Abstract
Background
FA-2-b-β, an extract derived from traditional Chinese medicine (TCM), has been suggested as a potential neuroprotective agent.
Objective
This study aimed to elucidate its role in modulating HMGB1-mediated inflammation and pyroptosis in Alzheimer's disease (AD), with a particular emphasis on the interaction between FA-2-b-β and HMGB1.
Methods
AD cell and animal models were used to examine the effect of FA-2-b-β on HMGB1/NF-κB/NLRP3 signaling pathway. Protein expression levels were detected by western blotting, and enzyme-linked immunosorbent assay (ELISA), respectively. Immunofluorescence staining was performed to determine the cellular localization of key proteins. The role of HMGB1 in amyloid-β (Aβ)-induced neuroinflammation and pyroptosis was examined through siRNA-mediated HMGB1 knockdown. Behavioral tests were conducted in AD animal models to evaluate cognitive improvements following FA-2-b-β treatment.
Results
In cellular models, FA-2-b-β significantly suppressed Aβ-induced overexpression HMGB1 and inhibited the activation of NF-κB, which consequently led to a reduction in the formation of the NLRP3 inflammasome. This suppression resulted in decreased of activation caspase-1 and lower levels of IL-1β and IL-18, thereby alleviating pyroptosis and neuroinflammation. The knockdown of HMGB1 further corroborated its role in mediating Aβ-induced inflammatory responses. In AD animal models, treatment with FA-2-b-β attenuated neuroinflammation, preserved neuronal integrity, and enhanced cognitive function.
Conclusions
FA-2-b-β exhibits a capacity to modulate the HMGB1/NF-κB/NLRP3 signaling pathway, thereby mitigating neuroinflammation and pyroptosis, highlighting its potential as a therapeutic intervention for AD.
Introduction
Alzheimer's disease (AD), a progressive neurodegenerative disorder affecting the central nervous system, is the leading cuase of dementia worldwide.1,2 With its prevalence rising annually, AD has emerged as a significant global health challenge. Despite extensive research, the pathogenesis of AD remains incompletely understood, and effective therapeutic strategies are still lacking.3,4
Pathologically, AD is characterized by three cardinal features: the extracellular aggregation of amyloid-β (Aβ) peptides, the intracellular accumulation of hyperphosphorylated tau proteins, and the progressive deterioration of synaptic connections accompanied by neuronal loss. 5 Although these pathological processes have traditionally dominated AD research paradigms, emerging evidence increasingly highlights the role of neuroinflammation as a crucial driver of disease progression. The deposition of Aβ peptides triggers the activation of microglia and astrocytes, initiating a cascade of inflammatory responses that exacerbate neurodegenerative processes and accelerate cognitive impairment.6,7
Among the various inflammatory mechanisms implicated in AD, pyroptosis—a highly inflammatory form of programmed cell death—has attracted increasing scholarly attention. 8 Unlike apoptosis, pyroptosis is initiated by the activation of inflammasomes, particularly the NLRP3 inflammasome, leading to the release of pro-inflammatory cytokines such as interleukin-1β (IL-1β) and IL-18. The pathological features of Aβ and tau proteins can trigger pyroptosis, thereby exacerbating neuroinflammation and accelerating neuronal degeneration.9,10 Notably, the inhibition of caspase-1-mediated pyroptosis has shown neuroprotective effects in experimental models, indicating that targeting this pathway may represent a promising therapeutic strategy for AD.
Agaricus blazei Murill (AbM), a medicinal mushroom belonging to the Basidiomycetes family, has garnered interest for its potent anti-inflammatory, antioxidant, and immunomodulatory properties.11,12 Research has demonstrated that AbM confers protection against intestinal ischemia-reperfusion injury by modulating gut microbiota and suppressing excessive inflammation. 13 Its bioactive constituents, particularly polysaccharides and β-glucans, have exhibited anti-tumor, antimicrobial, and immunoregulatory effects.14,15 Among these components, FA-2-b-β has been identified as a key anti-inflammatory compound, capable of regulating natural killer cells and macrophages. 16 Notably, while FA-2-b-β’s anti-inflammatory properties are well-documented in peripheral systems, its potential neuroprotective roles in AD, particularly in the context of pyroptosis (a pro-inflammatory programmed cell death pathway), remain underexplored.
Damage-associated molecular patterns (DAMPs), with high mobility group box 1 (HMGB1) serving as a prominent example, as pivotal modulators of neuroinflammation in AD. HMGB1 is released by cells under stress or damage cells and activates immune responses through receptor interactions, thereby exacerbating neuroinflammation and accelerating disease progression.17,18 Targeting HMGB1 and its downstream signaling pathways has demonstrated efficacy in reduceing Aβ-induced neuroinflammation, decreasing disease burden, and enhancing cognitive function in experimental models. 19 Nontheless, the regulatory role of Aβ in pyroptosis via HMGB1 and the contribution of this mechanism contributes to AD pathogenesis remains unclear.
In this study, we investigate the role of the HMGB1/NF-κB/NLRP3 signaling pathway in Aβ-induced pyroptosis and neuroinflammation associated with AD. By employing HMC3 microglial cells and an in vivo AD mouse model, we evaluate the potential of FA-2-b-β, derived from AbM, to inhibit Aβ-induced pyroptosis and mitigate neuroinflammation by targeting the HMGB1 signaling axis. Through this investigation, we propose a novel mechanism for understanding the inflammatory processes contributing to AD pathogenesis. We explore the efficacy of FA-2-b-β for neuroinflammation-related neurodegeneration.
Methods
Drug preparation
The water-soluble active compound FA-2-b-β was isolated and purified at the Key Laboratory of Natural Drugs, Lanzhou Institute of Chemical Physics, Chinese Academy of Sciences. From an initial 500 g of water-soluble polysaccharide, a total of 4.86 g of the FA-2-b-β acid RNA protein complex was successfully obtained through separation and purification processes. The compound was subsequently dissolved in double-distilled water, sterilized, and stored at 4°C.
Cell treatment
HMC3 (human microglial cell line) cell was routinely cultured in Dulbecco's Modified Eagle Medium (DMEM) supplemented with 10% heat-inactivated fetal bovine serum (FBS), 100 U/mL penicillin, and 100 μg/mL streptomycin. Cells were seeded at a density of 5 × 104 cells/cm2 in 6-well plates and maintained at 37°C in a humidified incubator with 5% CO2. Upon reaching 70-80% confluence, cells underwent subjected to experimental treatments following a 12-hour serum starvation period to synchronize the cell cycle phase.
Cell transfection
Small interfering RNA (siRNA) sequences targeting human HMGB1 (siHMGB1-1, siHMGB1-2, and siHMGB1-3) were designed by Shanghai GenePharma Co., Ltd. HMC3 cells were seeded in six-well plates devoid of antibiotics and incubated for 12 hours. Upon reaching 50-60% confluency, transfection of HMC3 cells was performed using Lipofectamine® 2000 (Invitrogen, Thermo Fisher Scientific, Inc.) with either 20 nM negative control (si-NC) or 20 nM siHMGB1. The transfection process was maintain lasted for 6 hours at 37°C, after which the cells were incubated for an additional 48 hours prior to further experimentation. Transfection efficiency was confirmed by quantitative real time (qRT-PCR) and western blot. To induce microglial activation, Aβ42 was applied to HMC3 cells for 24 hours. Subsequently, the cells were categorized into five groups: Control, Aβ42, Aβ42 + si-NC, Aβ42 + si-HMGB1, and FA-2-b-β+Aβ42.
Cell viability assay (CCK-8)
HMC3 cells were seeded in 96-well plates at a density of 1 × 104 cells/well and allowed to adhere overnight. After a subsequent 12-hour serum starvation, cells were treated with either FA-2-b-β or siHMGB1 for a duration of 24 hours. After treatment, 10 μL of CCK-8 reagent was added to each well, and the plates were incubated at 37°C for 2 hours in the darkness. Absorbance was recorded at 450 nm, with a reference wavelength of 630 nm, utilizing a microplate reader (TECAN, Switzerland).
Transmission electron microscopy (TEM)
The ultrastructure of pyroptotic cells was investigated using TEM. Cells were fixed with glutaraldehyde, subjected to dehydration, and subsequently embedded. Ultrathin sections were mounted onto copper grids, stained with uranyl acetate, and analyzed via TEM.
Animal model and treatment
The study utilized thirty APP/PS1 transgenic male mice, aged 22 weeks. Stereotactic brain injections of the drug were performed on days 12, 14, and 16 following the establishment of the model. Prior to anesthesia with 3% sodium pentobarbital, mice underwent a fasting period of four hours. Lenti siHMGB1 lentiviral particles (5 × 10^7 transducing units per mouse) were injected into the hippocampus at a rate of 0.25 μl/min using a Hamilton microsyringe, with the needle remaining in situ for three minutes post-injection. The experimental groups comprised Control, AD, Lenti Ctrl + AD, FA-2-b-β+AD, and Lenti siHMGB1 + AD. All animal experiments were conducted in strict accordance with the ARRIVE guidelines and adhered to the following regulations: (i) the U.K. Animals (Scientific Procedures) Act 1986 and its associated regulatory guidelines; (ii) EU Directive 2010/63/EU on the protection of animals used for scientific purposes; and (iii) the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals.
Behavioral analysis
The Morris water maze test was employed to evaluate cognitive function in AD mice. This assessment comprised the hidden platform trial, spatial exploration phase, and visible platform trial, evaluated spatial memory and anxiety levels. 20
Hematoxylin and eosin (H&E) staining
Brain tissues were collected and fixed in 4% paraformaldehyde (PFA) for 24 hours, followed by dehydration through a graded ethanol series and clearance with xylene. Subsequently, the tissues were embedded in paraffin, sectioned coronally at a thickness of 5 μm using a microtome, and mounted on slides. Following deparaffinization and rehydration, the sections were stained with hematoxylin for 5 minutes and then with eosin. The stained sections underwent dehydrated, xylene clearance, and were mounted with neutral balsam. Pathological changes were observed under a light microscope.
Enzyme-linked immunosorbent assay (ELISA)
The concentrations of TNF-α, IL-1β, IL-18, IL-6, iNOS, and caspase-1 in cell supernatants and mouse brain tissues were quantified using ELISA in accordance with the manufacturer’s instructions. Absorbance was recorded at 450 nm using a microplate reader.
Immunohistochemical staining
Brain sections were deparaffinized with xylene, rehydrated through a series of graded alcohols, and subsequently incubated with 3% H2O2 at 37°C for 10 minutes. Following antigen retrieval in a 0.01 M citrate buffer, the sections were blocked with sheep serum for one hour. This was followed by an overnight incubation with primary antibodies overnight at 4°C, and a subsequently incubation with secondary antibodies at 37°C for 30 minutes. DAB/H2O2 staining was performed, and the sections were counterstained, dehydrated, and mounted for microscopic analysis.
Immunofluorescence
Immunofluorescence was used to visualize the localization of Aβ42 and HMGB1. Cells were fixed with 4% paraformaldehyde for 15 minutes. After washing with PBS, the sections were blocked with 5% BSA for one hour. Primary antibodies (mouse anti-Aβ, rabbit anti-HMGB1) were applied overnight at 4°C. Fluorescence-labeled secondary antibodies were subsequently added, followed by PBS washes. The slides were sealed and examined under a fluorescent microscope.
Western blot analysis
Total protein was extracted from brain tissues or cells using RIPA lysis buffer and quantified by the BCA assay. Samples (20 μg) were denatured at 100°C, resolved by SDS-PAGE, and transferred onto nitrocellulose membranes (Millipore, USA). The membranes were blocked for one hour before incubation with primary antibodies overnight at 4°C. Afterward, HRP-conjugated secondary antibodies were applied for one hour at room temperature, and protein bands were visualized using an enhanced chemiluminescence (ECL) substrate kit (Beyotime, Shanghai, China) according to the manufacturer’s protocol. The chemiluminescent signals were captured using a gel imaging system (Bio-Rad, USA), and the gray value of each protein band was quantified using ImageJ software (Version 1.53, NIH, USA). β-actin (1:5000, 20536-1-AP, Proteintech, Wuhan, China) was used as the internal reference to normalize the relative expression levels of target proteins. The primary antibodies were as follows, HMGB1 (1:5000, 10829-1-AP), NLRP3 (1:2000, 30109-1-AP), phosphorylated NF-κB (p-NF-κB, 1:2000, 82335-1-RR), NF-κB (1:5000, 80979-1-RR), MyD88 (1:1000, AF5195, Affinity, Jiangsu, China), phosphorylated IκBα (p-IκBα, 1:1000, 82349-1-RR), IκBα(1:5000, 10268-1-AP), TLR4 (1:1000, 19811-1-AP), caspase-1 (1:1000, AF5418), and ASC (1:1000,DF6304).
Statistical analysis
Data are presented as the mean ± standard deviation (SD). Statistical analyses were conducted using one-way analysis of variance (ANOVA) with Tukey's post-hoc test for multiple comparisons, implemented in SPSS version 23.0 software. A p-value of less than 0.05 was considered statistically significant.
Results
HMGB1 gene silencing efficiency
To evaluate the efficiency of HMGB1 knockdown, a stable cell line was established using siHMGB1-1, siHMGB1-2, and siHMGB1-3. Gene silencing was confirmed by qRT-PCR and Western blot analyses, as shown in Figure 1A and 1B. Among the three siRNAs, siHMGB1-1 demonstrated the highest silencing efficiency and was thus selected for subsequent experiments (p < 0.05).
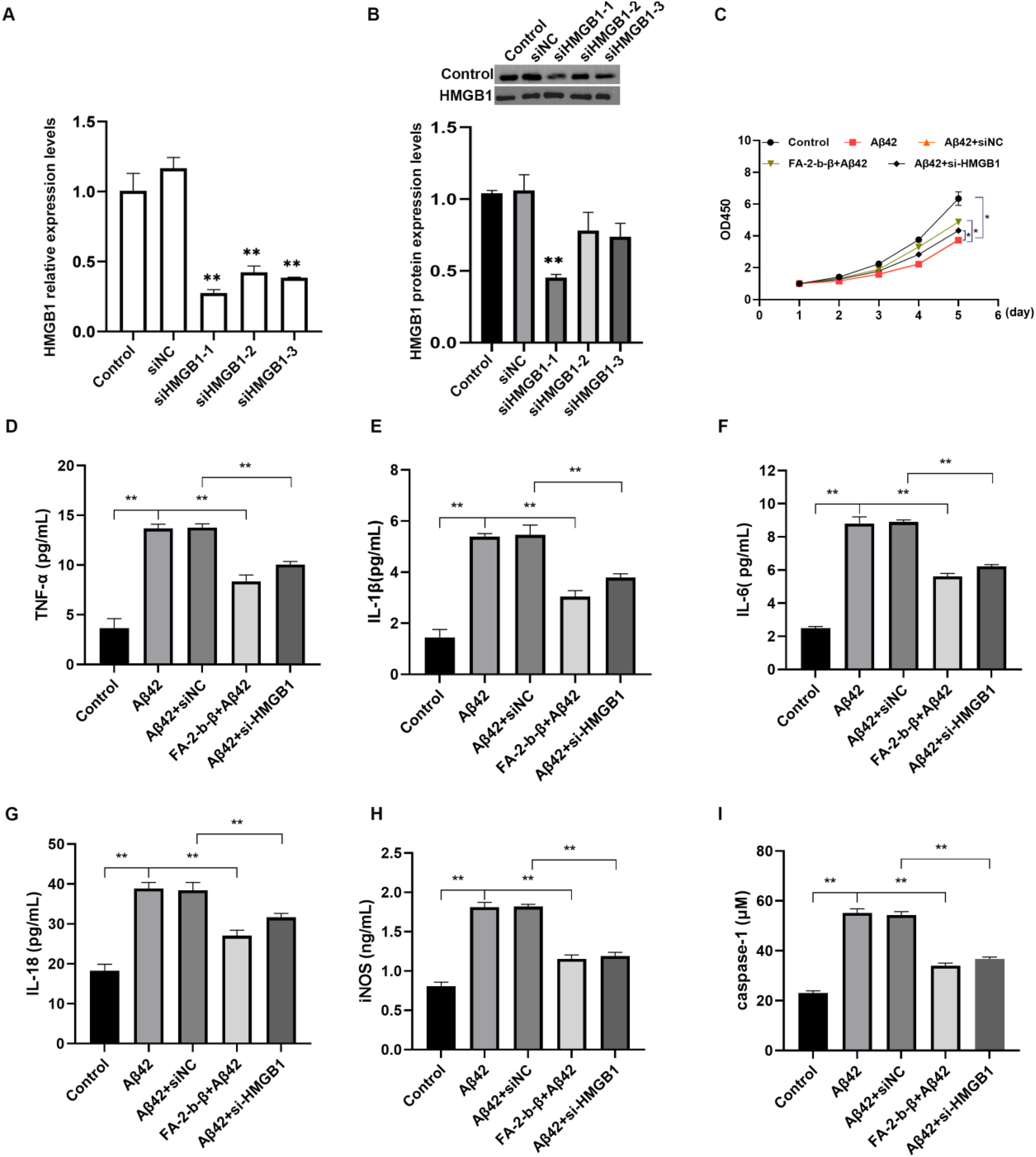
Effects of FA-2-b-β and HMGB1 on cellular viability and levels of TNF-α, IL-1β, IL-18, IL-6, and iNOS in HMC3 cells. The efficiency of siHMGB1 transfection was confirmed by qRT-PCR (A) and western blot assay (B). (C) CCK-8 was used to detect the OD values of cartilage cells in each group at 1, 2, 3, 4, and 5 days; The effect of FA-2-b-β and HMGB1 on the levels of TNF-α (D), IL-1β (E), IL-18 (F), IL-6 (G), iNOS (H), and caspase-1 (I). (*p < 0.05; **p < 0.01). CCK-8: cell counting kit-8; OD: optical density.
FA-2-b-β enhances HMC3 cell viability
To activate microglia, Aβ42 was diminstered to HMC3 cells, and cell viability was assessed using the CCK-8 assay (Figure 1C). Exposured to Aβ42 significantly decreased cell viability in both the Aβ42 and Aβ42 + siNC groups compared to the Control group (p < 0.05). Conversely, treatment with FA-2-b-β significantly enhanced cell viability in the FA-2-b-β + Aβ42 group relative to the Aβ42 group (p < 0.05). Furthermore, si-HMGB1 transfection reduced cell viability compared to the Aβ42 + siNC group (p < 0.05).
FA-2-b-β attenuates inflammatory markers in HMC3 cells
Compared to the Control group, the levels of TNF-α, IL-1β, IL-18, IL-6, and iNOS were significantly elevated in both the Aβ42 group and the Aβ42 + siNC group (Figure 1D-H, p < 0.05). Treatment with FA-2-b-β significantly decreased these inflammatory markers relative to the Aβ42 group, while si-HMGB1 further reduced their levels compared to the Aβ42 + siNC group (p < 0.05).
FA-2-b-β inhibits pyroptosis in HMC3 cells
Pyroptosis, an inflammatory form of programmed cell death reliant on caspase-1 was assessed using ELISA analyses. Results indicated that caspase-1 levels were significantly elevated in both the Aβ42 and Aβ42 + siNC groups compared to the Control group (Figure 1I, p < 0.05). Notably, FA-2-b-β treatment markedly inhibited caspase-1 activation, indicating its efficacy in suppressing Aβ42-induced pyroptosis. Interestingly, si-HMGB1 exhibited a comparable protective effect.
FA-2-b-β reduces HMGB1 and Aβ levels in HMC3 cells
Co-staining analysis revealed a significant increase in HMGB1 and Aβ double-positive cells in the Aβ42 and Aβ42 + siNC groups (Figure 2A). However, treatment with either FA-2-b-β or si-HMGB1 substantially reduced the co-localization of HMGB1 and Aβ.
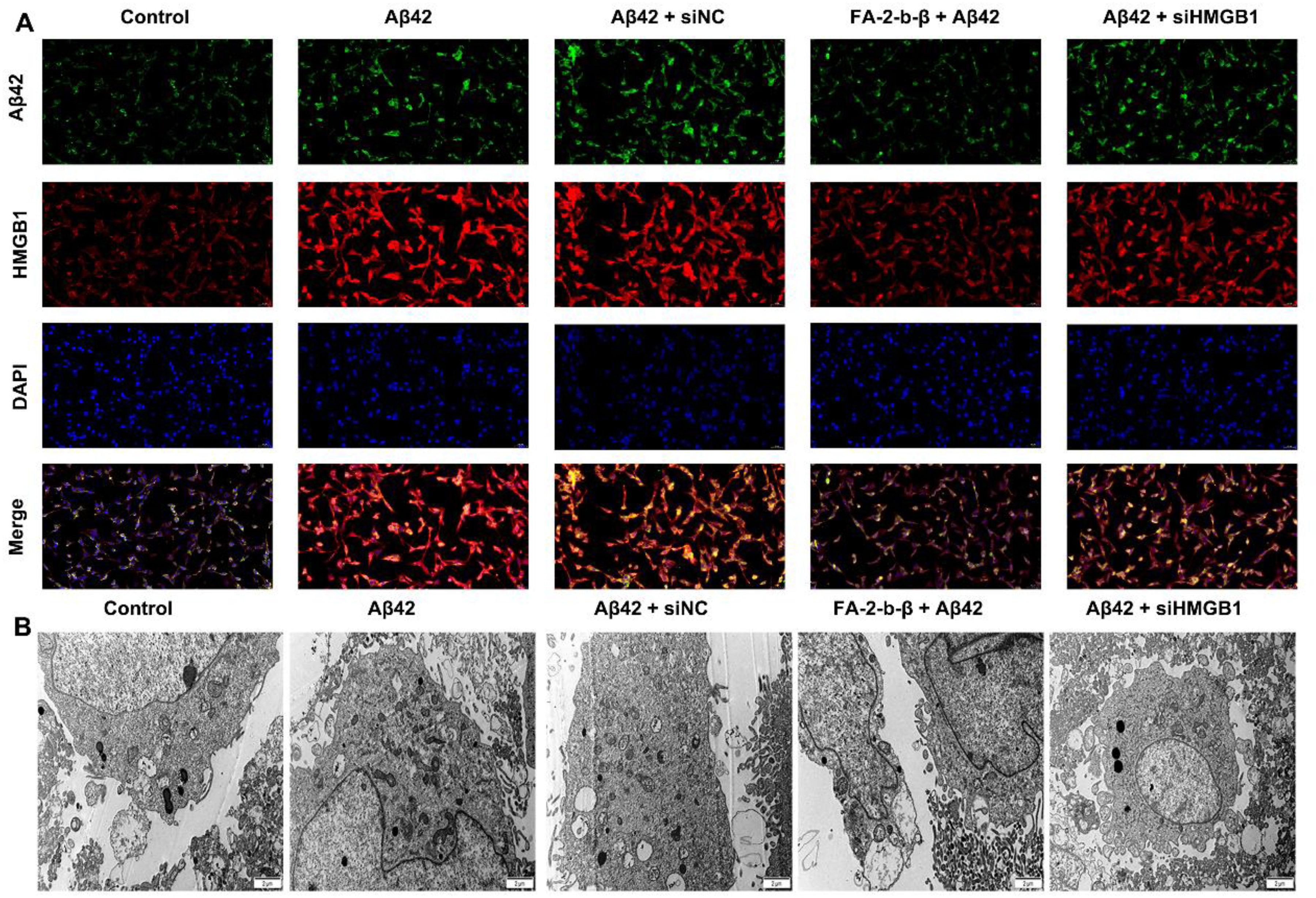
FA-2-b-β adiministration influenced the expression of HMGB1 and improved the cellular ultrastructure in HMC3 cells. (A) Co-staining of Aβ42 and HMGB1 in HMC3 cells (scar bar = 50 μm). (B) Transmission electron microscopy was used to observe the changes in cellular ultrastructure of HMC3 cells.
FA-2-b-β reduces pyroptosis and modulates HMGB1/NF-κB/NLRP3 signaling in HMC3 cells
TEM results identified pyroptotic features, such as organelle swelling and pyroptosome formation in Aβ42 and Aβ42 + siNC groups, which were alleviated by FA-2-b-β and si-HMGB1 treatments, indicating their role in reducing pyroptosis (Figure 2B). Western blot analysis revealed increased expression of HMGB1, p-NF-κB-65, NF-κB-65, NLRP3, ASC, and caspase-1 in the Aβ42 and Aβ42 + siNC groups compared to the Control (Figure 3A-G, p < 0.05). FA-2-b-β or si-HMGB1 treatments significantly decreased these protein levels, suggesting inhibition of the HMGB1/NF-κB/NLRP3 pathway.
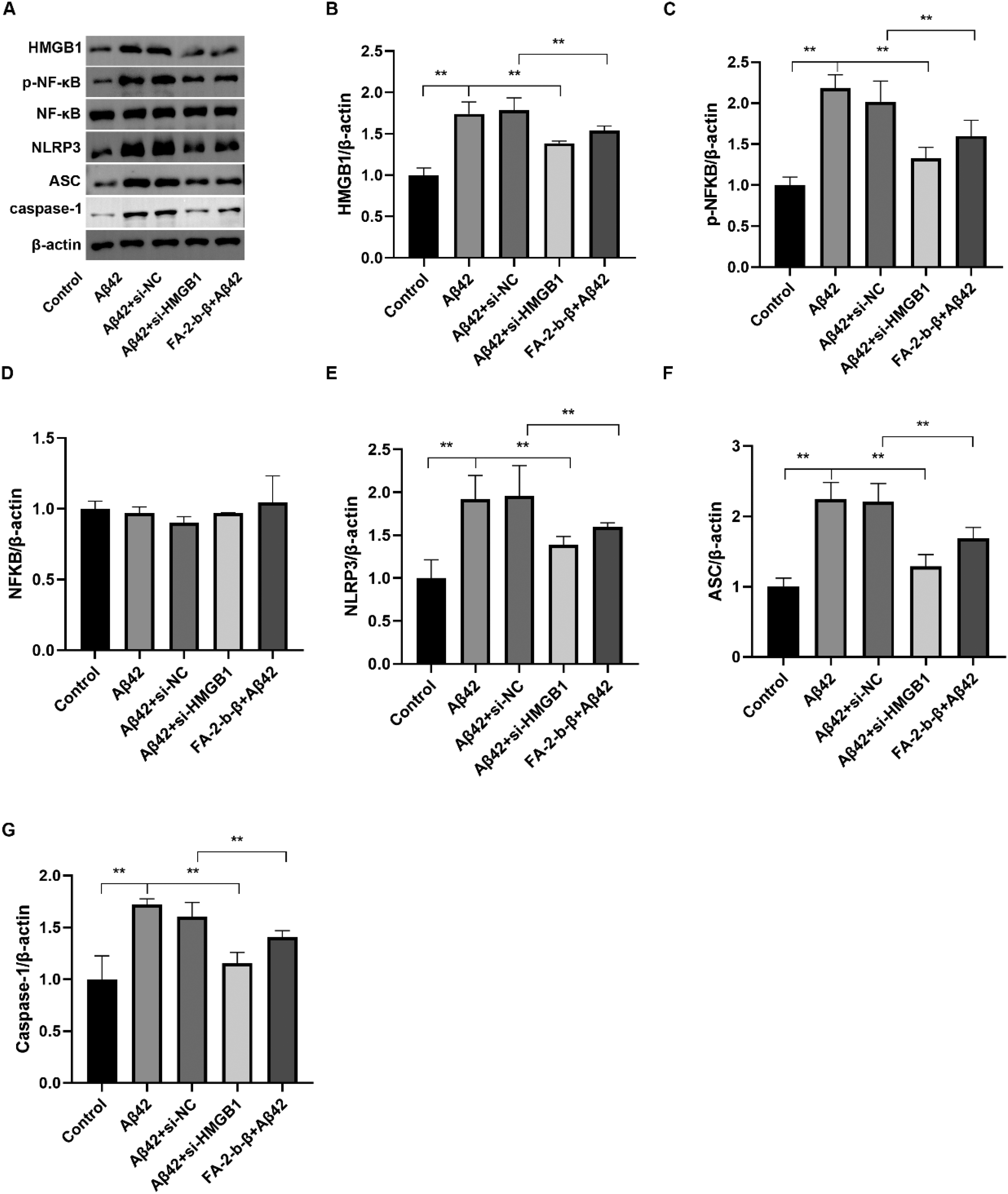
The effects of FA-2-b-β and HMGB1 on HMGB1/NF-κB/NLRP3 pathway-related protein levels. (A) Changes in the protein levels of HMGB1/NF-κB/NLRP3 pathway-related proteins. The protein expression levels of HMGB1 (B), p-NF-κB-65 (C), NF-κB-65 (D), NLRP3 (E), ASC (F), and caspase-1 (G) were assessed. **p < 0.01.
FA-2-b-β enhances spatial learning and memory in AD mice
To assess the neuroprotective effects of FA-2-b-β, APP/PS1 transgenic male mice were utilized. The Morris water maze indicated significant impairments in swimming distance, platform crossings (NCP), time spent in the platform quadrant (STP), and total distance in the AD and Lenti-control + AD groups compared to the Control group (Figure 4, p < 0.05). Treatments with FA-2-b-β and HMGB1 knockdown significantly enhanced cognitive performance, indicating potential neuroprotective effects.
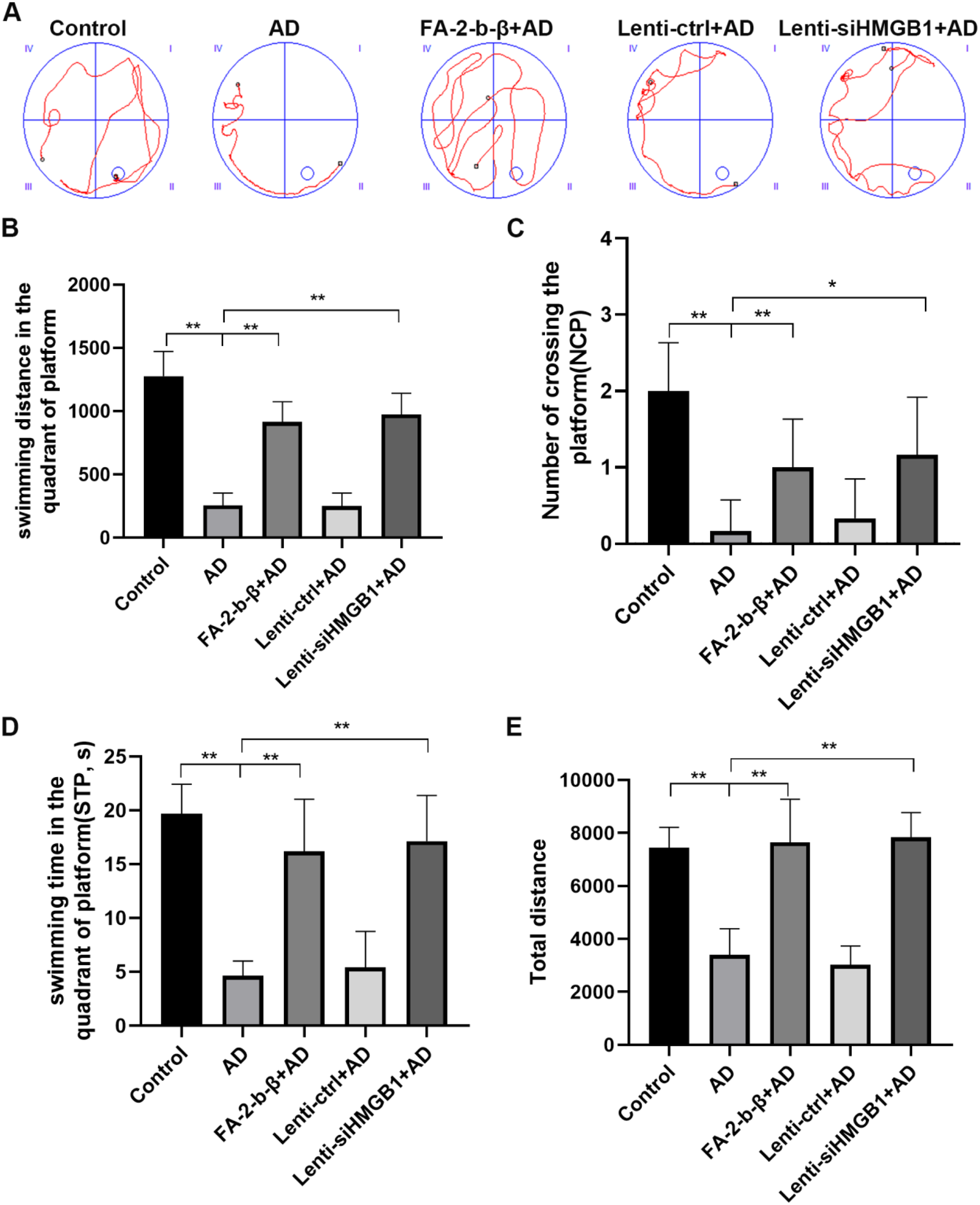
Effects of FA-2-b-β and HMGB1 knockdown on spatial learning and memory in rats. (A) Trajectory of open field test; (B) The swimming distance in the platform quadrant; (C) The number of platform crossings (NCP); (D) The swimming time in the platform quadrant (STP, s); (E) The total distance. *p < 0.05; **p < 0.01.
FA-2-b-β alleviates hippocampal pathology in AD mice
Histological analysis of the hippocampal CA1 region demonstrated structural disruption, neuronal loss, nuclear pyknosis, and increased glial infiltration in AD mice (Figure 5A). Treatments with FA-2-b-β and HMGB1 knockdown ameliorated these pathological changes, preserving neuronal morphology and reducing glial activation.
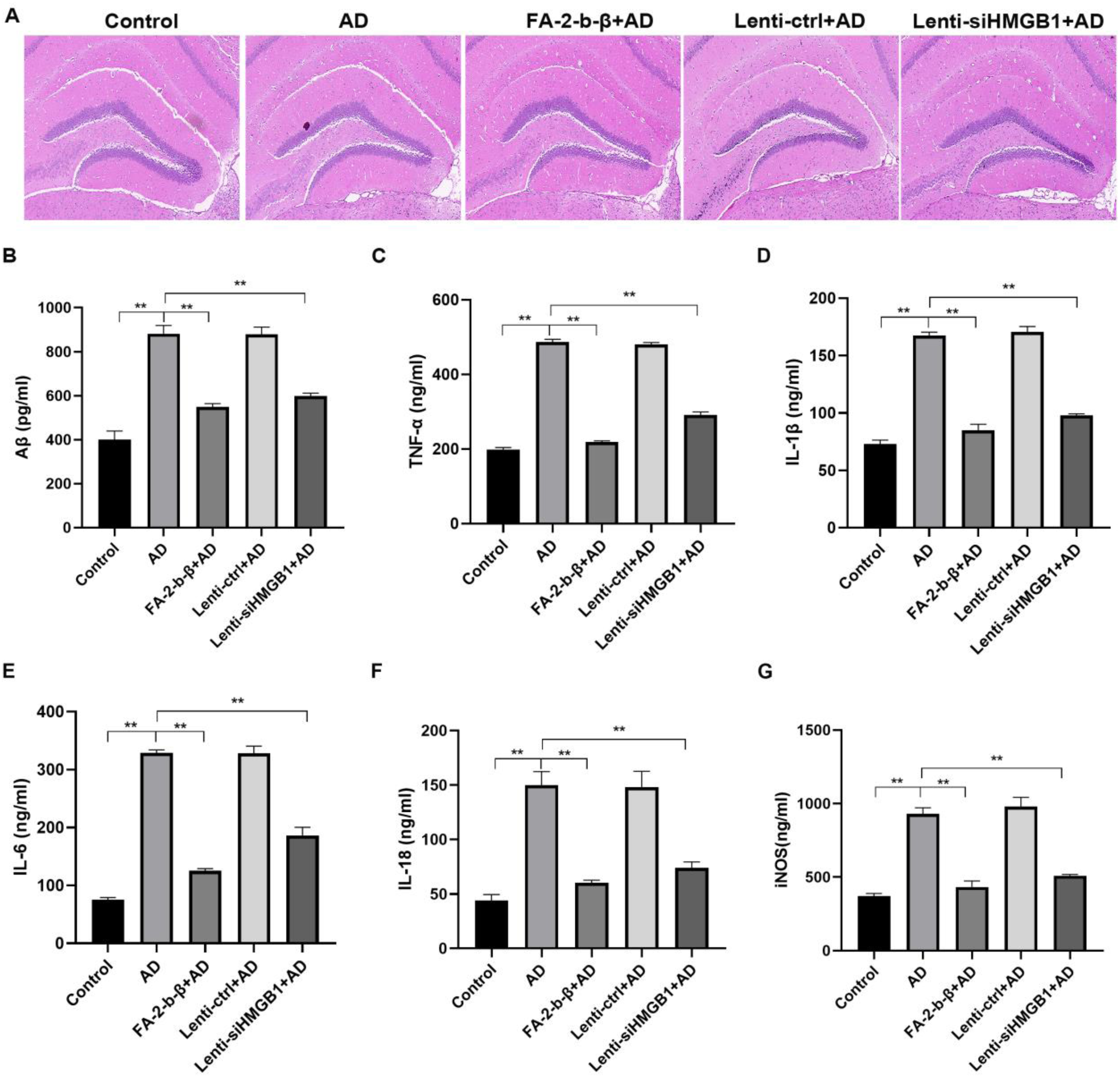
Effects of FA-2-b-β and HMGB1 on histopathological evaluation and changes in the inflammatory factors in the serum. (A) Histopathological evaluation was detected by HE staining (scar bar = 20 μm). The effect of FA-2-b-β and HMGB1 on the levels of Aβ42 (B), TNF-α (C), IL-1β (D), IL-6 (E), IL-18 (F), and iNOS (G). **p < 0.01.
FA-2-b-β reduces inflammation and microglial activation in AD mice
In the AD and Lenti-control + AD groups, the levels of Aβ42, TNF-α, IL-1β, IL-18, IL-6, and iNOS were significantly elevated (Figure 5B–5G, p < 0.05). Treatments with FA-2-b-β and HMGB1 knockdown substantially reduced these inflammatory markers, underscoring their anti-inflammatory properties.
FA-2-b-β attenuates Aβ42, NLRP3, and caspase-1 expression in the hippocampus of AD mice
Co-staining analysis demonstrated a significantly increase in HMGB1 and the microglial marker Iba1 double-positive cells in the AD group compared to the Control group (Figure 6A). Treatments with FA-2-b-β and si-HMGB1 significantly reduced the co-localization of HMGB1 and Iba1, indicating microglial deactivation. Immunohistochemical staining revealed elevated levels of Aβ42, NLRP3, and caspase-1 expression in the AD and Lenti-Ctrl + AD groups (Figure 6B). The adiminstration of FA-2-b-β and HMGB1 knockdown significantly decreased the expression of these proteins, suggesting a protective effect against neuroinflammation and pyroptosis.
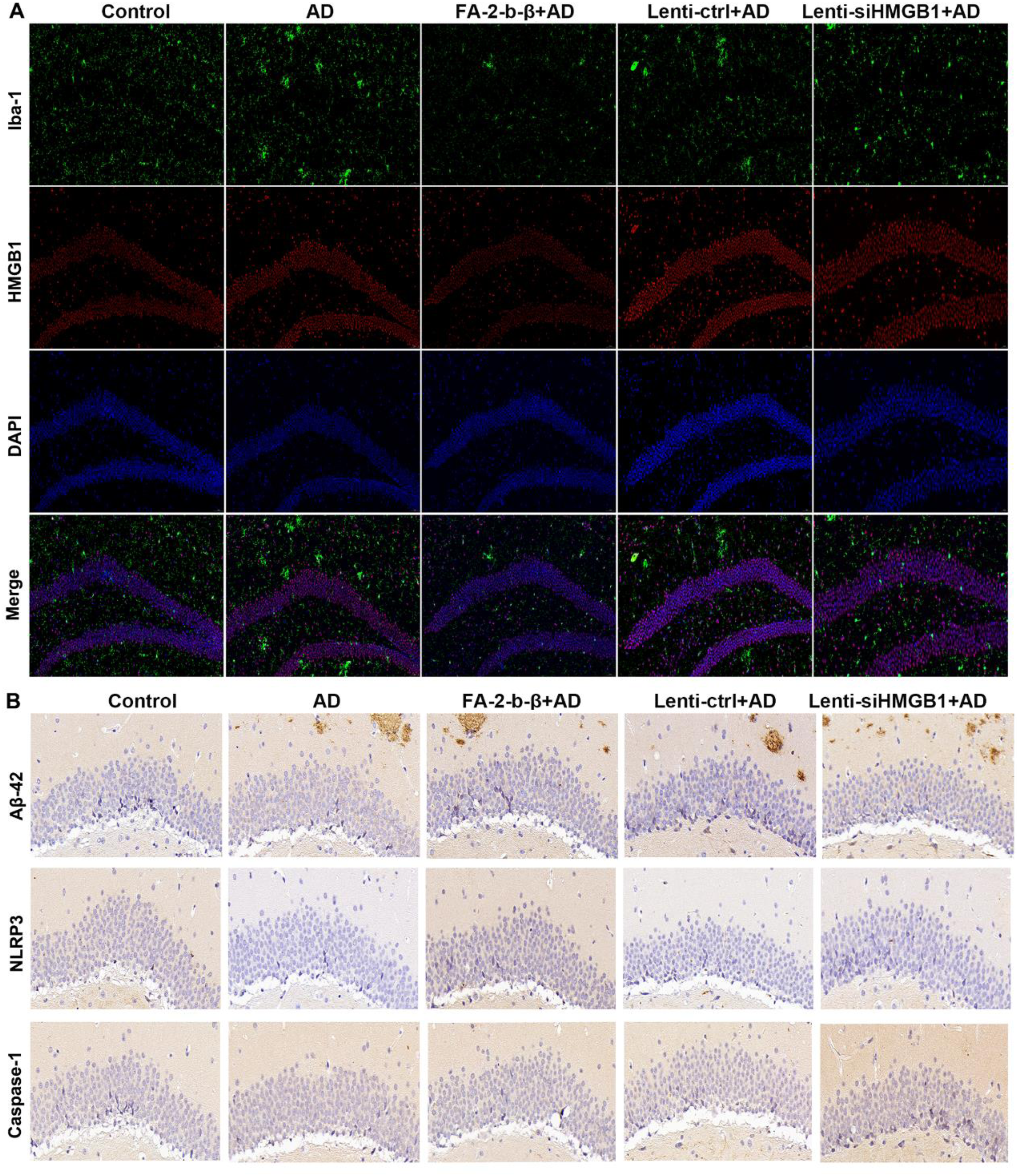
Effects of FA-2-b-β and HMGB1 on Iba1 + HMGB1, Aβ42, NLRP3 and caspase-1 expression. (A) Co-staining of Iba1and HMGB1 in AD mice (scar bar = 20 μm). (B) Localization of Aβ42, NLRP3, and caspase-1 expression by immunohistochemistry (scar bar = 10 μm).
FA-2-b-β inhibits HMGB1/NF-κB/NLRP3 signaling pathway in AD mice
Western blot analysis indicated increased expression of HMGB1, NLRP3, p-NF-κB, NF-κB, MyD88, p-IκBα, IκBα, TLR4, caspase-1, and ASC in AD and Lenti-control + AD groups compared to the Control (Figure 7A-K, p < 0.05). FA-2-b-β and HMGB1 knockdown treatments significantly downregulated these proteins, which indicates that both agents can inhibit the HMGB1/NF-κB/NLRP3 pathway.
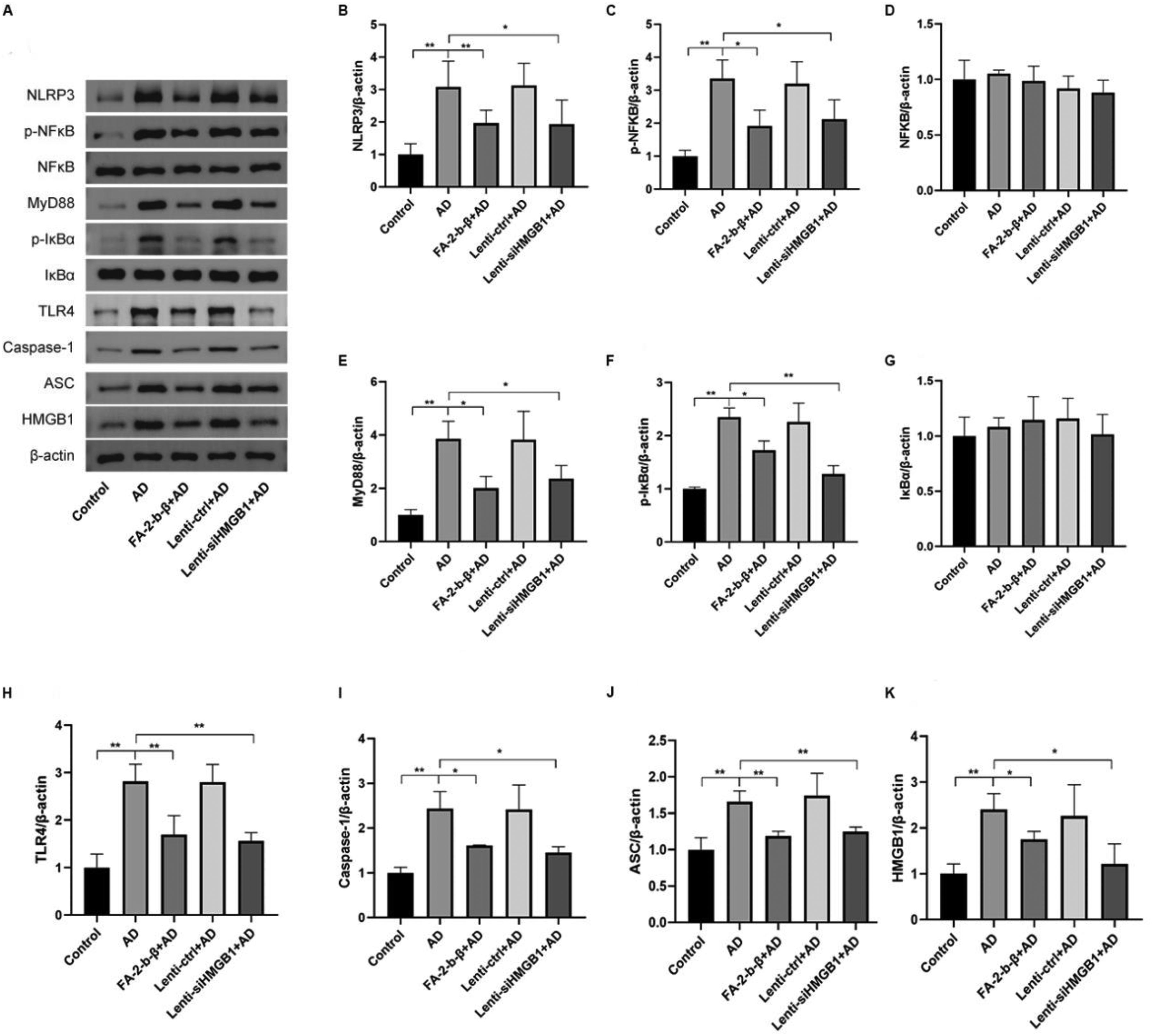
The effects of FA-2-b-β and HMGB1 on HMGB1/NF-κB/NLRP3 pathway-related protein levels. (A) Changes in the protein levels of HMGB1/NF-κB/NLRP3 pathway-related proteins. The protein expression levels of NLRP3 (B), p-NF-κB (C), NF-κB (D), MyD88 (E), p-IκBα (F), IκBα (G), TLR4 (H), caspase-1 (I), ASC (J), and HMGB1 (K) were assessed. *p < 0.05; **p < 0.01.
Discussion
AD is a complex neurodegenerative disorder characterized by the pathological accumulation of extracellular Aβ plaques and intracellular neurofibrillary tangles composed of hyperphosphorylated tau protein. 21 In this study, we demonstrated that FA-2-b-β inhibits Aβ-induced HMGB1 expression and binds to HMGB1, thereby stabilizing its function. Excessive HMGB1 interacts with TLR4 on the cell membrane, initiating the NF-κB signaling cascade and promoting the expression of NLRP3. Subsequently, NLRP3 recruits ASC and caspase-1 to form inflammasomes, ultimately leading to caspase-1-dependent pyroptosis. Our findings suggest that FA-2-b-β effectively mitigates neuroinflammation and influences the progression of AD by modulating this pathway. Specifically, we propose that HMGB1 plays a central role in Aβ-induced neuroinflammation and pyroptosis in AD, and that FA-2-b-β can modulate this interaction.
HMGB1 emerges as a critical mediator of neuroinflammation in AD, functioning as a prototypical damage-associated molecular pattern (DAMP) molecule. 22 HMGB1 activates the NLRP3 inflammasome, thereby exacerbating inflammation in AD. 23 Targeting HMGB1 and its downstream pathways, including NF-κB and NLRP3, offers a promosing therapeutic strategy to attenuate neuroinflammation and decelerate AD progression.24,25 Previous studies have associated HMGB1 with pyroptosis-related inflammatory responses in various pathological conditions, including heat stroke-induced hepatocyte injury, although the exact mechanisms are not fully understood. This study provides evidence that Aβ influences pyroptosis thourgh HMGB1.24,26 By employing siRNA-mediated HMGB1 knockdown, we showed that inhibiting HMGB1 decreases inflammatory factor expression and pyroptosis in both cellular and animal models. These results underscore HMGB1’s role as a mediator of Aβ-induced neuroinflammation and pyroptosis in AD.
Emerging evidence underscores pyroptosis as a pivotal contributors to neuroinflammation in neurodegenerative diseases, including AD. 27 As a pro-inflammatory form of programmed cell death, pyroptosis is distinguish by rapid plasma membrane permeabilization and the release of intracellular pro-inflammatory mediators, setting it apart from other cell death modalities such as apoptosis or necrosis. 28 This process primarily initiated through the classical caspase-1 pathway, as well as alternative pathways involving caspases-4, -5, and -11 pathways. 29 Within the classical pathway, caspase-1 cleaves gasdermin D (GSDMD), generating an N-terminal fragment that forms membrane pores, thereby increasing permeability and facilitating the release of cytoplasmic contents, and inflammation.30,31 Simultaneously, activated caspase-1 converts pro-IL-1β and pro-IL-18 into their active forms, which are subsequently secreted to recruit immune cells and amplify the inflammatory response.32,33 Caspase-1 mediated pyroptosis is intricately linked to the initiation and progression of AD, with caspase-1 inhibition shown to significantly mitigate neuroinflammation and confer protection in AD models. 34 Research has highlighted the critical role of the NLRP3/caspase-1 axis in the pathogenesis of AD, suggesting that inhibition of the NLRP3 inflammasome may serve as a promising therapeutic strategy for this neurodegenerative disorder. 35 In the present study, we examined the levels of IL-1β, IL-18, and caspase-1 in animal models of AD. Our results demonstrated that FA-2-b-β suppresses NLRP3 inflammasome activation, thereby alleviating neuroinflammation and neuronal damage associated with AD.
The NF-κB signaling pathway, known for its regulatory role in inflammation, is persistently activated in AD, facilitating the transcription of various pro-inflammatory cytokines and enzymes that exacerbate neuroinflammation. Thus, targeting NF-κB presents a viable approach to mitigating inflammation in AD.36,37 The interaction between NF-κB and the NLRP3 inflammasome is particularly crucial, as NF-κB activation enhances NLRP3 expression, promoting inflammasome assembly and subsequent pyroptosis. 38 Our findings suggest that Aβ influences pyroptosis and neuroinflammation in AD cells via the HMGB1/NF-κB/NLRP3 signaling axis, and that FA-2-b-β effectively attenuates these effects, thereby influencing the pathogenesis and progression of AD.
FA-2-b-β, an extract derived from Agaricus blazei Murill, has demonstrated significant bioactivity in inflammation-related processes, with well-documented antitumor and antioxidative properties. Research has confirmed its efficacy in inhibiting tumor growth and promoting apoptosis across various cancer types, including acute myeloid leukemia (AML) and acute lymphoblastic leukemia (ALL). 16 Notably, FA-2-b-β has been observed to inhibit the proliferation of diffuse large B-cell lymphoma cells by inducing ferroptosis through the Nrf2/HO-1 signaling pathway, thereby establishing its potential as a promising therapeutic candidate. 39 This study seeks to further elucidate its mechamisms in AD by examining FA-2-b-β's role in modulating HMGB1-mediated inflammation and pyroptosis, with a particular emphasis on its interaction with the HMGB1/NF-κB/NLRP3 signaling axis. In cellular and animal models of AD, FA-2-b-β reduced Aβ-induced HMGB1 overexpression and attenuated NF-κB activation, leding to a decreasing NLRP3 inflammasome assembly. This cascade resulted in reduced activation of caspase-1 and decreased levels of the pro-inflammatory cytokines IL-1β and IL-18, thereby mitigating pyroptosis and neuroinflammation. Importantly, the knockdown of HMGB1 further confirmed the role of HMGB1 in modulating Aβ-induced inflammatory responses. Administration of FA-2-b-β was found to attenuate neuroinflammation, preserve neuronal integrity, and improve cognitive performance.
Although our study focused on the neuroprotective effects of FA-2-b-β, its safety profile remains insufficiently explored. As a bioactive compound with immunomodulatory properties, excessive exposure to FA-2-b-β may pose risks, such as dose-dependent immunosuppression or off-target effects. Future studies should investigate its toxicity thresholds, long-term effects, and interactions with other AD therapies to ensure its clinical applicability. While our study provides valuable insights into the neuroprotective effects of FA-2-b-β via the HMGB1/NF-κB/NLRP3 axis in AD, there are certain limitations that merit consideration. Specifically, the detailed molecular basis underlying the interaction between FA-2-b-β and HMGB1 remains to be fully characterized, and the sequential causal links between FA-2-b-β-mediated HMGB1 modulation, NF-κB inhibition, and subsequent NLRP3 inflammasome suppression require further rigorous validation. These aspects will be the focus of our future research.
Conclusion
In conclusion, this study elucidates the effect of FA-2-b-β in suppressing Aβ-induced neuroinflammation and pyroptosis via the HMGB1/NF-κB/NLRP3 signaling axis. Through comprehensive evaluations at the cellular, molecular, and animal levels, we have uncovered novel insights into its therapeutic potential for AD. The findings suggest that FA-2-b-β may represent a promising therapeutic strategy for AD treatment by targeting neuroinflammatory and pyroptotic pathways.
Footnotes
Acknowledgements
The authors have no acknowledgments to report.
Ethical considerations
Ethical approval for this study was obtained from the Ethics Committee of Gansu Provincial Hospital (2025029).
Consent to participate
Not applicable
Consent for publication
Not applicable
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Gansu provincial joint research fund of science and technology planning project (24JRRA890).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data used to support the findings of this study are available from the corresponding author upon request.
