Abstract
Background
Soluble tau oligomers (tauO) are early, synaptotoxic drivers of dysfunction in tauopathies. While selective vulnerability is well documented at the cellular level, emerging evidence suggests that synaptic subtypes may differ in their susceptibility to tau pathology. Still, the key factors that shape synaptic vulnerability to toxic tauO, particularly in humans, remain poorly understood.
Objective
To define the synaptic compartments and subtypes most vulnerable to tauO and identify molecular correlates underlying this susceptibility.
Methods
Synaptosomes were isolated from cognitively normal human autopsy specimens and acutely challenged with preformed recombinant tauO. Flow cytometry with multiplexed immunophenotyping resolved tauO engagement across intact pre- and postsynaptic compartments and excitatory versus inhibitory subtypes. Functional effects were assessed by microtransplanting synaptic membranes into
Results
TauO preferentially engaged presynaptic compartments and showed elevated association with GABAergic synapses. Functionally, acute tauO exposure selectively enhanced GABAAR-mediated responses, with no effect on AMPAR-mediated currents. The PART BDTO interactome was enriched for presynaptic vesicle-associated proteins involved in vesicle cycling and neurotransmitter release, consistent with a presynaptic axis of vulnerability.
Conclusions
This integrative analysis identifies a compartment- and subtype-specific vulnerability of human synapses to tauO, highlighting a presynaptic inhibitory bias as a potential driver of synaptic dysfunction and tau propagation in early-stage tauopathies.
Introduction
Tau is a microtubule-associated protein canonically implicated in axonal transport and cytoskeletal stability, but is now recognized also as a key modulator of synaptic architecture, signaling, and plasticity.1–5 In tauopathies—neurodegenerative disorders characterized by aberrant tau accumulation—tau adopts misfolded conformations that assemble into oligomers, paired helical filaments, and ultimately neurofibrillary tangles. 6 Among these, soluble tau oligomers (tauO) have garnered particular attention as early, synaptotoxic species that correlate strongly with cognitive decline.7–11 TauO have also been implicated in the trans-synaptic propagation of pathology via prion-like propagation, further implicating the synapse as both a vulnerable target and a conduit for early tau-mediated injury.12–16
Within this framework, emerging evidence suggests that the accumulation and functional consequences of tauO may not be uniformly exerted across all synapses. 17 While selective vulnerability has long been appreciated at the cellular level, where specific neuronal populations exhibit disproportionate susceptibility to tau pathology, recent studies indicate that distinct synaptic subtypes, distinguished by features such as cytoarchitecture, neurotransmitter signaling, or anatomic localization, may differ also in their capacity to engage and respond to pathological tau species.17–20 Yet, the structural, functional, and molecular determinants of this selective synaptic vulnerability, especially as they pertain to tauO, remain largely ill defined.
To begin addressing this gap, we recently examined autopsy brain specimens from individuals with primary age-related tauopathy (PART), leveraging PART as an amyloid-free model to isolate synapse-specific vulnerability to tau pathology in a companion study. 21 Using complementary biochemical and electrophysiological approaches, we quantified native tau species and assessed ligand-gated synaptic responses from the same tissue; we found that elevated tauO burden in PART was correlated with a reduction in the synaptic excitation-to-inhibition (sE/I) ratio. However, whether this shift reflects diminished excitation, enhanced inhibition, or a more complex interplay remains unresolved. In particular, it is unclear whether tauO preferentially engage and modulate specific functional synaptic subtypes (excitatory or inhibitory) or synaptic compartments (presynaptic or postsynaptic).
To further elucidate the structural, functional, and molecular determinants of synaptic vulnerability to tauO, here we implemented a translationally relevant platform using human synaptosomes derived from autopsy brain specimens of aged, cognitively normal individuals. Synaptosomes were acutely challenged with preformed recombinant tauO, and synapse-specific tauO engagement was quantified via flow cytometry using a multiplexed synaptic immunophenotyping panel. To evaluate changes in synaptic function, synaptic membranes from tauO-treated synaptosomes were microtransplanted into
Our findings suggest that tauO preferentially engage presynaptic terminals, with additional preference for inhibitory synapses. Functionally, tauO exposure selectively potentiated GABAergic currents, while glutamatergic (i.e., kainate-sensitive AMPAR) responses remained unaffected. Proteomic analysis of PART BDTO revealed a vesicle-associated interactome enriched in presynaptic release machinery. Together, these findings delineate a synapse-type- and compartment-specific pattern of oligomeric tau engagement and implicate presynaptic inhibitory terminals as selectively vulnerable targets in early-stage tauopathy (Figure 1).
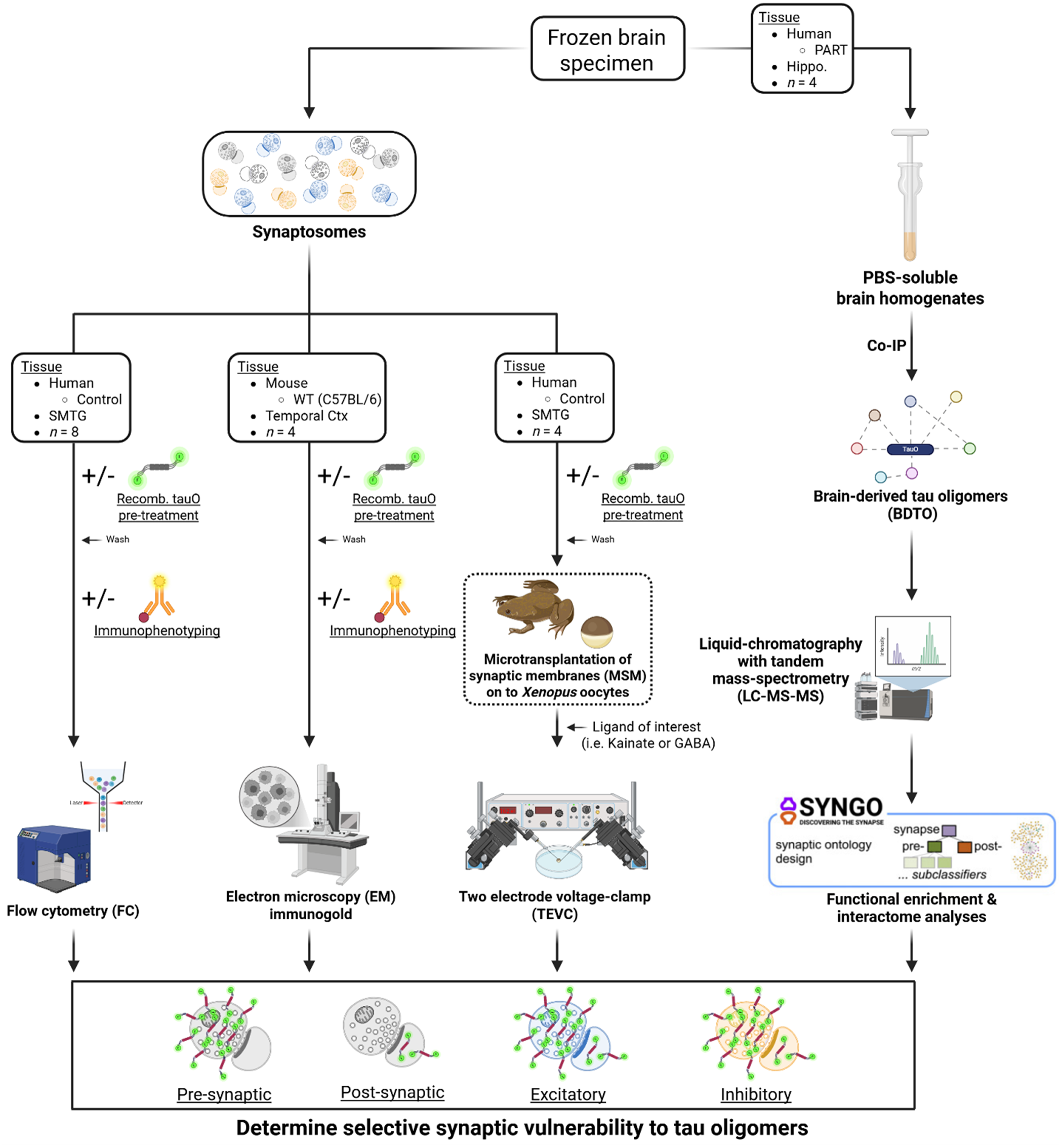
Schematic overview of the experimental workflow. This study investigates selective synaptic vulnerability to tauO using a multi-faceted approach integrating cellular, molecular, structural, and functional techniques. (Left panel) Synaptosomes isolated from Control human SMTG or WT mouse cortex were treated ± rtauO and analyzed via FC, EM, and TEVC electrophysiology. (Right panel) BDTO were isolated from PART hippocampal tissue and analyzed via LC-MS/MS proteomics and SynGO enrichment to identify synapse-specific pathways and interactomes. This integrative strategy enables the characterization of selective synaptic vulnerability to tauO across pre- versus postsynaptic and excitatory versus inhibitory compartments/subtypes.
Methods
Clinical and pathological profiling of human postmortem samples
Human postmortem brain tissue was obtained through the Alzheimer's Disease Research Center (ADRC) at the Sanders-Brown Center on Aging, University of Kentucky, under established material transfer agreements. All participants were enrolled in longitudinal studies of aging and dementia, with informed consent obtained prior to enrollment. All procedures adhered to institutional IRB guidelines.
Annual clinical assessments were conducted by experienced clinicians trained in cognitive and neurological evaluation, including administration of the Mini-Mental State Examination (MMSE) and the Clinical Dementia Rating (CDR) scale. Standardized neuropathological evaluations were performed by board-certified neuropathologists using established criteria from the Consortium to Establish a Registry for Alzheimer's Disease (CERAD) and Braak staging to assess AD pathology. Control cases exhibited no cognitive impairment (CDR = 0, MMSE ≥ 26) and were free of AD-related neuropathological changes. Cases classified as PART demonstrated mild to moderate tau pathology (Braak stages I–II), predominantly confined to the hippocampus, in the absence of Aβ plaques, in accordance with established diagnostic criteria.22–24 Notably, previous work from our group confirmed the absence of detectable Aβ pathology in synaptosomal preparations derived from this PART cohort.
13
Individuals carrying the
All specimens were de-identified prior to transfer to the University of Texas Medical Branch (UTMB), rendering their use exempt from additional IRB oversight under CFR §46.101(a)(1). Comprehensive demographic, clinical, and neuropathological characteristics of all subjects are provided in Supplemental Table 1.
Isolation and preparation of synaptosomes from human brain tissue
Standardized protocols for synaptosome isolation, quality control, and downstream applications have been optimized and validated in our vast prior work.13,28–31 In the present study, synaptosome integrity and synaptic enrichment were confirmed by transmission electron microscopy and LC-MS/MS proteomic profiling with subsequent SynGO analysis (Supplemental Figure 1).
For FC experiments, synaptosomes were isolated from frozen autopsy samples of the superior middle temporal gyrus (SMTG) obtained from cognitively intact Control subjects (
For MSM (microtransplantation of synaptic membranes) experiments, synaptosomes were prepared as described above. Following the final centrifugation step, P2 pellets were resuspended in Syn-PER containing protease and phosphatase inhibitors (2 µL per mg of starting tissue). Total protein concentrations were quantified fluorometrically using a QFX fluorometer (DeNovix, New Castle, DE, USA), and volumes were adjusted to a final concentration of 2 mg/mL. Aliquots were immediately stored at −80°C to prevent degradation from repeated freeze–thaw cycles.
Preparation and fluorescent labeling of preformed recombinant tau oligomers
Preformed recombinant tau oligomers (rtauO), generated from monomers derived from the full-length (2N4R) tau construct, were kindly provided by the laboratory of Dr Rakez Kayed and prepared in accordance with previously established protocols.32–34 All procedures involving rtauO utilized low-retention plasticware to minimize protein loss.
Fluorescent labeling of rtauO was performed using the Microscale Protein Labeling Kit with Alexa Fluor™ 488 (Thermo Fisher Scientific, cat# A30006), following the manufacturer's instructions. Labeling conditions were optimized for full-length human tau (45.9 kDa) to ensure efficient conjugation and reproducibility across preparations. Briefly, tauO were diluted to 1 mg/mL in 1× Dulbecco's phosphate-buffered saline (PBS; Corning, cat# 46-013-CM). Alexa Fluor 488 dye was added at a dye-to-protein molar ratio of 50:1 to achieve an optimal degree of labeling (DOL). The reaction mixture was gently mixed and incubated for 30 min at room temperature. Following conjugation, excess unbound dye was removed via spin filtration using Bio-Gel P-6 fine resin columns, as recommended in the kit protocol. The DOL and post-labeling protein concentration were determined by measuring absorbance at 280 nm (A280), adjusted for fluorophore-specific absorbance, using a NanoDrop 2000c spectrophotometer (Thermo Fisher Scientific, Wilmington, DE, USA). Protein concentrations were further validated using the Pierce™ BCA Protein Assay Kit (Thermo Fisher Scientific, cat# 23227). Efficient fluorophore conjugation and successful removal of unreacted dye were confirmed by UV-Vis spectroscopy, which showed a strong absorbance peak at 495 nm in the purified rtauO fraction and negligible signal in the flow-through (Supplemental Figure 2a). To confirm structural integrity, western blot analysis was routinely performed to verify maintenance of the oligomeric conformation following labeling (Supplemental Figure 2b).
Flow cytometry
For synaptic rtauO challenge, synaptosomes were incubated with Alexa Fluor 488-conjugated rtauO for 1 h at room temperature (RT) in a final volume of 20 µL HBK. Samples were then washed three times with HBK and either analyzed immediately or stored at 4°C for no more than 24 h before analysis. Prior to reading, samples were diluted with 200 µL DPBS and gently vortexed. To determine optimal conditions for rtauO challenge, titration experiments were performed by fixing either the synaptosome concentration (100,000 synaptosomes/µL) while varying rtauO concentrations (2.5 nM to 2.0 µM), or fixing the rtauO concentration (25 nM) while varying synaptosome concentrations (3700 to 300,000 synaptosomes/µL). These experiments revealed that 25 nM rtauO produced a near half-maximal engagement response (∼60%), and that synaptosome concentrations below 33,000 synaptosomes/µL yielded maximal signal intensity; accordingly, these parameters were adopted for all subsequent experiments (Supplemental Figure 3).
Prior to initiating immunophenotyping experiments, the following solutions and buffers were prepared: HBK buffer (recipe outlined in
The antibody panel used for synaptosome immunophenotyping was selected based on four key criteria: (1) compartment specificity of the target protein, (2) commercial validation of the antibody for FC applications, (3) availability of pre-conjugated fluorophore formats to eliminate the need for secondary staining, and (4) minimal spectral overlap among selected fluorophores. All antibodies used in multiplexed panels, except for STX1A and VAMP2, were pre-conjugated. For STX1A and VAMP2, secondary antibodies were included where applicable. Antibodies were titrated against isotype-matched controls to confirm specificity and optimize signal-to-noise ratios (Supplemental Figure 4). To ensure reliable colocalization and distinguishability of synaptic subpopulations, fluorophores were chosen to allow resolution of both positive and negative populations across at least two, and preferably three markers per panel (Supplemental Figure 5). Co-staining experiments confirmed expected colocalization patterns for pre- and postsynaptic markers (Supplemental Figure 6), and final panel design was iteratively optimized to balance resolution, signal intensity, and fluorophore compatibility for multiplexed FC.
To assess rtauO engagement across specific synaptic compartments and functional subtypes, the rtauO challenge was integrated within the immunophenotyping workflow. Following CellTrace labeling and neutralization, synaptosomes were centrifuged and resuspended in 100 µL HBK (assuming a starting amount of ∼1 × 106 synaptosomes) in lieu of SET buffer, then divided equally (50 µL each) into rtauO-positive (rtauO+) and rtauO-negative (rtauO–; HBK-only) groups for incubation at RT for 1 h. Wash steps were performed in HBK as described above, followed by resuspension in SET buffer. Subsequent fixation, blocking, permeabilization, and antibody incubation steps adhered strictly to the standard immunophenotyping protocol and were performed in parallel for rtauO+ and rtauO– groups. When multiple samples were processed simultaneously, procedural steps through blocking and permeabilization were scaled proportionally. For example, to immunophenotype five samples, 5 × 106 synaptosomes were labeled in 500 µL HBK, then evenly divided into rtauO+ and rtauO– groups (250 µL each). Following blocking and permeabilization, each group was further split into five 50 µL aliquots for multiplexed antibody staining. All wash steps and reagent volumes were proportionally scaled to maintain consistent working concentrations.
Instrumentation and acquisition parameters for flow cytometry small particle measurement
FC experiments were performed using either the Guava EasyCyte 8 (EMD Millipore, Burlington, MA) or the BD FACSymphony A5 SE (BD Biosciences, San Jose, CA), with instrumentation selected based on experimental needs. With the exception of updated optical configurations for additional probes and fluorophores, all instrument settings and acquisition parameters were adopted directly from our recently published work. 13 These updates, implemented on the BD FACSymphony A5 SE, enabled expanded multiplexed detection of synaptic markers with minimal spectral interference.
The Guava EasyCyte 8 was used exclusively for synaptosome enumeration and rtauO titration experiments due to its built-in absolute particle counting capability, which obviates the need for reference beads. Synaptosomes were acquired at a flow rate of 0.5 µL/s (“Low” setting) using SSC triggering. A total of 10,000 events within the designated size gate were acquired per sample.
Synaptosome immunophenotyping was conducted on the BD FACSymphony A5 SE flow cytometer. This instrument was selected for its multispectral detection capabilities, supported by five spatially separated lasers and a dedicated array of detectors. These features enabled efficient multiplexing and precise resolution of diverse synaptic sub-populations. Detailed optical configurations are provided in Supplemental Table 3. Synaptosomes were acquired using dual-threshold triggering on side scatter (SSC) and CellTrace Blue fluorescence to ensure reliable detection of structurally intact particles, as fluorescence-based thresholding has been shown to significantly outperform standard FSC-based detection for small particle analysis. 37 Data were collected using BD FACSDiva Software v9.0, with fluorescence compensation applied at the start of each run using single-color compensation beads (ThermoFisher, cat# 01-2222-42) and matrix calculations performed according to standard cytometry practices. 38 Samples were acquired for 3 min per condition.
To ensure accurate and reliable detection of small particles such as synaptosomes, several methodological considerations were implemented across both platforms. Instrument sensitivity and size calibration were assessed on both instruments prior to each run using standardized ApogeeMix “Micro” (cat# 1493, Apogee Flow Systems) and Spherotech polystyrene (Spherotech Inc., Lake Forest, IL, USA) bead standards. These calibrations supported consistent definition of the synaptosome size gate (0.5–1.3 µm) (Supplemental Figure 7). Optimization of linear detection range and event rate fidelity for both instruments has been described previously. 13 Lastly, to minimize coincidence detection and particle aggregation, we implemented several additional quality control measures, including shortened centrifugation steps and spin time, optimal sample dilution, and the use of non-ionic buffers when feasible.13,37
Flow cytometry data analysis
FC data analysis, including gating, quantification, and the generation of histograms and density plots, was performed using FlowJo v10 (BD Life Sciences, Ashland, OR). Synaptic compartments and functional subtypes were differentiated using Boolean (conditional) gating strategies, defined by the co-expression or exclusion of established synaptic markers. Gating thresholds were determined using antibody-negative (antibody–) controls, ensuring identical implementation across rtauO+ and untreated rtauO− samples. Importantly, rtauO− samples served as internal gating references, facilitating accurate delineation of rtauO+ events within rtauO treated samples. A comprehensive list of the conditional gating strategies used to define individual synaptic populations is provided in Supplemental Figure 5, and a representative gating workflow is illustrated in Supplemental Figure 7.
While the identification of rtauO+ synaptosomes provided qualitative confirmation of oligomer engagement, quantitative analysis was based on the median fluorescence intensity (MFI; detector: B537; fluorophore: Alexa-488) of the entire synaptic subpopulation. This approach is particularly advantageous for analyzing small particles such as synaptosomes, where inherent signal heterogeneity and optical limitations frequently hinder accurate discrimination between dim and truly negative events—a potential limitation of percentage-based metrics in this context. 39 Furthermore, MFI was chosen as the primary readout due to its sensitivity in capturing the full range of fluorescence intensity, thus enabling robust detection of shifts in oligomer engagement.
To account for technical variability and background signal, MFI values for each synaptic subpopulation were normalized in two stages. First, MFI values were divided by the MFI of the total rtauO+ synaptosome population to control for inter-sample variation in rtauO delivery. Second, this value was further normalized to the MFI of the same synaptic subpopulation in rtauO− samples, thereby correcting for background fluorescence, autofluorescence, or other sources of optical noise. The resulting normalized MFI values were used for all downstream analyses. Samples were analyzed in technical duplicate within the same experimental day, and each experimental paradigm was repeated on separate days to confirm reproducibility. All MFI metrics and event counts were exported using FlowJo's Batch Export function, with subsequent statistical analyses conducted in GraphPad Prism v10.0 (GraphPad Software, La Jolla, CA).
Assessment of synaptic function via microtransplantation of synaptic membranes
Protocols for isolating synaptosomes, including detailed characterization and stringent quality control procedures, have been optimized and validated in our vast prior studies.13,28–31 Similarly, our group has rigorously established and validated methodologies for assessing synaptic functionality through microtransplantation of synaptic membranes (MSM) into
For MSM experiments, pooled synaptosomes (
rtauO challenge experiments were conducted in a manner analogous to our FC experiments, with minor modifications. Briefly, synaptosomes were incubated with either rtauO or HBK-only control for 1 h at room temperature in a final volume of 20 µL HBK, followed by three sequential washes in HBK (10,000 × g for 5 min at 4°C). To achieve optimal transplantation of synaptic receptors on to
Microinjection needles were generated from 3.5-inch borosilicate glass tubes (Drummond Scientific, PA, USA) using a micropipette puller (Model P-1000; Sutter Instruments, Novato, CA, USA), and tips were trimmed uniformly to a length of approximately 4 mm to facilitate consistent injection volumes. Prior to injection, oocytes were transferred individually to SBS. Each microinjection needle, freshly prepared for each sample, was prefilled with 0.5 mL/cc mineral oil and mounted onto a Nanoject II microinjector (Drummond Scientific, PA, USA). Synaptosome preparations were adjusted to a concentration of 2 ng/nL, homogenized by ultrasonication (3 cycles of 5 s each) in an ice-cold FS20D ultrasonic cleaner (Thermo Fisher Scientific, Waltham, MA, USA), and maintained on ice between cycles. Approximately 50.6 nL of the synaptosome preparation was injected per oocyte, with injections carried out under the oocyte's surface within a nylon mesh-lined glass Petri dish. Injected oocytes were incubated at 16°C in SBS within a 96-well conical-bottom plate (Thermo Fisher Scientific).
Stock solutions of GABA (Sigma-Aldrich, St Louis, MO, USA) and kainate (Tocris Bioscience, Bristol, UK) were prepared in deionized water and stored at −20°C. Prior to electrophysiological assessment, working solutions (final concentrations: 1 mM and 100 µM) were freshly diluted in a magnesium-free custom formulation of Normal Frog Ringer (NFR) solution (Ecocyte BioScience Inc.). Two-electrode voltage clamp (TEVC) recordings were performed using a Roboocyte2 recording system, consumable measuring heads, and glass electrodes pre-filled with 3 M KCl (Thermo Fisher Scientific) and embedded with Ag/AgCl-treated silver wires (Multi-Channel Systems, MCS GmbH, Reutlingen, Germany). Electrode resistances were maintained between 200 and 1000 kΩ, and a minimum resting membrane potential of −15 mV was confirmed prior to recordings. Oocytes were voltage-clamped at a holding potential of −80 mV, with a drug application rate set at 5000 µL/min controlled by a peristaltic pump. Experimental sequences for application and recording were programmed via JavaScript, with detailed scripts accessible at https://github.com/LimonLab-UTMB. Subsequent analysis, including drift correction, baseline adjustment, and peak amplitude measurements, was conducted using Roboocyte2 + software (Multi Channel Systems, MCS GmbH). Outliers from peak amplitude measurements were identified using the interquartile range (IQR) method with tail quantiles set at 0.25 and a multiplier (Q) of 1.5. Data points outside this quantile range were excluded from subsequent analyses.
Post hoc analysis of synaptic pathways within the PART BDTO interactome
Building on previously published proteomic data from individuals with Primary Age-Related Tauopathy (PART), we re-analyzed the brain-derived tau oligomer (BDTO) interactome to evaluate enrichment of synaptic components and processes.
44
This dataset was originally generated from hippocampal autopsy tissue (
For the present study, we performed
Electron microscopy (EM)
Following rtauO treatment, mouse synaptosomes were pelleted (10,000 × g for 10 min at 4°C) and fixed in 2% glutaraldehyde overnight at 4°C. Samples were stained en bloc with 2% aqueous uranyl acetate, dehydrated in 50% and then 75% ethanol, and embedded in LR White resin (medium grade; Electron Microscopy Sciences, Cat#14381). Ultrathin sections (70–80 nm) were prepared on a Leica EM UC7 ultramicrotome and mounted on Formvar-carbon-coated nickel grids (EMS, Hatfield, PA). Grids were incubated in a humid chamber on drops of blocking buffer (0.1% BSA and 0.01 M glycine in 0.01 M PBS) for 15 min, followed by incubation with T22 (1:50 in 1% BSA/PBS) for 1 h at room temperature and overnight at 4°C. After washing, grids were incubated for 1 h at room temperature in the dark with 6 nm gold-conjugated anti-mouse IgG secondary antibody (Electron Microscopy Sciences, Cat#25123) diluted 1:20 in 1% BSA. Following additional washes in blocking buffer, PBS, and distilled water, grids were fixed in 2% aqueous glutaraldehyde for 5 min at room temperature. Sections were stained with 2% uranyl acetate for 5 min, washed, air dried, and counterstained with lead citrate for 30 s. Final imaging was conducted using a JEM-1400 TEM (JEOL USA) at 80 kV, with images acquired on a bottom-mounted Orius SC-200 camera (Gatan).
Systematic literature review of tauO–synapse studies in human tissue
To contextualize our findings within the broader literature on tauO and synaptic pathology in human systems, we conducted a systematic PubMed search using defined Boolean operators. Of note, our search was focused on three main criteria: tau oligomers, synapses, and human/patient specimens. The search string was: (“tau oligomer"[Title/Abstract] OR “tau oligomers"[Title/Abstract] OR “oligomeric tau"[Title/Abstract]) AND (“synapse"[Title/Abstract] OR “synapses"[Title/Abstract] OR “synaptic"[Title/Abstract] OR “synaptic vulnerability"[Title/Abstract] OR “synaptic dysfunction"[Title/Abstract])
This search yielded 67 articles in total (see Supplemental File 2 for the full list), including 47 primary research articles and 20 reviews. After filtering for studies involving human tissue, 29 primary articles remained. Following individual review, key studies specifically addressing tauO-associated synaptic vulnerability in human specimens were curated and summarized in Table 1 of the Discussion.
Summary of prior studies examining tau oligomer interactions with human synapses.

Survey of PubMed-indexed studies assessing tauO localization and synaptic impact in human tissue. Entries are ranked (row-wise) by publication date and organized (column-wise) by diagnostic classification, methodological approach, and key findings relevant to selective synaptic vulnerability. AD: Alzheimer's disease; AT: array tomography; Alz50: antibody against misfolded and/or aggregated tau species; BDTO: brain-derived tau oligomers; co-IP: co-immunoprecipitation; dSTORM: direct stochastic optical reconstruction microscopy; sE/I: synaptic excitatory-to-inhibitory ratio; EM: electron microscopy; hiPSC: human induced pluripotent stem cells; IF: immunofluorescence; IHC: immunohistochemistry; LC-MS/MS: liquid chromatography–tandem mass spectrometry; MSM: microtransplantation of synaptic membranes; NDAN: non-demented with Alzheimer's neuropathology; PART: primary age-related tauopathy; PHF: paired helical filament; PSD95: postsynaptic density protein 95; SYPH: synaptophysin; TOC1 and T22: anti-tau antibody specific to oligomeric conformation; VGLUT1: vesicular glutamate transporter 1; WB : western blot.
Data analysis and visualization software
All statistical analyses and graphical representations were performed using GraphPad Prism 10.0 (GraphPad Software, La Jolla, CA, USA). Figures were prepared with BioRender (BioRender.com) and Adobe Illustrator (Adobe Inc., San Jose, CA, USA).
Results
To assess whether tauO selectively engage specific synaptic compartments and functional subtypes, SMTG synaptosomes isolated from autopsy specimens of Control individuals were treated with rtauO (25 nM) and analyzed via FC immunophenotyping. Experimental conditions were rigorously optimized for accurate detection of small particles and synaptic resolution (see
Synapses were delineated by compartment (presynaptic versus postsynaptic) and functional subtype (excitatory versus inhibitory). rtauO engagement was significantly greater (∼1.73-fold) in the presynaptic compared to the postsynaptic compartment (Figure 2(a);
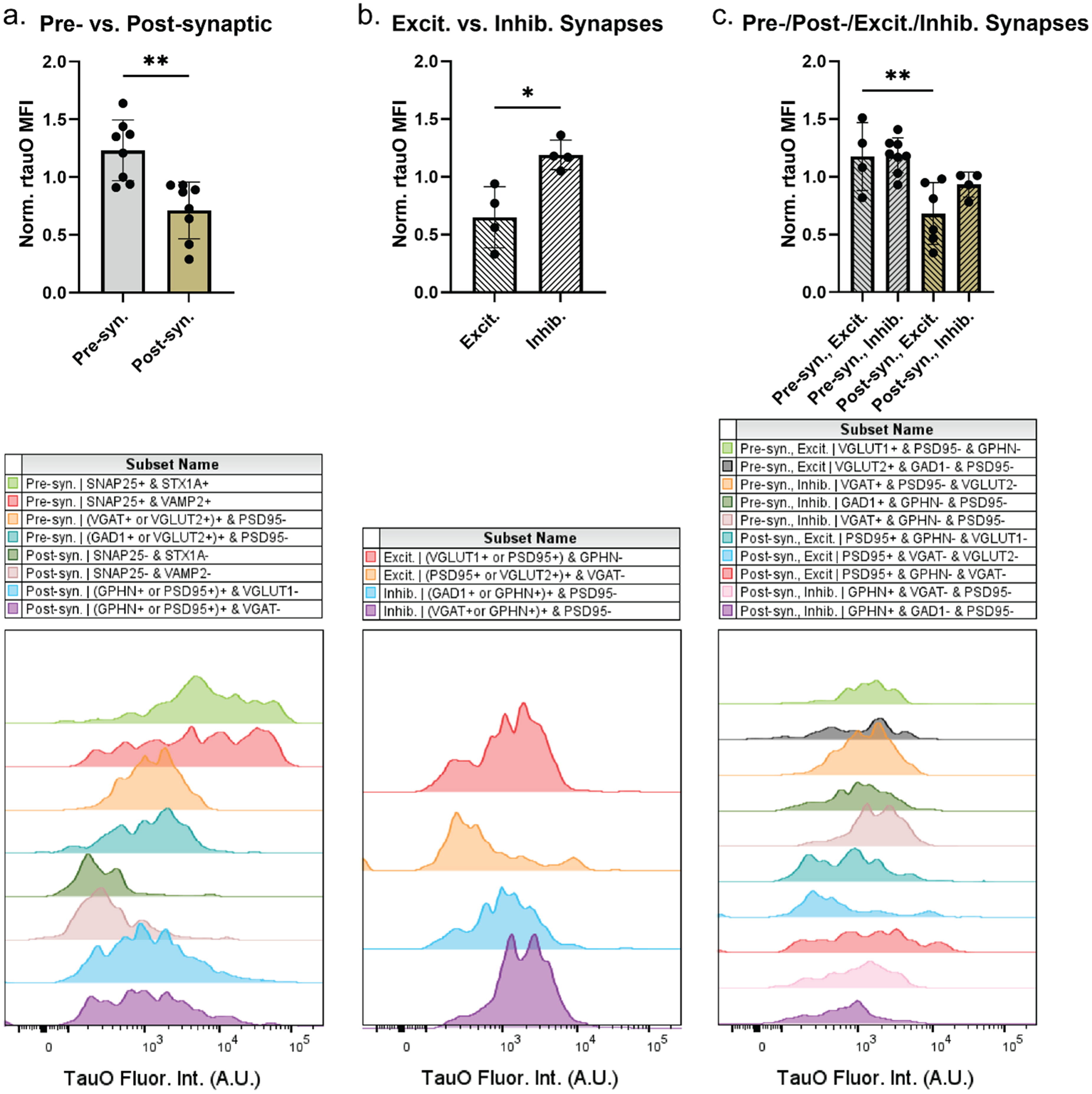
rtauO preferentially engage presynaptic compartments and inhibitory synapses. FC immunophenotyping was performed on pooled Control SMTG synaptosomes (
To determine whether tauO modulate synaptic receptor function, we performed TEVC recordings in
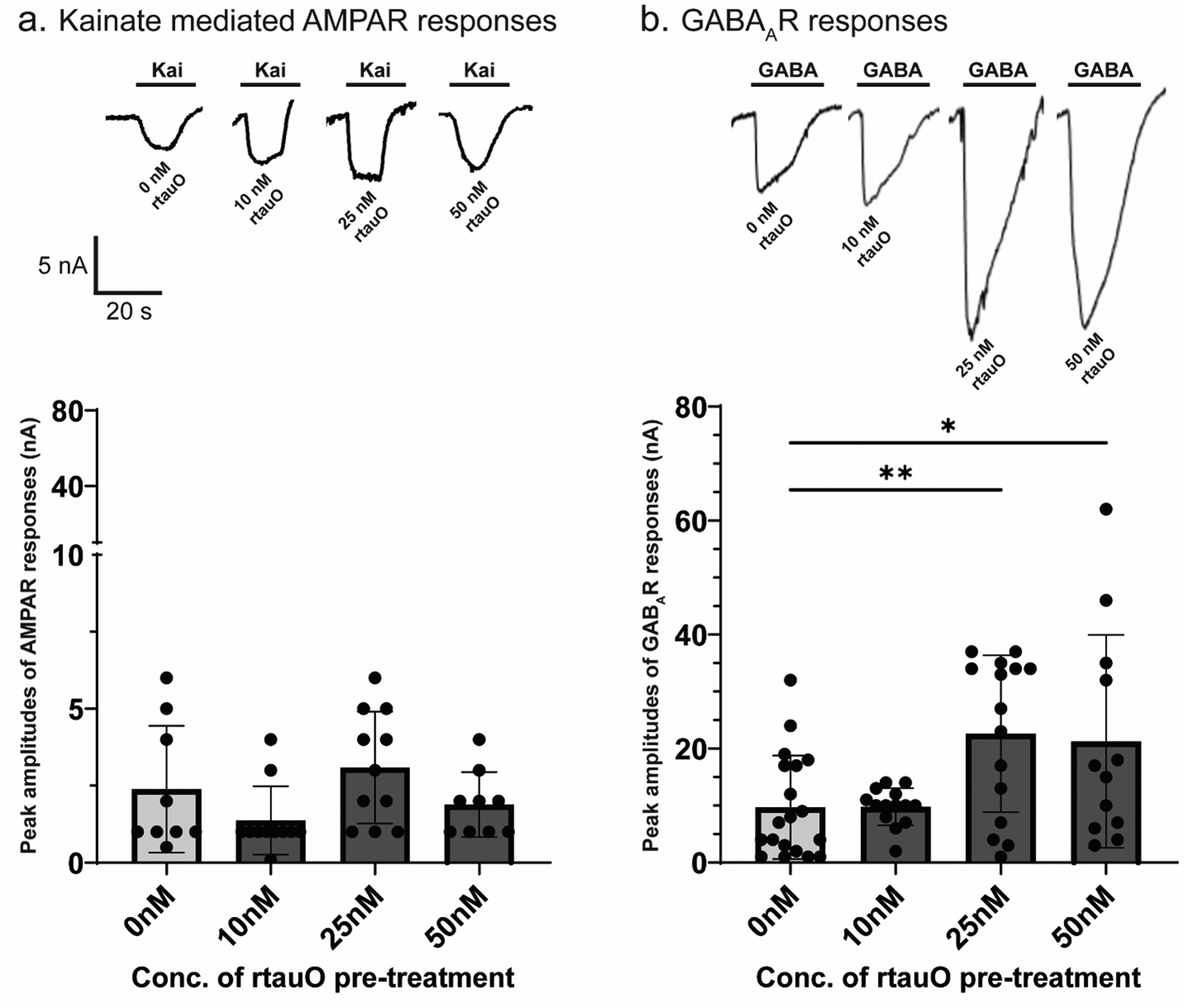
rtauO selectively potentiates GABAergic synaptic currents in
To examine selective synaptic vulnerability to tauO from a molecular perspective, BDTOs and their associated proteins were isolated from PBS-soluble fractions of PART hippocampal autopsy tissue via co-immunoprecipitation using the tau oligomer-specific antibody T18. This approach enabled the identification of synaptic proteins associated with endogenous BDTOs in their native structural conformation. Associated proteins were characterized by LC-MS/MS proteomic analysis, and the resulting interactome was analyzed using SynGO to assess enrichment across structural (Cellular Component, CC) and functional (Biological Process, BP) ontologies. In addition, isolating BDTO from the PBS-soluble total homogenate, rather than a pre-enriched synaptosomal fraction, offered two methodological advantages. First, this approach selectively targets “soluble” tauO, which are widely considered to be the most neurotoxic species.33,54–56 Second, it supports unbiased profiling of protein interactors by not limiting detection to synaptic compartments.
BDTO-associated proteins demonstrated broad enrichment across synaptic pathways in both the CC and BP ontologies (–log10 Q = 5.7 and 4.2, respectively; Figure 4(a)–(d)), as indicated by the red central node in the CC map and the purple central node in the BP map. Within the CC ontology, proteins were enriched across both pre- and postsynaptic compartments, with a bias towards presynaptic components (–log10 Q = 6.4; Figure 4(a)). Notably, substructural enrichment was observed for synaptic vesicles (SVs) and related machinery (Figure 4(b)). Seven proteins were identified within this vesicle-enriched category: amphiphysin (AMPH), ATPase H+ transporting V1 subunit A (ATP6V1A), clathrin heavy chain (CLTC), Parkinsonism-associated deglycase (PARK7), Ras-related proteins (RAB14 and RAB2A), and synapsin-1 (SYN1). Of these, six (AMPH, ATP6V1A, CLTC, RAB14, RAB2A, SYN1) localized to the synaptic vesicle membrane, and four (AMPH, ATP6V1A, CLTC, SYN1) were specifically annotated to the extrinsic component of that membrane.
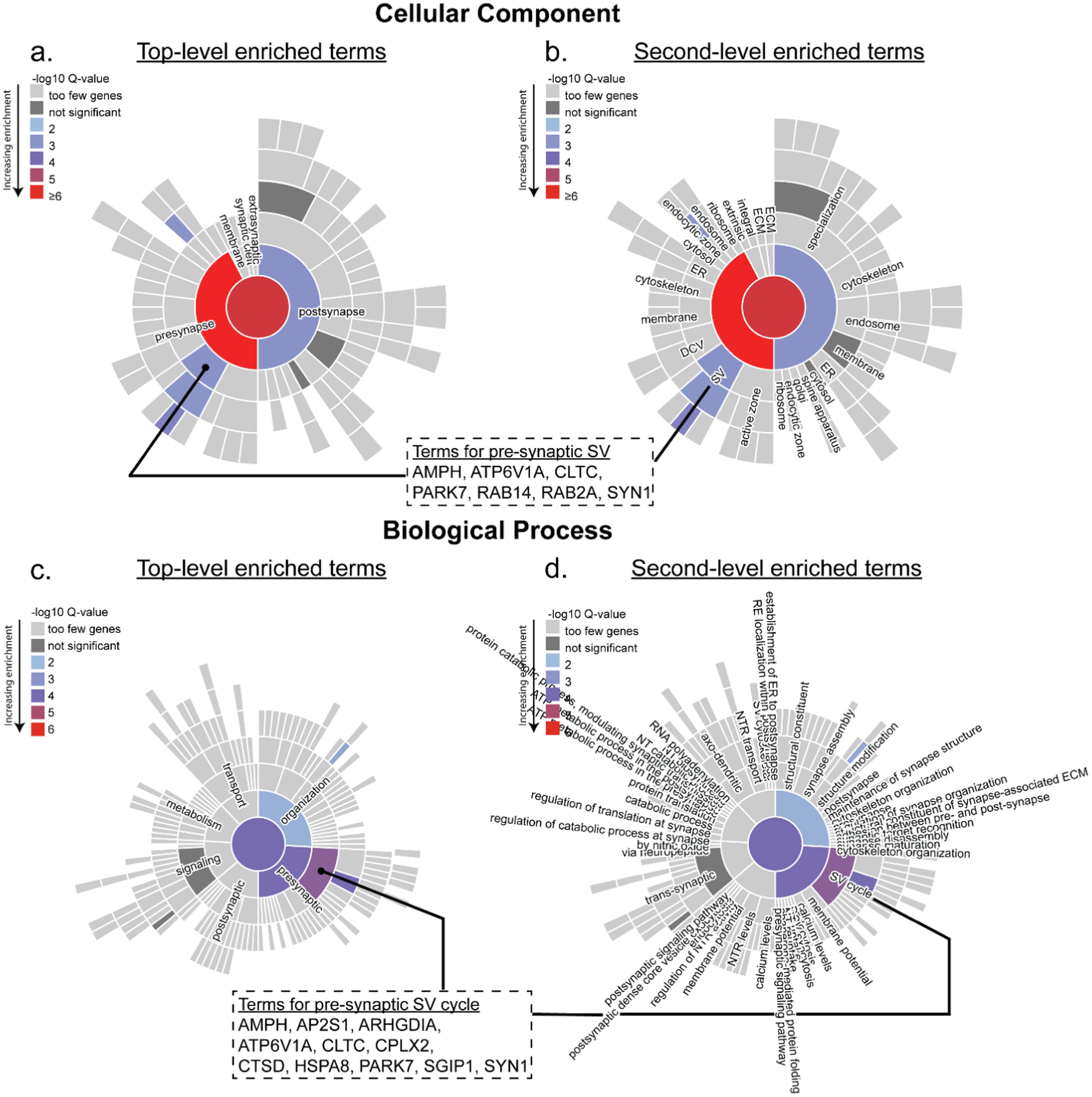
Proteins within the PART BDTO interactome are enriched in presynaptic components and synaptic vesicle-associated processes. BDTO were co-immunoprecipitated (antibody: T18) from PBS-soluble fractions of PART hippocampal autopsy tissue (
BP ontology analysis revealed significant enrichment in processes related to presynaptic vesicle cycling and synaptic terminal organization, with vesicle cycling exhibiting the strongest enrichment (Figure 4c,d). Eleven proteins were associated with this pathway, including previously noted AMPH, ATP6V1A, CLTC, PARK7, and SYN1, along with adaptor-related protein complex 2 subunit sigma 1 (AP2S1), Rho GDP dissociation inhibitor alpha (ARHGDIA), complexin-2 (CPLX2), cathepsin D (CTSD), heat shock protein 8 (HSPA8), and SH3GL interacting endocytic adaptor 1 (SGIP1). Among these, six (AMPH, AP2S1, CLTC, HSPA8, PARK7, SGIP1) were specifically associated with synaptic vesicle endocytosis. Together, these findings define the molecular landscape of the PART BDTO interactome and highlight preferential association with presynaptic compartments and synaptic vesicle-associated processes, particularly those involved in vesicle cycling and endocytosis.
To corroborate these proteomic findings at the ultrastructural level, we performed EM immunogold labeling on murine synaptosomes acutely challenged with rtauO. Synaptosomes were derived from the temporal cortex of WT mice to minimize off-target detection of endogenous human tau and to leverage prior demonstrations of conserved rtauO engagement profiles across rodent and human synapses. 29 Samples were labeled with the oligomer-selective T22 antibody to determine the subcellular localization of rtauO. In vehicle-treated synaptosomes, minimal immunogold labeling was detected, consistent with background signal (Supplemental Figure 8a–(d)). In contrast, synaptosomes exposed to rtauO exhibited robust labeling, with immunogold particles distributed along vesicle-rich zones and presynaptic membranes (Supplemental Figure 8e–(h)).
Discussion
Synapses are early and selectively vulnerable targets in tauopathies, prone to both tau accumulation and functional impairment; yet the molecular mechanisms underlying this vulnerability, particularly in relation to soluble tauO, remain poorly understood. Notably, whether tauO preferentially associate with specific synaptic compartments (e.g., presynaptic versus postsynaptic) or functional subtypes (e.g., excitatory versus inhibitory), and how such targeting might influence synaptic function is especially unclear. In this study, we implemented a novel flow cytometry–based immunophenotyping panel for human synaptosomes, which, in combination with electrophysiological assessment of ligand-gated synaptic responses, enabled us to delineate the vulnerability of specific synaptic compartments and functional subtypes to tauO. To anchor our findings in clinical biology, we profiled the synaptic interactome of BDTO isolated from PBS-soluble hippocampal lysates of PART autopsy specimens as a surrogate for early-stage AD, thus capturing native binding partners of soluble tauO in the absence of Aβ pathology. Our results reveal a convergent pattern in which tauO a) preferentially engage presynaptic and inhibitory synapses, b) selectively potentiate synaptic GABAergic responses, and c) interact with a molecular network enriched in presynaptic proteins involved in synaptic vesicle dynamics. Collectively, these findings provide key insights into how tauO selectively engage and modulate human synapses, advancing our understanding of synapse-specific vulnerability in early tauopathy.
The present study introduces a novel experimental strategy enabling direct, real-time assessment of tauO engagement and its functional impact at native human synapses—offering a new framework for linking these interactions with synaptic outcomes in human tissue. To accurately contextualize our findings within the existing literature, we performed a systematic PubMed search using defined Boolean operators (see
Pre- versus postsynaptic vulnerability to tauO
Using multiplexed FC immunophenotyping, we found that tauO engage both pre- and postsynaptic compartments in human synapses, yet with a notable preference for presynaptic association. This asymmetry was further reflected in our proteomic analysis of the PART BDTO interactome, which showed greater enrichment of presynaptic proteins, particularly those related to synaptic vesicle cycling (e.g., components of the vesicle docking and fusion machinery and neurotransmitter release), as compared to postsynaptic elements. EM immunogold labeling further confirmed rtauO localization to presynaptic, vesicle-containing structures.
Our findings are consistent with numerous prior studies utilizing
Mechanistic studies in tauopathy models have illuminated how tau might perturb presynaptic function. In Drosophila and rodent models, tau accumulates in presynaptic terminals and binds synaptic vesicles via its N-terminal domain, impairing vesicle mobility and neurotransmission.
61
This interaction disrupts vesicle mobility and impairs neurotransmitter release, mirroring the role of Synapsins, which also tether vesicles to the actin cytoskeleton to maintain reserve pools.
62
Consistent with this, Synapsin-1 emerged as a prominent interactor in our PART BDTO dataset, potentially reflecting a shared mechanistic pathway. Collectively, converging evidence from human
Excitatory versus inhibitory synaptic vulnerability to tauO
FC immunophenotyping further revealed that rtauO preferentially engage GABAergic more than glutamatergic synapses. Complementary electrophysiological recordings from MSM experiments showed a selective enhancement of GABAAR currents following acute rtauO exposure, with no corresponding change in kainate-mediated AMPAR responses. Such preferential engagement of inhibitory synapses contrasts intriguingly with the predominant narrative in AD pathology, which has frequently emphasized heightened vulnerability of excitatory neurons to NFT accumulation and overt degeneration.20,59,63–65 However, prior investigations into selective neuronal vulnerability have predominantly focused on AD, inherently confounded by co-existing Aβ pathology (Table 1). It is well established that Aβ pathology independently promotes neuronal hyperexcitability,66–68 complicating interpretations regarding tau-specific vulnerability.
Given conflicting evidence regarding tau's role in neural excitability,68,69 our findings underscore the need to disentangle tau pathology from Aβ effects at human synapses. Recent evidence suggests that tau, particularly in its soluble form, contributes to neural network hypoexcitability; several studies in tau transgenic models observe reduced hippocampal excitability70–72 as well as cortical hypoactivity.73–75 For instance, Busche et al. (2019) illustrated neuronal hypoactivity in rTg21221 mice, which express non-fibrillar wild-type human tau at levels comparable to NFT-forming models (rTg4510), reinforcing the notion that soluble tau alone can drive significant neuronal dysfunction independent of overt NFT formation. 73
Focusing on the role of tauO, we recently found that aggregation-prone tau species—particularly tauO and PHF-tau—were associated with a reduced synaptic E/I ratio in PART autopsy brain specimens. 21 These observations are further supported by studies performed in human-derived model systems, wherein iPSC-derived neuronal cultures seeded with rtauO exhibited an increase in basal GABA release, while glutamate release remained unaffected. 52 Likewise, treatment of organotypic slice cultures with oligomeric tau generated from the pro-aggregant TauRDΔ construct led to increased inhibitory filipodia density, which selectively innervate interneurons. 76
Suggestive mechanistic implications of selective synaptic vulnerability to tauO
Our findings, in conjunction with prior literature, offer novel insights into potential mechanisms underlying selective vulnerability of human synapses to tauO. We speculate two non-mutually exclusive mechanistic frameworks: (1) tauO promote presynaptic GABA release, leading to increased postsynaptic GABAAR activity, or (2) tauO engage presynaptic terminals—both GABAergic and glutamatergic—without necessarily promoting vesicle release, and instead modulate GABAAR function, perhaps directly (e.g., conformational or allosteric mechanisms) or indirectly (e.g., receptor clustering via gephyrin stabilization). While these models remain conjectural and necessitate further empirical validation, our data favor the latter scenario.
In our MSM electrophysiology paradigm, synaptosomes were pretreated with rtauO, thoroughly washed, and microtransplanted into
Independent studies reinforce this hypothesis. Hill et al. (2019) elegantly demonstrated that rtauO, derived from the same full-length (2N4R) tau construct utilized in the current study, injected selectively into the presynaptic compartment led to a marked reduction in synaptic strength—an effect not observed with postsynaptic delivery. 9 Unlike classical inhibition of neurotransmitter release (e.g., mediated by receptors such as adenosine A1 or GABAB), which typically reduces release probability and thereby diminishes synaptic depression across a train of excitatory postsynaptic potentials (EPSPs), presynaptic rtauO paradoxically increased synaptic depression. 9 One interpretation is that tau, following activity-dependent presynaptic release,15,78,79 may bind to postsynaptic GABAAR in a sustained agonistic or allosteric fashion, leading to prolonged receptor activation. Alternatively, given tau's well established association with microtubules, 80 tauO may modulate GABAAR clustering or trafficking by influencing gephyrin–microtubule dynamics, thereby stabilizing receptor localization at inhibitory synapses.81,82 Of note, these postulated frameworks remain speculative and warrant further mechanistic investigation.
Limitations and future directions
While our study provides novel evidence of the selective vulnerability of human synapses to tauO, several limitations should be noted. First, we cannot definitively determine whether the observed tauO–synapse interactions are inherently pathogenic or reflect physiological roles of tau at the synapse. However, multiple independent investigations support a synaptotoxic role for tauO,7,8 and their absence from synapses in cognitively intact individuals with high NFT burden further reinforces a pathogenic interpretation. 51 Second, although the use of human synaptosomes enhances the translational relevance of our findings, the experimental system remains reductionist. Isolated exposure of synaptosomes to rtauO does not fully recapitulate the complex environment of the human brain, where endogenous tauO may exist in diverse structural conformations and carry disease-relevant posttranslational modifications. Finally, our analyses were limited to synaptosomes from the temporal cortex of Control cases and BDTO from the hippocampus of PART cases; however, susceptibility to tauO may vary across brain regions or disease stages.18,20
Future studies should aim to identify the specific synaptic targets of tauO, including potential interactions with GABAAR subunits and scaffolding proteins such as gephyrin. Proteomic pull-downs and super-resolution imaging may help map these interactions and guide functional validation. It will also be important to examine regional and cell-type specificity, as tauO may display distinct patterns of synaptic engagement across brain areas and neuronal subtypes. Investigating synapses from the locus coeruleus and hippocampus—regions known to be highly vulnerable—as well as from more resilient areas such as the cerebellum, could reveal intrinsic factors underlying selective vulnerability. Additionally, the interplay between tauO and other amyloidogenic proteins—including Aβ, α-synuclein, and TDP-43—remains poorly understood. In AD, for instance, it is unclear whether AβO modulate tauO's synaptic binding profile or vice versa. Co-incubation experiments, combined with the synaptic immunophenotyping platform developed here, could help determine whether and how these oligomers interact to drive synaptic dysfunction and contribute to the transition from early Aβ-driven hyperexcitability to later tau-associated hypoactivity.
Conclusion
In summary, our study provides novel insights into the selective synaptic vulnerability to tau oligomers in human synapses, revealing a pronounced presynaptic and inhibitory bias. Through integrative structural, functional, and proteomic analyses, we delineate specific synaptic compartments and pathways preferentially targeted by oligomeric tau, with particular emphasis on presynaptic vesicle machinery and GABAergic signaling. These findings refine our understanding of tau-driven synaptic dysfunction, help reconcile prior inconsistencies in the literature, and lay the groundwork for future therapeutic strategies aimed at promoting synaptic resilience across the tauopathy spectrum.
Supplemental Material
sj-txt-1-alz-10.1177_13872877261416539 - Supplemental material for Selective vulnerability of human synapses to soluble tau oligomers
Supplemental material, sj-txt-1-alz-10.1177_13872877261416539 for Selective vulnerability of human synapses to soluble tau oligomers by Shrinath Kadamangudi, Laura Sanchez-Sanchez, Rakez Kayed, Agenor Limon and Giulio Taglialatela in Journal of Alzheimer's Disease
Supplemental Material
sj-docx-2-alz-10.1177_13872877261416539 - Supplemental material for Selective vulnerability of human synapses to soluble tau oligomers
Supplemental material, sj-docx-2-alz-10.1177_13872877261416539 for Selective vulnerability of human synapses to soluble tau oligomers by Shrinath Kadamangudi, Laura Sanchez-Sanchez, Rakez Kayed, Agenor Limon and Giulio Taglialatela in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
This work was supported by the NIH/NIA grants F30AG085974 to SK, R21AG089708 to GT, R01AG070255 to AL, R01AG054025 to RK, and R01AG073133 to GT and AL. We extend our sincere gratitude to the donors and families that make these studies possible. We thank Dr Peter Nelson from the University of Kentucky for dissection and neuropathological evaluation of the
Ethical considerations
Postmortem frozen human brain tissues used in this study were obtained through established material transfer agreements with the ADRC at the Sanders-Brown Center on Aging, University of Kentucky. All procedures involving human specimens were conducted in accordance with IRB guidelines at the originating institutions. Donor samples were fully de-identified prior to transfer to the University of Texas Medical Branch (UTMB); therefore, the use of these specimens did not constitute human subjects research and did not require UTMB IRB approval, in accordance with 45 CFR §46.101(a).
Consent to participate
All donors were enrolled in longitudinal brain aging studies at the contributing institutions, and written informed consent for participation and postmortem tissue donation was obtained prior to death.
Consent for publication
Not applicable
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NIH/NIA grants F30AG085974 (SK), R21AG089708 (GT), R01AG070255 (AL), R01AG054025 (RK), R01AG073133 (GT and AL), and P30AG072946 (University of Kentucky).
National Institute on Aging (grant number F30AG085974, R01AG070255, R01AG073133, R21AG089708).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Raw proteomic data used for the ![]() .
44
All other forms of raw data will be provided by the authors upon reasonable request.
.
44
All other forms of raw data will be provided by the authors upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
