Abstract
Background
Olfactory dysfunction and neuropsychiatric symptoms are well-established hallmarks of Alzheimer's disease (AD) and serve as predictors of progression from mild cognitive impairment to AD. However, the literature remains unclear on whether olfactory dysfunction is already evident in the prodromal form of subjective cognitive decline (SCD).
Objective
This study aimed to characterize olfactory function and neuropsychiatric symptoms, including anxiety and depression, in individuals with SCD compared to healthy controls (HC).
Methods
A total of 110 community-dwelling participants aged 60 years and older were recruited, with 59 classified as SCD (42 women) and 51 as HC (35 women). Participants completed validated anxiety and depression questionnaires (Geriatric Anxiety Inventory and Geriatric Depression Scale) and underwent a comprehensive olfactory assessment using the complete Sniffin’ Sticks battery (odor threshold, discrimination, and identification).
Results
The SCD group exhibited lower global olfactory scores (small to moderate effect size; Cohen's d = 0.41), and higher depression (large effect size; Cohen's d = 1.07) and anxiety (moderate effect size; Cohen's d = 0.64) scores compared to HC. Notably, global olfactory performance enhanced group status prediction and classification (SCD versus HC) when combined with anxiety and depression measures.
Conclusions
These findings highlight subtle but significant lower olfactory performance in individuals with SCD. The neurophysiological underpinnings are unclear and may be unveiled with electrophysiological methods such as olfactory-evoked potentials.
Introduction
Dementia poses an increasing burden on society, affecting over 50 million people worldwide, with projections indicating that this number will increase to 152 million by 2050. 1 Of these cases, it is estimated that 60 to 80% are, or will be, at least partially attributable to Alzheimer's disease (AD). 2 AD is a neurodegenerative disorder characterized by the presence of extracellular amyloid-β (Aβ) plaques and intracellular neurofibrillary tangles of tau protein. 3 The accumulation of these factors and others (e.g., mitochondrial dysfunction) is believed to trigger a gradual and substantial loss of synapses and neurons in the brain, ultimately resulting in cognitive and behavioral changes. 4 In its most prototypical form, episodic memory dysfunction is a defining feature of AD and often among the earliest cognitive symptoms, while impairments in learning, language, executive function, and visuospatial abilities typically become more evident as AD progresses.5,6 In turn, behavioral changes, such as depression and anxiety, are common and usually emerge early in the onset of AD. However, these symptoms tend to diminish as cognitive functions decline. In contrast, agitation, paranoia and hallucinations tend to increase.7,8
Neurodegeneration caused by AD typically progresses over more than a decade, leading to distinct stages of clinical and pathological development. 9 The accumulation of tau neurofibrillary tangles first impacts the transentorhinal cortex and entorhinal cortex, spreading to the limbic system, including the hippocampus and eventually reaching the entire cortex. 10 This pattern of atrophy aligns with the clinical progression of the disease. 11
Mild cognitive impairment (MCI) is defined by measurable deficits in at least one cognitive domain on standard neuropsychological testing, while daily functioning remains largely intact. 12 This stage is recognized as a clinical condition that represents a transitional phase between normal cognitive aging and dementia.13,14 In fact, between 6 and 47% of individuals with MCI will convert to dementia within the following four years. 15 Of those, the majority will develop AD.16,17 The conversion to AD compared to other neurodegenerative diseases is better predicted when cognitive deficits predominantly affect episodic memory—a condition known as amnestic MCI. 18 This stage has therefore been the focus of significant research due to its importance in early detection and intervention. However, even at this stage, underlying neurodegenerative processes have already advanced considerably. 19
More recently, there has been a growing effort to identify individuals at risk of developing AD earlier, with the goal of enabling more effective management and earlier intervention, including the exploration of potential drug therapies. 20 To this end, subjective cognitive decline (SCD) has been established, an even more preclinical stage of AD. Two central features define SCD: (1) a self-reported, persistent decline in cognitive function, unrelated to any acute incidents, and (2) normal results on standardized cognitive tests typically administered to diagnose MCI, adjusted for sex, education, and age.21,22 As such, the individual's cognition is unimpaired from an objective standpoint. A meta-analysis indicates that within five years, 27% of individuals identified with a SCD will progress to MCI, while 14% will develop dementia. Similar to amnestic MCI, subjective memory impairment rather than in other cognitive domains increases predictive validity of SCD to preclinical AD.21,23 Nevertheless, the specificity of SCD is limited.24,25 Better characterizing SCD is important for establishing a baseline profile that can later be used to predict which individuals are more likely to progress to MCI or AD.
One possible avenue is the evaluation of sensory impairment and affective changes that predate memory dysfunction in AD. 26 Olfactory impairment appears to offer the greatest potential for predictive insights, compared to other sensory modalities like vision and hearing. 27 A possible explanation for the advantage of olfaction over other sensory modalities is its anatomical proximity and interconnection with brain regions implicated in memory function, such as the entorhinal cortex, and hippocampus, which are among the first areas to be affected by neurodegeneration in AD.26,28
Olfactory function is generally assessed by standardized tests such as the Sniffin’ Sticks, which measure threshold, discrimination, and identification or by the University of Pennsylvania Smell Identification Test (UPSIT) which focuses on odor identification.29,30 Olfactory impairment is very common in AD, with nearly 90% of individuals experiencing difficulties in olfactory identification. 31 Additionally, there is a high prevalence of deficits in olfactory discrimination and threshold, further highlighting the extent of olfactory dysfunction associated with the disease. 32 Olfactory changes manifest very early and are readily apparent in individuals with MCI. A meta-analysis revealed that individuals with MCI show greater olfactory impairments across all olfactory domains compared to controls, with olfactory identification showing the most significant deficits. 33 Multiple studies indicate a rise in olfactory impairment through disease stages, with olfactory performance emerging as a significant predictor of verbal episodic memory deficit.26,34–36 Furthermore, olfactory identification more reliably predicts conversion from MCI to AD than verbal episodic memory, underscoring the importance of measuring olfaction as a biomarker. 37
Olfactory deficits in AD and MCI are substantiated by both neuroanatomical and neurophysiological evidence. Individuals with MCI and AD show a decrease in the volume of the primary olfactory cortex and the olfactory bulb, which correlates with reduced olfactory function and impaired activity in olfactory regions.38–40 Further, AD and MCI affect the latencies 41 and amplitudes 42 of olfactory event-related potentials (OERP).
In turn, studies on olfactory function are very scarce in SCD. Olfactory identification is slightly altered in SCD, but the confidence interval included an effect size of 0 in a meta-analysis, 43 in line with several studies not having found any significant differences in olfactory function.44,45 One possible explanation is that olfactory dysfunction at this stage may be mild, requiring more sensitive measures to detect it. Such additional measures may include olfactory discrimination and/or olfactory threshold measurements. In MCI, although olfactory identification shows the greatest decline, both discrimination and threshold also exhibit important reductions. 33 To our knowledge, no studies have assessed olfactory function in SCD combining threshold, discrimination, and identification (TDI). This approach offers a more accurate reflection of overall olfactory function, while retaining the ability to explore individual components without losing valuable variability. 46 Additionally, although olfactory identification is the most studied component of olfaction in relation to AD, combining it with other olfactory tasks enhances the specificity of olfaction for predicting transitions between cognitive states.47,48
Similar to olfactory dysfunction, anxiety and depression emerge early in the development of AD and increase the risk of progression from MCI to dementia.49,50 These neuropsychiatric symptoms may signal early neurobiological changes associated with AD. 51 In fact, the limbic system is essential for emotional regulation and comprises several key brain regions that are significantly impacted by neurodegeneration and are intricately connected to both memory and olfactory function.10,52 Individuals presenting with SCD or MCI combined with elevated anxiety and/or depression are more likely to progress through stages of cognitive decline.55–57 Depression, in particular, shows robust associations with declines across multiple cognitive domains, including episodic memory, executive function, attention, language, and global cognition. 58 Prevalence of these symptoms has been well established in both MCI and SCD.55,59 Identifying and characterizing neuropsychiatric symptoms such as anxiety and depression in individuals with SCD is therefore crucial, as early intervention may mitigate the impact on cognitive function and delay or influence progression to MCI or dementia. Furthermore, anxiety and depression can exacerbate cognitive deficits and interfere with treatment adherence and outcomes.53,54 However, few studies have investigated the interplay between neuropsychiatric symptoms and olfactory function in SCD. Individuals diagnosed with major depression and SCD scored lower on measures of global olfaction, threshold, and discrimination, but not of olfactory identification. 60 Among community-dwelling adults without cognitive deficits, both depression and impaired olfactory identification independently predict an increased risk of dementia over a 12-year period, with their co-occurrence further amplifying this risk. 61 Furthermore, individuals with late-life depression and olfactory dysfunction exhibit more severe cognitive impairment—particularly in memory—and brain structural abnormalities similar to those observed in AD, compared to those with late-life depression but without olfactory dysfunction. 62 Olfactory assessment may thus provide a valuable clinical marker to differentiate late-onset depression from early-stage dementia. Nonetheless, findings remain mixed. Recent research involving populations with MCI and SCD reported no significant differences in olfactory identification between depressed and non-depressed groups. Remarkably, in the same study, patients with AD and depression performed better on olfactory tasks than those without depression, whereas cognitively healthy individuals with depression performed worse than their non-depressed counterparts. 63 These conflicting findings underscore the complex relationship between affective symptoms and olfactory function and highlight the need for further, and more detail investigation.
Symptoms of depression and anxiety are closely intertwined with SCD, as they are integral to its manifestation and play a significant role in its characterization. 22 Olfactory dysfunction may therefore provide additional insights into the features of SCD. Further research is essential to determine whether olfactory dysfunction can serve as a complementary marker at this stage, particularly given that individuals with SCD who exhibit elevated neuropsychiatric symptoms and olfactory deficits may be at increased risk of progression to MCI or AD. Notably, the assessment of these markers can be easily integrated into clinical practice through non-invasive and cost-effective measures.
To clarify the questions we raised, we aimed to characterize olfactory function and neuropsychiatric symptoms including anxiety and depression in individuals with SCD compared to HC. We hypothesized that: (1) individuals with SCD exhibit lower olfactory function and higher levels of anxiety and depression compared to HC; (2) anxiety and depression scores predict group status (SCD versus HC) and (3) when combined with anxiety and depression, olfactory function strengthens group status prediction.
The novelty of this study lies in its detailed behavioral assessment of olfactory function—including threshold, discrimination, and identification—while also evaluating and accounting for anxiety and depression in community-based sample of individuals with SCD. This aligns with current perspectives emphasizing the importance of multidimensional strategies for early detection.
Methods
Participants
A total of 115 participants aged 60 years and older were recruited on a voluntary basis from the community in Trois-Rivières, Quebec, Canada and surrounding regions. Recruitment was conducted through multiple channels, including a database maintained by the Chemosensory Neuroanatomy Laboratory at Université du Québec à Trois-Rivières, as well as social media announcements, word-of-mouth referrals, and outreach to organizations with members in the targeted age group. The process aimed to reach individuals with mild memory concerns as well as older adults from the general population. All participants with memory concerns were self-referred and were not contacted through the laboratory database.
The inclusion criteria were as follows: individuals aged 60 and above, with no history of concussion or loss of consciousness, no nasal polyps or history of nasal surgery, no congenital anosmia, and no mental illnesses or neurological disorders. We assessed inclusion criteria and verified them at three distinct time points: initially through instructions sent via email, then through an online sociodemographic questionnaire, and finally during the initial in-person meeting to confirm continued eligibility across all criteria. We assessed global cognitive abilities with the Montreal Cognitive Assessment (MoCA) and excluded participants with scores below 26, adjusted for education, as they would be categorized as MCI. Of the 115 participants, 110 met these criteria and continued with the study, while 5 were excluded due to potential signs of MCI.
A final sample of 110 participants (77 women) aged 60 to 79 years (mean age = 67.3 years, SD = 5.0 years) successfully completed the study. Participants were instructed to refrain from wearing perfume and to avoid eating, drinking, or smoking for one hour prior to their testing session. The classification for SCD was based on established criteria 21 which define SCD as a self-perceived persistent decline in cognitive function, unrelated to any acute incidents, and characterized by normal results on standardized cognitive tests typically used to diagnose MCI—in this case, the MoCA. As a result, 59 participants (42 women) were classified into the SCD group due to their reported memory concerns, while 51 participants (35 women) were assigned to the HC group, which consisted of individuals with no reported cognitive concerns.
All participants provided written informed consent prior to testing and received $10 per hour as financial compensation and their parking fees were covered by the laboratory. This study was approved by the Ethics Committee for Research Involving Humans at Université du Québec à Trois-Rivières.
Questionnaires
SCD memory complaints criteria
Individuals were considered to have SCD status if they responded “yes” to the question, “Do you have memory complaints?”. Memory complaint questions are the most predictive for future progression to amnestic MCI or AD, particularly when distinguishing these conditions from non-AD dementias.64,65 This single question has been effectively employed in studies assessing SCD. 66
Geriatric anxiety inventory
We used the validated French version of the Geriatric Anxiety Inventory questionnaire (GAI) to measure levels of anxiety. This questionnaire was designed by the authors to limit confusion between somatic symptoms due to anxiety and those due to other health problems or normal aging. This questionnaire consists of 20 items to which participants must indicate agreement or disagreement. The sum of each “agree” response indicates the score out of 20. A score above 9 suggests the presence of clinically significant anxiety; this threshold was used as the cut-off in our study.67,68
Geriatric depression scale
To measure our participants’ level of depression, we used the validated French-Canadian version of the Geriatric Depression Scale (GDS). This questionnaire was designed to limit confusion between somatic symptoms due to depression and those due to other health problems or normal aging. This questionnaire consists of 30 items to which participants must indicate agreement or disagreement. The sum of each “agree” response indicates the score out of 30. A score above 11 suggests the presence of mild depression; this threshold was used as the cut-off in our study.69,70
Cognitive assessment
Montreal cognitive assessment (MoCA)
To assess our participants’ overall cognitive abilities and ensure that they did not meet the criteria for MCI, we administered the French version of the MoCA. This test was designed as a screening tool for MCI. It generates a total score and six domain-specific index scores: (1) Memory, (2) Executive Functioning, (3) Attention, (4) Language, (5) Visuospatial, and (6) Orientation. 71
Olfactory assessment
Sniffin’ sticks test battery
To comprehensively evaluate participants’ sense of smell, the Sniffin’ Sticks Test Battery was employed. This battery encompassed multiple tests designed to assess different aspects of olfactory ability. The Sniffin’ Sticks have been validated across various cultures and is widely and is used in clinical and research settings. 72 They offer good reliability, with test–retest correlations of up to 0.92 for odor threshold, 0.69 for odor discrimination, 0.86 for odor identification and 0.91 for the total score. 73 To determine the olfactory threshold score, participants were blindfolded and presented with sets of three pencils under their nose and tasked with identifying the scented pencil among the three options. These pencils were organized into triplets, where triplet 1 had the highest concentration of odor and triplet 16 had the lowest. By utilizing a staircase procedure, the participant's threshold was determined. For evaluating discrimination abilities, participants were blindfolded and exposed to 16 triplets of scented pencils. Each triplet consisted of two pencils with similar odors and one pencil with a different odor. Participants received a score out of 16, with one point deducted for incorrectly identifying the pencil with the different odor. To assess odor identification ability, participants were presented with 16 pencils, each infused with a unique odor. With their eyes uncovered, participants had to identify the specific odor associated with each pencil by selecting from four response choices. A score out of 16 was obtained, with one point deducted for each incorrect identification. The total score — the TDI score — was obtained by summing the scores from each sub-test of the olfactory assessment battery, yielding a maximum possible score of 48.72,74
Procedures
After participants expressed their interest, we sent them an email with the instructions of the Geriatric Depression Scale (GDS), Geriatric Anxiety Inventory and questions about memory complaints and socio-demographic information (e.g., age, education, etc.) via the Lime Survey platform. Before they could access the questionnaires, participants were asked to read an information letter and provide their consent to participate. Participants were subsequently invited to the Chemosensory Neuroanatomy Laboratory at Université du Québec à Trois-Rivières to complete the cognitive and olfactory assessments, provided they still met the eligibility criteria.
During the testing session, participants were not discouraged from making spontaneous remarks, including those concerning their cognitive health. Importantly, experimenters—primarily trained research assistants—were blinded to group assignment, questionnaire responses, and study hypotheses. After obtaining written consent, the MoCA was administered, followed by the Sniffin’ Sticks battery. The session lasted approximately 1 h and 30 min. Participants who scored below the specified threshold (below 26) on the MoCA were excluded from the study. They received a brief letter outlining their MoCA score and a recommendation of a more thorough cognitive evaluation. Participants were encouraged to share this letter with their healthcare provider or seek a second opinion from another professional.
Statistics
Statistical analyses were conducted using R version 4.4. To examine group effects (SCD versus HC), we computed independent samples t-tests for education, age, global cognition, global olfaction (TDI score), anxiety (GAI score) and depression (GDS score), as well as a chi-square test for sex. We calculated effect sizes using Cohen's d for continuous variables and Phi for categorical variables (e.g., sex). We interpreted Cohen's d values of 0.20, 0.50, and 0.80 as small, medium, and large effects, respectively. 75 We interpreted Phi values using the same criteria: 0.10 for a small effect, 0.30 for a medium effect, and 0.50 for a large effect. 75 The frequency of participants with scores above the cut-off—9 on the GAI and 11 on the GDS, indicating clinically significant of anxiety and/or mild depression—was specified for each group. We performed a mixed ANOVA to assess potential interactions between group and olfactory task (threshold, discrimination, identification) reporting partial eta squared (η²) as the effect size. Values of 0.01, 0.06, and 0.14 were interpreted as small, medium, and large effects, respectively. 75 Additionally, we performed a hierarchical logistic regression model to predict group classification. The first block included anxiety and depression scores as predictors, while the second block added TDI scores. We reported Nagelkerke's R² as an index of model fit in the logistic regression analyses, with values closer to 1 indicating a better fit. 76 Receiver Operating Characteristic (ROC) analyses were conducted to assess the classification performance of each predictor and regression model. We evaluated several classification metrics to assess the models’ and predictors’ performance, including the Area Under the Curve (AUC) for overall accuracy, sensitivity, and specificity for identifying true positives and true negatives respectively; and Positive Predictive Value (PPV) and Negative Predictive Value (NPV) to assess the predictive value of the model's outputs. We calculated likelihood ratios, with Positive Likelihood Ratios (LR+) values above 5 indicating moderate, and above 10 strong, diagnostic value, while Negative Likelihood Ratios (LR−) between 0.1 and 0.3 suggested moderate, and below 0.1 strong, effectiveness in ruling out a condition. 77 We also reported Youden's index, with values ranging from 0 to 1, where higher values reflect better discriminative ability. 78
Results
We observed a difference in olfaction, with the SCD group exhibiting significantly lower TDI scores than the HC group (t(101.53) = 2.19, p = 0.03, Cohen's d = 0.41; see Figure 1). Additionally, participants with SCD reported significantly higher levels of anxiety (t(97.59) = 3.46, p < 0.001, Cohen's d = −0.64) and depressive symptoms (t(88.80) = 5.86, p < 0.001, Cohen's d = −1.07) compared to HC participants. In the SCD group, 4 participants (9%) scored above the GAI cut-off of 9, indicating elevated anxiety, and the same number (9%) exceeded the GDS cut-off of 11, suggesting mild depressive symptoms. In the HC group, 2 participants (4%) were above the GAI cut-off, while 1 participant (2%) exceeded the GDS cut-off.
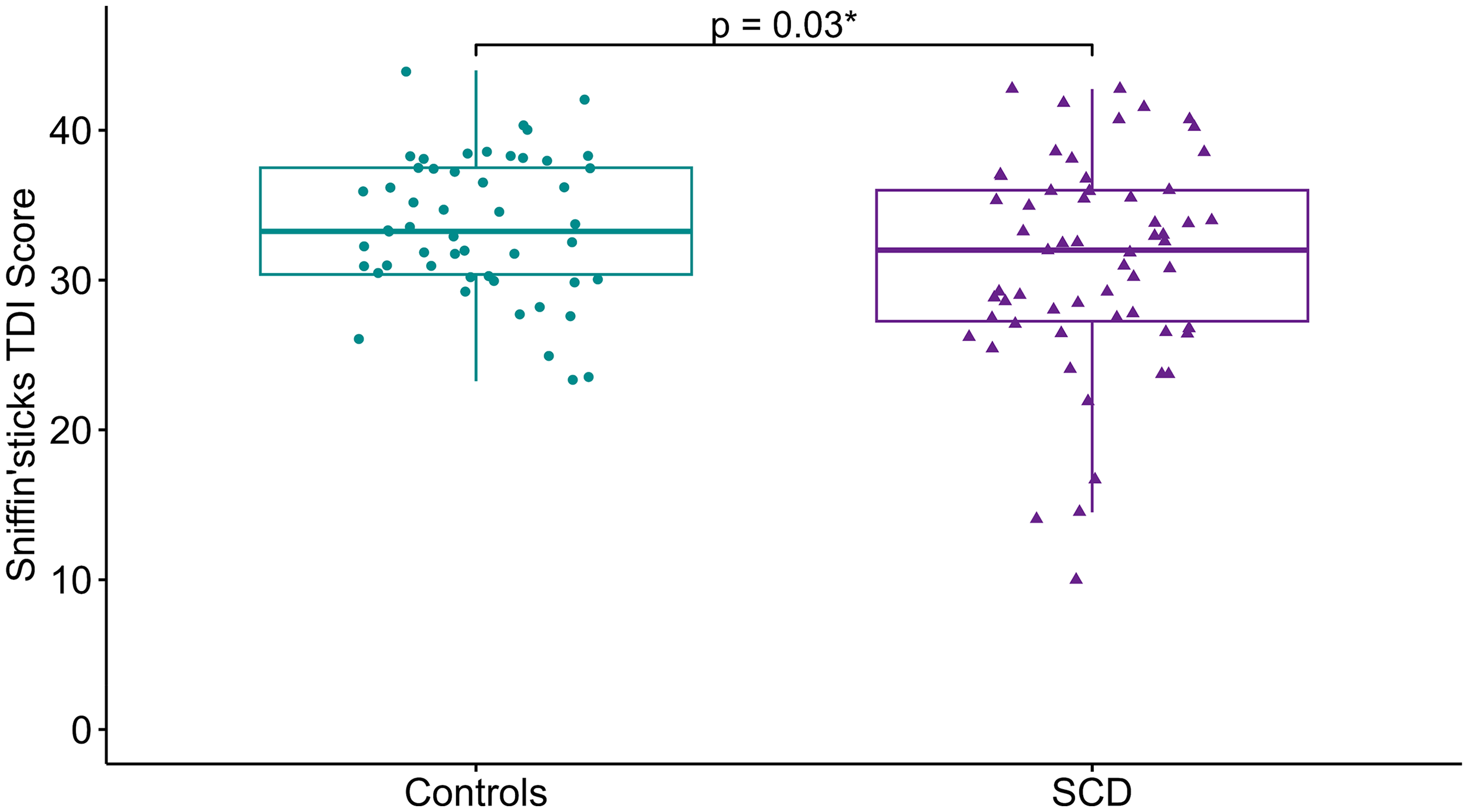
Global olfactory scores by groups. Box plot comparing the mean Sniffin’ Sticks TDI scores between the HC group (green) and the SCD group (purple). Individual data points are overlaid, with green circles representing scores for each HC participant and purple triangles representing scores for each SCD participant.
The subsequent mixed ANOVA revealed a significant main effect of group ((1108) = 4.33, p = 0.04, η² = 0.02) and of task ((2216) = 48.36, p < 0.001, η² = 0.17), but no significant group x task interaction (F(2216) = 0.96, p = 0.386, η² = 0.004). Exploratory post hoc analyses were conducted for threshold, discrimination, and identification. Although these analyses did not yield statistically significant results, a trend was observed toward higher threshold scores in the SCD group compared to the HC group (t(107.65) = 1.91, p = 0.06, Cohen's d = 0.36; see Table 1).
Demographic, neuropsychiatric, and olfactory characteristics of study participants by group.
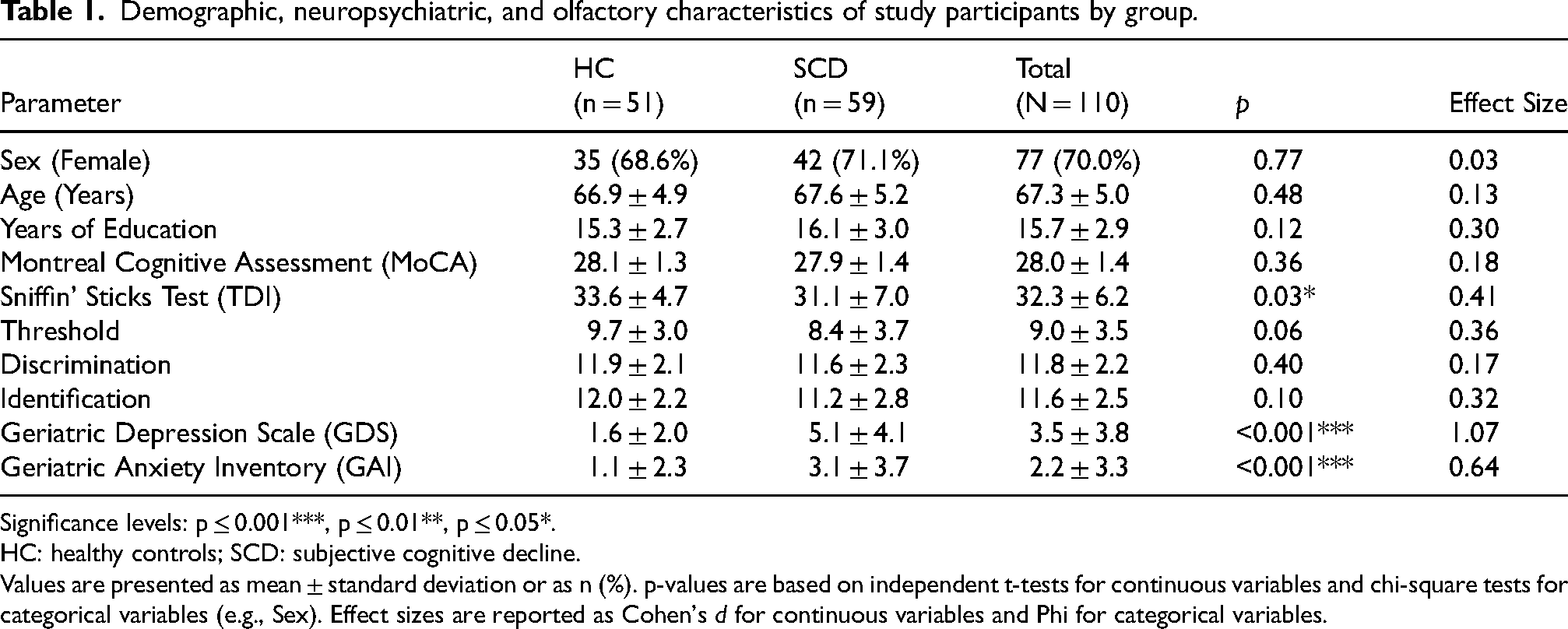
Significance levels: p ≤ 0.001***, p ≤ 0.01**, p ≤ 0.05*.
HC: healthy controls; SCD: subjective cognitive decline.
Values are presented as mean ± standard deviation or as n (%). p-values are based on independent t-tests for continuous variables and chi-square tests for categorical variables (e.g., Sex). Effect sizes are reported as Cohen's d for continuous variables and Phi for categorical variables.
Next, we conducted a hierarchical logistic regression analysis to evaluate the predictive value of anxiety, depression, and olfaction on group (SCD versus HC). In the first block, GAI and GDS scores were entered as predictors. This yielded a significant effect of depression (b = 0.52 (SE = 0.13), z = 4.0, p < 0.001, OR = 1.68, 95% CI [1.33, 2.22]), but not of anxiety (b = −0.10 (SE = 0.13), z = −0.80, p = 0.43, OR = 0.90, 95% CI [0.70, 1.15]). The model accounted for approximately 22.7% of the variance (Nagelkerke's R² = 0.23) and was statistically significant (χ²(1) = 34.52, p < 0.001). In the second block, olfaction was added to the model. Once again, depression showed a significant effect (b = 0.63 (SE = 0.15), z = 4.2, p < 0.001, OR = 1.89, 95% CI [1.44, 2.62]), while anxiety remained non significant (b = −0.16 (SE = 0.14), z = −1.80, p = 0.24, OR = 0.85, 95% CI [0.64, 1.11])). Additionally, olfaction contributed significantly to the model (b = −0.15 (SE = 0.050), z = −3.0, p < 0.01, OR = 0.86, 95% CI [0.78, 0.94]). The addition of olfaction increased Nagelkerke's R² to 0.30 with a significant model chi-square statistic (χ²(1) = 45.89, p < 0.001; see Table 2)). An analysis of deviance indicated a significant increase in explained variance between the two models (χ²(1) = 11.38, p < 0.001; see Table 3).
Hierarchical logistic regression analysis of predictors for SCD.
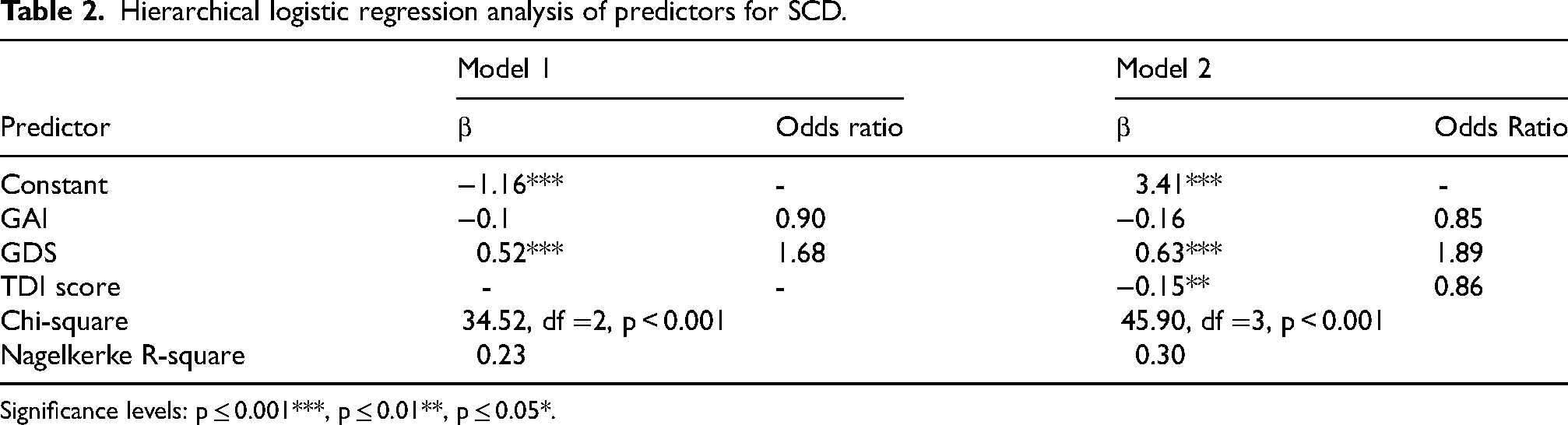
Significance levels: p ≤ 0.001***, p ≤ 0.01**, p ≤ 0.05*.
Analysis of deviance for hierarchical logistic regression models.

Significance levels: p ≤ 0.001***, p ≤ 0.01**, p ≤ 0.05*.
Follow-up ROC analyses were conducted to evaluate the classification performance of each individual predictor and the hierarchical models in distinguishing SCD from HC. depression alone yielded an AUC of 0.81 (optimal threshold = 2.5; sensitivity = 67.8%; specificity = 82.4%; PPV = 81.6%; negative predictive value NPV = 68.9%; Youden's index = 0.50; LR + = 3.84; LR− = 0.39). anxiety had an AUC of 0.66 (optimal threshold = 1.5; sensitivity = 52.5%; specificity = 78.4%; PPV = 73.8%; NPV = 58.8%; Youden's index = 0.31; LR + = 2.44; LR− = 0.61). Olfaction was inversely associated with SCD status. After adjusting for directionality, the AUC was 0.60 (optimal threshold = 40.5; sensitivity = 10.2%; specificity = 96.1%; PPV = 75.0%; NPV = 48.0%; Youden's index = 0.06; LR + = 2.59; LR− = 0.93). For the hierarchical logistic regression models, Model 1 (depression and anxiety only) achieved an AUC of 0.82 (optimal threshold = 0.59; sensitivity = 64.4%; specificity = 88.2%; PPV = 86.4%; NPV = 68.2%; Youden's index = 0.53; LR + = 5.47; LR− = 0.40), and Model 2 (depression, anxiety, and olfaction) achieved a higher AUC of 0.86 (optimal threshold = 0.53; sensitivity = 77.9%; specificity = 82.4%; PPV = 83.6%; NPV = 76.4%; Youden's index = 0.60; LR + = 4.42; LR− = 0.27; see Figure 2).
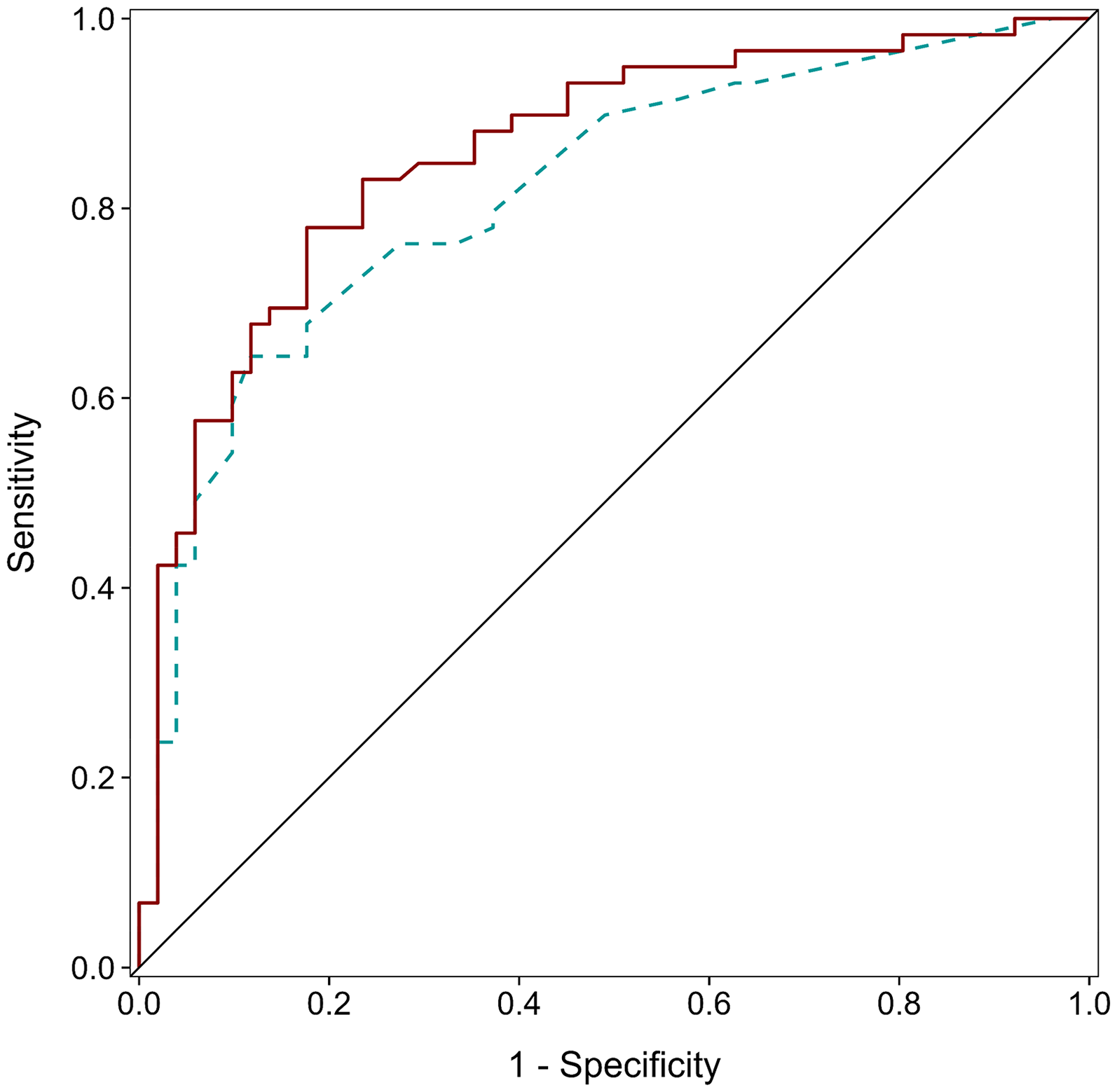
SCD classification models. ROC curves illustrating the classification ability of Model 1 (depression and anxiety; dashed blue line, AUC = 0.82) and Model 2 (depression, anxiety and olfaction; solid red line, AUC = 0.86) for distinguishing individuals with subjective cognitive decline (SCD). The diagonal black line represents chance-level performance.
Discussion
This study aimed to characterize olfactory function and neuropsychiatric symptoms, including anxiety and depression, in individuals with SCD. As hypothesized, participants with SCD exhibited lower global olfactory scores and higher levels of depression and anxiety than HC. Regarding olfactory function, we did not observe any significant interaction between group status and individual olfactory tasks (identification, discrimination, threshold). However, the global olfactory score significantly contributed to the predictive model for group status alongside with depression symptoms, while anxiety symptoms did not.
Individuals with SCD exhibited lower global olfactory function. This aligns with the notion that olfactory impairment is one of the earliest symptoms of AD and precedes objective memory and cognitive impairments.37,79 In fact, previous studies found lower olfactory identification scores in participants with SCD,43,80 but inconsistently.44,45 Although global olfactory scores were lower in the SCD group, we did not find significant group differences for the olfactory subtasks, such as identification, threshold, and discrimination. One may hypothesize that the lack of robustness of olfactory identification impairment in SCD, and the absence of significant differences between olfactory tasks in our study, may be due to limited test variability, as well as the subtlety of the olfactory impairment. 45 Moreover, our sample consists of community-dwelling individuals with SCD, rather than individuals with SCD referred through a memory clinic. This could suggest a less advanced stage of the condition in our sample, which, by extension, may involve fewer olfactory abnormalities and therefore more subtle changes. 81 Building on this perspective, one could argue that not all individuals with SCD will progress to MCI, AD, or dementia. If olfactory impairment is specifically associated with progression, combining SCD individuals who will progress with those who will not, may dilute the observed effect. Longitudinal studies that specifically track these trajectories are needed to fully understand this relationship.
While olfactory identification is often considered the most sensitive task for early detection due to its reliance on higher-order cognitive functions, all three domains are affected in AD and MCI.82,83 Additionally, the patterns of degeneration across olfactory tasks may vary across AD stages or even among its subtypes. This raises questions about whether olfactory identification offers the same advantage over other tasks and retains the same predictive value in SCD as it does in later stages of the disease. For instance, one study found that the olfactory discrimination and the TDI score, but not identification,—predicted cognitive decline over three-years in community-dwelling older adults. 84 Furthermore, individuals carrying the APOE ε4 allele—a genetic risk factor for AD—have been shown to exhibit reduced olfactory sensitivity (threshold) years before declines in identification.85,86 Interestingly, we observed a trend toward higher olfactory threshold scores in the SCD group, with a small effect size, though this result should be interpreted with caution given its exploratory nature and lack of statistical correction.
Taking all of this into consideration, evaluating global olfactory performance—including threshold, discrimination, and identification—may offer greater sensitivity than focusing solely on identification. The evolution of deficits across olfactory domains remains poorly understood and may follow complex trajectories. Evidence suggests that threshold and discrimination are affected early in the disease. Our findings support the idea that a comprehensive olfactory battery is better suited to detect subtle early alterations in SCD.
The progression of olfactory deficits in AD is still not well understood. Key regions involved in olfactory processing—including the olfactory bulb, olfactory tract, primary olfactory cortex, entorhinal cortex, insula, hippocampus, and frontal cortex—are reduced in volume in AD and MCI, with limited research on connectivity.38,79,87 In SCD, neuroanatomical changes in olfaction regions may already be occurring. While some studies report gray matter atrophy in medial temporal structures such as the hippocampus and entorhinal cortex, findings are heterogeneous. 88 Functional connectivity may offer insight into early pathomechanisms that may precede structural atrophy at this stage.45,89,90 This highlights the need for further research to better understand subtle early manifestations of olfactory decline in SCD. Nonetheless, our observation of olfactory impairment in a SCD sample is particularly compelling. It reaffirms that olfactory assessments can play a crucial role in detecting early, subtle changes even in non-clinical populations, extending their relevance as a marker for AD risk.
Anxiety and depression symptoms were more prominent in the SCD group than in the HC group, replicating findings from previous studies that associate these neuropsychiatric factors with SCD.55,59 Although anxiety symptoms were more pronounced in the SCD group, anxiety was not a significant predictor of group status, unlike depression. One possible explanation is an overlap between anxiety and depression constructs, which can complicate the isolation of their distinct contributions to group status. 91 We evaluated multicollinearity to address this issue and found it not to be a concern in our analyses. Both anxiety and depression symptoms have been linked to cognitive decline in the elderly. However, depression symptoms are more robustly associated with SCD, poorer memory performance and reduced performance in other cognitive domains in older adults, whereas anxiety yields mixed results.58,59,92,93 This is also consistent with the large effect size observed for group differences in depression, compared to a moderate effect size found for anxiety. Ultimately, the roles of anxiety and depression in SCD remain poorly understood, highlighting the need for further investigation.
A second major finding of the present study is the added value of olfactory score in predicting group status when combined with depression and anxiety. This reinforces the notion that olfactory performance holds predictive value within the AD continuum. Specifically, olfactory scores significantly improved our first model predictive power, highlighting the potential of olfactory function as an important biomarker for distinguishing between SCD and HC. This Align with previous studies that have shown promising results using olfactory scores as a predictor for differentiating between various stages of AD.41,80
While depressive symptoms emerged as the strongest individual predictor for classification and olfaction the weakest, their combination led to a more accurate reflecting by improvements in AUC, sensitivity, PPV and NPV, as well as a higher Youden's index—despite a minor reduction in specificity. These results are consistent with findings from a recent study showing that combining olfactory identification with depressive symptoms predicts dementia progression over a 12-years more effectively than either factor alone. 61
To contextualize our findings, it is worth comparing our model's performance to that of standard cognitive screening tests. Studies with memory clinic samples have found that tools like the MoCA and the Mini-Mental State Examination have limited accuracy in distinguishing individuals with SCD from cognitively healthy controls. 94 When more specific neuropsychological tests are used, classification improves. For instance, the Rey Auditory Verbal Learning Test combined with demographic variables have shown AUCs up to 0.84 but drops to 0.67 when used alone. 95 Other tasks such as the Stroop task and the Boston Naming Test have also shown moderate discriminative ability in community-based SCD samples.96,97 In this context, our community-based SCD model compares favorably reinforcing the added value of including sensory and psychological assessments alongside cognitive testing for early detection of AD. These results support a growing consensus advocating for multidimensional screening strategies.
Our study presents certain limitations. As a cross-sectional study, the findings suggest that combining global olfactory testing with neuropsychiatric assessments could serve as a complementary approach to existing diagnostic tools for preclinical AD. However, longitudinal studies are required to confirm and evaluate the sustained predictive value of these findings. For this reason, we have obtained consent from participants who consented for future follow-up, tentatively scheduled within five years, to assess conversion to MCI or AD.
Although single-question assessments of memory complaints have been used effectively for SCD categorization, incorporating broader criteria, such as those outlined in SCD-plus, may improve the identification of individuals at risk of progression to MCI or AD. 22 Adding questions related to spatio-temporal orientation and executive function, as well as obtaining input from an informant, has also been considered a valuable complement to memory-based questions for enhancing early detection. 65 Future research may benefit from investigating whether olfactory deficits are more pronounced in individuals who meet these expended criteria.
Additionally, the predictive value the global olfactory score in our model appears limited, likely due to the constraints of behavioral measures in detecting subtle olfactory changes. This aligns with the small to modest effect size observed when comparing global olfactory performance between our groups, as well as with the effect size reported in studies showing reduced olfactory identification in individuals with SCD. 43 Notably, AD status may be better predicted by combining objective measures such as OERP and behavioral measures with a classification accuracy of up to 100%. 41 OERP have also shown promising result when comparing MCI with HC. 42 With its excellent temporal resolution, these EEG based signals could offer deeper insights into differences in the time course of olfactory processing, potentially revealing distinct patterns of neural activity associated with early neurodegenerative changes. 98 These patterns may occur before behavioral deficits become measurable and are less influenced by higher-order cognitive functions typically involved in behavioral assessments of olfaction (e.g., semantic memory), offering an opportunity to better isolate early olfactory dysfunction.98,99
Conclusion
We show that individuals with SCD exhibit reduced global olfactory performance, heightened anxiety, and more depressive symptoms. Combining olfactory performance with measures of anxiety and depression improves the prediction and classification between HC and individuals with SCD. Longitudinal research is essential to evaluate olfaction as a biomarker alongside SCD in the transition through different stages of AD. Finally, OERP studies could provide valuable insights to complement behavioral measures, which may be limited in detecting specific olfactory alterations at this stage.
Footnotes
Acknowledgements
The authors thank all participants for their participation.
Ethical considerations
This study was approved by the Ethics Committee for Research Involving Humans at Université du Québec à Trois-Rivières (CER-23-298-07.01). All procedures followed institutional and Helsinki Declaration ethical standards.
Consent to participate
Written informed consent was obtained from all participants before their inclusion in the study.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by The Canadian Institutes of Health Research (CIHR; JF: AFF 173514; OFL: 187458), Fonds de Recherche du Québec–Santé (FRQS; JF: 352197) and by the Natural Sciences and Engineering Research Council of Canada (NSERC; JF: 2022-04813).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy restrictions and requirements by the REB.
