Abstract
Today, approximately 7.2 million people have Alzheimer's disease in the United States. That number is expected to double by 2060 if a preventive approach is not undertaken. Globally, those figures are also predicted to increase. As we’ll soon discover, air pollution and other aspects of climate change are a predominant cause of this remarkable surge. The purpose of this article is to examine the scientific basis of pollution and suggest that an evolutionary lifestyle medicine program may help mitigate and slow down its negative effects on brain health. This review examines existing literature on pollution, and lifestyle as well as other aspects of a lifestyle medicine program, including diet, exercise, stress reduction especially via yoga and meditation, good sleep and essential and spiritual well-being, thus investigating their potential to enhance awareness of the issue and prevent the increase in Alzheimer's disease predicted to arise from air pollution and other forms of climate stress. Air pollution, and the potential for global warming, presents a critical emergency. Scientific evidence indicates that these factors contribute to an elevated risk of Alzheimer's disease, along with other neurological, mental, and physical health challenges. Our hypothesis is that an evidence-based lifestyle medicine program can potentially prevent, slow, or reverse multiple medical problems that are tied to Alzheimer's disease prevention. It is hypothesized that once practitioners of this program become aware, they can reach out to their family, friends, communities, country and globally to attend to the issue with strength, wisdom and clarity.
Keywords
Introduction
An 11-year-old boy tragically lost his life after being struck by a vehicle while crossing the street in Mexico City, one of the most polluted places on earth. At autopsy it was seen that he had multiple brain findings consistent with someone much older and prone to Alzheimer's disease (AD) such as positive neurites and tangles stained for phosphorylated tau. 1
The theory that AD may start many decades before symptoms develop should now be adjusted to include the new reality that AD may, in fact, begin in childhood in the many highly polluted areas of the globe, which essentially puts hundreds of millions more people at increased risk. This article explores how environmental stressors such as pollution, nanoparticles, and climate change impact AD and overall well-being. Secondly, a plan to prevent pathological changes and reduce the risk of AD and other neurodegenerative disorders will be presented.
According to the preface of the Journal of Alzheimer's Disease book (IOS Press) on Alzheimer's disease and air pollution, “Our only hope for prevention lies in protecting millions of people exposed to air pollutants and ameliorate their brain effects, we will be on the right path to have a clean planet free of Alzheimer's disease.” Following this plan will also decrease dramatically health care costs at this critical time and perhaps most importantly stop the progression of climate stress. 1
That is our goal and the purpose of this article.
The science
Pathological basis of air pollution and climate change on brain health
Climate change is a pressing global concern, frequently highlighted as a direct threat to the future of human health and society. Average surface temperatures are steadily rising, with predictions of a 2–3°C increase by 2090. 2 This global warming phenomenon has already been linked to extreme climatic events, including droughts, floods, heat waves, and wildfires, resulting in devastating societal and economic consequences.3,4 Despite persistent climate skepticism, the expansive effects of climate change are undeniable. The 2024 annual report from the United States National Oceanic and Atmospheric Administration (NOAA) recounts the toll of extreme weather and climate disasters, indicating that the country has sustained over 400 major weather and climate disasters since 1980, with overall damages exceeding $2.9 trillion and resulting in thousands of deaths. 5 Globally impactful events, such as the recent Los Angeles wildfires in January 2025, exemplify this reality, with the Palisades and Eaton fires causing catastrophic losses. Preliminary estimates suggest the destruction of 16,240 properties, translating to capital losses ranging from $95 to $164 billion. 6 Moreover, recent wildfires have unusually occurred in North and South Carolina, Georgia, and Texas. 7 Beyond the financial toll, the physical, emotional, and mental health burden of the fires on affected communities will likely be profound and enduring. Systematic reviews have extensively documented that the environmental consequences of climate change, such as wildfires, significantly worsen human health outcomes. 8 Specifically, climate change has been associated with increased mortality, neurological diseases, and various other bodily problems.8–10 Over the past three decades, the global incidence of neurological disorders has steadily risen, particularly in regions with higher socioeconomic development. 11 As the global population ages and life expectancy continues to increase, neurological disorder diagnoses are expected to rise significantly in the coming decades. 11 The underlying mechanisms driving these disorders are diverse, including: (1) proteinopathies, (2) vascular pathologies, and (3) alterations in neurotransmitter levels. Proteinopathies, for instance, involve the accumulation of misfolded proteins in brain regions, such as amyloid-β plaques and neurofibrillary tangles in AD, abnormal intracytoplasmic α-synuclein protein accumulation in Lewy body dementia and Parkinson's disease, or mHTT aggregation in the striatal region in Huntington's disease. 12 Similarly, vascular pathologies compromise blood flow, leading to localized brain parenchymal injuries, as seen in vascular dementia. 12 Finally, depletion of neurotransmitters such as acetylcholine, dopamine, and serotonin leads to specific symptoms of neurodegenerative diseases, including movement disorders, tremor, rigidity, bradykinesia, memory dysfunction, and cognitive defects. 13 While these mechanisms are well-studied, mounting evidence suggests that air pollution and climate change may accelerate and intensify these processes.
As described at the outset of this article, air pollution plays a critical role in neurodegenerative disease progression. Oxidative stress, heavy metals, and environmental pollution have been reported to significantly reduce neurotransmitters in the central nervous system, such as acetylcholine and dopamine, increasing the incidence of AD, Parkinson's disease, and vascular dementia. 13 Common air pollutants such as carbon monoxide, lead, nitrogen oxides, tropospheric ozone, fine particulate matter (PM), and sulfur dioxide often results from vehicle exhaust, the burning of vehicle exhaust, fires, and power plants among other sources (Table 1).
Common air pollutants.
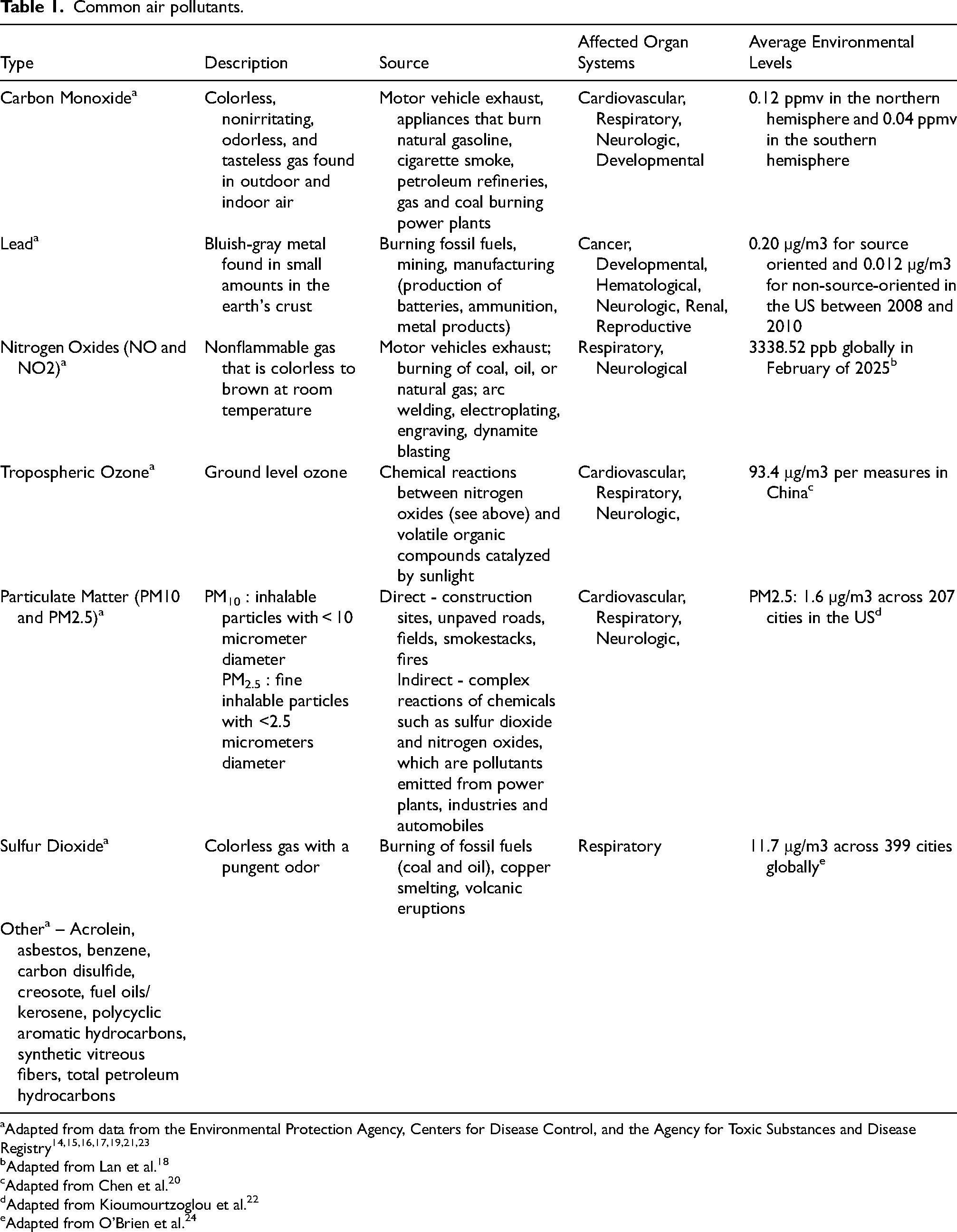
Adapted from data from the Environmental Protection Agency, Centers for Disease Control, and the Agency for Toxic Substances and Disease Registry14,15,16,17,19,21,23
Adapted from Lan et al. 18
Adapted from Chen et al. 20
Adapted from Kioumourtzoglou et al. 22
Adapted from O’Brien et al. 24
Many of these air pollutants have direct effects on neurological health. More specifically, air pollutants, including lead, ozone, polycyclic aromatic hydrocarbons, and particulate matter in urban environments, can negatively impact neuronal hormones and neurons. 13 Fine particulate matter PM can induce nervous system injury by crossing the blood-brain barrier, triggering inflammatory responses, and promoting processes such as amyloid-β accumulation, tau protein phosphorylation, and synaptic dysfunctions, which contribute to AD pathology.25,26 Fu et al. found a 95% increase in AD risk with higher levels of PM 2.5. 27 Building on this Zhang et al. reported that higher PM 2.5 levels from agriculture and wildfires were associated a robust increase in the rates of dementia in the U.S. 28 Studies based out of China, the United Kingdom, and Germany have also linked fine PM to cognitive decline.29–31 For this reason air pollution specifically has been identified as a modifiable risk factor for AD by the Lancet Commission Report in 2020, and the reduction of exposure to air pollution and second-hand tobacco smoke have been set as specific actions for across the life course. 32 Recognizing this danger, governmental agencies, such as the United States Environmental Protection Agency (EPA), had implemented stricter air pollution regulations, aiming to prevent thousands of premature deaths annually. 33 However, these regulations have since been rescinded. 34
Through the same mechanisms, and depending on individual susceptibility, microplastics could contribute to the increasing incidence of neurodegenerative diseases. In recent years, plastic pollution has become ubiquitous, and what is more concerning is that such contamination is enduring, taking between 100 to 1000 years to decompose. 35 Microplastics are small plastic particles that come from the degradation of plastics and these tiny particles of plastic can be found in the air we breathe, the water we drink, and the food we consume. 36 Scientists have even identified microplastics in the atmosphere, where they are exposed to ultraviolet radiation and contribute to the production of toxic greenhouse gases. 36 Studies have indicated that increased levels of environmental plastics have been linked to higher levels of micro- and nano-plastics (MNP) found in human tissue samples.37,38 Specifically, polyethylene shards and flakes have been identified in the brain tissue of American adults, with a significant increase in the presence of MNPs observed between 2016 and 2024. 39 While the data does not prove a causative relationship, there was an association documented between dementia diagnosis and the level of MNP accumulation. Specifically, a high level of deposition was found in the cerebrovascular walls and immune cells. 39 Mechanistically, microplastics may induce an inflammatory response and thereby may be at the origin of oxidative stress and neurotoxicity, which is related to neurodegenerative diseases. In animal models, microplastics have been shown to impact neuronal function and behavior, as microplastics are reported to cause inhibition of acetylcholinesterase, oxidative stress with increase in lipid peroxidation levels and an increase in the anaerobic pathway of energy production. 40 Furthermore, microplastics have been linked to strokes, and Huang et al. suggest that microplastics can induce cerebral thrombosis by causing cells to obstruct capillaries in the brain cortex. 41 While research in the field of MNPs is still in its early stages, these early results highlight the need to further explore the consequences of plastics on brain health.
Finally, when faced with the dire realities of climate change, it is somewhat unsurprising that psychological distress related to climate change has been documented across countries and generations.42,43 Qualitative studies reveal concerns that cover themes such as fear of environmental changes, grief over the loss of natural environments, and a sense of despair or hopelessness. 44 At sufficiently high levels, this distress can manifest as mental health disorders such as depression and anxiety. 45 Mental health is intricately linked with the progression and prognosis of neurodegenerative diseases. Depression can cause a significant decline in cognition, affecting domains such as memory, executive function, and language performance, 46 and has been independently linked with increased rates of dementia. 47 When depression and dementia co-occur, patients experience reduced quality of life, increased chances of early institutionalization, greater health care utilization, and higher mortality rates. 48 The intricate link between climate-induced psychological distress and neurodegenerative diseases underscores the urgent need for comprehensive strategies to address both environmental and mental health challenges.43,49,50
Current predictions indicate that many of these air pollution and climate change challenges, as well as, their consequent health effects will intensify over time, particularly impacting urban populations in crowded cities with large manmade structures. 51 Addressing climate change through policy and individual-level interventions is crucial. Global solutions to reduce emissions and pollutants are crucial, but adopting brain-healthy lifestyles—through diet, supplements, exercise, sleep, and stress reduction—can help mitigate and even reverse these effects. 52 This article will delve into these mechanisms and interventions, highlighting the connection between environmental factors and brain health.
The evidence-based plan
As described above, research shows that pollution and climate stress damage brain function, increasing the risk of cognitive decline, anxiety, depression, and cardiovascular, cerebrovascular, and pulmonary diseases.
This review offers an evidence-based plan to prevent and reverse these medical conditions, including proper diet, supplements, mental and physical exercise, stress management through yoga and meditation, and spiritual fitness. This concept of linking neurodegeneration and AD to environmental pollution and climate stress, alongside a preventive and healing plan, is unprecedented. Obviously since it is hypothetical but backed by other proven brain health programs hopefully future research will pointedly address this situation.
Published in 1997, the book Brain Longevity introduced a lifestyle medicine plan to prevent AD through a holistic approach. 53 This was markedly enhanced in 2017 with the first publication of the now world-famous FINGER study, from Finland, which has resulted in a multitude of significant positive papers and has been expanded to become World-Wide Fingers. 54
The participants in FINGER followed a healthy Nordic diet, which included fish, participated in physical and cognitive exercise, enjoyed social activity and managed vascular and metabolic risk factors. According to FINGER's lead investigator, Miia Kivipelto, MD, PhD, “The idea was that several risk factors needed to be targeted at the same time to get an optimal preventive effect.” Additionally, new FINGER-based studies will include stress management and meditation (Miia Kivipelto, 2024, personal communication).
Furthermore, FINGER outcomes suggested that after 24 months, global cognition was 25% higher than the control group. It also resulted in a 13–20% reduction in cardiovascular and cerebrovascular disease. 55 The study also indicated that individuals with more subjective memory complaints might experience greater benefits from the intervention.56–58 FINGER also assessed quality of life or essential well-being (EWB), which is a component of Spiritual Fitness, and found it to be enhanced. 59
In 2024, The Ornish Study, a randomized lifestyle medicine approach, suggested strongly that cognitive decline and AD may be prevented, and in some cases reversed. Ornish differed from FINGER in that it included a vegan diet as well as yoga, and meditation. It was one of the first randomized controlled trials to compare a whole foods plant-based diet with a control diet on early dementia. Amongst other plant-based diet observational studies, this study stands out because it also included strategic supplementation. The intervention participants were given these supplements: omega-3 fatty acids, curcumin, a multivitamin, coenzyme Q10, vitamin C, vitamin B12, magnesium L-threonate, lion's mane mushroom extract, and a probiotic—a total of 14 pills a day. 60
The study concludes that “comprehensive changes can significantly improve cognition and function after 20 weeks in many patients with mild cognitive impairment or early Alzheimer's disease.” 60
It demonstrated improvement in cognitive tests, cognitive function, and showed less progression relative to the usual care- control group. Additionally, although there was a significant improvement in the plasma Aβ42/40 ratio, the microbiome, and telomere measurements, no scans were utilized. 60 FINGER also did not use scans, and their telomere measurements suggested that subjects with the shortest telomeres at the beginning of the study had the most significant improvements in telomere length, thus signifying the importance of a multimodal program in boosting not only brain health but also an improvement in immune function and the possibility of enhanced longevity.
As suggested, research indicates that a lifestyle program may help in preventing neurodegeneration, memory loss, AD, prostate and cardiovascular diseases, stroke, anxiety, depression, diabetes, menopausal symptoms like brain fog, and sleep disorders. It's currently said that “what's good for the heart is good for the brain.” 61 Clearly, it's evident that the brain, mind, body, and spirit are interconnected. What impacts one impacts them all down to the cellular level via second and third messengers such as Cyclic CAMP, Cyclic GMP and hormones including cortisol, testosterone, estrogen, human growth hormone and various peptides. 62 Clearly health care costs can be reduced by the utilization of a lifestyle medicine program. 63
Nutrition
Brain-protective dietary patterns
There is more than one dietary pattern proven to protect the brain from AD and other neurodegenerative diseases. All the science-based brain-protective dietary patterns, thus far, however, are spin-offs of the Mediterranean diet (MeDi). The MeDi is characterized by a high intake of vegetables, beans and legumes, whole grains, fruit, fish and seafood, and unsaturated fatty acids (mostly in the form of olive oil), a low intake of dairy products, meat, and processed foods. The MeDi is also a lifestyle that includes enjoying meals with friends and family, moving naturally throughout the day, and socializing often.
Scarmeas et al. (2006) were the first to suggest that a community of people outside the Mediterranean countries could lower their risk of AD by following a MeDi diet. 64 In this longitudinal study of community-based individuals in New York, adherence to the MeDi diet reduced AD risk in a dose-response effect; the more closely participants follow a MeDi diet, the lower their risk of AD.
PREDIMED
One of the first randomized controlled nutrition intervention trials, the PREDIMED study in 2018, paved the way for looking at brain health through the lens of nutrition. 65 PREDIMED showed that following a MeDi diet could reduce the risk of cardiovascular events by as much as 30%. Previously, the PREDIMED-NAVARRA trial, a sub-study of the larger PREDIMED trial, showed a protective role in following a MeDi diet on cognition and brain health. 66 These findings lend strong support to the idea that while the MeDi could enhance longevity and protect from cardiovascular events, it may also prevent or delay age-related cognitive decline.
MIND
Dr Martha Clare Morris, a nutritional epidemiologist from Rush University who had been studying the impact of nutrition on brain health for over 20 years, and her colleagues created the first brain-specific MeDi spin-off. The MIND (Mediterranean-DASH Intervention for Neurodegenerative Delay) is a hybrid of the MeDi and the DASH (Dietary Approaches to Stop Hypertension) diet. 67 This dietary intervention was applied to 923 participants of the Rush Memory and Aging Project (MAP), ages 58 to 98, and followed for 4.5 years to assess cognitive health and the development of AD.
The MIND diet defines 10 brain-healthy food groups: berries, leafy greens, vegetables, nuts, fish and seafood, poultry, legumes, whole grains, olive oil, and red wine. The MIND diet differs from the MeDi one by carving out a few specific food groups as neuroprotective, namely berries and green leafy vegetables. In addition, the MIND diet is more specific than MeDi regarding which foods to limit or avoid: fast and fried food (up to one serving per week), pastries and sweets (up to five servings per week), butter (under one tablespoon per day), cheese (less than one ounce per week), and red meat (under four three-ounce servings per week).
Upon completion, the MIND diet results were published as two papers in 2015. In the first, those who followed the guidelines most closely reduced their AD risk by 53% after 4.5 years. Even those who ate from the diet just sometimes reduced their risk by 35%. And, in a separate paper, MIND diet eaters actually improved memory and thinking skills over time. 68 This was an astoundingly positive finding for an AD prevention study. Researchers determined that close adherence to the MIND diet slows cognitive aging by 7.5 years.
In a postmortem examination of MIND and MeDi eaters, researchers found that following either diet closely had almost 40% reduced risk of having either beta-amyloid or tau pathology. 69 In fact, close adherence to the MeDi resulted in brains that appeared 18 years younger at autopsy, while MIND diet guidelines yielded brains that looked 12 years younger.
The DIRECT-PLUS trial and green MED diet
The Green MED diet is another brain-specific spin-off of the MeDi, this time by boosting it with polyphenol-rich foods. Polyphenols are a class of bioactive substances in plant foods that perform specific brain health functions, such as reducing oxidative stress, enhancing blood vessel health, and nurturing a healthy gut microbiome.
The Green MED diet strategically includes these foods known to be high in polyphenols in a specific daily dose (number of servings): 3 to 4 cups of green tea, ¼ cup walnuts, and a smoothie made with a protein-rich leafy green. In addition, the Green MED diet emphasizes getting protein from plant sources instead of animal products and it specifically recommends avoiding poultry, red and processed meat.
This polyphenol-boosted MeDi diet was evaluated as part of the Dietary Intervention Randomized Controlled Trial Polyphenols Unprocessed Study (DIRECT-PLUS), a randomized controlled trial of metabolic and brain health comparing a traditional MeDi diet, a Green MED diet, and another healthful diet. 70 Participants were encouraged to exercise, limit meat consumption (especially processed meat), and were followed with cognitive testing and brain magnetic resonance imaging to assess total brain volume and hippocampal volume over 18 months.
The brains of the MeDi and Green MED diet followers both had significantly reduced brain shrinkage over 18 months. The group who maintained the most brain volume, however, was the Green MED group. Not only did they hold onto more gray and white matter, they had a more robust volume in the hippocampus. Researchers were able to demonstrate a linear positive effect on the hippocampus with each serving of polyphenol rich foods consumed.
The trial of the MIND
In 2015 when Dr Martha Clare Morris published data from the MIND diet study, plans were subsequently made to conduct a randomized controlled trial comparing the MIND diet with another healthful diet. The Trial for the MIND aimed to compare the MIND diet with another diet combined with other lifestyle interventions, tracking a group of participants over three years to assess changes in cognitive health. 71
For this new study, 300 people were asked to follow MIND diet guidelines, but with 250 fewer calories. Another 300 people—the control group—were asked to eat the way they usually do but also with the same reduction in calories. Each group met with a nutritionist at frequent intervals, and the MIND diet arm was also provided with blueberries, mixed nuts, and olive oil. Everyone was asked to exercise as they usually do.
All the study participants had measurable health gains by the end of the study—they all ate healthier, lost weight, and had improvements in brain health parameters. The MIND diet arm of the study started to break away from the control group at year 2 with higher cognitive scores, just as was seen in the FINGER study. 54 When the global pandemic disrupted the study resulting in a shift to virtual meetings with some participant dropout, researchers saw a general reduction in cognitive scores in both groups. By the study's completion, both groups’ cognitive function scores had started to improve but the MIND group had only a slight edge over the control group.
The brain health kitchen food pyramid
The Brain Health Kitchen Food Pyramid, published in The Brain Health Kitchen: Preventing Alzheimer's Through Food by Annie Fenn, MD (@Artisan Books 2023) reflects all the science-based brain-protective dietary patterns in one user-friendly graphic. It aligns most closely with MeDi and MIND diet guidelines, while reflecting recent data about the importance of consuming more fiber-rich and fermented foods and fewer servings of animal products. Red wine is dropped as a brain-healthy food group based on recent data that shows moderate drinking is associated with accelerated brain atrophy and there is no safe level of alcohol for the brain (Figure 1).72,73
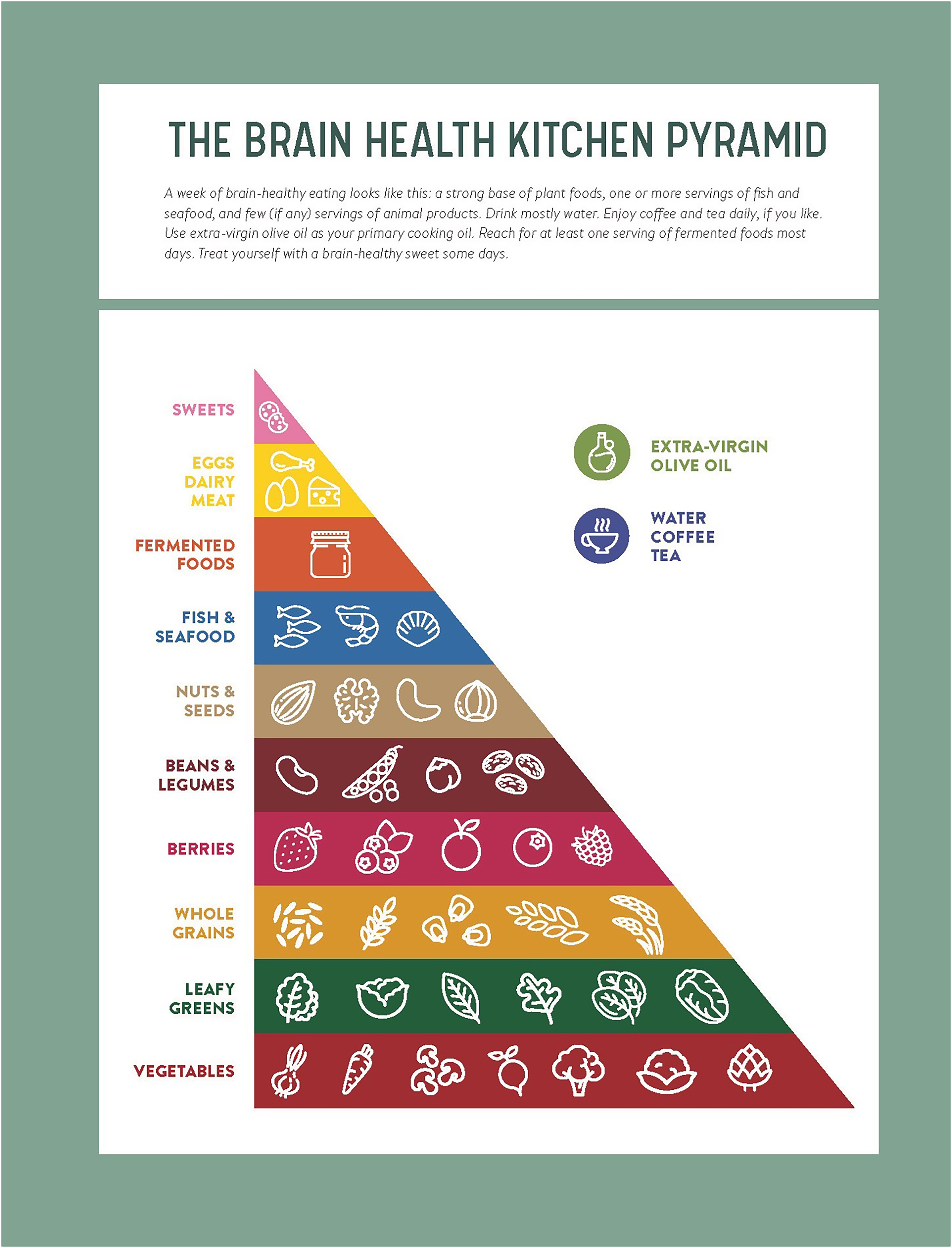
The brain health kitchen pyramid.
Instead, polyphenol-rich coffee and tea (especially green tea) make up the tenth brain healthy food group. This Brain Health Kitchen approach is inclusive of many dietary proclivities, including pescatarians, omnivores, vegetarians, vegans, whole-food plant-based, and traditional ethnic dietary patterns. Strategic supplementation is encouraged for those limiting or totally avoiding animal products. 74
Physical and cognitive exercise
It is known that frailty is a risk factor for AD as is reduced grip strength, balance and mobility. Recent studies indicate that a significant proportion of AD cases may be preventable through increased levels of physical activity. 75 Moreover, people who are more physically active can maintain cognition for a longer time than sedentary people. 76 In a large meta-analysis, including several cohort studies and 33,816 individuals, higher physical activity levels were associated with a significant reduction in the onset of dementia in a linear dose–response manner. 77 Additional research has also reported that exercise can delay the transition from mild cognitive impairment (MCI) to dementia. 77
Exercise was able to significantly improve global cognition and specific cognitive tests in MCI. 78 In people already affected by dementia, exercise improves global cognition because exercise improves the management of cardiovascular risk factors (e.g., diabetes, hypertension, dyslipidemia, and obesity), which are traditionally associated with poor cognitive performance. 79 Aerobic exercise is also associated with an increase in brain-derived neurotrophic factor, which stimulates neuronal cell growth and helps maintain neurons in an optimal status. 80 Moreover, using neuroimaging techniques, additional evidence for the impact of physical activity on brain function and structure has been reported. 81 Exercise is also a good predictor of long-term changes in brain structure, such as brain volume.82,83
For people with dementia, exercise interventions may also reduce the risk of disability, falls, and neuropsychiatric symptoms by approximately 31%. Since falls significantly contribute to disability, physical exercise can reduce fall risk and improve daily activities. Physical activity also reduces the presence and severity of neuropsychiatric symptoms, especially depression.
As seen above, physical exercise is critically important to maintain cognition even in frank AD. According to an article in Scientific Reports in 2023, exercising outdoors augments its many positive effects. 84 Due to rising pollution, it is important to check air quality before exercising outdoors, particularly for those with heart or lung conditions.
Exercise regimens have made great strides recently with the advent of high intensity interval training, the use of treadmills and elliptical machines and strength training regimens. Walking may be the best exercise for many to begin and stay active.
Beyond physical exercise, it is also clear that staying mentally engaged by incorporating brain exercises, or brain aerobics, results in positive brain changes and help in memory maintenance. Physical and mental exercises can be combined through awareness walking, which involves walking outdoors while being consciously aware of your surroundings.
Sleep
Restorative nightly sleep is a first-line defense against neurodegeneration, yet up to 50–70 million Americans live with a chronic sleep disorder such as insomnia or obstructive sleep apnoea. 85 Roughly half of adults aged 65 years and older report frequent sleep problems, and prevalence exceeds 70 % in some community samples.86,87 These already high background rates are aggravated by environmental pollution: each 10 µg/m³ rise in annual PM2.5 is linked to a 6-point reduction in sleep-quality indices and a 13 % higher obstructive sleep apnea incidence 88 ; long-term traffic noise above 55 dB elevates odds of insomnia and self-reported “high sleep disturbance” by roughly 25% 89 ; and wildfire smoke, nitrogen dioxide, and urban sky-glow shorten slow-wave sleep or fragment sleep architecture. 90
COVID-19 compounded the problem. Although early lockdowns briefly reduced outdoor traffic emissions, stay-at-home mandates increased time spent in poorly ventilated dwellings, heightened exposure to indoor volatile organic compounds and biomass cooking smoke, and unleashed unprecedented psychosocial stress—collectively producing the global surge in “coronosomnia” documented in 2020. 91
Mechanistic studies clarify why pollutant-induced sleep loss matters for dementia prevention. Deep slow-wave sleep facilitates glymphatic clearance of amyloid-β and tau; its erosion accelerates cortical deposition of these neurotoxic proteins.93,95 Sleep fragmentation also amplifies pollutant-primed inflammatory cascades: older adults with disturbed sleep exhibit up-regulation of IL-6, TNF-α, and NF-κB signalling. 94 These same inflammatory pathways are independently linked to cognitive decline and AD pathology.32,95,96 Prospective cohort data therefore converge on the conclusion that habitual short or poor-quality sleep doubles the risk of incident dementia.92,93
Pharmacologic responses may inadvertently worsen long-term brain health. Benzodiazepines and Z-drug hypnotics are used nightly by 11.9 % of U.S. women aged ≥ 80 years; chronic exposure nearly doubles AD risk and impairs memory via down-regulation of GABA-A receptors.97,98 Before initiating hypnotics, clinicians should first target modifiable environmental drivers—installing HEPA air filtration, minimizing nocturnal noise and light, and addressing evening screen use.
Lifestyle-medicine strategies offer non-pharmacologic leverage. The multimodal program detailed in this review, daylight physical activity, Mediterranean–MIND nutrition, strategic supplementation, and twice-daily Kirtan Kriya meditation—has been shown to normalize sleep architecture or improve insomnia severity. 99 Implemented together, these interventions can restore sleep, counteract pollutant-triggered inflammation, and thereby reduce downstream dementia risk.
Yoga and meditation: a brief history and modern research
To fully appreciate the concept of yoga and meditation, it is important to know that Dr Walter Hess won the Nobel Prize in 1949 for revealing two spots in the hypothalamus: a stress center and a relaxation center. 100
In 1975, Harvard researcher Herbert Benson, MD, demonstrated that anyone could activate the relaxation center by following 4 steps: a comfortable position, a quiet environment, a mental device, and focused concentration. He called this “the elicitation of relaxation response.” 101 Over four decades this simple technique has been shown to affect positively many parts of the body, mind, immune system, and genes.102–104 Further research indicates yoga and meditation offer numerous benefits and are part of advanced lifestyle medicine programs.60,105
The medical textbook The Principles and Practice of Yoga in Health Care covers multiple AD risk factors such as insomnia, post-traumatic stress disorder, chronic stress, anxiety, 106 depression, 107 cardiovascular disorders, diabetes, and neurological issues, 108 especially improving attention and verbal memory. Yoga may also improve cognitive functioning by enhancing mood, sleep, neural connectivity, and reducing stress, 109 which is a known risk factor for cognitive decline and was inextricably and mistakenly not included in the recent Lancet journal on AD risk. 63 Chronic stress is linked to cognitive decline and decreased personal well-being, mainly due to its connection with cortisol. This connection results in toxicity to the hippocampus and limbic system, as well as epigenetic damage. These pathological findings lead to neuronal inflammation, loss or decrease in synaptic transmission, and brain cell death in significant brain areas such as the hippocampus, which are crucial for maintaining high levels of cognition and perhaps preventing cognitive decline and AD. Outcomes include inhibition of memory consolidation, memory retrieval, and working memory all of which may lead to an increased risk for AD.
Like yoga, meditation has been studied in relation to disorders associated with AD risk.110–125 Kirtan Kriya, a very easy 12-min singing meditation, which includes specific sounds, as well as prescribed fingertip movements in sequence from the thumb to the little finger, has shown many positive effects. Kirtan Kriya involves singing these specialized sounds for 2 min out loud, 2 min in a whisper, 4 min silently, then 2 min whispering and 2 min out loud. The sounds activate the hypothalamus and pituitary gland, while fingertip touching stimulates the brain's homunculus, thereby significantly enhancing brain activity and providing optimal protection against neurodegeneration. 126
Kirtan Kriya has been researched for three decades at leading universities including UCLA, West Virginia University and The University of Pennsylvania, with findings widely published, as mentioned above. This work suggests many positive benefits on subjective cognitive decline, MCI, and early AD. Studies suggest that long-term Kirtan Kriya practitioners have more gray matter than non-meditators.114,118,127 Kirtan Kriya provides similar benefits to a comprehensive lifestyle medicine program with just 12 min of daily practice. According to PROTECT, a long-term study involving 1000 participants, the musical aspect of Kirtan Kriya may also have many positive effects on brain health. 128 Kirtan Kriya has been associated with improved telomere health, increased amyloid levels in plasma, suggesting lower amyloid in the brain, decreased inflammatory markers, and reversed memory loss.115,123
Multiple studies with Kirtan Kriya utilizing a variety of scans, along with cognitive and mental health evaluations, suggested positive changes in multiple brain areas associated with memory and mood including the dentate gyrus, anterior and posterior cingulate gyrus, hippocampus, amygdala, frontal lobe and pre-frontal cortex. 129 Synaptic transmission is also enhanced with important neurotransmitters such as acetylcholine, dopamine, serotonin also increasing as well as anandamide, the bliss chemical. This is significant because a decrease in synaptic transmission is a hallmark of AD. Moreover, our research suggests an activation of the brain's glymphatic system as evidenced by an increase in plasma amyloid concentration.120,130,131
A recent study, The Pink Brain Project conducted at UCLA, found that menopausal women with MCI and cardiovascular risk factors also showed increased gray matter, improved mood, and better memory after an 8-week yoga and Kirtan Kriya program compared to a Memory Enhancement Training control group. Kirtan Kriya lowered the age marker exotoxin-1, enhanced brain connectivity, and reduced inflammatory markers, aligning with our prior research.132–135
Spiritual fitness
As previously described by Khalsa, Spiritual Fitness, although once discussed by the US military, is a new concept in medicine combining multiple aspects of personal religious involvement (if there is one), psychological well-being, and spirituality or a search for the sacred. 136 Having a spiritual connection may reduce AD risk and slow its progression. 137 Spiritual Fitness has also been shown to reduce risk factors for AD including cardiovascular disease, strokes, and diabetes. It promotes healthy behaviors such as proper diet, exercise, and stress management, and reduces negative behaviors such as smoking, drug use, alcohol abuse, and is associated with enhanced cognitive function with age (Figure 2).138,139

Benefits of Kirtan Kriya chart.
Essential wellbeing
EWB, as enhanced by Kirtan Kriya and other aspects of a lifestyle medicine program, helps create a buffer against illness, inflammation, anxiety, depression, and cognitive decline.139,140
It includes the following:141–143
Spiritual Fitness integrates EWB with four new non-sectarian elements, promoting enhanced cognitive health and elevated consciousness.
Environmental wellness: our new future
This article explores the possibility that air and other aspects of environmental pollution and climate stress—resulting from climate change and potentially global warming—could accelerate the onset of AD at a younger age than traditionally believed. Recent policy changes, including the substitution of paper straws with plastic ones and various other actions that counter environmental protection efforts, may pose risks to both environmental integrity and human health. Autopsy studies, in fact, show that levels of microplastic and other nanoparticles in human brains are rapidly rising and beyond that, plastics were found in increasing numbers of the brains of AD patients. This evidence-based lifestyle medicine program promotes health and psychological well-being, creates spiritual fitness, prevents AD, and helps address environmental pollution and climate stress. By committing to global conservation efforts, we can address climate concerns and advance environmental sustainability for present and future generations. We do not have much time to lose.
Footnotes
Acknowledgements
We are grateful to Kirti Khalsa, CEO of Alzheimer's Research and Prevention Foundation for her help in manuscript preparation.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Kirtan Kriya Meditation studies and some of the FINGER studies were partially funded by The Alzheimer's Research and Prevention Foundation of which Dr Khalsa is President/Medical Director and receives a stipend. Dr Lavretsky currently receives grant funding from the NCCIH/NIH; NIA/NIH, Dept of Defense
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DSK is an Editorial Board Member of this journal but was not involved in the peer-review process of this article nor had access to any information regarding its peer-review. AF is the author of The Brain Healthy Kitchen, from which the pyramid comes. The remaining authors received no financial support for the research, authorship, and/or publication of this article.
