatment may be delayed both due to patient's delay and doctor's delay.
It has been estimated that 5 to 10% of patients with asymptomatic PAOD progress to CLI within 5 years, and that 1–3% have evidence of CLI at the first investigation (7). The progress to CLI is individual and does not follow a standardised pattern. In addition, due to differences in the definition of CLI, the number of patients suffering from CLI is difficult to estimate. An objective test to verify the hemodynamic changes in the limb and foot is needed to confirm PAOD/CLI. In the TASC-documents, the latest from 2007 (7), investigations of local microcirculation in the ischemic skin area are recommended in addition to measurements of ankle and/or toe blood pressures. In a recently published document (5), it is recommended that toe blood pressure is measured instead of ankle blood pressure in all patients (not only patients with diabetes) in order to avoid falsely elevated ankle blood pressure. In addition, the need for investigations of local microcirculation and tissue viability is stressed, and for this purpose the authors suggest measurement of forefoot transcutaneous oxygen tension (tcPO2) (5). TcPO2 is a non-invasive method reflecting the local arterial blood flow and skin oxygenation (8). Correctly used tcPO2 has a good reproducibility (9) and is useful for predicting ulcer healing in diabetic patients (5, 10). Measurements of toe blood pressure and tcPO2 are not commonly used in clinical practice, and as a consequence treatment may be delayed. However, palpation of pedal pulses should not be forgotten, as it is a quick and simple method for evaluating arterial blood supply to a foot. If pulses are palpable the risk of ischemic foot problems is markedly reduced.
NON-HEALING DIABETIC FOOT ULCERS
Foot ulcers are severe complications in diabetic patients (11). Ten to 25% of the diabetic patients develop a chronic foot ulcer. Fifty to 60% of all non-traumatic amputations are performed in diabetic patients, and 75 to 85% of these amputations are caused by chronic foot ulcers.
The foot ulcers in diabetic patients can be divided into neuropathic, ischemic and neuroischemic ulcers of which the ischemic and neuroischemic together are the most common. In 90% of the patients undergoing amputation ischemia is the main cause (12). The healing of neuropathic foot ulcers is most often successful if local pressure to the area can be avoided. However, a real challenge is to improve healing of ischemic or neuroischemic foot ulcers due to the low distal perfusion pressure, which is superimposed to the already existing disturbances in microvascular function. Patients with ischemic or neuroischemic foot ulcers should be considered for vascular surgery or angioplasty in order to optimise the peripheral circulation and ulcer healing, and to avoid amputation. Due to the neuroischemic pattern of the disease, dysfunction in microcirculation and challenges in diagnostics, the threshold for revascularization should be lower in diabetic patients with foot ulcer.
As earlier mentioned palpation of pedal pulses and measurement of toe blood pressure are methods useful for confirmation of PAOD/CLI, while measurement of local tissue perfusion is necessary for estimating tissue viability and the prognosis of ulcer healing. In a prospective study investigating outcome of ulcer healing in 50 diabetic patients (10), tcPO2 levels > 40 mm Hg indicated improved ulcer healing or healing with intact skin. Poor healing and risk of amputation was seen when tcPO2 was < 10 mm Hg, while levels between 10–30 mm Hg indicated impaired ulcer healing. In contrast, toe blood pressure did not predict ulcer healing (10).
Unfortunately, all diabetic patients with neuroischemic foot ulcers are not available for vascular surgery or angioplasty. In these patients, treatment with low molecular weight heparin (dalteparin 5000 U daily) in addition to the traditional medication/treatment has shown significant positive effects on hemostasis, forefoot skin microcirculation and ulcer outcome (13, 14).
SKIN MICROVASCULAR DYSFUNCTION
Diabetes mellitus is associated with severe vascular complications involving both micro- and macrocirculation. Poor glycemic control is an important contributing factor for the development of the microangiopathy seen in skin, nerves, retina and kidney (DCCT 1993). Among the diabetic complications, the non-healing foot ulcer constitutes a real challenge for the medical profession due to the combination of several factors, i.e. neuropathy, macro- and microvascular dysfunction, and infection (5).
Skin microcirculation can be studied non-invasively by capillaroscopy and laser Doppler techniques. It is regulated by several mechanisms of which the sympathetic nerve system, endothelial- (nitric oxide, prostanoids, hyperpolarising factor) and non-endothelial dependent (smooth muscle cells, basement membrane, extracellular matrix) factors are most well known. Neurogenic factors play a prominent role in the regulation of skin microcirculation due to the presence of thermoregulating arteriovenous shunt vessels which are innervated by the sympathetic nerves. In the diabetic foot, the skin capillary blood circulation is reduced and disturbances in microvascular function have been shown early after debut of type 1 diabetes. It is more pronounced when other complications are present, and in patients with poor glycemic control (15, 16). The disturbances in foot skin capillary circulation were found despite a normal or even increased total blood supply to the region in diabetic patients without PAOD. Similar disturbances have been found in patients with type 2 diabetes and neuropathy (17). The reason for this markedly reduced capillary blood flow is primarily due to an arterio-venous shunting, bypassing the nutritional skin capillaries. This impaired capillary circulation is even more pronounced in diabetic patients with PAOD (18). The findings in the diabetic patients were compatible with a state of “chronic capillary ischemia”, despite that both patient groups were matched for age, gender and toe blood pressure (18). Our findings also indicated that the capillary circulation in diabetic patients with CLI is already very low during resting conditions, and cannot be improved in situations where an increased nutritional circulation is needed (Fig. 1), such as in the healing process of ischemic foot ulcers. These microvascular disturbances are of great importance for the reduced skin oxygenation, poor tissue survival and impaired foot ulcer healing.
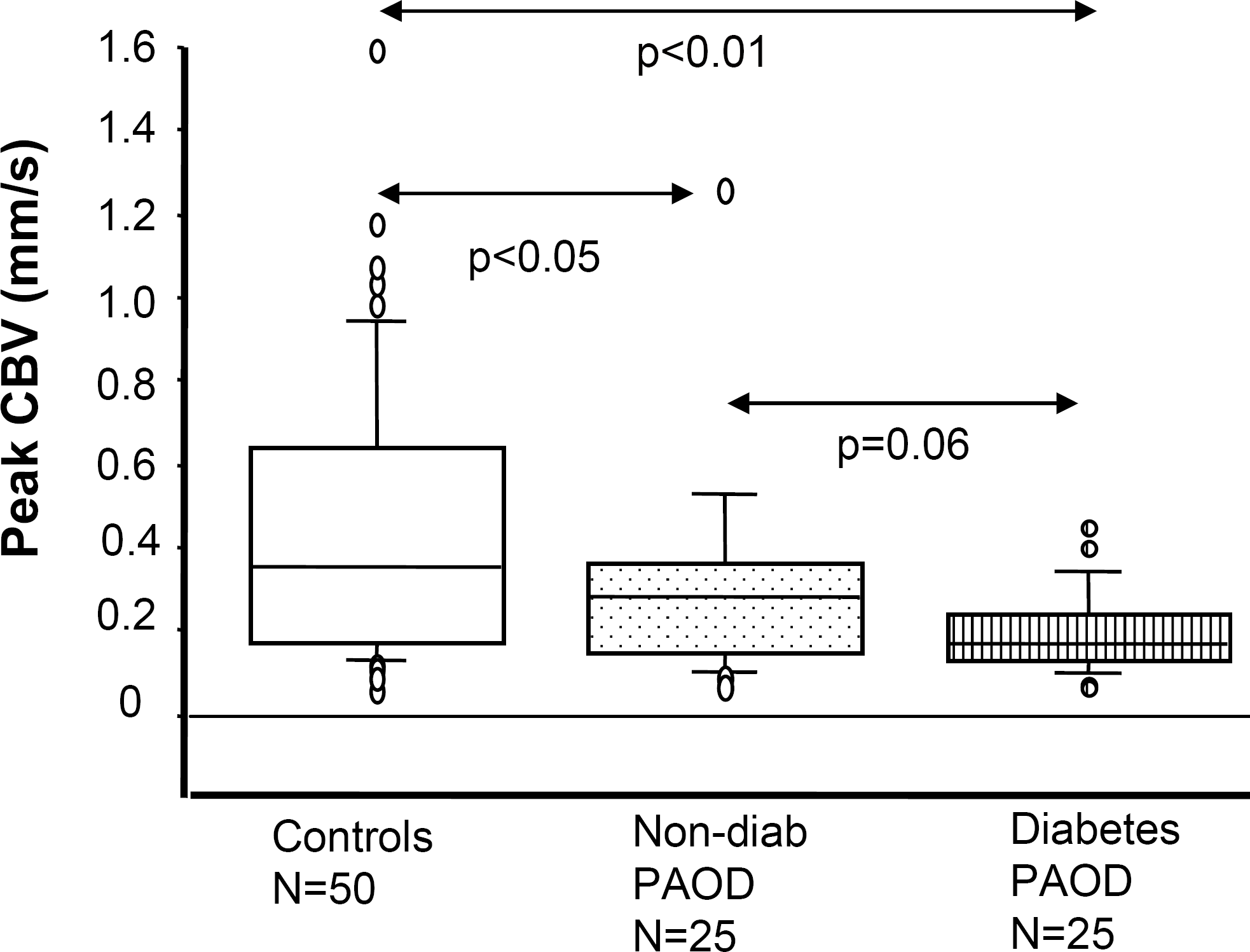
Peak capillary blood flow (CBV) during postocclusive reactive hyperemia showing severe capillary ischemia in patients with peripheral arterial occlusive disease (PAOD), most pronounced in the diabetic patients. The patients with PAOD are matched for age, gender and systolic toe blood pressure.
Although the pathophysiological mechanisms behind “the chronic capillary ischemia” in the diabetic foot are not fully known, some possible explanations are described in Fig. 2. Thermoregulating arteriovenous (AV) shunts are common in skin microcirculation and these channels are mainly innervated by the sympathetic nervous system. Autonomic neuropathy, which is common in patients with diabetes, causes sympathetic denervation and loss of vessel autoregulation. The denervated AV-shunts loose their normal contraction and stay open leading to blood bypassing the capillaries. This also transforms the arteriolar blood pressure into the subcapillary venules resulting in a decreased AV pressure difference leading to a reduced capillary circulation. Interestingly, and in line with our hypothesis, an increased capillary blood pressure has been measured by direct microcannulation of nailfold capillaries in diabetic patients (19). Thus, in diabetic patients, especially those with neuropathy, the total skin microcirculation is normal, or even overperfused, while the capillary circulation is markedly reduced. In line with the hypothesis of AV shunting as a mechanism behind chronic capillary ischemia, diabetic patients with peripheral neuropathy have warm and red feet indicating a good total skin blood flow, in spite of the fact that the skin capillary flow is markedly reduced in the same region.
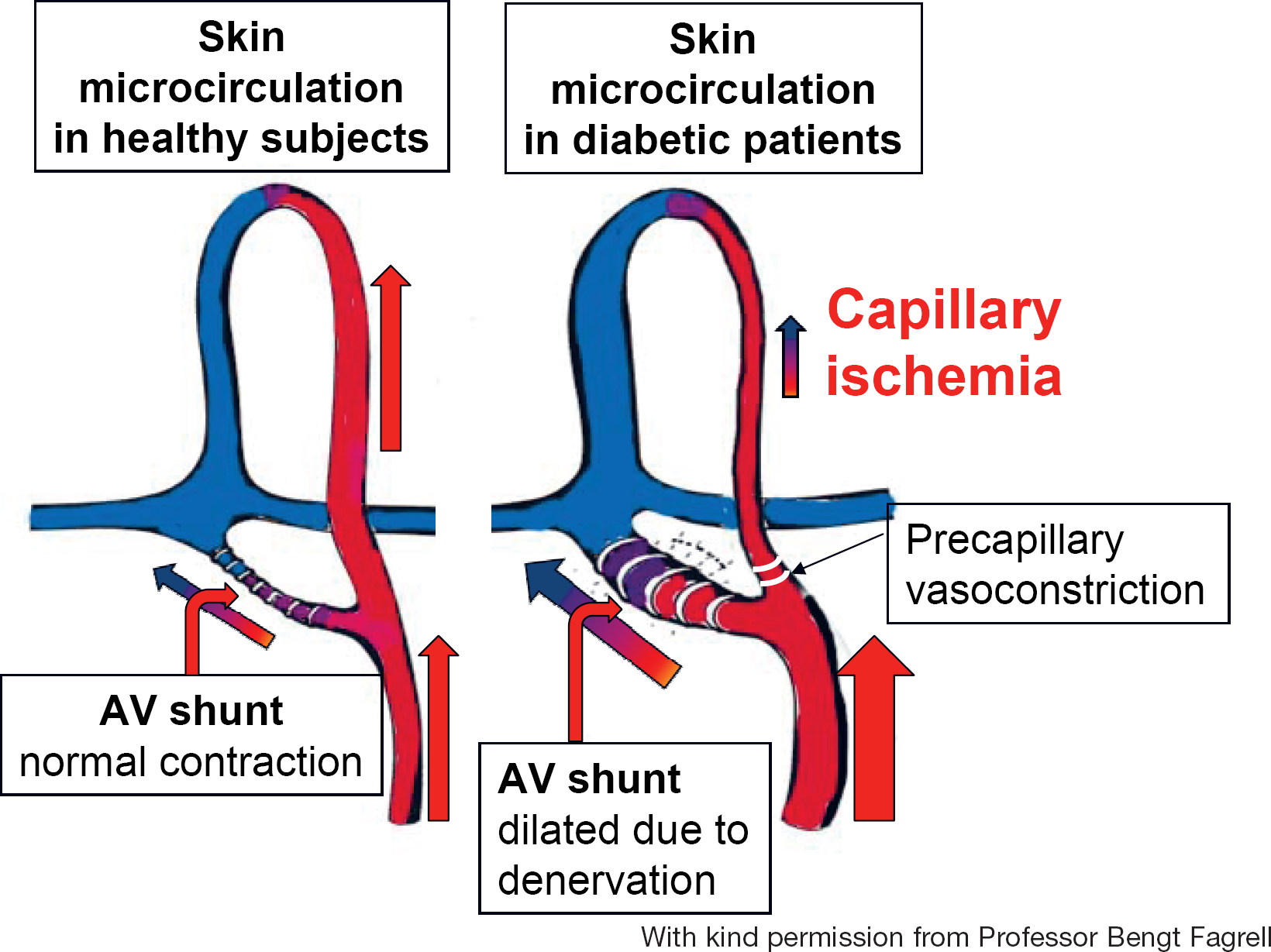
Possible explanations behind “chronic capillary ischemia” in the diabetic foot: 1) Thermo-regulating arteriovenous (AV) shunts are innervated by the sympathetic nerve system. In diabetes, autonomic neuropathy may lead to denervation of the AV shunts, which loose their normal contraction leading to blood passing through these shunts instead of the capillaries, 2) Endothelial dysfunction with a disturbed balance between endogenous vasodilators and vasoconstrictors leading to precapillary vasoconstriction, 3) Hemorheological alterations such as elevated levels of plasma fibrinogen.
Another contributing factor to the chronic capillary ischemia might be an altered balance between endogenous vasodilators (nitric oxide, hyperpolarising factor, prostanoids) and vasoconstrictors (angiotensin II, endothelin-1) at the precapillary level leading to reduced capillary circulation. Blocking the endothelin-A-receptors increases skin microcirculation and oxygenation in the diabetic foot with CLI (20). A third factor contributing to the maldistribution of blood between nutritive and deeper non-nutritive blood circulation, is altered hemorheology, such as elevated levels of plasma fibrinogen (15, 18). Fibrinogen is an acute phase reactant and an independent predictor of cardiovascular disease. Concentration of plasma fibrinogen is often increased in patients with diabetes and even more so in the presence of PAOD. Fibrinogen increases blood viscosity and elevated levels have been associated with impaired forefoot tcPO2 in diabetic patients with CLI (14).
CONCLUSIONS
The microvascular disturbances in the diabetic foot with ischemia, which are superimposed on the already existing structural diabetic microangiopathy, are compatible with a state of “chronic capillary ischemia” and an increased shunting of blood through arteriovenous channels. This maldistribution of blood within the skin microcirculation cannot be detected by measurement of peripheral blood pressure (systolic ankle blood pressure, systolic toe blood pressure). Consequently, the disturbances in peripheral tissue perfusion of the diabetic foot may be underestimated leading to delayed vascular surgery, angioplasty and/or medical treatment. Thus, measurements of peripheral blood pressure, e.g. toe blood pressure, should be combined with investigations of local tissue perfusion in order to get an adequate estimation of peripheral tissue perfusion in diabetic patients. For this purpose local skin microcirculation can be investigated by transcutaneous oxygen tension of the forefoot.
Footnotes
ACKNOWLEDGEMENT
I wish to thank Professor Bengt Fagrell for his kind support.
