Abstract
Background
Cognitive impairment (CI) and its related risk factors (e.g., diabetes and stroke) are highly prevalent among Hispanic/Latinos (H/L); however, prior research in H/L focused on aging individuals (≥65 years old).
Objective
To comprehensively assess the associations between a wide-range of cardiometabolic health indicators and CI using a prospective study design in a younger cohort of H/L (majority <65 years old) from the Cameron County Hispanic Cohort (CCHC).
Methods
We identified a total of 1240 CCHC subjects with complete Mini-Mental Status Exam (MMSE) data at study baseline and at 5-year follow-up. The outcome (i.e., CI) was based on MMSE scores of less than 24. We conducted univariate associations for multiple cardiometabolic indicators with CI; and mixed logistic regression models to estimate odds ratios for the associations between cardiometabolic indicators and CI adjusted for age, education, prior stroke, and APOE gene.
Results
The majority (89.9%) of the participants were <65 years old. A total of 117 subjects had CI at baseline (9.4%). Baseline study cohort showed that Individuals with CI were older with a lower education performance, and were more likely to be diabetic with lower mean levels of Low-density Lipoprotein, and a higher mean systolic blood pressure. Diabetes significantly increased the odds for CI (OR:2.11, 95%CI:1.26–3.52) from the adjusted multivariate mixed logistic models.
Conclusions
This analysis showed that diabetes was an important indicator for CI regardless of age, education, or APOE gene status. These findings highlight the higher burden of cardiometabolic risk factors on CI in the CCHC cohort.
Keywords
Introduction
Hispanic/Latino (H/L) populations will represent 30% of the entire US population and 23% of the projected 13.9 million Americans with Alzheimer's disease and related dementias (ADRD) by 2060. 1 While ADRD impairs daily living activities; 2 mild cognitive impairment (MCI) does not interfere with daily living activities.2–4 MCI increases the risk for ADRD by over 3 folds 3 and shares similar risk factors to ADRD such as APOE 4 haplotype and cardiometabolic risk factors such as diabetes and hypertension and sedentary lifestyle.5,6 Understanding the driving risk factors for MCI in H/L will help reduce the risk of ADRD knowing that 35% of ADRD cases are preventable by controlling modifiable risk factors such as cardiometabolic risk factors and increasing physical activity.1,5,7
H/L populations tend to have a higher prevalence and incidence of dementia and MCI when compared to Non-Hispanic (NH) Whites.8–11 H/L 65 years and older showed 50% increased risk for ADRD compared to NH White.9,11 Furthermore, H/L tend to have a different genetic background compared to NH white populations. In particular, H/L populations showed weaker APOE gene associations with dementia, as far less cases of dementia among H/L were explained by the APOE haplotype compared to NH Whites.12–14 Additionally, genetic risk scores for diabetes have not been associated with increased risk for dementia in H/L, as was the case with NH White. 15 These differences in genetic backgrounds make the case for a higher influence of cardiometabolic risk factors as drivers for the high incidence of cognitive impairment in H/L. 16 Lastly, H/L, mainly Mexican Americans, tend to develop cognitive impairment at a younger age group when compared to NH Whites.11,14 Prior research studies in dementia in the H/L population have focused on those over 65 years of age and mainly addressed the relationship between dementia and other cardiovascular risk factors in cross-sectional or case-control study designs. 16
The objective of this study was to investigate the associations of multiple demographics and cardiometabolic indicators with MCI in the Cameron County Hispanic Cohort (CCHC), a longitudinal cohort study of individuals living in Cameron County in Brownsville, Texas mainly consisting of Mexican origin H/L who are selected randomly to participate in this cohort. The contributions of APOE haplotypes and cardiometabolic factors to cognitive decline in this unique cohort of H/L are still unknown. Therefore, we designed a longitudinal study in which each individual was tested at two different points, five years apart, to reduce residual confounding in our study. We hypothesize that cardiometabolic risk factors, not APOE haplotypes, are the major drivers for the high incidence of cognitive impairment in H/L populations of Mexican origin and the CCHC in particular.
Methods
The CCHC, a longitudinal study that collects data from the local H/L community in the Rio Grande Valley, is mainly consisted of Mexican American participants. Details regarding the CCHC's randomization, recruitment, and data collection procedures have been described previously. 17 Briefly, Mexican-American households in Brownsville, Texas were randomly selected based on data from the US census. Individuals within selected households were invited to participate in the CCHC study. Sampling bias was adjusted by applying weights to the data based on age, sex, census tract or block, and household clustering to achieve a representative sample of the local community.17,18 Our longitudinal study analyses were based on a subset of the participants in the CCHC cohort who received a Mini-Mental State Examination (MMSE) assessment at the baseline interview and a follow up MMSE assessment at their second visit (i.e., 5-year follow-up interview).
Consented CCHC participants provided their medical and socioeconomic information through standardized questionnaires which included the MMSE assessment at baseline and each following visit. Participants answered those questionnaires in either English or Spanish, based on their preferred language. Blood samples were also collected at the baseline interview and at each following visit. This study was approved by the Institutional Review Board (IRB) of the University of Texas Health Science Center at Houston.
Sample selection
Currently, there are about 5000 CCHC participants with data points of follow-up for 20 years (2004–2024). To construct our prospective cohort, we limited the current analysis to include those who had their second follow up visit after 5 years from their baseline enrollment (i.e., each patient in this cohort will have two visits: a baseline visit, and a second visit after 5 years). The earliest baseline visit in our selected prospective cohort was in April 2004 and the latest second follow-up visit was in May 2024 (Figure 1). At baseline and 5-year follow up visits, subjects received full assessments which included MMSE questionnaire and cardiometabolic assessments (Figure 1). The outcome (i.e., cognitive impairment) of this study was defined based on MMSE score where MMSE of less than 24 was considered as an indication of cognitive impairment.19–21 This MMSE score was determined based on clinical trials practices which applied MMSE scores to select participants with AD. 19 Clinical trials excluded participants with cognitive impairment from receiving disease modifying treatment for AD using MMSE scores of a mean of 24 points. 19 Additionally, subjects were excluded from our cohort based on age (<18 years of age were excluded), and the presence of MMSE scores data (subjects with missing MMSE scores were excluded).
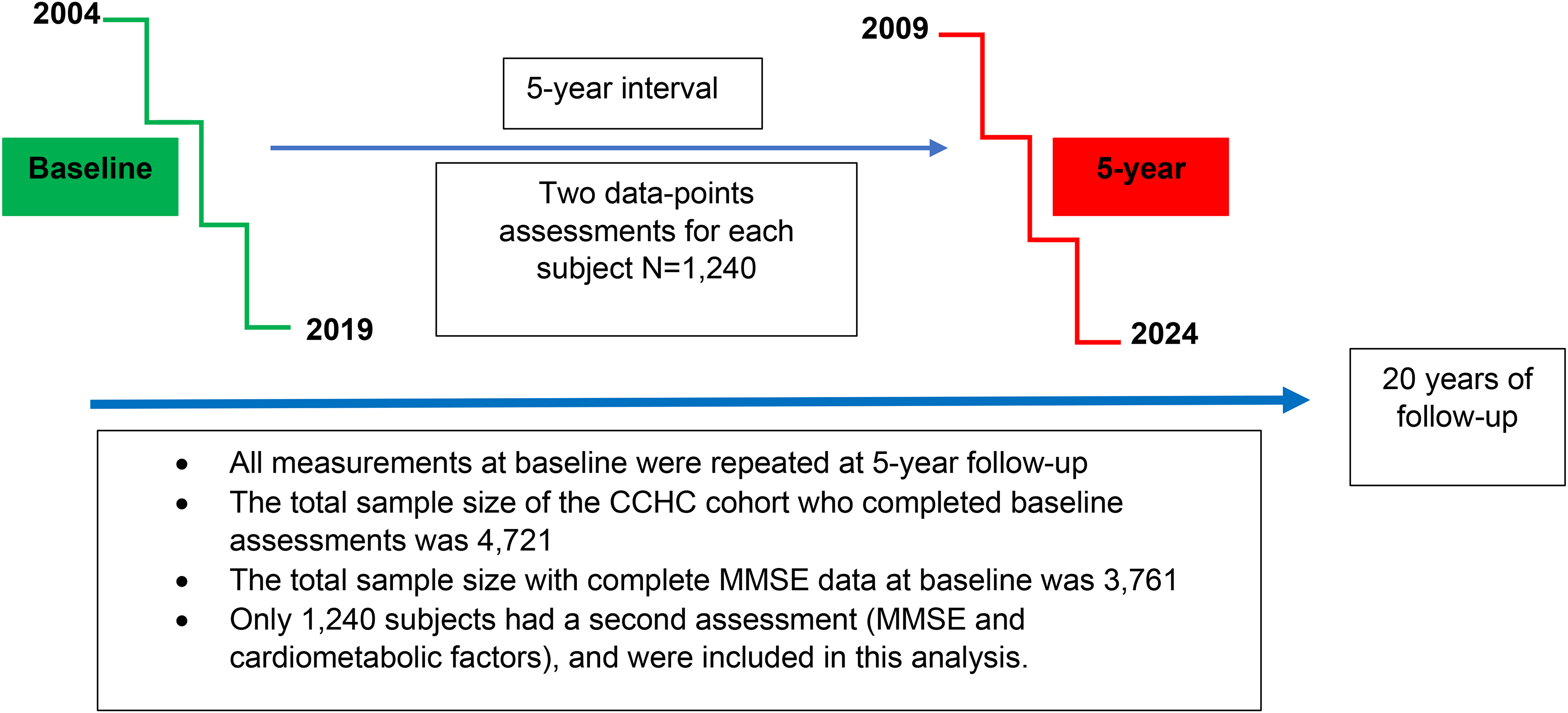
An overview diagram showing cohort enrollment and analytic processes for the current analyses.
Statistical analysis
All analyses were conducted using R software (version 4.4.2). We included independent variables and risk factors that were tested in prior research studies on cognition and AD from Hispanic and Non-Hispanic populations. We conducted univariate associations at study baseline for multiple cardiometabolic indicators (e.g., hypertension [systolic and diastolic], diabetes [diabetes history and HbA1c], lipids [Low Density Lipoproteins (LDL-c), High Density Lipoproteins (HDL-c), Triglyceride, Total Cholesterol], echocardiogram measurements [Left Ventricular Ejection fraction (LVEF), Left Ventricular End Diastolic Diameter (LVIDd), Left Ventricular End Systolic Diameter (LVIDs), and Left Ventricular (LV) mass], inflammatory markers [C reactive protein (CRP), IL1beta, IL6, IL8, Tumor Necrosis Factor (TNF) alpha), and sociodemographic factors (e.g., age, education level, Body mass index, physical activity, annual income) with cognitive impairment using Chi square, Fisher exact, and student T-test. Next, we ran mixed logistic regression models to estimate the odds ratios for the associations between nine cardiometabolic indicators with missingness rate of <5% and cognitive impairment adjusted for age, education, prior stroke, and APOE haplotypes (Table 2). We included a random intercept to account for within-person correlation and a fixed effect of visit (baseline versus 5-year). To construct the 95% confidence intervals and p values that accounts for the random effect from the mixed logistic model, we estimated the random standard error using the variance-covariance matrix from the mixed logistic regression analysis. The definitions and criteria used in reporting those risk factors in the CCHC cohort are provided previously. 19 We excluded risk factors with more than 5% of missing information from our mixed logistic models (Tables 1 and 2). To identify the level of significance with multiple testing, we used False discovery rate (FDR) method to estimate the threshold of significance for multiple testing (p value<0.05). Finally, we looked into the dose-response relationship between Hemoglobin A1c and MMSE scores using adjusted and unadjusted mixed logistic modeling.
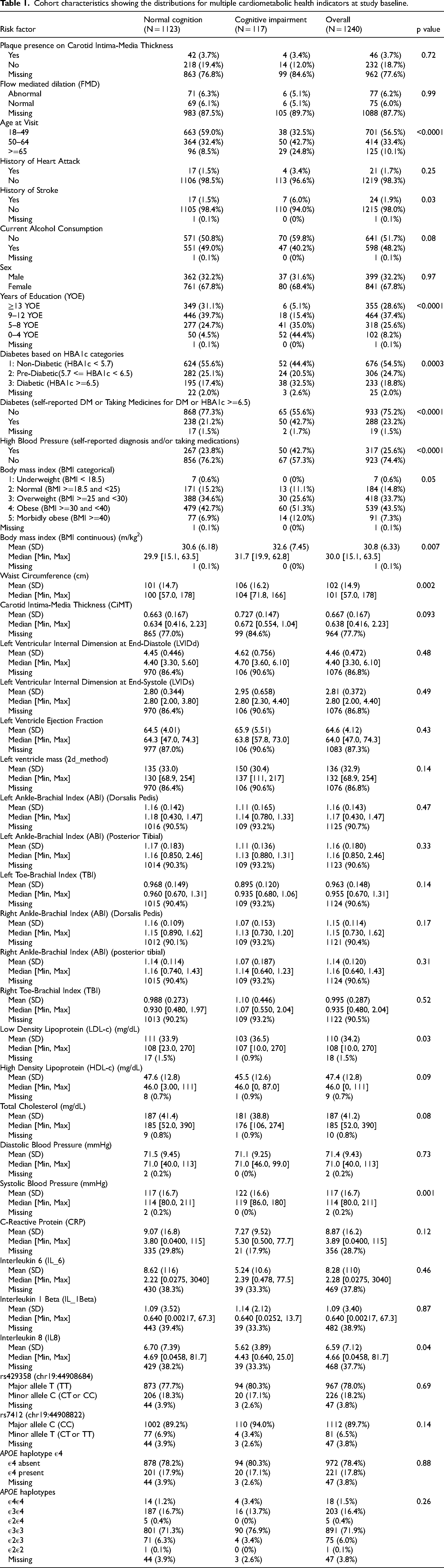
Cohort characteristics showing the distributions for multiple cardiometabolic health indicators at study baseline.
Associations from several cardiometabolic indicators with cognitive impairment from crude and adjusted mixed logistic regression models. False Discovery Rate (FDR) was used to adjust for multiple testing. All models were adjusted for age, level of education, prior stroke, and presence of APOE ɛ4 haplotype.
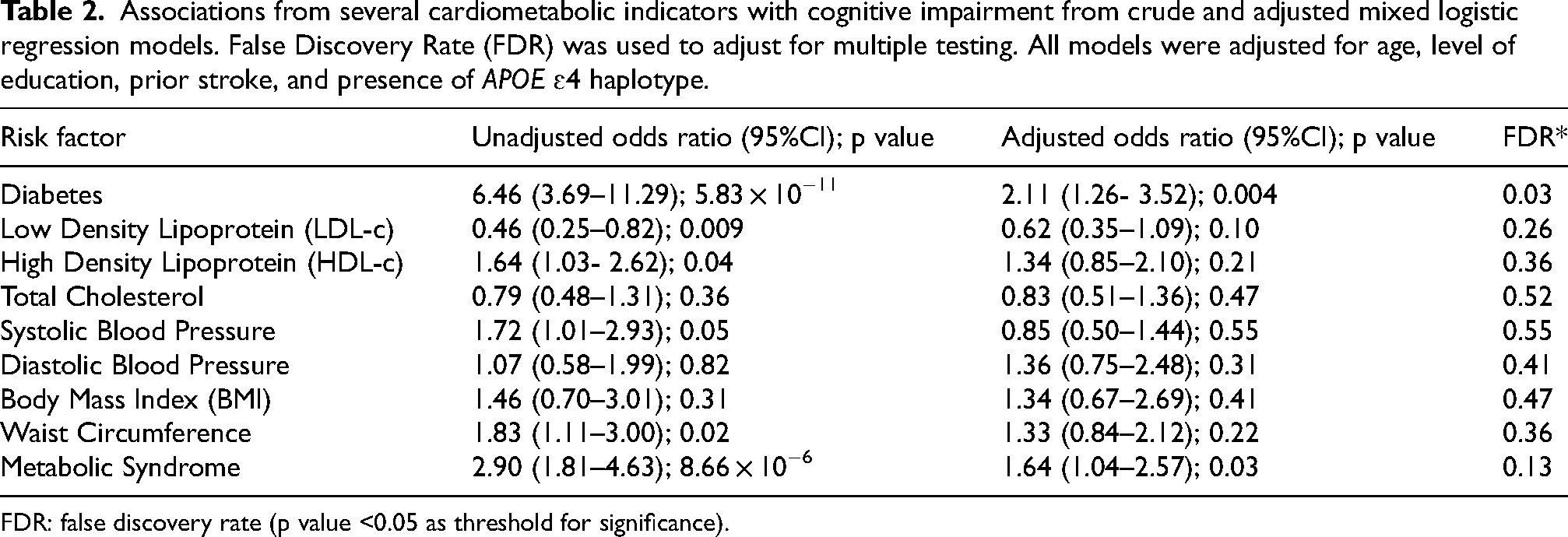
FDR: false discovery rate (p value <0.05 as threshold for significance).
DNA genotyping and imputation
DNA genotyping and imputation procedures for the CCHC cohort were previously described. 22 Briefly, Illumina chip (Multi-Ethnic Genotyping Array – MEGA) was used to provide genotypes specific to Hispanic population. After quality control, genotypes were imputed using the TOPMed reference panel and the Michigan Imputation Server. 22 For the APOE haplotype analyses, we used two imputed SNPs (rs429358 and rs7412) in the APOE region (imputation R2 > 0.95) to estimate the APOE haplotype in our cohort using PLINK and R. 23
Results
The total number of subjects 18 years and older with complete follow-up data on MMSE at baseline of the study was 3761. A total of 1240 out of 3761 subjects completed the 5-year interview and were available for the current analysis (Figure 1). Except for a higher percentage of cognitive decline (12% versus 9%) and slightly younger average age (44 years versus 46.5 years), those who did not complete their 5-year follow-up had similar distributions for education and cardiometabolic risk factors to those completed their 5-year follow-up and were included in the current analysis. Baseline study cohort characteristics are shown in Table 1. A total of 117 out of 1240 subjects showed cognitive impairment (MMSE<24) at study baseline. The majority of the subjects involved from this cohort were less than 65 years old (89.9%) and the male-to-female ratio in this cohort was about 2 to 1 (Table 1). Subjects with cognitive impairment were more likely to be older in age, have lower education levels, have a history of myocardial infarction or stroke (Table 1). Having heterozygous (APOE4E3) or homozygous (APOE4E4) APOE haplotypes did not significantly increase the risk for cognitive impairment in our cohort (Table 1). APOE haplotypes distributions are shown in Table 1.
The mixed logistic regression models’ estimates for crude and adjusted odds ratios are shown in Table 2. We adjusted our modes for age (categories:18–49, 50–64, ≥65), education level (categories: ≥13, 9–12, 5–8, ≤4), history of incident stroke, and APOE4 haplotype status (Table 2). Diabetes mellitus significantly increased the odds for cognitive impairment by 2 times (OR:2.11, 95%CI (1.26- 3.52)) when compared to those without diabetes and after adjusting for age, education, history of stroke, and APOE4 haplotype status (Table 2). Lower LDL-c levels, higher HDL-c levels, and higher systolic blood pressure decreased the odds for cognitive impairment; while higher diastolic blood pressure, higher BMI, and metabolic syndrome increased the odds for cognitive impairment. However, besides diabetes, none of those cardiometabolic risk factors remained significant after adjusting for multiple testing (Table 2). We tested for a dose-response relationship between glycosylated Hemoglobin A1c (HBA1c) and cognitive impairment using unadjusted and adjusted mixed logistic regression modeling (Table 3). Our dose-response analyses showed a significant dose-response association between increasing levels of HBA1c and having cognitive impairment (Table 3).
Shows the dose-response associations between HBA1c and mild cognitive impairment (MMSE<24) using unadjusted and adjusted mixed logistic regression modeling.

Discussion
We showed that the cognitive impairment process in the majority of our Mexican American H/L subjects develops below the age of 65, and is significantly associated with diabetes. This important observation opens possibilities for establishing early biomarkers and preventive approaches to lower the burden of cognitive impairment in H/L populations, mainly Mexican Americans. The current analysis was a longitudinal study to investigate the associations of several cardiometabolic risk factors with cognitive impairment in a relatively young Hispanic population of mainly Mexican Americans living in the Rio Grande Valley. We adjusted our analyses for age, level of education, the presence of a self-reported incident stroke, and APOE haplotype status. Our adjusted models and, after correction for multiple testing (FDR p value <0.05), showed that diabetes was positively associated with incident cognitive impairment in our population. LDL-c, HDL-c, total cholesterol (TC), metabolic syndrome, and both systolic and diastolic blood pressures were not associated with cognitive impairment in our adjusted analyses.
The diabetes association with cognitive impairment is well-established in prior research studies of H/L and non-H/L populations. Our results showed that participants with diabetes had two times the odds of incident cognitive impairment compared to those without diabetes after adjusting for age, education level, history of incident stroke, and APOE haplotype (OR: 2.11, 95%CI: 1.16–2.63). Prior research by Gonzalez et al. reported a 74% increase in the odds of having prevalent diabetes among those with cognitive impairment in middle-aged and elderly Hispanics from the Study of Latinos cohort (odds ratio 1.74 [95% CI 1.34; 2.26]; p < 0.001), 16 and a weaker association with incident diabetes and cognition. 16 Similarly, prior research on diabetes and cognition in older H/L (≥70 years of age) showed increased risk for cognitive impairment among those with diabetes when compared to non-diabetics. 24 Prior research on diabetes and cognitive impairment in H/L, retrospective or prospective, was focused on older Hispanics over the age of 65 years. For example, H/L participants from retrospective,25–27 and prospective studies such as the Study of Latinos–Investigation of Neurocognitive Aging (SOL-INCA), 16 the Health and Aging Brain among Latino Elders (HABLE),28,29 and the San Antonio Longitudinal Study of Aging (SALSA), 30 had an average age of over 60 years. Our younger Hispanic population (89.9% below that age of 65, mean age = 46 years) showed that over 40% of those with impaired cognitive function were between the age of 50 to 64 years-old (Table 1). Furthermore, we were able to show a significant (p value <0.05) dose-response relationship between increasing levels of HBA1c and having cognitive impairment, a novel finding in Mexican American H/L (Table 3). 31
Genetic studies on APOE and diabetes genes and their associations with cognitive impairment and AD have been inconclusive among Hispanics. 9 This inconsistency in the APOE-cognitive impairment association may be attributed to the heterogenous nature of the H/L population and it was shown that H/L from different geographic regions showed different APOE-cognitive impairment associations.9,32 A study from the Million Veteran Program showed no associations between genetic risk score for diabetes genes and dementia among Hispanics, regardless of the APOE allele status. 15 APOE haplotypes showed the weakest associations with AD when compared to APOE haplotypes associations with AD from Asians, Non-H/L Whites, and Non-Hispanic Black populations.12,30 This attenuated association between APOE ɛ4 alleles and dementia could be attributed to the lower prevalence of APOE alleles in H/L populations. 14 Our findings from analyzing the APOE haplotype in our cohort showed no association between having an APOE ɛ4 haplotype and risk for cognitive impairment and were in line with previous analyses in Hispanic/Latino populations33–35 (Table 1). This may indicate that the increased risk for cognitive impairment in diabetic Hispanics is mainly driven by the cardiometabolic effects of diabetes on brain vasculature. 30
Our analyses investigated the associations of several lipid profiles (e.g., LDL-c, HDL-c, and TC) with cognitive impairment. LDL-c mean and median were significantly lower among those with cognitive impairment compared to those with normal cognition in our cohort. Although statistically insignificant, our adjusted models showed that a higher LDL-c to have protective effect on cognition (OR:0.62, 95%CI: 0.35–1.09). Prior reports on lipid profiles and cognition in H/L were inconsistent. Similar to our results, Reitz at el., reported sex and age adjusted associations between LDL-c level and incident cognitive impairment that were protective for incident cognitive impairment among Hispanics 65 years and older. 36 However, this protective LDL association with cognitive impairment was attenuated after adjusting for education, APOE4, age, sex, and vascular risk factors. 36 Reitz at el., concluded that there were no associations between lipids (LDL-c, HDL-c, TC) and incident cognitive impairment among those 65 and older. A more recent cross-sectional study from the Study of Latinos cohort showed that higher total cholesterol and LDL-c levels were associated with improved learning and verbal cognition, 37 an unusual finding that is in line with our report on the association between LDL-c and cognition in our longitudinal analyses. Furthermore, this study showed an effect modification of Hispanic background in the association of LDL-c and cognition suggesting that different H/L backgrounds have different effects on this association. 37 Adjusting for antihyperlipidemic medications did not change the association between LDL-c and cognition. 37 Similar to our findings on the association between HDL-c and cognition from our cohort, Lamar et al., showed that HDL-c to be insignificantly associated with cognitive test performance in learning, memory, and verbal fluency domains. 37
Hypertension is an important risk factor for vascular health and vascular cognitive impairment. Few studies had examined the associations of having hypertension on cognitive impairment among Hispanics. Márquez et al., reported that having persistent hypertension (i.e., prevalent hypertension) was significantly associated with cognitive decline in the Study of Latinos Hispanic cohort; however, incident hypertension in the same cohort was not associated with cognitive impairment. 38 Although our study did not differentiate between persistent and incident hypertension, our adjusted model did not show a significant association of either systolic or diastolic blood pressure measurements with incident mild cognitive decline. This lack of association between blood pressure readings and incident MCI in our cohort may be explained by the fact that our observations for blood pressure were applied at one time point per visit with no continuous or serial blood pressure readings in-between the visits. Furthermore, Marquez et al., included a mixture of Hispanic/Latino populations in their analyses with only 34.4% were Mexican Americans, and did not stratify their hypertension associations with cognitive impairment by country of origin. 38 Different H/L populations might contribute to the differences in the reported associations between hypertension and cognitive impairment from our cohort and those reported by Marquez et al.
The use of the MMSE score for assessing cognitive impairment in H/L showed that H/L usually scored lower on the MMSE test compared to NH White and that might be attributed to lower education levels or environmental factors.39–42 A study by Espino et al., showed that the main reason for variability in MMSE scores regardless of ethnicity and language is the MMSE scoring method and recommended the use of the serial seven as the best scoring technique. 43 Moreover, Espino et al., noted that MMSE becomes less sensitive to detect cognitive impairment in those with higher education levels. 43 Our scoring technique for the MMSE test reported the highest score from either the serial seven or the spelling of “WORLD” backwards, that might further reduced the MMSE test sensitivity to detect cognitive impairment in our population.
Strengths and limitations
Our study has several strengths such as the longitudinal study design and the random recruitment from the population that was not based on the presence of cognitive impairment. Several limitations in our analyses included the use of the MMSE which is known for its low sensitivity in detecting cognitive impairment, 44 may resulted in missed opportunities to identify cases of cognitive impairment. We are currently improving our cognitive testing screening procedures in the CCHC cohort through implementing the Montreal Cognitive Assessment test which is proven for its higher sensitivity in detecting MCI over MMSE. 44 Although we used high quality imputed SNPs to estimate our APOE haplotypes; the attenuated APOE4 associations in our cohort may be related to the use of imputed SNPs instead of genotyped or sequenced SNPs to estimate APOE alleles frequencies. Missing values from several important lifestyle risk factors such as physical activity and nutrition, and cardiometabolic indicators such as echocardiography, carotid intimal thickness, and inflammatory markers measurements might result in missing opportunities in identifying associations with cognitive impairment. The relatively small sample size may also contribute to the low power of detecting significant associations.
Conclusions
Diabetes showed significant association with MCI in young and middle-aged Hispanics in our longitudinal study. This, in addition to the lower impact of APOE haplotypes on Hispanic populations, makes it possible that main drivers for cognitive impairment in this population most likely are driven by vascular pathologies related to diabetes and its related cardiometabolic outcomes. Future studies should examine the link between vessel health of the cerebrovasculature and cognition in Hispanic patients less than 65 years old.
Footnotes
Ethical considerations
This study was approved by the Institutional Review Board (IRB) of the University of Texas Health Science Center at Houston.
Author contribution(s)
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from Dr McCormick and Dr Fisher-Hoch upon reasonable request.
