Abstract
Aim:
This study aimed to translate the Living with Medicines Questionnaire, version 3 (LMQ-3) into Swedish for validation in an outpatient cohort regularly using medicines.
Methods:
Phase 1 translated the originator questionnaire into Swedish, and tested face and content validity through cognitive interviews. Phase 2 tested validation through a cross-sectional survey. Exploratory factor analysis (EFA) of the 41 items in the LMQ-3 was conducted.
Results:
Phase 1 involved 25 cognitive interviews. In phase 2, 779 respondents completed the questionnaire online. The total mean LMQ-3 score was 90.6 (95% confidence interval (CI) 89.0–92.2; range 43–176), and the mean visual analogue scale score was 3.1 (95% CI 2.9–3.3; range 0–10). The median age was 72 years (range 21–96) with 80.9% retired. The mean number of prescribed medicines was 8.5 (SD 3.8) per respondent. EFA analyses revealed most items loaded in their original domains, except three items: ‘My medicines allow me to live my life as I want to’ (question (Q)32), ‘I have to put a lot of planning and thought into taking medicines’ (Q37) and ‘I find getting prescriptions from the doctor difficult’ (Q1).
Conclusions:
Keywords
Introduction
Medicines have historically changed the face of public health. Although aimed at improving health and quality of life [1,2] medicines also bring multi-faceted burden [3] that increases with polypharmacy [4] and affects how patients use their medicines [5]. This burden and the cumulative workload of healthcare treatment can overwhelm patients in their everyday life [6,7]. However, treatment burden is not commonly considered in the often guideline-based prescribing process that primarily focuses on clinical outcomes [8].
Qualitative studies have investigated patients’ lived experience of medicines in their everyday life [9 –12], including ‘if and how’ patients find medicines burdensome. This research has shown that patients’ lived experiences affect their medicine-taking decisions [9,11,12]. Quantitative instruments exist that measure the individual patient experience of taking medicines specifically focused on medicine-related burden [13]. Such instruments can identify areas where a structured approach could address medicine-related issues and aim to improve both medicine prescribing and use through specifically tailored interventions. Robust qualitative investigation of patient experiences with medicines led Krska et al. to develop a patient-reported outcome measure (PROM) that measures medicine-related burden – the Living with Medicines Questionnaire (LMQ) [13,14].
The revised third version of the English LMQ [15] contains 41 statements about medicine use across eight domains. These domains cover the patients’ relationships/communication with healthcare professionals (five items), practical difficulties (seven items), daily interferences (six items), perceived effectiveness of medicines (six items), side-effects (four items), general concerns (seven items), cost-related burden (three items) and autonomy of medicines use (three items). This tool has been previously used in research, for example, in elderly populations of Bahrain, Kuwait and Ghana [16 –18] and in various populations with chronic conditions across the UK, Ethiopia, China, Malta and Qatar (HIV, diabetes mellitus, schizophrenia, or other co-morbidities) [19 –23], with translated versions used in those countries where English is not the most common language. However, to date, no tools are available in Swedish to quantify the patient’s perspective on medicine-related burden.
The aim of this study is to translate the Living with Medicines Questionnaire, version 3 (LMQ-3) into Swedish and to validate this questionnaire in an outpatient cohort regularly using medicines [15].
Methods
The original English LMQ-3 [15] contains 41 statements about medicine use across eight domains. These domains cover the patients’ relationships/communication with healthcare professionals (five items), practical difficulties (seven items), daily interferences (six items), perceived effectiveness of medicines (six items), side-effects (four items), general concerns (seven items), cost-related burden (three items) and autonomy of medicines use (three items).
Each statement is rated by the patient on a five-point Likert-type scale from 1=strongly agree to 5=strongly disagree. Negatively worded statements are appropriately reverse scored for analysis. Higher total item scores (range 41–205) indicate greater medicine-related burden equivalent to a worse experience of medicine use. Data from English populations suggest that patient scores are categorised as: minimal burden (41–87), moderate degree of burden (88–110) and high burden (⩾ 111) for those likely to benefit from an intervention. Overall medicine-use burden is assessed using a visual analogue scale (VAS) with the number 0 indicating ‘no burden at all’ and the number 10 indicating ‘extremely burdensome’.
Additionally, an open-ended question is included to capture any other patient’s concerns.
Last, the LMQ-3 collects patient characteristics and demographic variables such as gender, age, education level, employment status, number of medicines, medicine-use frequency, assistance in using medicines and payment for prescriptions.
Study design
This research occurred in two phases: phase 1 involved translating the English version of the LMQ-3 into Swedish and cognitive interviews (face and content validation). Two native Swedish-speaking researchers (SW, LH) who are fluent in English separately translated the original LMQ-3 into Swedish. A consensus version was reached together with a third native Swedish-speaking researcher (IE). The translated Swedish consensus version was then back-translated into English by a native English-speaking researcher (JMF) fluent in Swedish. The back-translated English version was then compared with the original version. Discrepancies were discussed with one of the English to Swedish translating team (SW) and a further native English-speaking researcher (NB). For discrepancies where a consensus could not be reached within this smaller group, the discrepancy was lifted to the larger research team for discussion and a final decision made. The final consensus version was tested through cognitive interviews with people who regularly took multiple prescribed medicines (polypharmacy). This group (N=25) included both native Swedish-speakers and permanent residents in Sweden who had Swedish as a second language. These respondents were recruited from local divisions of the National Organisation for Retired Persons (Pensionärernas riksorganisation) and the National Health & Lung Patient Association (Riksförbundet hjärtlung) as well as an informal network of people with Swedish as a second language.
Phase 2 was conducted as a cross-sectional survey distributed to various patient organisations electronically, via email, to minimise direct patient contact during the COVID-19 pandemic (July to September 2020).
Settings, participants and data collection
For phase 1, cognitive interviews were conducted face-to-face after receiving written informed consent. Participants received a paper copy of the Swedish LMQ-3 (LMQ-Swe) and were asked to read each statement aloud and verbally reason about their response, any questions or thoughts they may have regarding the question or words that are used. Interviews were recorded in case the researchers needed to listen to the interview to clarify or discuss any discrepancies with the research team.
For phase 2, a link to the online LMQ-Swe was emailed to patient representatives at the Swedish Kidney Patient Association (Njurförbundet), the Swedish Heart & Lung Patient Association (Riksförbundet hjärtlung) and the National Organisation for Retired Persons. Two of the organisations distributed the link to all their members via email. One organisation opted to forward the link to members of two regional districts only in order to limit research fatigue as, nationally, their members were participating in several research surveys during the ongoing COVID-19 pandemic.
Sample size calculation and inclusion criteria
Previous validation studies of the LMQ-3 instrument in other populations involved a sample size of at least 500 completed questionnaires to enable psychometric testing/factor analysis (at least 5–10 participants per item). Swedish population studies typically have a response rate from 44% to 60% [24,25]. Hence, assuming a 50% response rate, the invitation email including the link to the online questionnaire was sent to at least 1000 persons. Respondents were aged ⩾ 18 years, Swedish-speakers and had experience of taking prescribed medicines on a regular basis for at least six months.
Data entry and analysis
The LMQ-Swe (phase 2) was hosted on Swedish University Computer Network (SUNET) Survey&Report in Artologik®, which is a secure digital survey platform used by Swedish universities (SUNET Survey) [26]. The anonymous survey was open from July to September 2020, during which consenting patients entered their responses directly online.
Quantitative data were analysed using SPSS® version 27. Descriptive statistics included percentages (%), means (standard deviation (SD)) and medians (range). Exploratory factor analysis (EFA) of the 41 items in the LMQ-Swe was conducted to verify whether the Swedish translation fitted the eight-factor structure hypothesised in the English LMQ-3, as a form of construct validation. For the EFA, principal axis factoring was used with promax rotation based on the inter-related domains underpinning the originator questionnaire [15]; the number of factors to extract was set to eight as originally hypothesised, and standard criteria for item screening were used (poor loadings < 0.32 and cross-loadings on two or more factors > 0.4). Cronbach’s alpha was assessed for all eight subscales/domains to evaluate internal consistency of the Swedish translation.
Known-groups validity
Known-groups validity was assessed in the overall sample using the demographic data collected through the LMQ-3. Medicine-related burden was hypothesised to correlate with a higher number of medicines, frequency of administration, and the need for help when using medicines [15]. Relationships between characteristics and medicines burden were explored using independent t-tests or analysis of variance in responses with two or more groups. Statistical significance was set at p-values less than 0.05.
Ethics
All participants received written study information and provided their signed informed consent. The study was approved by the Regional Ethical Review Board in Gothenburg (DNo. 063-17) and the Swedish Ethical Review Authority (DNo. 2019-04134, DNo. 2020-02763) and conforms to the principles outlined in the Declaration of Helsinki.
Results
Participants
During phase 1, 13 female and 12 male respondents completed 25 cognitive interviews over a six-month period (November 2019 to May 2020), mostly in-person. Because of the COVID-19 pandemic, the final five participants were interviewed over the telephone with the questionnaire posted prior to interviews. Recruitment ceased after the 25th interview as no new interpretation issues emerged from completing the questionnaire, thus reaching data saturation. Of the participants, 24% (n=6) had Swedish as a second language. Problems regarding comprehension were minor. Some respondents commented that the statements regarding side-effects were repetitive, which was similar to the original English validation study. Several respondents commented how relevant the questionnaire was to their life and how they wished to see it used in further research.
During phase 2, a total of 1373 accessed the online questionnaire, and the number of completed responses was 779, yielding a completion rate of 57% (779/1373). It was not possible to calculate the actual number of recipients of the survey owing to varying memberships of the patient organisations involved and lack of this information.
Medicine-related burden
The total mean LMQ-Swe score was 90.6 (95% confidence interval (CI) 89.0–92.2; range 4–176) and the mean VAS score was 3.1 (95% CI 2.9–3.3; range 0–10). From the total LMQ-Swe scores, overall burden was categorised into three categories as follows: 49.6% (n=386) reported minimal burden, 30.9% (n=241) moderate burden and 19.5% (n=152) high burden.
Respondent characteristics and questionnaire responses
Table I describes population characteristics. The median age was 72 years (range 21–96) with a mean of 69.7 (SD 10.4). A large proportion of respondents were retired (80.9%). The mean number of prescribed medicines was high at 8.5 (SD 3.8) per respondent (polypharmacy). Most respondents did not report needing help in taking their medicines (92.9%). Four hundred and eight (52.4%) respondents belonged to the National Organisation for Retired persons, 326 (41.8%) to the Swedish Kidney Patient Association and 45 (5.8%) to the Swedish Heart & Lung Patient Association.
Characteristics of the respondents in phase 2.
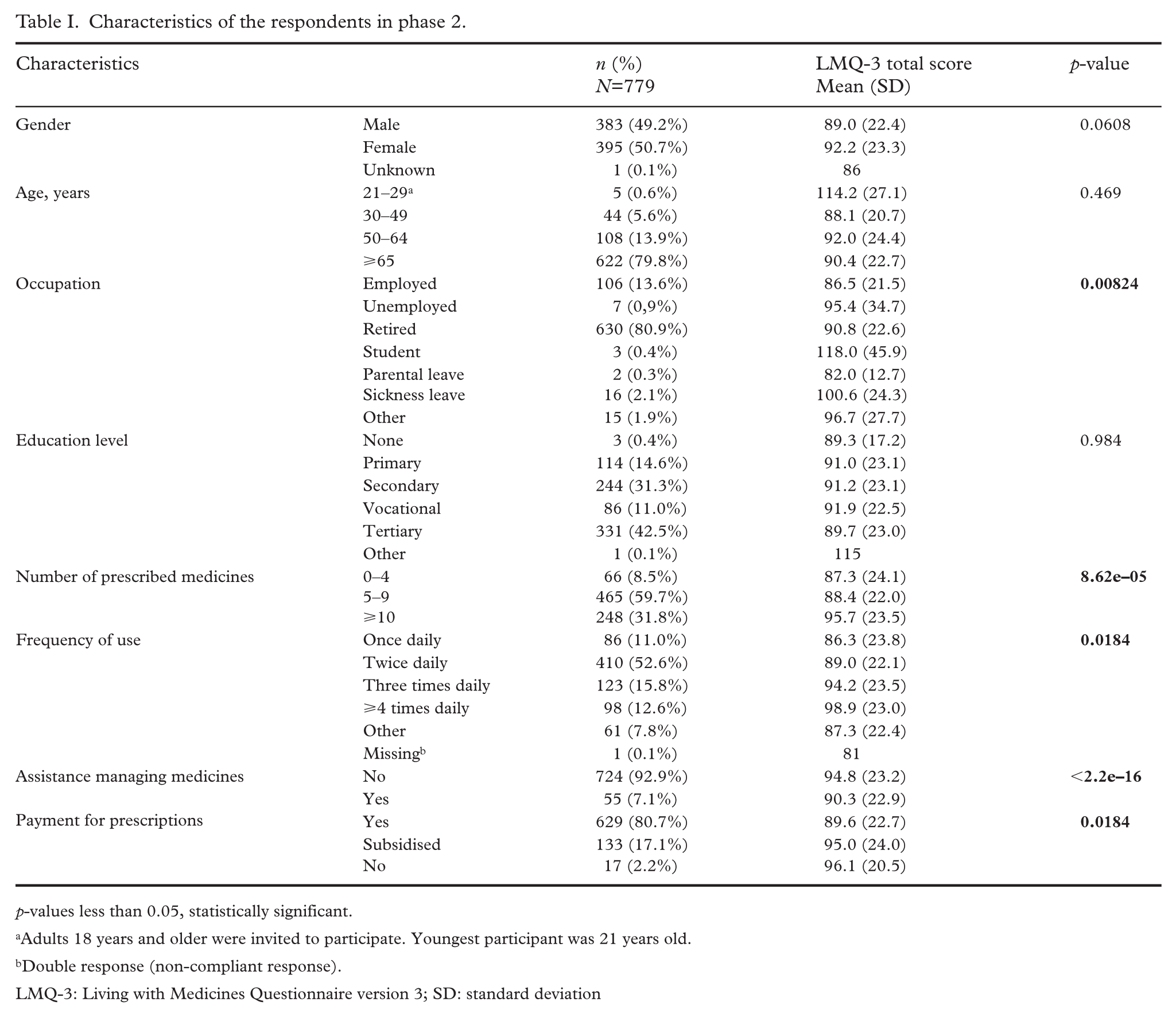
p-values less than 0.05, statistically significant.
Adults 18 years and older were invited to participate. Youngest participant was 21 years old.
Double response (non-compliant response).
LMQ-3: Living with Medicines Questionnaire version 3; SD: standard deviation
Values and differences in mean LMQ-Swe scores per characteristic are described in Table I. There were statistically significant differences in mean LMQ-Swe total scores dependent on occupation: higher burden scores were shown in respondents who were full-time students, followed by respondents on sickness benefits or when unemployed. Other factors that showed statistically significant differences in medicine burden scores were: the number of medicines prescribed, frequency of medicine use, managing medicines independently versus needing support, and prescription payment. Gender, age and education level did not impact on overall burden scores.
More than eight medicines were prescribed to 40.0% of respondents, of whom 41.7% reported a minimal burden, 36.2% a moderate burden and 22.0% a high burden.
The responses for each of the 41 statements are shown in Figure 1. The 10 questions with the highest mean burden scores pertained to the domains ‘General concerns about medicines’ (question (Q)8, Q9, Q12, Q18), ‘Lack of autonomy/control of medicine use’ (Q11, Q13, Q26), ‘Side effects’ (Q30, Q38) and ‘Interferences with day-to-day life’ (Q23) (Supplemental material Table 2 online). The 10 questions with the lowest mean LMQ-Swe scores pertained to the domains ‘Practical difficulties’ (Q4, Q28, Q29), ‘Lack of effectiveness’ (Q15, Q39, Q40), ‘Patient–doctor relationships and communication about medicines’ (Q1, Q7), ‘Cost-related burden’ (Q17) and ‘Interferences with day-to-day life’ (Q27).
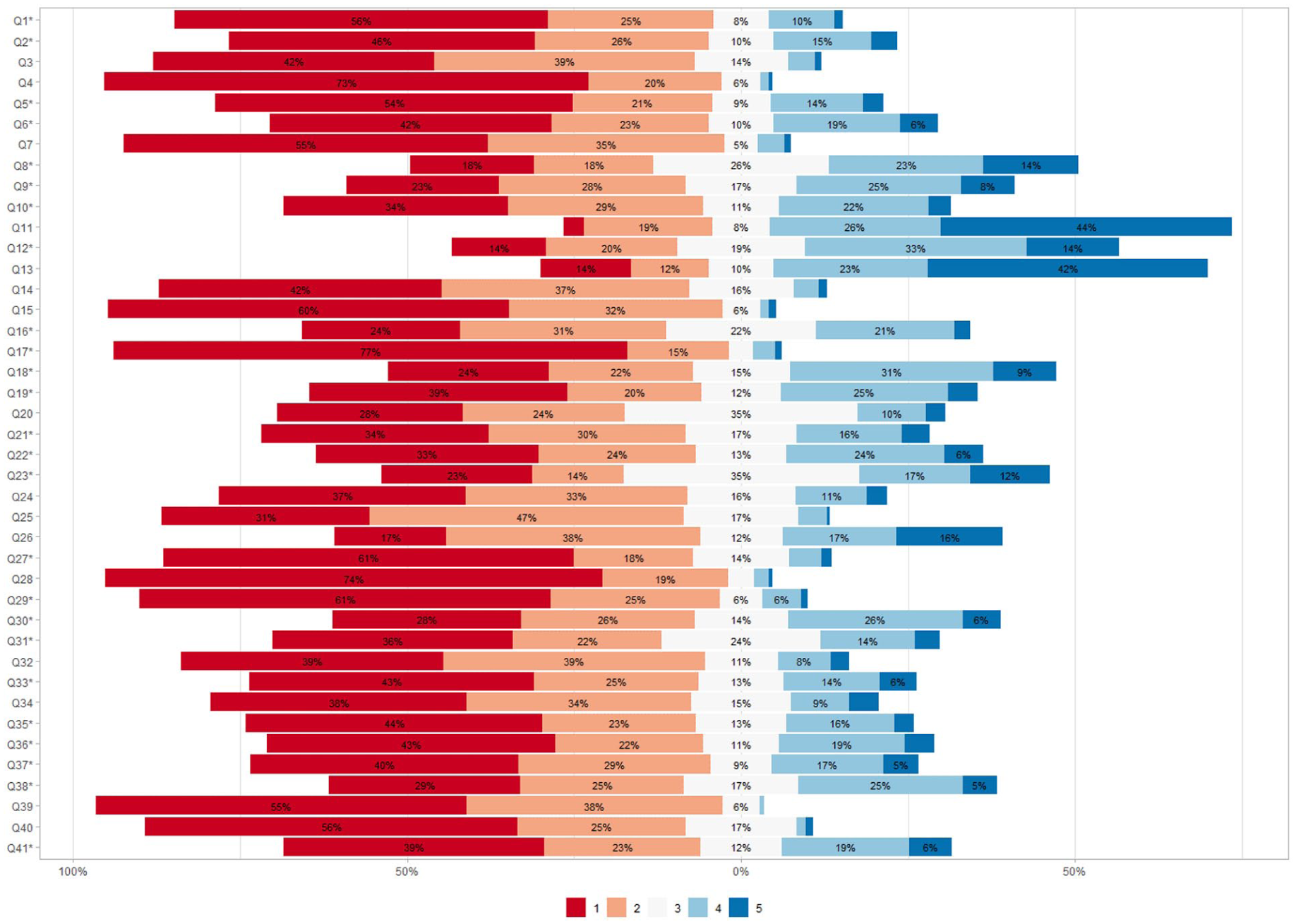
Responses to each of the 41 statements in the Living with Medicines Questionnaire version 3.
Construct validity of the Swedish translation of the LMQ-3
Data were suitable for EFA as confirmed by the results of Kaiser–Meyer–Olkin Measure of Sampling Adequacy (KMO = 0.932) and Bartlett’s test of sphericity (approximate Chi-square = 13,531.545, df = 820, p < 0.001). Based on Kaiser’s criterion (eigen values over 1), the Swedish translation (LMQ-Swe) generated eight eigenvalues >1 (range 1.061 for the eighth factor to 11.309 for the first factor), which together explained 57.2% of the total variance. Internal consistency of the eight subscales/domains of the translation ranged from 0.594 to 0.878 for the ‘autonomy’ and ‘side-effects’ domains respectively. EFA analyses of the Swedish translation revealed that the vast majority of items loaded in their original domains proposed in the English version, excepting three items: ‘I find getting prescriptions from the doctor difficult’ (Q1), ‘My medicines allow me to live my life as I want to’ (Q32) and ‘I have to put a lot of planning and thought into taking medicines’ (Q37). These statements were originally assigned to ‘practical difficulties’ ‘effectiveness’ and ‘practical difficulties’ respectively, but now loaded under related domains: ‘patient–doctor relationships and communication about medicines’, ‘interferences with day-to-day life’ and ‘interferences with day-to-day life’ respectively (Supplemental Table 3).
Two items loaded significantly in two domains: ‘I have to put a lot of planning and thought into taking medicines’ (Q37) and ‘The side effects I get from my medicines interfere with my day-to-day life (e.g. work, housework, sleep)’ (Q22) (Supplemental Table 3). Analyses of intercorrelations among the eight domains showed that the highest correlation (r = 0.618) was between ‘effectiveness’ and ‘patient–doctor relationships and communication about medicines’. The lowest correlation (–0.013) was between ‘autonomy’ and ‘interference’ domains, suggesting that lower perceived autonomy to vary medicine use regimes was associated with higher interference to an individual’s everyday life.
Discussion
This study successfully translated the LMQ-3 into Swedish (LMQ-Swe) and cross-culturally validated it in an outpatient cohort regularly using medicines.
The results revealed adequate construct validity of the translation, with minor differences from the originator instrument. The three statements that loaded in different domains theoretically make sense and differ possibly because of issues around translation. The meanings of statements in the instrument were overall well-understood by the Swedish patients, suggesting that the tool can serve as a useful patient-reported measure of medicine-related burden in this population. Only minor differences were identified through analyses, hence showing no rationale for further assessment through confirmatory factor analysis. Future work could involve testing of second-order factors of both instruments to assess model stability. Additionally, future test–retest reliability might help assess stability of scores over time and usefulness of the translation in longitudinal studies. Internal consistency was adequate for all subscales/domains, with the ‘autonomy’ (α=0.594) displaying the lowest Cronbach’s alpha, similar to the originator instrument (α=0.692) [15].
The loading of the statement regarding ‘getting prescriptions from the doctor’ (Q1) in a differing domain (from ‘practical difficulties’ to ‘patient–doctor relationships/communication about medicines’) might reflect the current state of frustration with waiting times in primary healthcare in Sweden, where many patients are experiencing longer waiting times than previously [9,27]. Similarly, longer waiting times have led to repeat prescriptions often being re-issued without direct patient–doctor communication, meaning that a patient’s opportunity to ask questions about their medication is forgone [(9] and may also influence the loading of the ‘communication around medicines’ domain. Similarly, both ‘I have to put a lot of planning and thought into taking medicines’ (Q37) and ‘The side effects I get from my medicines interfere with my day-to-day life’ (Q22) statements loaded in two domains in the LMQ-Swe, contrasting with loading in a single domain in the originator questionnaire, which might also be indicative of frustration with accessing primary healthcare doctors. However, we cannot exclude that the difference in loading might also reflect a difference in prescribing culture that is beyond the instrument’s scope as a PROM that measures the patient’s perspective. The statement ‘The side-effects I get from my medicines interfere with my day-to-day life’ (Q22) loaded under the ‘side-effects’ domain, as in the originator instrument. In the Swedish version the word ‘negatively’ was included in the statement, which might have led the focus more to side-effects.
Notwithstanding these specific differences, the LMQ-Swe questionnaire is, overall, structurally comparable to the originator questionnaire, meaning that the translation is scientifically sound and valid.
Strengths and limitations
The originator LMQ instrument was developed from the patient perspective and the following modified versions (2 and 3) derived and tested by long-term users of medicines [15]. The LMQ-Swe was also tested by long-term users of multiple medicines, although mostly retired older persons. Whilst this patient group is not representative of the entire general population, it is particularly of interest owing to the significance in our growing aging population. During the cognitive testing phase of the LMQ-Swe, several patients stated how much they liked the instrument and wanted to see it used in practice owing to its relevance to issues important from their perspective, suggesting cultural acceptability. Favourable questionnaire completion rates generated a larger sample size, which enabled adequate psychometric testing and relevant analyses. We attempted to assess test–retest reliability following the main study; however, only one of three patient organisations was willing to redistribute the survey link, impacting response rates, and it was challenging to match the test–retest scores across participants. No specific cultural modifications to the instrument were deemed necessary for the Swedish context. In 2018 the out-of-pocket payments represented 16.7% of the UK total expenditure on health, and 13% in Sweden in 2020 [28]. Although the systems for patient out-of-pocket prescription payments differ between the UK and Sweden, both countries have capped out-of-pocket prescription charges, meaning that most prescription medicines do not cost full market price. However, in contrast to the UK, all patients in Sweden, regardless of income amount or source, pay up to the maximum capped threshold over a 12-month period.
Conclusion
In conclusion, the Swedish version of the LMQ-3 (LMQ-Swe) is now available following successful translation and adequate psychometric testing. This instrument provides a person-centred measure of medicine-related burden as experienced by individuals who regularly take multiple prescribed medicines.
Practice implications
Medicines burden is correlated with problematic medication taking. The LMQ-Swe provides a person-centred research outcome measure of medicines burden which could facilitate discussions around problematic medication taking, particularly in patients with polypharmacy, and be used to demonstrate improvements following intervention [29] and hence contribute to better public health.
Supplemental Material
sj-docx-1-sjp-10.1177_14034948261437594 – Supplemental material for Translating the Living with Medicines Questionnaire from English to Swedish: a validation study
Supplemental material, sj-docx-2-sjp-10.1177_14034948261437594 for Translating the Living with Medicines Questionnaire from English to Swedish: a validation study by Amélie Cransac, Barbra Katusiime, Hanna Gyllensten, Lilas Ali, Nicky Britten, Inger Ekman, Andreas Fors, Lina Hellström, Janet Krska, Karl Swedberg, Sara Wallström and Joanne M. Fuller in Scandinavian Journal of Public Health
Supplemental Material
sj-docx-2-sjp-10.1177_14034948261437594 – Supplemental material for Translating the Living with Medicines Questionnaire from English to Swedish: a validation study
Supplemental material, sj-docx-2-sjp-10.1177_14034948261437594 for Translating the Living with Medicines Questionnaire from English to Swedish: a validation study by Amélie Cransac, Barbra Katusiime, Hanna Gyllensten, Lilas Ali, Nicky Britten, Inger Ekman, Andreas Fors, Lina Hellström, Janet Krska, Karl Swedberg, Sara Wallström and Joanne M. Fuller in Scandinavian Journal of Public Health
Footnotes
Acknowledgements
The authors gratefully acknowledge the individuals that participated in the cognitive interviews, the organisations that distributed the digital questionnaire to their members and the individuals who completed the questionnaire online. None of this research would be possible without their invaluable contribution.
Author contributions
All authors were responsible for the study’s conception and design. SW, LH, IE, JMF and NB were involved in the translation process. JMF, LA and SW conducted the cognitive interviews. JMF constructed the online questionnaire. AC conducted descriptive statistical analyses with the support of HG. BK performed the psychometric analyses (EFA), supported interpretation of results and reviewed drafts. JMF and AC drafted the article. All other authors critically revised the manuscript for intellectual content.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Swedish Heart-Lung Foundation (DNr. 20180183, Principal Investigator (PI) Inger Ekman (IE), https://www.hjart-lungfonden.se/om-oss/in-english/; The Swedish Research Council (DNr. 2017-01230), PI IE, https://www.vr.se/english.html; The Centre for Person-Centred Care at the University of Gothenburg (GPCC), Sweden, in separate grants PI IE, PI NB, PI JMF, ![]() ; and The Hjalmar Svensson Foundation, Sweden (HJSV2020090), Main-applicant JMF. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
; and The Hjalmar Svensson Foundation, Sweden (HJSV2020090), Main-applicant JMF. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
