Abstract
Background
Emerging studies implicate the microtubule-associated protein tau as a key modulator of neuronal excitability and synaptic dysfunction in human tauopathies. How distinct tau forms influence synaptic excitability across brain regions with differing susceptibility to tau accumulation remains unclear. Primary age-related tauopathy (PART), defined by hippocampal-restricted tau pathology in the absence of amyloid-β, offers a tractable model to investigate tau-specific effects on synaptic physiology.
Objective
To determine how regionally enriched tau species in PART relate to synaptic excitation–inhibition balance and to identify molecular pathways linking tau oligomers to synaptic dysfunction.
Methods
Autopsy-derived hippocampal and superior middle temporal gyrus tissues from neuropathologically validated PART specimens were analyzed. Tau species, including monomers, oligomers, and paired helical filaments (PHFs), were quantified by western blot. Synaptic function was assessed by microtransplantation of synaptosomal membranes into Xenopus laevis oocytes, followed by electrophysiological recordings of glutamatergic (kainate-evoked AMPAR) and GABAergic (GABAAR) currents to calculate the synaptic excitation-to-inhibition (sE/I) ratio. Proteomic and enrichment analyses of brain-derived tau oligomer (BDTO) interactomes from PART hippocampi were performed.
Results
PART specimens showed hippocampal accumulation of aggregation-prone tau assemblies (oligomeric and PHF-tau) that were negatively correlated with sE/I. Proteins within the BDTO interactome linked to reduced sE/I were enriched for pathways related to vesicle-mediated transport, synaptic endocytosis, and neurotransmitter receptor regulation.
Conclusions
In PART, oligomeric and fibrillar tau are associated with shift toward synaptic inhibition, predominantly within the hippocampus. Proteomic correlates implicate vesicle trafficking pathways as mediators of tau oligomer-associated alterations in synaptic function, providing mechanistic insight into early-stage tauopathy.
Keywords
Introduction
Tau is a microtubule-associated protein best known for supporting axonal transport and maintaining cytoskeletal integrity, though it is now understood to participate in a broader array of cellular functions.1–3 Under pathological conditions, tau misfolds and aggregates into soluble oligomers, paired helical filaments (PHFs), and eventually neurofibrillary tangles (NFTs); such lesions define tauopathies, with Alzheimer's disease (AD) being the most common.4–6 Among the two pathological hallmarks of AD, namely extracellular amyloid-β (Aβ) plaques and intracellular NFTs, the burden and regional distribution of tau pathology correlates with cognitive decline best.7–11 This association has prompted growing interest in tau's contribution to synaptic dysfunction, as mounting evidence implicates pathological tau in both synaptic impairment, as well as trans-synaptic propagation of pathology, positioning the synapse as a central locus of vulnerability in tau-mediated neurodegeneration.12–18
Synaptic dysfunction often precedes overt neurodegeneration and is a key driver of cognitive decline in tauopathies.17,19–22 Of particular interest is the balance between excitatory and inhibitory (E/I) signaling at the synaptic interface, which is critical for maintaining neural circuit stability.21,23–27 While E/I balance typically describes large-scale network dynamics, here we use the synaptic E/I ratio (sE/I)—a quantifiable measure of receptor-mediated excitatory and inhibitory responses—as a proxy for detecting synaptic changes that may underlie or reflect broader circuit-level shifts. In AD, Aβ pathology has been consistently shown to perturb this balance by promoting neuronal hyperexcitability.24,28–32; however, the role of tau in modulating neuronal excitability, particularly at the synaptic interface, remains less clearly defined.33,34
Primary age-related tauopathy (PART), a common tauopathy in older adults, offers a compelling model to disentangle tau-specific effects from those of mixed pathology. Neuropathological assessments of PART reveal that tau pathology, comprising hyperphosphorylated tau and NFTs, is largely confined to the hippocampus and entorhinal cortex, without extending into neocortical regions as observed in advanced AD.9,35,36 In this manner, PART occupies the early stages of Braak progression.,37–39 and, unlike other primary tauopathies such as frontotemporal dementia or progressive supranuclear palsy, recapitulates the early anatomical distribution of AD tau pathology while remaining devoid of Aβ. Importantly, we recently demonstrated that PART-derived synaptosomes are completely devoid of Aβ pathology. 16 Thus, PART provides a uniquely positioned, human-centric model to investigate tau-driven synaptic mechanisms independent of Aβ. Here, we leveraged this opportunity to address key knowledge gaps, including how distinct tau species differentially associate with excitatory and inhibitory signaling, whether these effects are region-specific, and which molecular pathways underlie the selective vulnerability associated with tau-mediated synaptic dysfunction.
To address these gaps, we examined how physiologically and pathologically relevant tau species, namely monomeric tau, hyperphosphorylated tau (p-tau), PHF-tau, and soluble tau oligomers, relate to synaptic physiology in PART cases, with particular attention to regional vulnerability. Using human autopsy brain specimens, we analyzed the hippocampus and superior middle temporal gyrus (SMTG)—two regions that differ in their susceptibility to pathological tau accumulation.9,37,40 Tau density was quantified via immunoblotting, and synaptic function was assessed by microtransplantation of synaptosomes into Xenopus laevis oocytes, with excitation, inhibition, and sE/I calculated from receptor-mediated responses. We found that aggregation-prone tau assemblies, particularly tau oligomers and PHF-tau, were associated with reduced sE/I in the hippocampus of PART cases. Given their putative role as precursors to fibrillar aggregates and established synaptotoxicity, tau oligomers were prioritized as a candidate mediator to investigate the molecular link between pathological tau and reduced sE/I. Proteomic profiling further identified vesicle trafficking proteins within the PART brain-derived tau oligomer (BDTO) interactome as candidate modulators of this shift, suggesting a mechanistic link between soluble tau oligomers and altered synaptic signaling.
Methods
Clinical and neuropathological characterization of human brain specimens
Human postmortem brain specimens utilized in this investigation were provided by the Alzheimer's Disease Research Center (ADRC) at the Sanders-Brown Center on Aging, University of Kentucky, through established material transfer agreements. Participants were part of ongoing longitudinal studies focused on brain aging, and informed consent was obtained from individuals prior to enrollment. All experimental procedures conformed to the Institutional Review Board (IRB) requirements of the respective institutions.
Participants received thorough annual evaluations conducted by clinicians experienced in neurological and cognitive assessments, including administration of the Clinical Dementia Rating (CDR) scale and Mini-Mental State Examination (MMSE). Expert neuropathologists conducted standardized neuropathological examinations, assessing AD pathology through criteria set forth by the Consortium to Establish a Registry for Alzheimer's Disease (CERAD) and Braak staging (0 – VI) to quantify Aβ plaque deposition and NFT pathology. PART cases were defined as having mild-to-moderate tau pathology predominantly within the hippocampus without concurrent Aβ plaques.41–43 Notably, we have recently demonstrated that these same PART specimens lack detectable Aβ pathology in both the PBS-soluble total homogenate 44 (Supplemental Figure 2) and synaptosome fractions 16 (Figure 1) by western blot analysis.
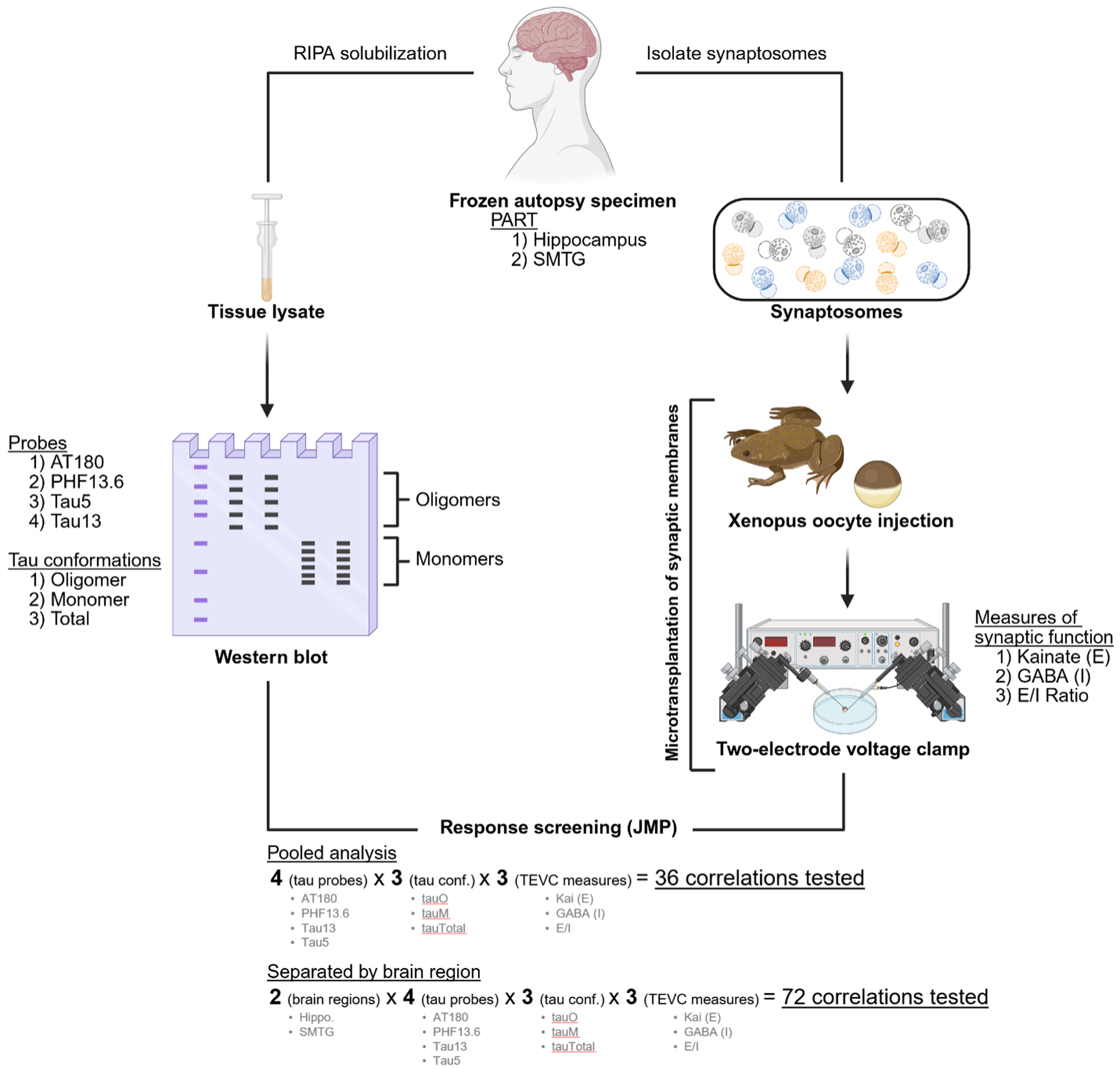
Schematic of experimental design and techniques. Overview of the experimental workflow used to investigate associations between tau pathology and synaptic function in PART brain specimens. Frozen autopsy-derived hippocampal and superior middle temporal gyrus (SMTG) samples were processed for either synaptosome isolation (right pathway) or tissue lysate preparation (left pathway).
Brain specimens were anonymized prior to transfer to the University of Texas Medical Branch (UTMB), thus exempting the use of these tissues from additional IRB oversight under CFR §46.101(a)(1). Relevant demographic, clinical, and histopathological information for all subjects are summarized in Supplemental Table 1.
Western blot analysis of tau
Fresh frozen brain tissue specimens (25 mg) from the hippocampus and SMTG of PART (n = 7 per region) underwent western blot analysis to evaluate tau pathology, employing methods previously validated by our group.16,45,46 Provided here is a brief overview of key steps in this analysis. Low retention plasticware, including tips and tubes, was used to minimize sample loss. Tissues were homogenized in ice-cold RIPA buffer (Cell Signaling Technology, cat# 9806) supplemented with protease (SigmaAldrich, cat# P8340) and phosphatase inhibitor (Thermo Fisher Scientific, cat# PI78426) cocktails, using 15–20 strokes with a dounce homogenizer. Homogenates were vortexed every 5 min over a 30-min period on ice to ensure thorough lysis. Supernatants containing soluble protein fractions were collected, and protein concentrations were quantified using the Pierce™ BCA Protein Assay Kit (Thermo Fisher Scientific, cat# 23227). Aliquots of these soluble fractions were stored at −80°C for subsequent analyses.
Protein lysates (15 μg/sample) were prepared for electrophoresis by combining samples with 1X NuPAGE™ LDS Sample Buffer and 1X NuPAGE™ Sample Reducing Agent (Thermo Fisher Scientific), followed by heat denaturation at 75°C for 10 min. Proteins were separated by electrophoresis using pre-cast NuPAGE 4–12% Bis-Tris gels (Thermo Fisher Scientific, cat# NP0323BOX) and transferred onto nitrocellulose membranes (Cytiva, cat# 10600001) via wet-tank transfer at 100 V for 1 h at 4°C. Membranes were blocked in Intercept® (TBS) Blocking Buffer (LI-COR Biosciences, cat# 927-60001) for 1 h at room temperature and incubated overnight at 4°C with primary antibodies diluted in Intercept® T20 (TBS) Antibody Diluent (LI-COR, cat# 927-65001). IRDye-conjugated secondary antibodies (LI-COR Biosciences; 680RD anti-mouse, cat# 926-68070; 800CW anti-rabbit, cat# 926-32211; both 1:10,000 dilution) were subsequently applied for 1 h at room temperature. Membranes were washed three times for 10 min each with TBS-T (0.1% Tween-20 in TBS) and visualized using the Odyssey LI-COR Classic Infrared imaging system.
Western blot analyses employed four distinct tau antibodies: AT180 (Thermo Fisher Scientific, cat# MN1040), PHF13.6 (Thermo Fisher Scientific, cat# 35-5300), Tau13 (BioLegend, cat# 835201), and Tau5 (BioLegend, cat# 806401). Each antibody targets a unique epitope spanning a different region of the tau protein, as illustrated in Supplemental Figure 1. Together, these antibodies enabled comprehensive detection of multiple structural and pathological states of tau. The phosphorylation-specific antibody AT180 detects tau phosphorylated at Thr231 within the proline-rich region, 47 associated with early pathological tau modifications.48–50 PHF13.6 specifically recognizes tau phosphorylated at Ser396, indicative of intermediate to advanced pathological tau aggregation stages.48,50,51 Pan-tau antibodies Tau13 and Tau5 target epitopes within residues 2–1852–55 and 210–241,52,54,56,57 respectively, and detect all six tau isoforms agnostic to phosphorylation or aggregation status.
To validate ensure reliable detection of native tau forms, including oligomeric aggregates, we conducted a series of optimization experiments using RIPA-soluble frontal homogenates from Control (Braak stage 0) and AD (Braak stage VI) cases (Supplemental Figure 2). We systematically tested the effects of denaturing conditions, namely heating and reduction (via dithiothreitol, DTT), on protein separation and detection. Blots were probed with Tau5, β-tubulin, and β-actin. Combined heat denaturation and reduction yielded the clearest separation of tau monomers from higher-order oligomers, thereby enabling quantitation of each species, while also minimizing nonspecific smear bands (particularly evident for β-tubulin; Supplemental Figure 2a). These conditions also preserved proper migration of β-actin, supporting their use in subsequent immunoblotting. Importantly, cryo-EM studies of AD tau filaments have shown that cysteine residues (Cys291 and Cys322) lie outside the ordered fibril core and are not required for structural stability, which explains why reducing conditions do not disrupt the detection of amyloidogenic tau aggregates. 58 To further validate detection fidelity, we evaluated linearity of signal for all tau antibodies used in the study (AT180, PHF13.6, Tau13, and Tau5) under optimized conditions. AD homogenates were loaded at two protein concentrations (15 µg and 20 µg) and probed with each antibody. Quantification confirmed a proportional signal increase corresponding to the 1.33× difference in input, demonstrating that detection remained within a linear range across the selected input levels (Supplemental Figure 2b).
Quantification of tau density was performed by applying defined areas of quantification (AOQ) to western blots, delineating monomeric (M), oligomeric (O), and total (T) tau bands. For clarity in data interpretation, each signal was labeled using the antibody clone name followed by the tau aggregation profile (e.g., AT180-M, AT180-O, AT180-T). Band intensities were quantified using ImageJ software (NIH) and normalized to β-actin to control for loading variability. For phosphorylated tau species (AT180, PHF13.6), values were additionally normalized to total tau levels (Tau5 or Tau13).
Assessment of synaptic function via microtransplantation of synaptic membranes
The protocol for synaptosome isolation, including associated characterization and quality control measures, has been optimized and validated in our previous work.16,25,45,59,60 Likewise, all procedures involved in the microtransplantation of synaptic membranes (MSM) into Xenopus laevis oocytes for functional analyses also have been systematically validated previously.25,61–64 A brief overview of key methodological steps for these procedures is provided below.
Synaptosome enriched preparations were isolated from 25 mg of fresh-frozen SMTG and hippocampus brain tissue by gently homogenizing the sample in Syn-PER buffer (ThermoFisher, cat# 87793) containing an EDTA free protease inhibitor (Thermo Fisher Scientific, cat# 78425). Following homogenization, samples were pelleted at 1200 g for 10 min at 4°C. The supernatant, containing the synaptosomal enriched fraction (P2), was collected and pelleted by centrifugation at 15,000 g for 20 min at 4°C. Resulting P2 fractions were resuspended in Syn-PER and underwent protein quantification via fluorometry using a DeNovix QFX unit (DeNovix, New Castle, DE, USA).
Oocytes were extracted from Xenopus laevis frogs in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals, and the Institutional Animal Care and Use Committee (IACUC) at UTMB (IACUC:1803024). Briefly, frogs were anesthetized by immersion in 0.17% MS-222 bath, euthanized and the ovaries were surgically removed. Oocytes were isolated and defolliculated by gentle rotation in 1x Barth's solution (SBS) (Ecocyte BioScience Inc, Dortmund, Germany) to which was added 2 mg/mL collagenase (Sigma, Sant Louis, MO, USA) at 30°C for 4 h. Healthy oocytes, upon inspection via light microscopy, were manually selected and stored at 16°C in fresh SBS until injection of synaptosomal enriched preparations.
Needles for microinjection were prepared by pulling 3.5 in. Borosilicate tubes (Drummond Sci., PA, USA) using a micropipette puller P-1000 (Sutter instruments, Novato, CA, USA). To ensure consistent sample loading, needle tips were trimmed to a final length of 4 mm. Prior to injection, oocytes of stages V and VI were individually selected and placed in 1x Barth's solution (SBS).65,66 For each sample, a new cut needle was used and filled with 0.5 mL/cc of mineral oil and placed in the nanoinjector. Before loading, synaptosomal preparations were adjusted to a final concentration of 2 ng/nl and sonicated in ice-cold water 3 × 5 s using a FS20D ultrasonic cleaner (Thermo Fisher Scientific, Walthman, MA, USA) with a 1-min cool down on ice. Oocytes were placed in a glass Petri dish containing a nylon mesh and penetrated underneath the surface, injecting 50.6 nl of preparation per oocyte using the Nanoject II system (Drummon Sci., PA, USA). 63 Following injection, oocytes were maintained in fresh 1x SBS at 16°C in a 96-well conical bottom plate (Thermo Fisher Scientific, Waltham, MA, USA).
γ-aminobutyric acid (GABA) (Sigma -Aldrich, St Louis, MO, USA) and kainate (Tocris Bioscience, Bristol, UK) were prepared as stock solutions in DI water and stored at −20°C until use. As previously demonstrated, only microtransplanted oocytes generated measurable electrophysiological responses to these agonists, with GABA selectively activating GABAA receptors (GABAAR) and kainate selectively activating AMPA receptors (AMPAR).
61
Furthermore, kainate was selected to activate AMPAR, given its capacity to suppress desensitization and its minimal activation of endogenous kainate receptors in conditions suitable for the two-electrode voltage clamp (TEVC) experiments.
61
Experimental solutions were prepared at 1 mM and 100 µM final concentrations in 1x Customized NFR without Mg solution (i.e., Ringer's solution) (Ecocyte Bioscience Inc, Dortmund, Germany). Ion currents were recorded using Roboocyte2 system and consumable TEVC measuring heads. Measuring head glass needles were filled with 3 M KCl (Thermo Fisher Scientific, Waltham, MA, USA) and ready-to-use Ag/Cl-treated silver wires were placed inside. All Roboocyte2 products were purchased in Multi-Channel Systems, MCS GmbH, (Reutlingen, Germany). For TEVC, electrode resistance was set between 200 and 1000 kOhm, and minimal resting membrane potential per oocyte was ensured to be under −15 mV. Peristaltic pump velocity for drug application was set at 5000 µl/min. Holding voltage was maintained at −80 mV throughout recording. The application and recording protocol were scripted using JavaScript and can be found at https://github.com/LimonLab-UTMB. Drift correction, baseline adjustment, and peak measurements were performed using Roboocyte2 + software (Multi Channel Systems, MCS GmbH, Reutlingen, Germany). Synaptic excitation-to-inhibition (sE/I) ratio was determined within individual oocytes as follows:
Statistical analysis
All statistical analyses were performed in JMP Pro 18 (SAS Institute Inc., Cary, NC). Continuous variables derived from western blot and TEVC data were assessed for normality using Shapiro–Wilk tests. Outliers were identified using the interquartile range (IQR) method with tail quantiles set at 0.25 and a multiplier (Q) of 1.5. Data points outside this quantile range were excluded from subsequent analyses (Supplemental Table 2).
Regional comparisons between the hippocampus and SMTG were carried out using paired t-tests if data met the assumptions of normal distribution and equal variance. When such assumptions were violated, the Wilcoxon signed-rank test was employed. All t-tests were performed using the Y-by-X module in JMP.
Correlations between measures of tau pathology (AT180, PHF13.6, Tau13, and Tau5; delineated as tau oligomers, tau monomers, and total tau) and variables of synaptic function (kainate-mediated excitatory [E] currents, GABA-mediated inhibitory [I] currents, and the excitation-to-inhibition [sE/I] ratio) were evaluated using Pearson correlation for normally distributed data or Spearman's rank correlation under non-parametric conditions. Correlations were performed with hippocampus and SMTG data initially pooled (panels a–b), followed by region-specific analyses (panels c–d) to evaluate whether associations were driven by one region over the other. Potential confounding variables including age at death, postmortem interval (PMI), and sex were assessed for correlations with synaptic function; none of these variables demonstrated significant correlations with synaptic function metrics; hence, they were not included as covariates in further analyses (Supplemental Table 4). Multivariate correlation methods were initially utilized and subsequently confirmed using the response screening function in JMP. We implemented an a priori criterion, whereby correlations with fewer than six data points would be excluded to ensure reliability across all analyses.
A significance threshold of p < 0.05 (two-tailed) was consistently applied for all statistical tests. Detailed descriptive statistics, confidence intervals, test statistics, p-values, and additional relevant metrics for each analysis are provided in the Supplemental Information (Supplemental Tables 2–5).
Assessment of synaptic interactome of PART BDTO
Detailed methodologies employed for isolation of brain-derived tau oligomers (BDTO) from PART autopsy specimen, proteomic analyses via liquid chromatography-tandem mass spectrometry (LC-MS/MS), and bioinformatic enrichment analyses have been outlined previously.16,25,64,67,68 The BDTO interactome dataset analyzed here was originally published as part of a broader study, which details quality control and validation procedures, and includes the raw proteomic data. 44 Importantly, while the raw proteomic data were drawn from the prior study, all analyses presented here are entirely novel and were conducted independently for the purposes of this investigation. A summary of the key methodological steps used for BDTO analysis is provided below.
PBS-soluble homogenates were prepared from fresh frozen hippocampal autopsy tissues (75 mg) from PART cases (n = 4). Tissue was homogenized gently in ice-cold DPBS supplemented with protease and phosphatase inhibitors, followed by centrifugation (9168×g, 10 min, 4°C). Supernatants were immediately utilized or stored at −80°C. BDTO were isolated using co-immunoprecipitation with a tau oligomer-specific antibody (TTC18/T18) coupled to resin.68–71 Detergent-free PBS buffers were used throughout to maintain protein complex integrity. Samples underwent overnight incubation at 4°C, column washing, and elution of BDTO complexes followed by immediate neutralization with Tris buffer.
Eluted BDTO underwent proteomic processing via S-Trap digestion, including protein denaturation, reduction, alkylation, enzymatic digestion (trypsin), and peptide purification. Peptides were subsequently analyzed using nanoLC-MS/MS (Orbitrap Fusion) employing data-independent acquisition (DIA). Data analysis was conducted with MSFragger and DIA-NN software, and spectral libraries were generated from pooled samples.
Linear regression analysis was performed to identify proteins within the PART BDTO interactome that significantly correlated (p < 0.05) negatively with excitatory currents, positively with inhibitory currents, or negatively with the sE/I ratio. Proteins meeting these criteria were selected for further enrichment analyses. Pathway enrichment analysis was conducted using Metascape (Express Analysis), integrating gene ontology (GO) terms and pathways from multiple databases. 72 Protein-protein interaction (PPI) networks of identified sE/I-associated proteins were constructed using the STRING database module within with Metascape web platform. Synaptic functional enrichment was assessed using SynGO (v1.2), employing ‘Stringent’ filtering to robustly annotate synapse-specific biological processes and cellular compartments based on experimentally validated data. 73
Data visualization
All graphs were generated in GraphPad Prism 10.0 (GraphPad Software, La Jolla, California, USA). Figures were prepared with BioRender (BioRender.com) and Adobe Illustrator (Adobe Inc., San Jose, CA, USA).
Results
Regional differences in tau pathology in PART and control brain specimens
To assess whether tau pathology differs between the hippocampus and SMTG in PART, total homogenate tissue lysates from each region were analyzed by western blot using four distinct tau antibodies: AT180, PHF13.6, Tau13, and Tau5. Specific AOQ were applied to delineate oligomeric (O), monomeric (M), and total (T) tau (denoted by red, green, and blue boxes, respectively). Tau forms detected by these probes are referred to by the antibody clone name combined with the corresponding aggregation profile (e.g., AT180-M, AT180-O, AT180-T). Detection and separation of monomeric and oligomeric tau were optimized in preliminary experiments using Control and AD brain homogenates (see Methods, Western blot analysis of tau and Supplemental Figure 2).
Both AT180-O (S = -13, p = 0.03) and AT180-T (S = -12, p = 0.04) were significantly elevated in the hippocampus compared to the SMTG, whereas AT180-M showed no significant regional variation (Figure 2(e)). PHF13.6 immunoreactivity exhibited consistent hippocampal enrichment across all tau species, with PHF13.6-O (S = -13, p = 0.03), PHF13.6-M (S = -12, p = 0.04), and PHF13.6-T (S = -13, p = 0.03) all significantly increased in the hippocampus compared to the SMTG (Figure 2(f)). Interestingly, pan-tau markers demonstrated a mixed pattern. Tau5-M was significantly elevated in the SMTG relative to the hippocampus (S = -13, p = 0.03; Figure 2(h)), a finding corroborated by a comparable trend in Tau13-M (t-ratio = 4.49, p = 0.03; Figure 2(g)). However, a key distinction emerged in PART specimens, where a selective increase in Tau5-O levels was detected in the hippocampus relative to the SMTG (t-ratio = −2.18, p = 0.04; Figure 2(h)).
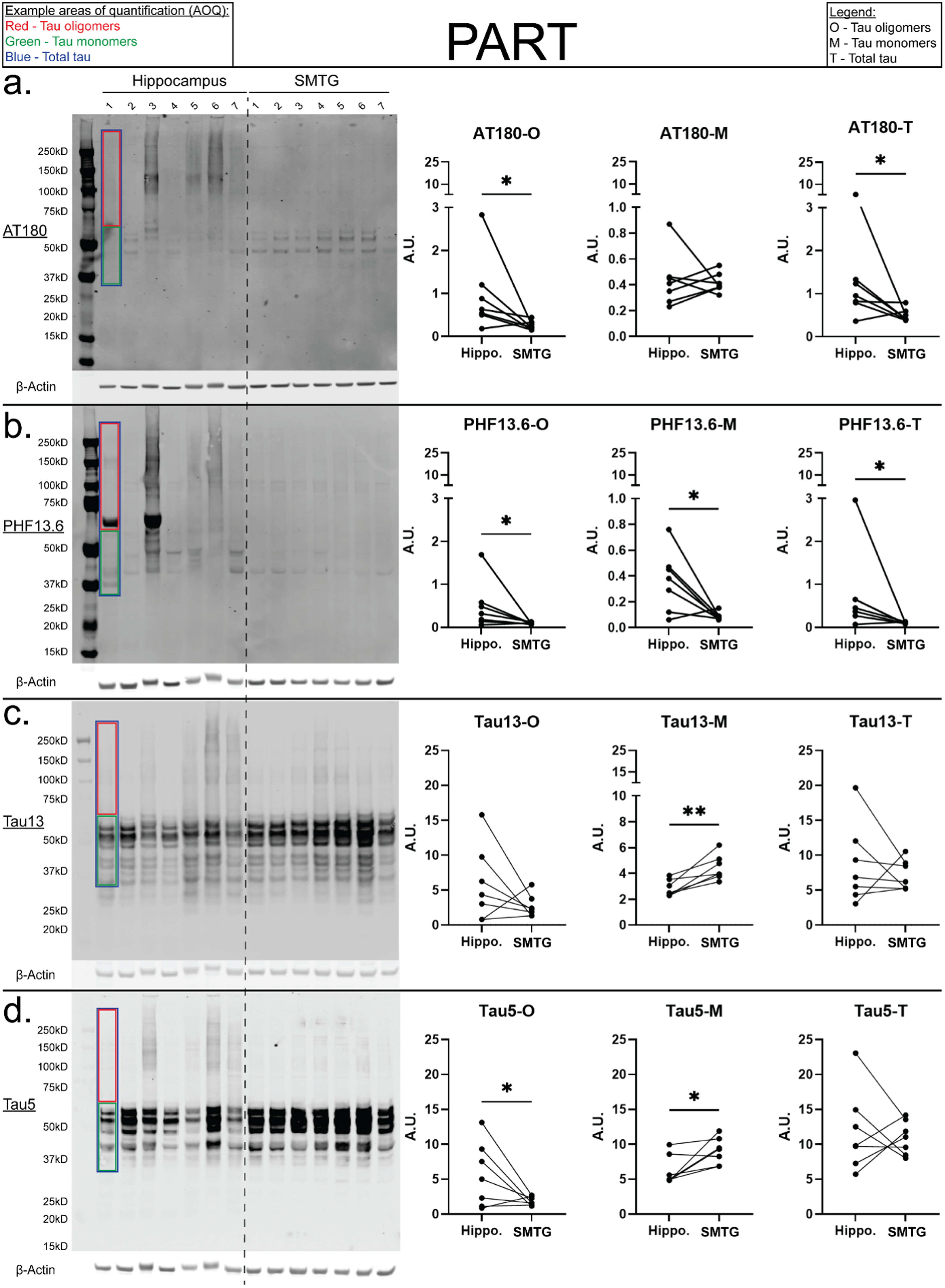
Regional differences in tau pathology in PART brain specimens. Tissue lysates from PART patients (n = 7) were analyzed via western blot to compare tau burden between the hippocampus and SMTG. Each lane (15 μg of protein) represents an individual patient, with paired comparisons across brain regions (lanes 1–7: hippocampus; lanes 8–14: SMTG from the same patient). Tau species were delineated using antibodies against hyperphosphorylated tau (AT180), PHF-tau (PHF13.6), and total tau (Tau13, Tau5). Specific areas of quantification (AOQ) were applied to distinguish tau oligomers (red), monomers (green), and total tau (blue), as outlined in the figure legend. Quantifications (right) display relative abundance for oligomeric (O), monomeric (M), and total (T) tau normalized to β-Actin. Phospho-tau signals (AT180, PHF13.6) were further normalized to total tau (Tau5 or Tau13). Normality was assessed using the Shapiro-Wilk test (Supplemental Table 3), and statistical significance was determined using paired t-tests or Wilcoxon signed-rank tests (α = 0.05), with *p < 0.05 indicating significant differences. (Color figure available online).
To assess regional differences in synaptic receptor function in PART, hippocampal and SMTG synaptosomes were analyzed via TEVC recordings in Xenopus laevis oocytes microtransplanted with human synaptic receptors. Representative traces illustrate ligand-gated currents evoked by kainate (excitatory, 100 µM) and GABA (inhibitory, 1 mM) application (Figure 3, top). The sE/I ratio was calculated by dividing the peak AMPAR current (evoked by kainate) by the peak GABAAR current recorded from the same microtransplanted oocyte.
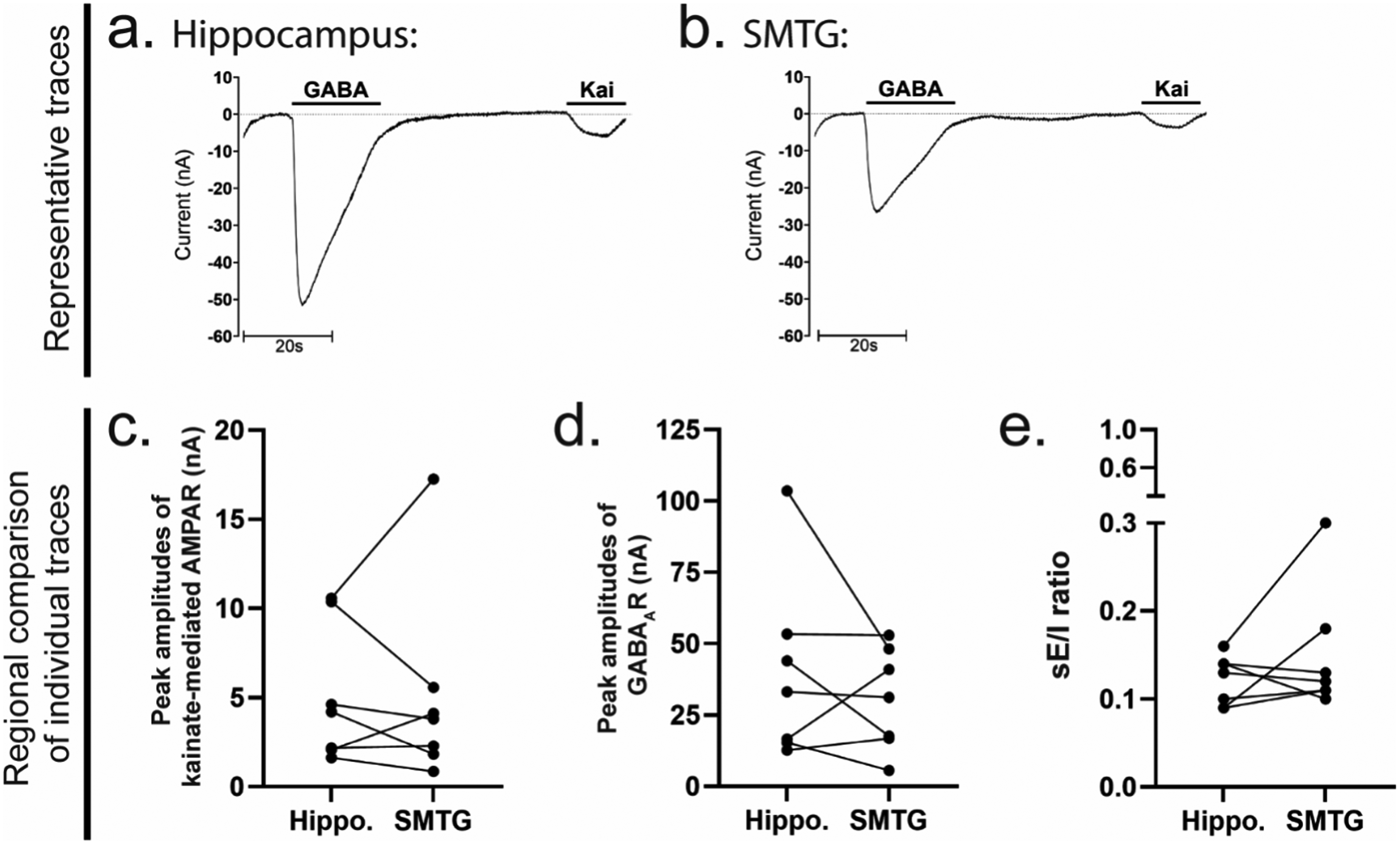
Regional differences in synaptic excitation and inhibition in PART brain specimens. TEVC recordings of glutamatergic and GABAergic synaptic responses in hippocampal and SMTG synaptosomes from PART (f – j; n = 7) patients. (Top) Representative traces depict ligand-gated currents evoked by kainate [100 µM] and GABA [1 mM] in Xenopus oocytes microtransplanted with human synaptic receptors. (Bottom) Quantification of kainate-activated AMPAR and GABA-activated GABAAR currents, along with the sE/I ratio, in hippocampal and SMTG samples. Paired comparisons reflect interregional differences within each patient. Normality was assessed using the Shapiro-Wilk test, two-tailed (Supplemental Table 3), and statistical significance was determined using paired t-tests or Wilcoxon signed-rank tests (α = 0.05), with *p < 0.05 indicating significance.
No significant differences were observed in excitatory or inhibitory currents between hippocampal and SMTG synaptosomes (Figure 3(c),d), and the sE/I ratio remained comparable across regions (Figure 3(e)). Notably, these findings broadly suggest conserved functional responses of glutamatergic and GABAergic synaptic receptor populations across hippocampal and neocortical regions.
Correlation analyses were performed to assess the relationship between tau abundance and synaptic function, specifically excitatory and inhibitory currents, as well as the sE/I ratio, in PART brain specimens (complete list of correlations is provided in Supplemental Table 5). Of note, potential confounding correlates with synaptic function, namely age at death, PMI, and gender, were also evaluated as a function of diagnosis, revealing no significant associations between these variables and measures of synaptic function (Supplemental Table 4). Notably, higher levels of oligomeric and fibril-associated tau were negatively correlated with the sE/I ratio, including Tau5-O (ρ = –0.69, p = 0.01; Figure 4(a)) and PHF13.6-M (ρ = –0.60, p = 0.03; Figure 4(b)). No significant correlations were found between any given tau species and excitatory or inhibitory currents individually; rather, significant relationships were exclusive to the sE/I ratio (Supplemental Table 5).
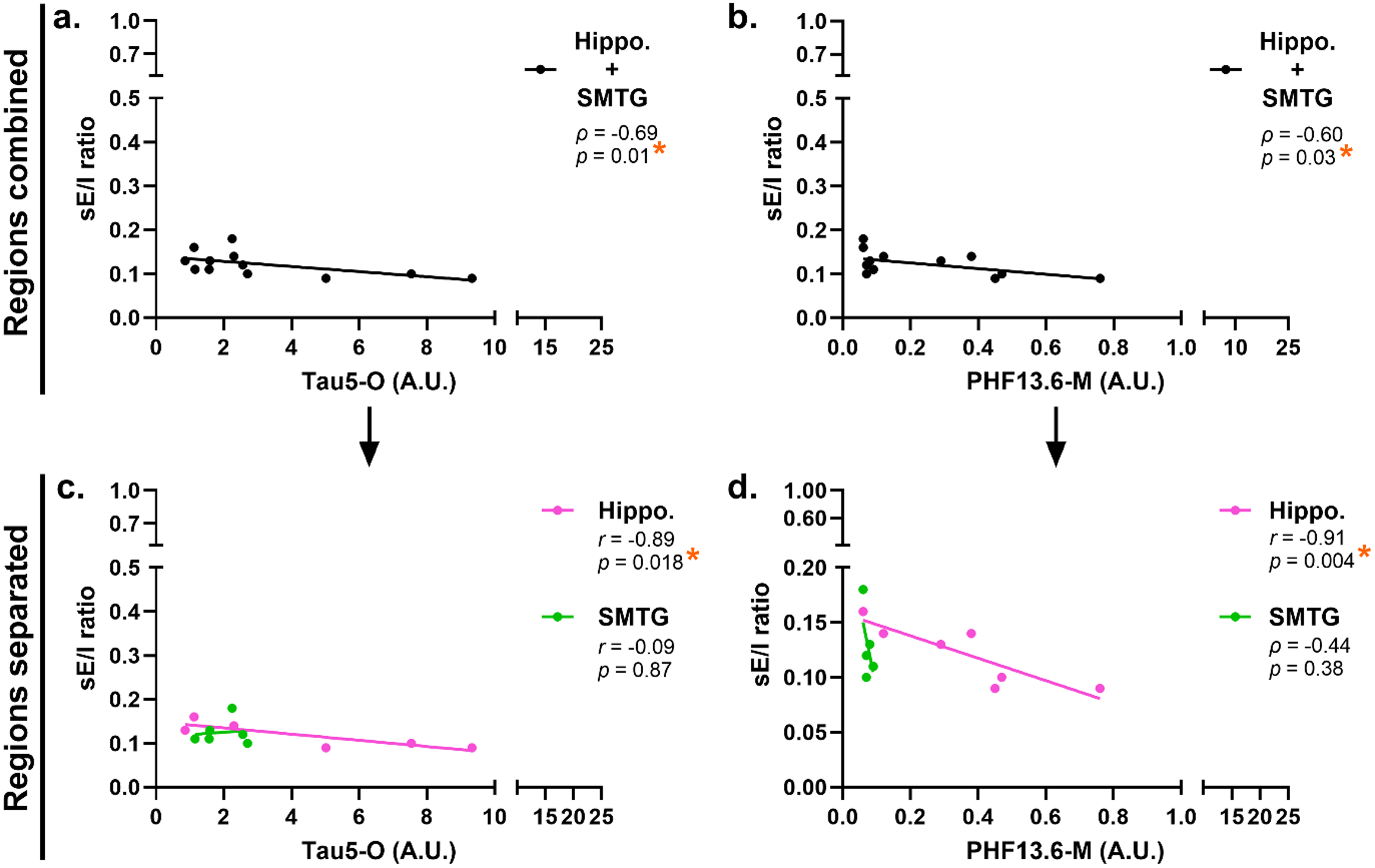
Associations between tau and synaptic function in PART. Correlation analyses were conducted to assess the relationship between tau burden and sE/I ratio in PART cases.
To assess regional specificity, correlations were repeated separately for hippocampal and SMTG samples (complete list of correlations is provided in Supplemental Table 5). Relevant confounding variables remained non-significant across regions and diagnoses (Supplemental Table 4). Of note, negative correlations between aggregation-prone tau assemblies and the sE/I ratio were observed exclusively in the hippocampus, including Tau5-O (r = –0.89, p = 0.018; Figure 4(c)) and PHF13.6-M (r = –0.91, p = 0.004; Figure 4(d)). These relationships were not detected in the SMTG (Figure 4(c), (d)). Again, no significant associations were observed with individual excitatory or inhibitory current amplitudes (Supplemental Table 5). Together, these findings suggest that the observed synaptic vulnerability is region-specific and primarily driven by the hippocampus.
Motivated by our finding that proteopathic tau were selectively associated with diminished sE/I in the hippocampus of PART (Figure 4), we sought to elucidate potential cellular and/or molecular mechanisms underlying this alteration in synaptic function. Of note, we focused our investigation on soluble tau oligomers, as substantial evidence suggests these pathological conformations exhibit heightened toxicity, enhanced seeding capacity, and are thought to precede fibrillar tau in the cascade of disease progression.74–78 To identify candidate mediators, pathway enrichment and PPI analyses were performed on proteins within the brain-derived tau oligomer (BDTO) interactome, co-immunoprecipitated from individual hippocampal PART specimens. Tau enrichment was confirmed by MS-based peptide quantification of MAPT in PART BDTOs relative to IgG isotype control eluates (Supplemental Figure 2). Linear regression analysis identified 38 proteins whose abundance either correlated negatively with excitatory currents, positively with inhibitory currents, or negatively with the sE/I ratio (complete protein list provided in Supplemental Table 6).
Pathway enrichment analysis revealed significant enrichment of several biological pathways; the top 20 pathways are displayed in Figure 5(a). Among these, the pathways with the highest enrichment significance (–log10(p) > 6) included vesicle-mediated transport, cellular oxidant detoxification, axon guidance, and cellular catabolic processes, indicating diverse cellular processes potentially influenced by tau oligomers. A PPI network was generated using the STRING database within Metascape (Figure 5(b)), revealing a cluster of proteins primarily involved in synaptic vesicle trafficking and cytoskeletal dynamics. Key proteins within this network included transferrin (TF), synaptojanin 1 (SYNJ1), adaptor-related protein complex 2 subunit mu 1 (AP2M1), and actin-related proteins ARPC3 and ARPC4. Further enrichment analysis within this PPI network identified clathrin-mediated endocytosis (log10(p) = –11.6), membrane trafficking (log10(p) = –8.4), and vesicle-mediated transport (log10(p) = –8.3) as the most significantly enriched pathways (Figure 5(b), right).
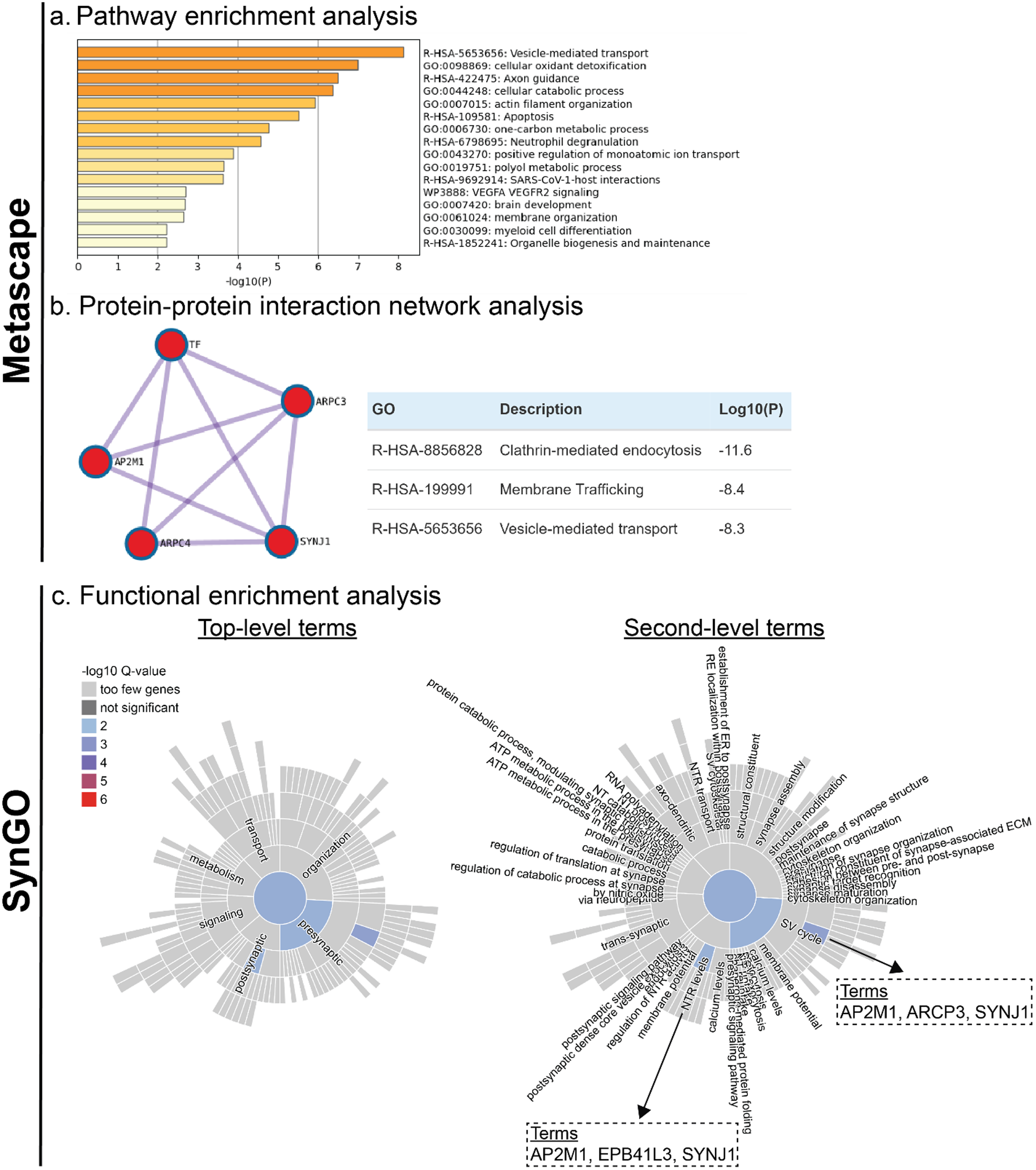
Pathway enrichment and protein-protein interaction analysis of sE/I-associated proteins in the PART BDTO interactome. Enrichment analysis of 38 proteins identified from the protein interactome of brain-derived tau oligomers (BDTO), isolated from individual PART hippocampal specimens (n = 4), that were associated with decreased sE/I. Proteins included in the analysis were those negatively correlated with excitatory currents, positively correlated with inhibitory currents, or negatively correlated with the sE/I ratio. The complete list of proteins is provided in Supplemental Table 6.
To more specifically evaluate the synaptic relevance of these proteins, functional enrichment analysis via SynGO was performed (Figure 5(c)), focusing on synapse-related biological processes and cellular components. This analysis revealed significant enrichment of two pathways: (1) synaptic vesicle (SV) cycling, particularly SV endocytosis, and (2) regulation of neurotransmitter receptor (NTR) levels. These pathways notably overlap with those identified in the broader pathway enrichment and PPI analyses (via Metascape), implicating a core subset of proteins—specifically AP2M1, ARPC3/4, and SYNJ1—as potential modulators of synaptic E/I balance in PART.
Discussion
Tau pathology is increasingly implicated in the modulation of neuronal and synaptic function, yet how distinct tau species relate to synaptic signaling across selectively vulnerable versus resilient brain regions, particularly during early stages of disease progression, remains poorly understood. In this study, we utilized neuropathologically validated PART specimens, comprising autopsy-derived hippocampal and neocortical tissue, to investigate the relationship between endogenous tau species and synaptic physiology. PART was leveraged as a surrogate model of early-stage AD, given its characteristic restriction of tau pathology to transentorhinal and limbic regions (per Braak staging), and the lack of Aβ pathology allows for focused investigation of tau-specific effects on synaptic signaling. By integrating quantitative immunoblotting of biologically relevant tau forms with electrophysiological recordings of human synaptic receptors, we identified region-specific associations between tau burden and the sE/I ratio. Specifically, oligomeric and fibrillar tau assemblies in the PART hippocampus were associated with a pro-inhibitory shift in sE/I. Further, proteomic analysis of BDTO complexes from PART hippocampi revealed tau-associated proteins linked to reduced sE/I, enriched in pathways related to vesicle cycling, membrane trafficking, and synaptic transport.
We observed region-specific differences in tau burden across PART cases, characterized by pronounced hippocampal accumulation of high–molecular-weight tau assemblies reactive to AT180, PHF13.6, and Tau5. These findings corroborate the operational definition of PART, originally based on AT8 immunoreactivity, 42 and extend prior work by demonstrating enrichment of additional pathological tau species. Specifically, AT180 detects tau phosphorylated at Thr231—a site implicated in early tau pathology—while PHF13.6 recognizes phosphorylation at Ser396, a residue linked to fibrillar tau and NFT.50,51 Enrichment of such species in the PART hippocampus suggests progressive tau pathology, consistent with reports of Gallyas-positive NFTs and PET-detectable tau burden in this region.41,79,80 Our findings further indicate a shift toward higher-order tau assemblies, with increased levels of oligomeric, detergent-soluble tau in the hippocampus. To our knowledge, this is the first study to report selective enrichment of soluble tau oligomers in the PART hippocampus—an important finding given their known synaptotoxic potential.13,15,81–85
Despite regional differences in select tau species, we observed no significant differences in individual excitatory responses, inhibitory responses, or the sE/I ratio between the hippocampus and SMTG. Such functional homogeneity may reflect intrinsic “synaptic homeostasis” or compensatory mechanisms that preserve sE/I ratio even in the presence of substantial hippocampal tau pathology32,86–88—a notion consistent with prior reports suggesting limited clinical impairment in PART.42,89,90 Supporting this hypothesis, studies in P301L tau mutant mice demonstrate that despite significant structural neuronal damage, surviving cortical pyramidal cells maintained synaptic function through compensatory mechanisms including enhanced intrinsic excitability and increased synapse formation. 91 Notably, the mean regional sE/I ratios observed in our PART cases (hippocampus: x̄ = 0.13; SMTG: x̄ = 0.17), as well as the corresponding gradient between regions, closely align with values reported in an independent MSM-based analysis of synaptosomes from individuals with mild cognitive impairment (MCI)—a condition with overlapping clinical and pathological features with PART (hippocampus: x̄ = 0.12; temporal cortex: x̄ = 0.22). 25 This concordance underscores the reliability of our measurements and lends further confidence to subsequent data interpretation. While seemingly at odds with the regional divergence in tau pathology, these findings emphasize the need to move beyond bulk functional measures and examine how specific tau forms relate to synaptic physiology.
We observed region-specific divergences in the relationship between tau species and synaptic function. In PART, hippocampal accumulation of oligomeric (Tau5-O) and fibrillar (PHF13.6-M) tau assemblies were negatively correlated with the sE/I ratio. These findings, while compelling, should be carefully contextualized within the existing literature—much of which has investigated tau's impact at the level of neuronal circuits or broader network activity.24,33,34 In contrast, our approach probes synaptic-scale phenomena, wherein the sE/I ratio serves as a proxy for local receptor-mediated signaling events that may either contribute to or reflect large-scale alterations in excitability. Notwithstanding these differences in resolution, our findings align with prior work in tau transgenic models, where hippocampal tau pathology has been associated with reduced excitability.92–94 Our group has also shown recently, using human autopsy tissue from Controls, MCI, and AD, that total tau burden in the temporal cortex, but not the hippocampus, correlates with increased sE/I ratio, further supporting a region-specific relationship between tau and synaptic function. 25 Additionally, phosphorylation at Ser396, detected here by the PHF13.6 antibody, has been linked to reduced excitability and network dampening. In 3xTg-AD mice, Ser396-phosphorylated tau suppresses hippocampal excitability and lowers seizure susceptibility, suggesting that individual phospho-epitopes can differentially regulate synaptic function. 94 This is consistent with experimental systems showing that NMDA receptor activation selectively increases phosphorylation at Ser396/404 and related sites in a reversible, dendritic, and postsynaptic manner, a pattern characteristic of physiological mechanisms that restrain NMDA receptor overexcitation. 95 Within this framework, the parallel associations that we observed between oligomeric tau, PHF-tau, and reduced sE/I ratio suggest that early tau assemblies may co-opt these signaling pathways, leading to depressed excitatory transmission.74–78 Nevertheless, regionally opposing patterns have also been reported, including cortical hypoactivity96–98 and hippocampal hyperexcitability in certain tauopathy models,99–101 likely reflecting heterogeneity in tau conformation, disease stage, and experimental paradigms. By directly interrogating native tau forms and synaptic physiology from the same brain specimen, our study helps reconcile these discrepancies and supports a model in which tau may modulate synaptic function in a subtype- and region-specific manner.
To explore mechanisms by which pathological tau may inform synaptic function, we profiled BDTO from hippocampal lysates of individuals with PART and identified a subset of interactome proteins whose abundance was correlated with functional changes driving reduced sE/I. Across both Metascape and SynGO analyses, these proteins were consistently enriched in vesicle-related pathways, including clathrin-mediated endocytosis, membrane trafficking, and vesicle-mediated transport—key processes that govern synaptic vesicle cycling and neurotransmitter receptor regulation.33,102–104 The convergence of enrichment results across multiple platforms reinforces the plausibility of vesicle dysregulation as a mechanistic link between oligomeric tau and synaptic signaling. Our interactome findings are consistent with a substantial body of work demonstrating that tau, across multiple aggregation and conformational states, localizes to synapses and associates with vesicle machinery. Studies in human AD tissue and model systems have shown that monomeric, 105 oligomeric, 16 hyperphosphorylated,106,107 and fibrillar tau 108 selectively engage presynaptic proteins involved in vesicle docking, fusion, and recycling. Imaging and biochemical analyses further indicate that oligomeric and pre-fibrillar tau accumulate at both pre- and postsynaptic compartments, where they interfere with neurotransmitter release, receptor cycling, and local calcium handling.13,18,82,109,110 Notably, a large-scale meta-analysis of human AD postmortem studies reported that presynaptic vesicle components are among the most consistently downregulated elements across tauopathic disease cohorts. 111 These observations align closely with our recent study exploring the synaptic interplay between Aβ and tau oligomers, where AD BDTOs were similarly enriched for proteins involved in clathrin-mediated endocytosis and cytoskeletal regulation. 16 Importantly, the fact that similar vesicle-enriched tau interactomes have been observed in AD brains containing both tau and amyloid pathology suggests that oligomeric tau can target vesicle systems even in the absence of Aβ, as is the case in PART. Taken together, these proteomic and functional data support a model in which oligomeric tau may modulate E/I balance by targeting vesicle trafficking pathways critical for maintaining synaptic homeostasis.
While our study provides important insights into tau-mediated synaptic dysfunction, key limitations must be acknowledged. First, although the association between oligomeric tau and reduced sE/I ratio is consistent with its proposed toxicity, the functional significance of this shift remains unclear. Given that hyperexcitability is often viewed as pathological, 34 a reduction in excitatory drive could reflect a beneficial, neutral, or compensatory response, particularly in early-stage tauopathy. Second, the cross-sectional nature of our study precludes causal inference. It remains unresolved whether tau drives changes in synaptic excitability, or whether altered excitability promotes changes to tau pathophysiology; both interpretations are biologically plausible, to include a feedback loop, and supported by prior work.33,34 Third, our electrophysiological approach captures ligand-gated receptor function but does not assess intrinsic excitability or network-level synchrony. As such, these data should be interpreted as synaptic correlates rather than definitive measures of neuronal activity.
To establish causality, future studies should test how specific tau species modulate E/I balance using experimental systems that permit controlled application of tau. For instance, delivering preformed tau oligomers onto synaptosomes, iPSC-derived neurons, or brain organoids, followed by TEVC or other physiological readouts, could help clarify direct effects on synaptic function. The consistent enrichment of BDTO-associated proteins in vesicle-related pathways across Metascape and SynGO analyses highlights promising candidates for mechanistic validation. Finally, linking tau conformer profiles to cognitive trajectories, ideally within longitudinal or biomarker-informed human cohorts, will be critical to contextualize these findings in disease progression.
Conclusion
In summary, our findings demonstrate that tau-mediated synaptic dysfunction in the human brain is influenced by both the molecular identity of pathological tau species and their regional distribution. By integrating biochemical, electrophysiological, and proteomic analyses of autopsy-derived specimens from PART cases, we show that soluble oligomeric and fibrillar tau assemblies in the hippocampus are associated with a reduced sE/I ratio. These associations implicate vesicle trafficking pathways as candidate mediators of synaptic vulnerability to tau oligomers. Collectively, these insights help reconcile prior inconsistencies and establish a foundation for future work aimed at targeting specific forms of pathogenic tau to preserve synaptic function in tauopathies along the AD disease spectrum.
Supplemental Material
sj-docx-1-alz-10.1177_13872877261416524 - Supplemental material for Regional correlates of tau pathology and synaptic function in primary age-related tauopathy
Supplemental material, sj-docx-1-alz-10.1177_13872877261416524 for Regional correlates of tau pathology and synaptic function in primary age-related tauopathy by Shrinath Kadamangudi, Laura Sanchez-Sanchez, Agenor Limon and Giulio Taglialatela in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
This work was supported by the NIH/NIA grants F30AG085974 to SK, R21AG089708 to GT, R01AG070255 to AL, and R01AG073133 to GT and AL. We extend our sincere gratitude to the donors and families that make these studies possible. We thank Dr Peter Nelson from the University of Kentucky for dissection and neuropathological evaluation of the postmortem autopsy specimens utilized in this study, facilitated in part by the University of Kentucky Alzheimer's Disease Research Center supported by P30AG072946. We also thank Mr Brice Miller for his assistance with methodology training and acquisition of the MSM data; Dr Lee Palmer, Dr William Russell, and the UTMB Mass Spectrometry Core Facility, for their assistance with acquisition and interpretation of mass spectrometry data; Dr Vsevolod Popov and the UTMB Electron Microscopy for their training and assistance with acquisition of electron microscopy data; Ara XIII of the Moody Brain Health Institute for assistance with manuscript revision.
Ethical considerations
Postmortem frozen human brain tissues used in this study were obtained through established material transfer agreements with the ADRC at the Sanders-Brown Center on Aging, University of Kentucky. All procedures involving human specimens were conducted in accordance with IRB guidelines at the originating institutions. Donor samples were fully de-identified prior to transfer to the University of Texas Medical Branch (UTMB); therefore, the use of these specimens did not constitute human subjects research and did not require UTMB IRB approval, in accordance with 45 CFR §46.101(a).
Consent to participate
All donors were enrolled in longitudinal brain aging studies at the contributing institutions, and written informed consent for participation and postmortem tissue donation was obtained prior to death.
Consent for publication
Not applicable.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NIH/NIA grants F30AG085974 (SK), R21AG089708 (GT), R01AG070255 (AL), R01AG073133 (GT and AL), and P30AG072946 (University of Kentucky).
National Institute on Aging, (grant number F30AG085974, P30AG07294, R01AG070255, R01AG073133, R21AG089708).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Raw proteomic data used for the post hoc PART BDTO synaptic interactome analysis was made available in Jamison et al. (2025), Supplemental File 1.
44
Raw western blot data are provided at the end of the ![]() document. All other forms of raw data will be provided by the authors upon reasonable request.
document. All other forms of raw data will be provided by the authors upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
