Abstract
Background
Cholinesterase inhibitors (ChE-Is), including donepezil, rivastigmine, and galantamine, and the N-methyl-D-aspartate receptor antagonist, memantine, are prescribed to decrease cognitive impairment symptoms.
Objective
This study examined prescriptions among patients with mild cognitive impairment (MCI) and/or Alzheimer's disease (AD) in the US by age, sex, and race/ethnicity.
Methods
This retrospective cohort study used Optum's de-identified Market Clarity Data containing electronic health records and insurance claims from January 2017 to September 2021. International Classification of Diseases, Ninth and Tenth Revisions diagnosis codes identified MCI and AD cases. Drug prescriptions were identified before, on, or after the date of first diagnosis. Cases were required to have ≥12 months of database enrolment prior to first diagnosis. Descriptive statistics were stratified by demographic groups.
Results
During the study period, 197,346 MCI and 144,321 AD cases were identified. Prescriptions were highest in patients with MCI aged 75–84 years before and after diagnosis for ChE-Is (8.1%, 20.4%) and memantine (2.6%, 7.8%) and among patients with AD aged 65–74 years for ChE-Is (25.0%, 38.7%) and memantine (11.2%, 22.1%). After AD diagnosis, the ChE-I prescriptions ranged from 31.3% of African American patients to 34.1% of Asian patients, and from 13.7% of African American patients to 18.5% of Hispanic patients for memantine.
Conclusions
ChE-I and memantine prescriptions generally increased to a certain age and were similar across sex and race/ethnicity groups. This information helps understand current prescriptions and how best to optimize in all demographic groups.
Keywords
Introduction
Mild cognitive impairment (MCI) and Alzheimer's disease (AD) constitute an enormous public health burden given both the number of cases and impact on patients and families. 1 In this study period, the most commonly prescribed treatments only alleviated symptoms of MCI and AD, being unable to stop the progression of neurodegeneration.1,2 The American Academy of Neurology guidelines state that although no empirical evidence demonstrates that these medications stop the progression of the disease, AAN leaves open the possibility that clinicians may offer medication treatments to their patients with proper consultation on the risks and benefits. 3 Published research has explored the utilization of available medications and their trends among different populations affected by AD,4–7 however, there is still a gap in assessing prescribed medications before and after the diagnoses of MCI and AD by age, sex, and race/ethnicity.
Three commonly used cholinesterase inhibitors (ChE-Is) in the United States (US) are donepezil (Aricept®), 8 rivastigmine (Exelon®), 9 and galantamine (Razadyne® ER [extended-release capsules, now discontinued but generic forms available]) 10 for the symptomatic treatment of AD in mild to moderate forms of the disease.11,12 The ChE-Is are usually choice of medication used in the early stage of AD. 13 They increase cell-to-cell communication by preserving depleted neurotransmitters in the synaptic cleft.11,12 The fourth medication commonly used for the treatment of AD is an N-methyl-D-aspartate receptor antagonist, memantine (Namenda XR®), 14 the medication of choice to be used later, in a moderate form of AD which prevents the excessive effects of glutamate produced by damaged neurons.11,15 A combination of memantine and donepezil (Namzaric®) 16 is used when patient transitions to moderate to severe AD 14 and delivers additional benefits to monotherapy, such as a simplified treatment regimen and reduced pill/swallowing burden. 17 A real-world analysis of treatment patterns within a two year period showed that treatment may change from initial monotherapy (either with ChE-Is or memantine) to a combination of the two or discontinuation of any treatment. 18
Previous research showed differences in the initiation and utilization of antidementia medication among race/ethnicity groups after the diagnosis of AD and related dementias (ADRD).5,7 The initiation of antidementia medication within 6 months after diagnosis of ADRD showed variability by race/ethnicity, with the highest initiation being among White patients and the lowest initiation among Hispanic patients. 5 On the contrary, in a study by Thorpe et al., 6 Hispanic patients with ADRD were more likely than white patients to initiate treatment. There was no significant difference in the likelihood of initiation between Black and White patients. In Thorpe et al. 6 and Olchanski et al., 7 adherence to treatment among patients with ADRD also varied by race/ethnicity, with both studies reporting higher discontinuation rates among non-Hispanic Black and Hispanic patients than non-Hispanic White patients.6,7
The aim of this observational retrospective cohort study was to characterize drug prescribing (ChE-Is and memantine) among patients with MCI and/or AD, in a large real-world database in the US and stratify by age, sex, and race/ethnicity.
Methods
This retrospective cohort study utilized insurance claims and electronic health records (EHRs) from Market Clarity for the period between 1 January 2017 to 30 September 2021 to identify a study population with MCI and AD and examine prescriptions of drugs to reduce the symptoms of cognitive impairment.
Data source
Market Clarity links medical and pharmacy claims with EHRs, including diagnosis codes and prescription information. At the time of analysis, data from more than 63 million people across the US with commercial insurance, Medicare, Medicaid, no insurance and those self-paying were included in this database.
Ethical approval and consent for publication is not applicable. Optum's de-identified Market Clarity Data deterministically links medical and pharmacy claims with electronic health record data from providers across the continuum of care. Market Clarity is certified as de-identified following the Health Insurance Portability and Accountability Act's statistical de-identification rules. The database is managed according to the Optum customer data use agreements and, therefore, this analysis is exempt from Institutional Review Board approval.
Participants
The study population included adults aged ≥18 years who were diagnosed with MCI and/or AD, identified using the International Classification of Diseases (ICD) codes (MCI – ICD-9: 331.83, ICD-10: G31.84; AD – ICD-9: 331.0, ICD-10: G30). To be considered a case, individuals had to have at least one corresponding ICD code in their EHRs or at least two diagnosis codes separated by at least 30 days in the insurance claims records.
The cohort entry date was defined as the date of first MCI and/or AD diagnosis within the study period. At least 12 months of enrolment (allowing for a gap of <45 days) was required for insurance claims before diagnosis. For EHRs, at least 12 months of active records prior to diagnosis was required. Individuals were able to be counted in both the MCI cohort and the AD cohort, assuming they met the required criteria for each of these diagnoses. Patients diagnosed with other types of dementia (i.e., Lewy body, frontotemporal dementia, vascular dementia, or dementia due to drug or medical conditions) were excluded and those with a general dementia diagnosis code occurring after their AD diagnosis were also excluded from the AD cases, as they could not be confirmed to be true AD cases.
Information on age on the date of diagnosis and sex were captured using insurance claims and EHRs. The variables of race and ethnicity taken from EHRs were combined to create the following categories: Hispanic (all races), Non-Hispanic Caucasian, Non-Hispanic African American, Non-Hispanic Asian, and Non-Hispanic Other/Unknown. These categories are referred to as Hispanic, Non-Hispanic Caucasian, African American, Asian and Other/Unknown in this publication.
Medication prescriptions
The medications analyzed in the study were as follows: 1) ChE-Is, including donepezil (Aricept®), galantamine (Razadyne® ER [extended-release capsules, now discontinued]), and rivastigmine (Exelon®); 2) memantine (Namenda®); and 3) memantine and donepezil (Namzaric®). These were identified in medical records and claims data using generic name, brand name and National Drug Codes (Supplemental Table 1).
Medication use in the 12 months before and 12 months after MCI or AD diagnosis were identified. Within electronic medical records, a prescription was noted when a healthcare practitioner administered or prescribed it. For insurance claims, prescribing information was not available, but reimbursement for a medication by insurance was used to establish that the medication was prescribed or administered.
Statistical analysis
Continuous data were summarized as mean (standard deviation [SD]) and median (min–max) while categorical variables were presented descriptively as frequency and percentages. The primary goal of the study was to describe the patient population and did not include comparative assessments; therefore, no statistical comparisons were performed. Descriptive statistics were calculated using SAS Studio 3.82 (SAS Institute, Cary, NC, US).
Results
Participant characteristics
Table 1 provides demographic information related to patients diagnosed with MCI and AD. The mean age at diagnosis was 79.3 years (SD 8.2) for AD compared with 70.0 years (SD 14.8) for MCI. The percentage of females diagnosed with MCI and AD was higher in this study population than that of males. The majority of individuals in the study with MCI and/or AD were Non-Hispanic Caucasian. Approximately 20% of study participants had Other/Unknown race/ethnicity. On average, individuals with MCI or AD had approximately 7 years of information in the database prior to their diagnosis and a mean of approximately two years of data after their diagnosis.
Baseline demographics of individuals with MCI/ AD, Market Clarity 1 January 2017–30 September 2021.
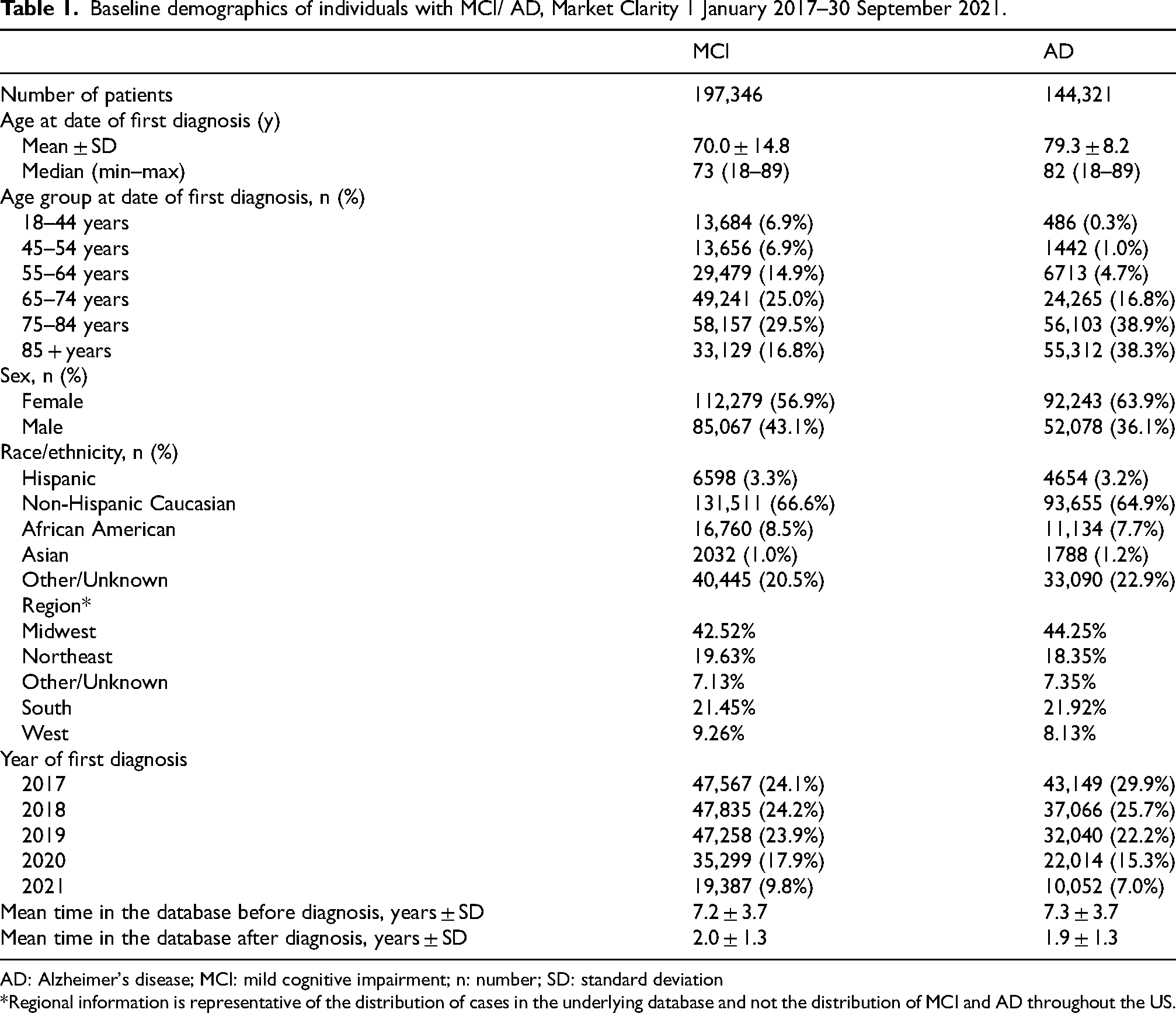
AD: Alzheimer's disease; MCI: mild cognitive impairment; n: number; SD: standard deviation
*Regional information is representative of the distribution of cases in the underlying database and not the distribution of MCI and AD throughout the US.
Medication prescriptions
Analysis of prescriptions among individuals with MCI in the 12 months before and after diagnosis demonstrated age-related trends (Figure 1(a)). ChE-Is were prescribed most frequently for patients aged 75–84, before, at, and after MCI diagnosis (Figure 1(a)). The proportion receiving ChE-I prescriptions was higher after diagnosis than before diagnosis for all age groups among individuals with both MCI and AD (Figures 1(a) and (b)). For example, in the 75–84 years group diagnosed with MCI, the percentage of patients with a prescription for ChE-Is was 20.4% after diagnosis and 8.1% before diagnosis (Figure 1(a)). Memantine was prescribed for fewer individuals than ChE-Is both before and after the diagnosis of MCI and AD but also demonstrated an increase following MCI and AD diagnoses (Figures 1(a) and (b)). In the age group with the largest proportion receiving a prescription, 2.6% of patients aged 75–84 years were prescribed memantine before MCI diagnosis and 7.8% received prescriptions after MCI diagnosis. For individuals diagnosed with AD, ChE-Is and memantine were prescribed most frequently in patients aged between 55 and 84 years (Figure 1(b)). In patients aged 65–74 years, the percentage of patients with ChE-I prescriptions were 25.0% before and 38.7% after diagnosis and the percentage of patients prescribed memantine were 11.2% before and 22.1% after AD diagnosis (Figure 1(b))
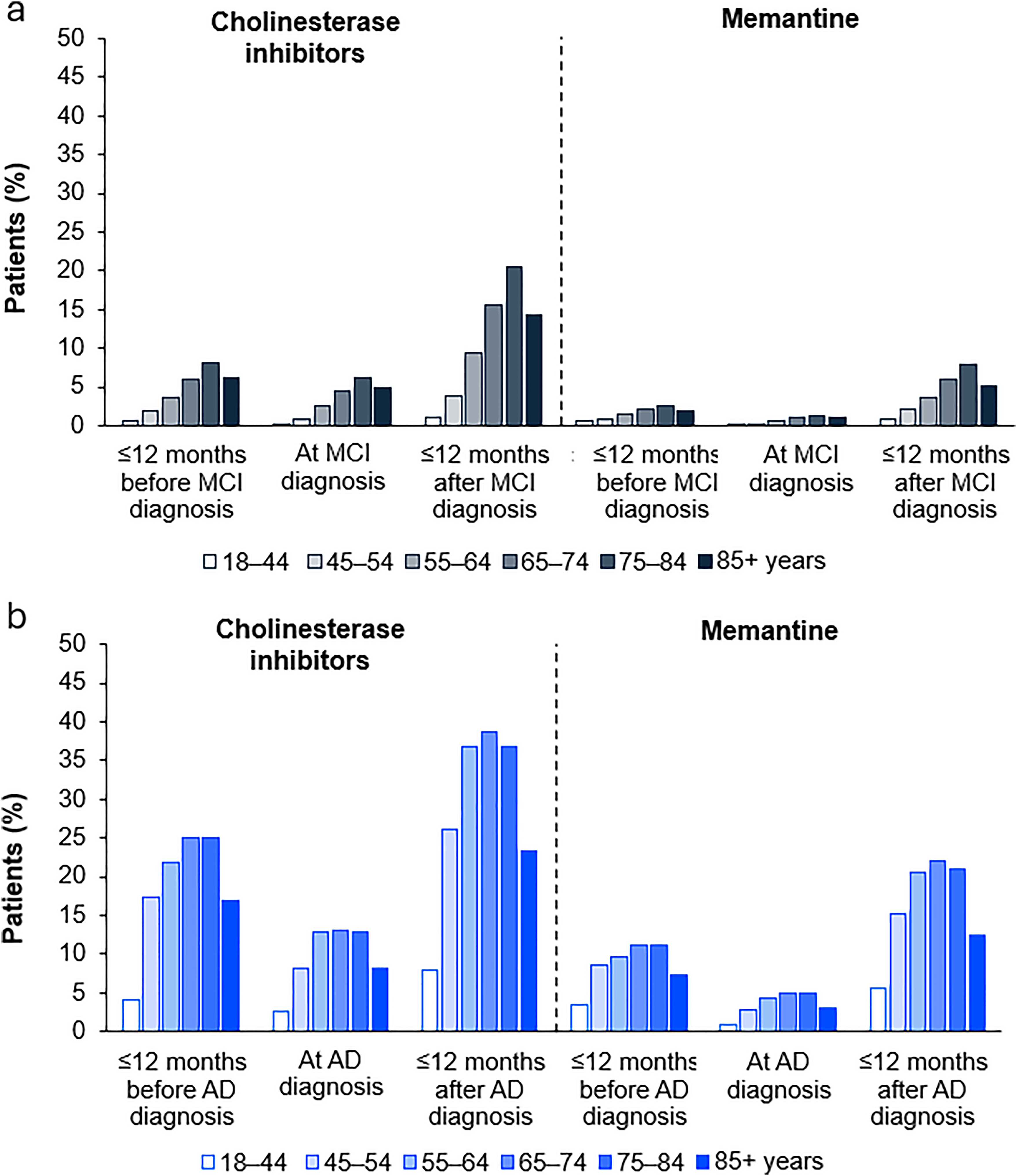
Prescription use stratified by age group among individuals diagnosed with MCI (a) and AD (b). AD: Alzheimer's disease; MCI: mild cognitive impairment.
When stratified by sex, the frequencies of ChE-I and memantine prescriptions were similar among males and females at all timepoints (Figure 2(a) and (b)).
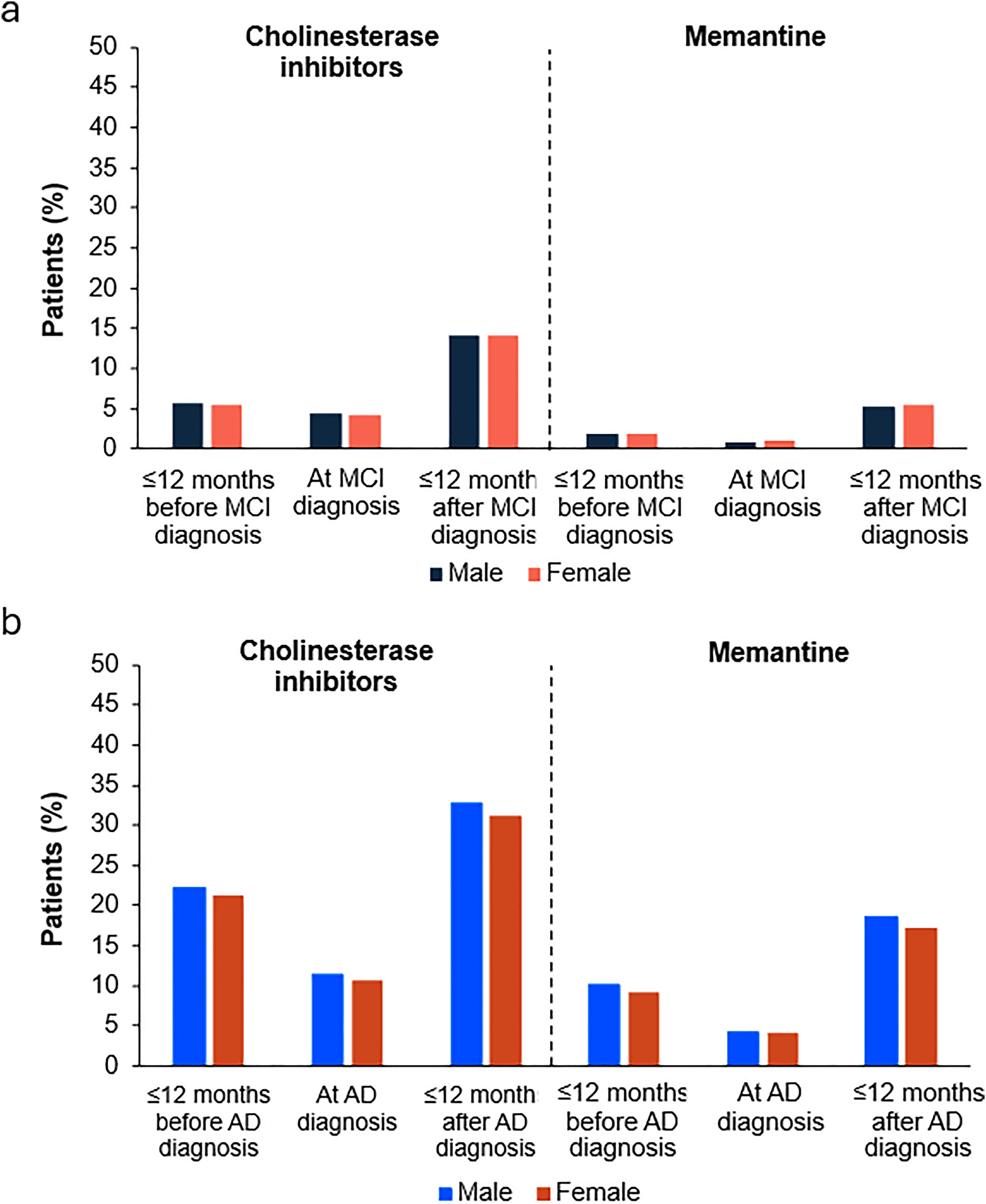
Prescription use among individuals diagnosed with MCI (a) or AD (b), stratified by sex. AD: Alzheimer's disease; MCI: mild cognitive impairment.
When stratified by race and ethnicity, proportions of patients with MCI or AD with prescriptions for ChE-Is or memantine were similar across all race/ethnicity groups. As observed among analyses stratified for age and sex, proportions of patients with prescriptions showed a pattern of being highest after diagnosis (Figure 3(a) and (b)). The percentage with ChE-I prescriptions prior to AD diagnoses was 22.3% in Non-Hispanic Caucasian patients, 22.1% in Hispanic patients, 21.0% in Asian patients, and 19.5% in African American patients. The percentage with memantine prescriptions was lower (10.0%, 9.6%, 8.8%, and 6.8%, respectively) than ChE-I prescriptions (Figure 3(b)). After AD diagnosis, ChE-I prescriptions were observed among 34.1% of Asian patients, 34.0% of Hispanic patients, 32.6% of Non-Hispanic Caucasian patients, and 31.3% of African American patients (Figure 3(b)). Memantine was prescribed among 18.5% of Hispanic and Non-Hispanic Caucasian patients, 17.1% of Asian patients, and 13.7% of African American patients (Figure 3(b)). Analysis of ChE-I and memantine prescriptions before and after MCI diagnosis by race/ethnicity showed a similar trend to AD, with lower proportions of patients receiving prescriptions before diagnosis (Figure 3(a)).
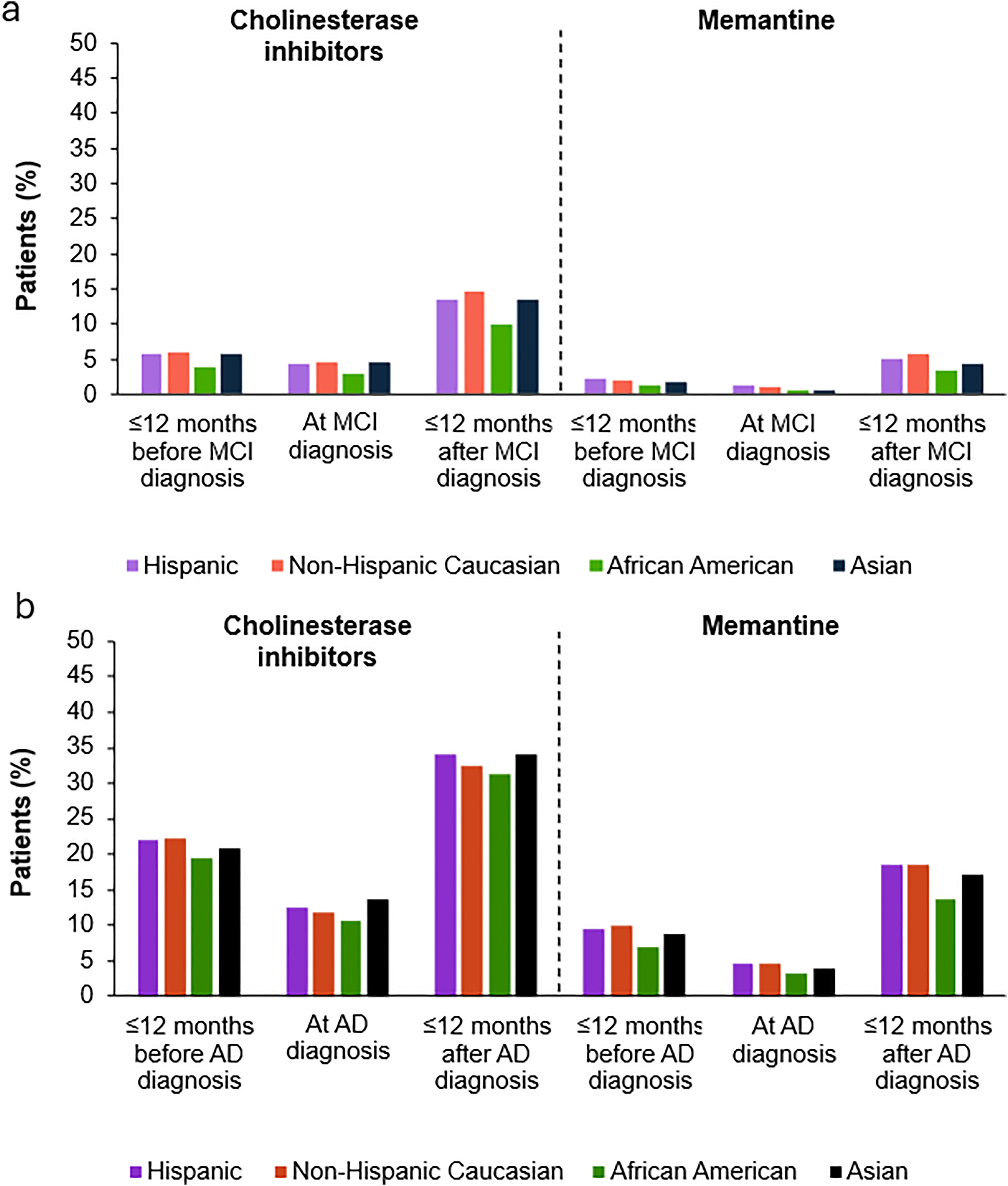
Prescription use stratified by race/ethnicity among individuals diagnosed with MCI (a) or AD (b). AD: Alzheimer's disease; MCI: mild cognitive impairment.
Use of combined memantine and donepezil (Namzaric®) was generally low, with the percentage of prescribed memantine and donepezil hydrochlorides reaching a maximum of 0.3% in any group of patients with MCI and 1.3% in any group of patients with AD (data not shown).
Discussion
This study demonstrates an increase in patient prescriptions for ChE-Is and memantine after the diagnoses of MCI and AD. In addition, prescriptions of ChE-Is and memantine generally increased with age and then decreased among the highest age group, 85 years and over. While not been approved for the treatment of MCI, this study observed that ChE-Is were prescribed to those with MCI.
The proportion of patients with MCI or AD receiving prescriptions was similar across sex and race/ethnicity groups.
Patterns of ChE-I use have previously been analyzed to assess sex-based treatment differences in older adults with dementia to optimize drug prescribing. 19 This population-based cross-sectional study in Canada found that 30% of participants with dementia were using ChE-Is (29.3% women, 28.6% men), and about 70% of those individuals with dementia were prescribed targeted therapeutic doses of ChE-I. 19 However, women were less likely to use the targeted therapeutic dose than men (67.8% women vs. 71.6% men, p < 0.001), and were more likely than men to concurrently use medication that impaired cognition such as antipsychotics or benzodiazepines while using medication to improve cognitive decline. 19 Our study results are similar, in that the proportion of patients with MCI or AD with a prescription were similar for men and women, and the proportions of patients with AD with a prescription for ChE-I were similar to those in Zhu's 19 study in individuals with dementia. In addition, a previous US study among Medicare patients with an ADRD or dementia-related symptom diagnosis found that women had similar odds for antidementia drug use to men (odds ratio [95% confidence interval] = 0.98 [0.97–0.99]). 5 Interestingly, a retrospective cohort study showed that adverse events related to the initiation of ChE-I therapy were related to the starting dose, especially a starting dose higher than recommended, and more commonly occurred in women (the number needed to harm [95% CI] was lower for women, 22 [18–29] vs. 36 [26–61] for men). 20 These findings indicate sex vulnerability and the need for attention while prescribing medications for AD and other dementias.
Studies have also been conducted assessing differences in the timing of antidementia treatment initiation after diagnosis among US patients of different race/ethnicity and sex groups.5,6 Analysis of Medicare claims (2008–2016, N = 613,970) showed that 33.3% of individuals diagnosed with ADRD or dementia-related symptoms used approved medication for AD during the study period, 16% prior to diagnosis, and 40% within 6 months after the diagnoses. Disparities were observed in this study regarding the initiation of AD medication prior to the diagnosis among race/ethnicity groups: Asian 28%, Hispanic 24%, African American 16%, and Non-Hispanic Caucasian 15%. 5 These findings differed from our study which showed the highest rates of medication prescription (ChE-Is and memantine) prior to AD diagnoses recorded in Non-Hispanic Caucasian patients and the lowest rates in African American patients, although statistical testing was not performed. In another previous study utilizing US Medicare and Medicaid claims linked to survey results, the initiation of medication was low across all race/ethnicity groups, with two-thirds of ADRD-diagnosed patients not having prescriptions for ChE-Is or memantine during the first year following diagnosis and only half of patients with a specific AD diagnosis using antidementia medication. 7 In addition, poor adherence to antidementia medication was observed among all race/ethnicity groups, adherence being higher for African American and Hispanic patients than for Non-Hispanic White patients (50% and 51% versus 42%, p = 0.01). 7 Discontinuation of the treatment was also higher among African American patients (32%) and Hispanic patients (27%) versus Non-Hispanic White patients (21%, p = 0.001). 7 It is noteworthy that in the study, Hispanic and African American patients had lower education, lower income, lower cognitive function, and more functional limitations at ADRD diagnosis compared with Non-Hispanic White patients (all p < 0.01), however, discontinuation rates did not differ between patient groups once patient characteristics were controlled for. 7 Another retrospective study using a sample of Medicare beneficiaries (N = 1,478,852) assessed the initiation and discontinuation of antidementia medications by race/ethnicity and did not find a difference in treatment initiation between Non-Hispanic Caucasian and African American patients. 6 Hispanic patients were more likely to start using medication than Non-Hispanic Caucasian and African American patients; however, Hispanic and African American patients showed faster rates of discontinuation of the treatment than Non-Hispanic Caucasian patients. 6
While memantine has been shown to improve cognition, it is licensed and recommended in moderate to severe stages of AD.15,21 The findings of this study showed that some patients with MCI received prescriptions for memantine before and after diagnosis, albeit at lower proportions than patients with AD, despite memantine not being indicated in the management of MCI. The clinical complexity of AD may justify a treatment combination with different medications but despite the clinical practice of starting memantine at an early stage of AD, there is no evidence of its clinical benefit in mild AD compared with moderate-to-severe AD. 21 This may be due to either a lack of recorded earlier diagnosis, a milder disease severity being included in the medical records, or it could also be a sign of individuals being prescribed medications at an earlier stage than appropriate.
Strengths and limitations
A benefit of this study database is the use of medical records to identify use and timing of medications instead of self-report among individuals with cognitive impairment or their caregivers. While a strength of this study was the large number of participants from across the US, a limitation was that some regions were represented more than others, preventing generalizability of the study population. Also, diagnoses of cognitive impairment were those that were recorded with diagnosis codes in insurance claims and EHRs; individuals who were experiencing these symptoms but did not receive an actual diagnosis from a physician were not included in this study. The current study does not have information on results of neuropsychiatric tests or imaging that would allow understanding of the stage of individuals at time of starting prescription drugs, regardless of whether it is before or after a formal diagnosis. Another limitation is that the study did not capture information on prescribed medication adherence or cause of treatment discontinuation. It was not possible to differentiate if treatment discontinuation was due to access or effectiveness/tolerability issues. Finally, the study determined that the proportion of the patient population prescribed medications after diagnosis but did not look at the change in prescriptions over time or the reasons for stopping or starting medications. The changes in use may be due to medication efficacy or adverse events. Although prescribed off-label for cognitive impairment, the progression to AD can discourage patients. Hogan 22 emphasized the importance of considering the adverse events in long-term treatment, individualized approaches for older patients, and discontinuation in severe dementia.
Of note, the number of diagnoses declined in the last two years of the study. This study did not plan to explore changes in diagnosis over time. This may be due to changes during the COVID pandemic but additionally the final year of the study only included the first nine months of the year and therefore would be expected to have a lower count of newly diagnosed cases.
Conclusions
This study provides an insight into currently approved symptomatic cognitive treatment trends in patients with MCI and AD in the US, before, at, and after these diagnoses. The study observed utilization of medication in the time period before the diagnosis of MCI or AD. The proportion of individuals prescribed memantine was lower than the proportion prescribed ChE-Is. Prescriptions of ChE-I and memantine increased after diagnosis of MCI and AD across all age, sex, and race/ethnicity groups. ChE-I and memantine prescriptions were observed in similar proportions among men and women diagnosed with MCI and AD and proportions of individuals with prescriptions tended to increase with age until 85 years. these findings have relevance in clinical practice and highlight the need to raise awareness of potential prescription differences and appropriate timing of initiation among physicians, patients, family members and caregivers. Future research into the difference of prescriptions by sex and race/ethnicity, as well as the timing of, and adherence to, certain prescriptions will be valuable in further understanding the patient populations and optimizing the use of medications for MCI and AD.
Footnotes
Acknowledgements
Medical writing and manuscript submission support was provided by Belen Gonzalez, PharmD, of AbbVie, according to Good Publication Practice guidelines. The listed authors have authorized the submission of their manuscript via a third party and have approved any statements or declarations.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AbbVie funded this study and participated in the study design, research, analysis, data collection, interpretation of data, reviewing, and approval of the publication. All authors had access to relevant data and participated in the drafting, review, and approval of this publication. No honoraria or payments were made for authorship.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employees of AbbVie and may hold AbbVie stock/options.
Data availability statement
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
