Abstract
Background
Subjective cognitive decline (SCD) is linked to a more rapid progression to the development of mild cognitive impairment (MCI) or Alzheimer's disease (AD). SCD has been correlated with affective symptoms such as depression and anxiety. Recent research aimed to shed light on the relationship between these affective symptoms and how they might correlate to a more rapid progression to objective cognitive impairment. No studies have assessed the presence, type, and intensity of depressive and anxiety symptoms between SCD individuals who progressed versus those who did not.
Objective
This study aimed to establish whether there are differences between subclinical depressive and anxiety symptoms in terms of presence, type, and intensity of symptoms presented by individuals with SCD who progressed to an objective cognitive decline.
Methods
The recruited participants originated from the Consortium for the Early Identification of Alzheimer's Disease - Québec (CIMA-Q) cohort. They were assessed twice, with an interval of 4 years separating the evaluations. Anxiety symptoms were assessed using the Geriatric Anxiety Inventory (GAI) and depression symptoms using the Geriatric Depression Scale (GDS-30).
Results
The presence, type and intensity of anxiety symptoms did not significantly distinguish the two groups. Only one type of hopelessness-related depressive symptom was significantly higher in SCD participants who had progressed to objective cognitive decline compared with those who had not.
Conclusions
Our results suggest that it may be beneficial to target hopelessness in non-pharmacological interventions aimed at preventing the progression of people with SCD to MCI or AD.
Keywords
Introduction
Alzheimer's disease (AD) is the leading cause of dementia, accounting for 60–80% of all dementia diagnosed annually. 1 AD, like other types of dementia, is a neurodegenerative disorder, meaning it progresses gradually. This progression is divided into several preclinical stages depending on the degree of cognitive impairment. 2 Subjective cognitive decline (SCD) constitutes one of the preclinical stages of AD increasingly recognized and studied. 3 Although the report of the personal perception/impression of cognitive decline is present in 50–80% of older adults,4,5 two conditions must be present to formally reflect the presence of SCD: a subjective impression of decline in memory abilities, and that this decline is not reflected in the person's performance on neuropsychological tests. 3
The next stage considered in the development of AD is mild cognitive impairment (MCI). At this stage, cognitive losses are detected by cognitive performance below what is expected according to the elderly person's age and level of education. However, this level of slight deterioration in cognitive abilities has little or no impact on the individual's daily functioning. 6 Although the subjective experience of cognitive losses associated with SCD is relatively common among older adults, longitudinal studies suggest this subjective perception predicts the eventual development of MCI and dementia,7–9 leading certain researchers to include this disorder on the AD development continuum.10,11 However, it should be noted that not all people with SCD systematically decline to mild cognitive impairment or dementia due to the heterogeneous etiology of SCD. However, a team of researchers at the Subjective Cognitive Decline Initiative determined based on the research that certain predictive factors are associated with subjective cognitive disorder that further predict decline. 3
Affective symptoms such as depression and anxiety, even at subclinical levels, can increase the risk of developing objective cognitive losses leading to a diagnosis of MCI or AD in SCD individuals.12–14 SCD has thus been strongly associated with depressive and anxiety symptoms in cross-sectional studies of older people from the community with or without cognitive decline.15,16 However, to date, relatively few prospective studies have examined these affective symptoms at subclinical level in samples of older individuals progressing to MCI or AD. 17 The few studies that did investigate it also found divergent results. One related study is a 4-year longitudinal study where depressive and anxiety symptoms were assessed in older adults with SCD. 18 Four groups were generated based on the presence or absence of anxiety and depressive symptoms. This study found that the group who only reported depressive symptoms showed objective cognitive decline in episodic memory and executive functions. The authors specified that this cognitive decline profile resembled AD. However, this study was unable to demonstrate an association between the presence of anxiety symptoms in those reporting only anxiety at the start of the study and objective cognitive decline 4 years later. The group with depressive and anxiety symptoms, however, was associated with a decline in their attentional abilities. A second study also supported these conclusions. Indeed, Geda et al. 19 concluded that the presence of depressive symptoms was one of the only predictors of MCI in cognitively normal older adults.
Conversely, other researchers have postulated that the presence of anxiety symptoms, even at a subclinical level, might be a risk factor for developing MCI. A longitudinal study by Liew 20 demonstrated that the association of the presence of SCD and anxiety symptoms in elderly people without objective decline resulted in the highest rate of objective cognitive decline in either MCI or dementia. Two other studies whose methodologies show similarities (number of participants, duration of follow-up) obtained contrary results. However, the Ahn et al. study 18 had the advantage of assessing both anxiety and depressive symptoms. Moreover, all the above studies defined the presence of affective symptoms by answering “yes” to a singular question. This convenient but rather simple method might not reflect a mild behavioral change as suggested by the National Institute on Aging and Alzheimer Association (NIA-AA) 21 to formally indicate the presence of affective symptoms in SCD as a preclinical stage of AD.
While the current state of research on the presence of either depressive or anxiety symptoms does not seem to converge on one conclusion, there is very little data on the types of depressive and anxiety symptoms that characterize individuals with SCD progressing to objective cognitive deficits (MCI or AD). Some of the questionnaires used in research to measure depressive and anxiety symptoms in elderly people with SCD could have been used to assess whether there is a clinical profile of affective symptoms differentiating them. For example, the Geriatric Anxiety Inventory (GAI) 22 and the Geriatric Depression Scale (GDS) 23 are validated measurement tools for older adults accounting for the specificity of the experience of anxiety and depression in these individuals, and they are also validated for elderly people with cognitive loss.24–26 These questionnaires were the subject of factor analyses which made it possible to subdivide them into dimensions that could be considered types of symptoms. For the GDS, Adams and colleagues 27 found six dimensions, including symptoms of dysphoria, withdrawal, anxiety, cognitive decline, hopelessness, and agitation. With the GAI, Bandari et al. 28 identified three dimensions: cognitive, somatic, and arousal symptoms. Although some studies have used the dimensions identified by these authors to establish a profile of affective symptoms of their sample,29–31 little research has used them with SCD individuals manifesting objective cognitive losses. The only two studies so far addressing this topic found that depressive symptoms of hopelessness were associated with greater load of amyloid-β protein—considered an important neurobiological correlate in the development of AD or higher rate of progression to dementia.32,33
A longitudinal study by Houde et al. 34 looked at the depressive symptoms characterizing individuals with MCI who developed AD versus those who did not. By analyzing the items most frequently endorsed by those who progressed from MCI to AD, the researchers created a list of 10 items they grouped under the term ‘melancholic depressive symptoms,’ corresponding to Adams et al.'s 27 dysphoric dimension. Knowing that SCD is considered on the continuum of AD development like MCI, the results of this study possibly indicate that the same type of depressive symptoms can be found in individuals presenting SCD who will develop MCI. Concerning the type of anxiety symptoms that could characterize people presenting with SCD and progressing towards an objective decline in cognitive abilities, the only studies listed analyzed anxiety relating to perceived cognitive losses.26,35 To measure the presence of this type of anxiety, participants who expressed a feeling of decline were asked if this worried them. Although those studies were able to draw interesting conclusions regarding the link between worry about perceived losses and progression toward objective decline, this characterization of the anxiety experience was quite limited given that worry about perceived losses is already part of the SCD identification criteria. Finally, the study by Liew 20 assessed anxiety by asking participants whether they had experienced what appear to be symptoms (e.g., nervousness, shortness of breath, etc.) that are part of the somatic type and arousal dimensions identified by Bandari et al. 28 This suggests that somatic- and arousal-type anxiety symptoms might differentiate SCD individuals progressing towards objective cognitive decline from those not progressing.
When we considered the intensity of depressive and anxiety symptoms, the aforementioned studies contradicted each other. First, higher levels of depressive symptoms were associated with increases in the rate of development of AD in SCD individuals.36,37 The higher intensity of these symptoms also appeared to increase the risk of developing a so-called “amnestic” MCI (i.e., an MCI whose main cognitive decline is in memory) in Sundermann et al. 38 Finally, even at low levels, depressive symptoms were associated with both the development of AD and amnestic MCI in an 8-year longitudinal study. 39
Regarding anxiety symptoms, a recent meta-analysis concluded that a higher intensity of depressive symptoms in people with SCD was not associated with higher rates of the development of objective cognitive losses. 40 They claimed the absence of conclusive results concerning the intensity of depressive symptoms and the risk of progressing towards objective cognitive losses may be due to the heterogeneous etiology of SCD and to the variability of the methodologies used to assess depressive symptoms, which were more numerous than anxiety symptoms. However, the author was still able to conclude that the report of a higher intensity of anxiety symptoms significantly increased the risk of developing objective cognitive losses, yet this conclusion was based on only four studies, which was mentioned as a study limitation. Thus, for the moment, the research does not appear to converge on one conclusion regarding the association between the presence and intensity of depressive and anxiety symptoms in individuals presenting with SCD who progress to objective cognitive impairment. However, depressive and anxiety symptoms are still considered in the definition of SCD according to the latest recommendations, and each of these symptoms are also considered independent risk factors for developing AD13,14 Overall, it appeared that no study had analyzed the presence, type, and intensity of subclinical depressive and anxiety symptoms characterizing people with SCD progressing (SCD-P) towards objective cognitive decline, as in the case of MCI or AD in people with non-progressing SCD (SCD-NP).
Objectives
This study aimed to characterize the depressive and anxiety symptoms present in SCD-P by comparing them to those present in SCD-NP individuals. The specific objectives were:
Compare the presence, type, and intensity of depressive symptoms of individuals with SCD-P with individuals with SCD-NP at baseline. Compare the presence, type, and intensity of anxiety symptoms of individuals with SCD-P with individuals with SCD-NP people at baseline.
Hypotheses
For the first objective, we hypothesized:
SCD-P individuals will report significantly more depressive symptoms than SCD-NP individuals at baseline. Depressive symptoms such as hopelessness and dysphoria will emerge significantly more in SCD-P individuals than SCD-NP individuals at baseline. The intensity of depressive symptoms will be significantly higher in SCD-P individuals than SCD-NP individuals at baseline. SCD-P individuals will exhibit significantly more anxiety symptoms than SCD-NP individuals at baseline. Somatic- and arousal-anxiety symptoms will emerge significantly more in SCD-P individuals than in SCD-NP individuals at baseline. The intensity of anxiety symptoms will be significantly higher in SCD-P individuals than in SCD-NP individuals at baseline.
For the second objective, we hypothesized:
Methods
Recruitment of participants
The data used in this manuscript were obtained through the Consortium for the Early Identification of Alzheimer's Disease - Québec (CIMA-Q). The primary objective of CIMA-Q is the longitudinal characterization of an observational cohort of more than 350 aging women and men who are cognitively healthy, with subjective cognitive impairments, with mild cognitive impairments or with a dementia resulting from a probable AD. CIMA-Q gathers clinical, cognitive, biological, radiological and pathological data from these participants in order to (1) establish early AD diagnosis; (2) make a cohort with specific characteristics available to the scientific community; (3) identify new therapeutic targets enabling to prevent or slow cognitive decline and AD; and (4) support new clinical studies of these targets. 41 Participants in the CIMA-Q cohort were recruited from three sources: the NuAge cohort (a cohort of elderly subjects which took shape as part of the Quebec Longitudinal Study on Nutrition as a Determinant of Successful Aging), 42 memory clinics participants, and from the community (announcements and bank of participants from participating sites) in the cities of Montreal, Quebec, and Sherbrooke (Canada). For full method details, see Belleville et al. 41 Access to the CIMA-Q data was provided following an agreement of the ethics committee (CER VN 20-21-24) and the data access committee of the consortium.
Ethical approval and informed consent
CIMA-Q research activities comply with Good Clinical Practice guides and requirements from the funding agencies. The study has received approval from the coordinating ethics committee of the Institut universitaire de geriatrie de Montreal. For this article, ethical approval was granted by Comité d’éthique de la recherche vieillissement-neuroimagerie - R2 (project # CER VN 20-21-24). Participants gave their written informed consent before enrollment in the study and for the different components of the project.
Inclusion/exclusion criteria
Inclusion criteria
To be included in the CIMA-Q research project, respondents had to be aged 65 or older and live in a community or residence. They had to understand and be able to read and write in French or English well enough and have sufficient visual and hearing acuity to pass neuropsychological tests.
Exclusion criteria
Respondents were excluded if they planned to move outside Quebec in the next 3 years. Respondents were also excluded if they had too-advanced dementia or if they had a clinical physical or psychological condition, such as clinical depression or an anxiety disorder, that could better explain their scores on questionnaires and scales measuring their cognitive state. Participants were also excluded if they were taking more than 1 mg of lorazepam per day. For all the general inclusion and exclusion criteria, please refer to Belleville et al. 41
Inclusion criteria for participants with subjective cognitive impairment who did not progress (SCD-NP)
The inclusion criteria for this group were based on criteria from the Subjective Cognitive Decline Initiative.43,44 The presence of subjective cognitive complaints among participants was identified with the question “Do you think your memory is getting worse?” This question was anchored with three answer options: (a) “Yes, but that does not worry me,” (b) “Yes, and that worries me,” and (c) “No.” Participants were required to answer (b) to this question. Their score on the Logical Memory II subtest of the Weschler Memory Scale (WMS III) 45 as well as the Montreal Cognitive Assessment (MoCA) 46 and the Clinical Dementia Rating (CDR) 47 measure had to be within the expected normal according to age and level of education.
Inclusion criteria for participants who progressed (SCD-P)
For assignment to this group, participants had to answer (b) to the question above regarding cognitive decline.43,44 These participants also had to meet NIA-AA diagnostic criteria for MCI 48 or probable AD 49 at follow-up. Their scores on the cognitive assessments mentioned above had to demonstrate a decline that suggested the presence of MCI or AD according to the measurement scales of these assessments.
Procedure
Pre-screening procedure
People who expressed interest in the CIMA-Q project were contacted by telephone for a pre-screening. This call aimed to confirm their interest in the study, to briefly inform them of its objectives, and to verify respondents’ eligibility based on the inclusion and exclusion criteria. The Telephone Mini-Mental State Examination was then administered, and it was also at this time that the subjective perception of memory decline was assessed by the question from Jessen et al. 43
Measuring time
Time 1 (T1)
Respondents meeting the eligibility criteria were met within 180 days after the first telephone contact. During this visit, participants completed questionnaires used in this study assessing their cognitive status and depressive and anxiety symptoms. Depending on the answer to Jessen's question 43 and the absence or presence of objective cognitive deficits, participants were identified as having SCD.
Time 2 (T2)
The participants met at time 1 (T1) completed the same questionnaires 4 years later. The results on the same measure of cognitive status administered at T1 were used to determine if the participants progressed to an objective cognitive deficit based on the NIA-AA diagnostic criteria for MCI 48 or probable AD. 49
Measures
Cognitive status
The MoCA is a test whose essential objective is to detect individuals living with mild to severe cognitive impairment. 46 This short-answer questionnaire assesses several areas of cognition like short-term memory, visuospatial skills, executive functions, attention, concentration, working memory, language, and orientation in time and space. The maximum score is 30. A score between 26 and 30 means no cognitive impairment. A score between 18 and 25 indicates a rather mild impairment, between 10 and 17, a moderate impairment, and a score of less than 10 indicates severe impairment. This test was developed and tested with a French-speaking Quebec population by Nasreddine et al. 46 Its sensitivity is excellent for detecting mild cognitive impairment at 90%, while for major cognitive impairment, the sensitivity is 100%. The specificity for mild cognitive impairment is 87%. The Cronbach's alpha was 0.83, demonstrating excellent internal consistency, and the test-retest reliability was also excellent, with a correlation coefficient of r = 0.92.
Depressive symptoms
The original English version of the GDS 23 or the French-Canadian translation, the Échelle de dépression gériatrique (EDG), were used depending on the participant's primary language. This is a self-report questionnaire assessing depressive symptoms, comprised of 30 items representing the manifestations of geriatric depression. The individual responds with “yes” or “no” depending on whether the statement corresponds to their state during the prior week. A score of 0 to 10 indicates no depressive symptoms, 11 to 20 indicates mild depressive symptoms, and 21 to 30 indicates moderate or severe depressive symptoms. For the original version, Yesavage et al. 23 reported excellent internal consistency (r = 0.94) and good test-retest reliability (r = 0.85). Convergent validity was measured by comparing the GDS to the Self-Rating Scale for Depression (SDS) and the Hamilton Rating Scale for Depression (HRS-D), two previously validated measures for assessing depression. The correlations between these tools and the GDS were found to be very good (r = 0.84 for the SDS, and r = 0.83 for the HRS-D). 23 The scale was also validated for a population of older people experiencing objective cognitive losses.25,26 The GDS was translated and validated in French by Bourque et al. 50 with a French-Canadian population. This translation demonstrated good internal consistency (r = 0.89) and acceptable temporal stability (r = 0.70).
For the GDS, Adams et al. 27 identified six dimensions: depressive symptoms of dysphoric type (items 1, 3, 4, 7, 9, 15, 16, 23, 25), withdrawal (items 2, 12, 19, 20, 21, 28), worry-anxiety (items 6, 8, 13, 18), cognitive decline (items 14, 26, 29, 30), hopelessness (items 5, 10, 17, 22), and agitation (items 11, 24, 27).
Anxiety symptoms
The original English version of the GAI, 22 or the French-Canadian translated version, the Inventaire d’anxiété gériatrique (IAG), 51 were used, depending on the participant's primary language. This is a self-report questionnaire assessing anxious symptoms, comprised of 20 items covering major areas of worry (e.g., fears, anxious mood, and meta-worries, i.e., worries about one's own concerns). For each statement, the respondent is asked to answer “agree” or “disagree.” The psychometric qualities of the original version with a geriatric population in the community demonstrated excellent internal consistency (r = 0.91), good test-retest reliability (r = 0.91), and excellent interrater reliability (r = 0.99). 22 This inventory was also validated in a population of elderly people who demonstrated objective cognitive losses 24 The French-Canadian version used in the present study also possesses excellent psychometric properties, with a Cronbach's alpha of 0.94, a test-retest reliability of 0.89, and a convergent validity of 0.50. 51 For the GAI, three dimensions are described in Bandari et al., 28 namely cognitive anxiety (items 1, 2, 3, 5, 8, 9, 11, 14, 16), somatic anxiety (items 7, 12, 15, 18), and arousal (items 4, 6, 10, 13, 20).
Statistical analyses
All statistical analyses were calculated with IBM's SPSS (Version 27). To verify the existence of significant differences between the SCD-NP and SCD-P groups per sociodemographic variables, T-tests were used for the age variable and a chi-square test for the sex variable. The MoCA score was also compared between groups to see whether differences existed per their cognitive functions. To analyze the presence of the ratio of anxiety and depressive symptoms between the two groups, the chi-square test was used.
To analyze the presence of anxiety and depressive symptoms between the two groups, the chi-square test was used. Thresholds were chosen for each questionnaire following the NIA-AA 21 recommendation that, for the preclinical stage of dementia, minimal change due to affective symptoms should be present. For depressive symptoms, a cut-off score of 3 or more was chosen to reflect the presence of depressive symptoms. This choice was made based on the article by Lozupone et al., 52 showing that the endorsement of three or more items on the GDS-30 was the most specific and sensitive score to reflect behavioral change. Then, for anxiety symptoms, a cut-off score of 2 or more on the GAI was chosen according to the results of Boddice et al., 24 demonstrating that this threshold also reflected minimal changes due to anxiety symptoms.
Regarding the analysis of the types of affective symptoms (anxiety and depressive), first, the items of each questionnaire were grouped into dimensions according to the factor analyses presented in Measures. This decision was made to reduce the number of comparisons which might increase type I errors and bias the results. The dimensions were drawn from factor analysis studies of these questionnaires for cognitively healthy populations. Then, for each of these dimensions, the Mann and Whitney 53 U-test was once again used. If significant differences were found for certain dimensions, the chi-square test. 54 was used to identify the item with the difference.
To analyze the existence of significant differences in the intensity of anxiety and depressive symptoms at the start of the study between SCD-P and SCD-NP individuals, the Mann and Whitney 53 U-test was used due to the pronounced positive skewness of the group distribution in relation to the scores on the anxiety and depression questionnaires. This non-parametric test allows for the comparison of two independent groups per a variable when the distribution does not meet the normality criteria. It also has the advantage of not being sensitive to outliers in the distribution.
Results
At T1 of the study, 140 participants with SCD were part of the sample. From the initial sample, 80 were reassessed at time 2 (T2). The 4-year attrition rate was 43%, which was consistent with attrition rates found in similar longitudinal studies.55,56 No difference was noted between those who left the study and those who remained, both per sociodemographic data and initial MoCA scores, as well as on questionnaires measuring affective symptoms (GDS and GAI).
Among the 80 participants with SCD, 21 individuals progressed to objective cognitive losses (SCD-P) either to MCI or AD, and 59 did not develop any (SCD-NP). The average age of the group at T1 was 72.17 years. There were 58% women making up the total sample (i.e., 66% women making up the SCD-P group, and 79.6% making up the SCD-NP group). The average number of years of education for the sample was 15.12. Table 1 presents the sociodemographic characteristics of the sample at T1.
Sociodemographic characteristics of participants with SCD-P compared with participants with SCD-NP at T1.

SCD-P: subjective cognitive decline progressing towards objective cognitive decline; SCD-NP: subjective cognitive decline not progressing towards objective cognitive decline; SD: Standard deviation; χ2: chi-square test; U: Mann and Whitney U-test.
Cognitive function
The results demonstrated that the two groups (SCD-P: 27.62 ± 1.72 versus SCD-NP: 27.92 ± 1.37) did not differ significantly on cognitive impairment measured by the MoCA U(N of SCD-P = 21; N of SCD-NP = 59) = 481.5, z = −1 .55, p = 0.122 at T1.
Presence, type, and intensity of depressive symptoms
Concerning the presence of depressive symptoms, our analyses showed that 66.1% of SCD-NP individuals endorsed three or more items on the GDS compared to 71.4% of SCD-P individuals at T1 of the study. The relationship between cognitive decline and report of depressive symptoms was found to be non-significant x2(1; N = 80) = 0.200, p = 0.654.
Concerning the type of symptoms, the hopelessness dimension was significatively higher in the SCD-P group, U(N of SCD-P = 21; N of SCD-NP = 59) = 457.5, z = −1.977, p = 0.048. The chi-square test also demonstrated a significant difference between the groups on the item relating to the feeling of powerlessness x2(1) = 6.767, p = 0.009. Indeed, powerlessness appeared to be higher among the SCD-P group than the SCD-NP group.
Regarding the intensity of depressive symptoms, it was not significant, U(N of SCD-P = 21; N of SCD-NP = 59) = 517.5, z = −1.20, p = 0.263.
Presence, type, and intensity of anxiety symptoms
Concerning the presence of anxiety symptoms, our analyses showed that 52.5% of SCD-NP individuals endorsed two or more items on the GAI compared to 47.6% of SCD-P individuals, who reported at least one anxiety symptom at T1 of the study. The relationship between cognitive decline and the report of anxiety symptoms was found to be non-significant, x2(1; N = 80) = 0.150, p = 0.698.
Regarding the type of symptoms, all differences turned out to be non-significant (i.e., cognitive symptoms: p = 0.610; somatic symptoms: p = 0.630; arousal symptoms: p = 0.135).
Regarding the intensity of symptoms, it was also not significant, U(N of TCS-P = 21; N of TCS-NP = 59) = 599.5, z = −0.224, p = 0.823. For detailed results on differences in affective symptoms between groups, see Table 2.
Affective symptoms identified in participants with SCD-P compared to those in participants with SCD-NP at T1.
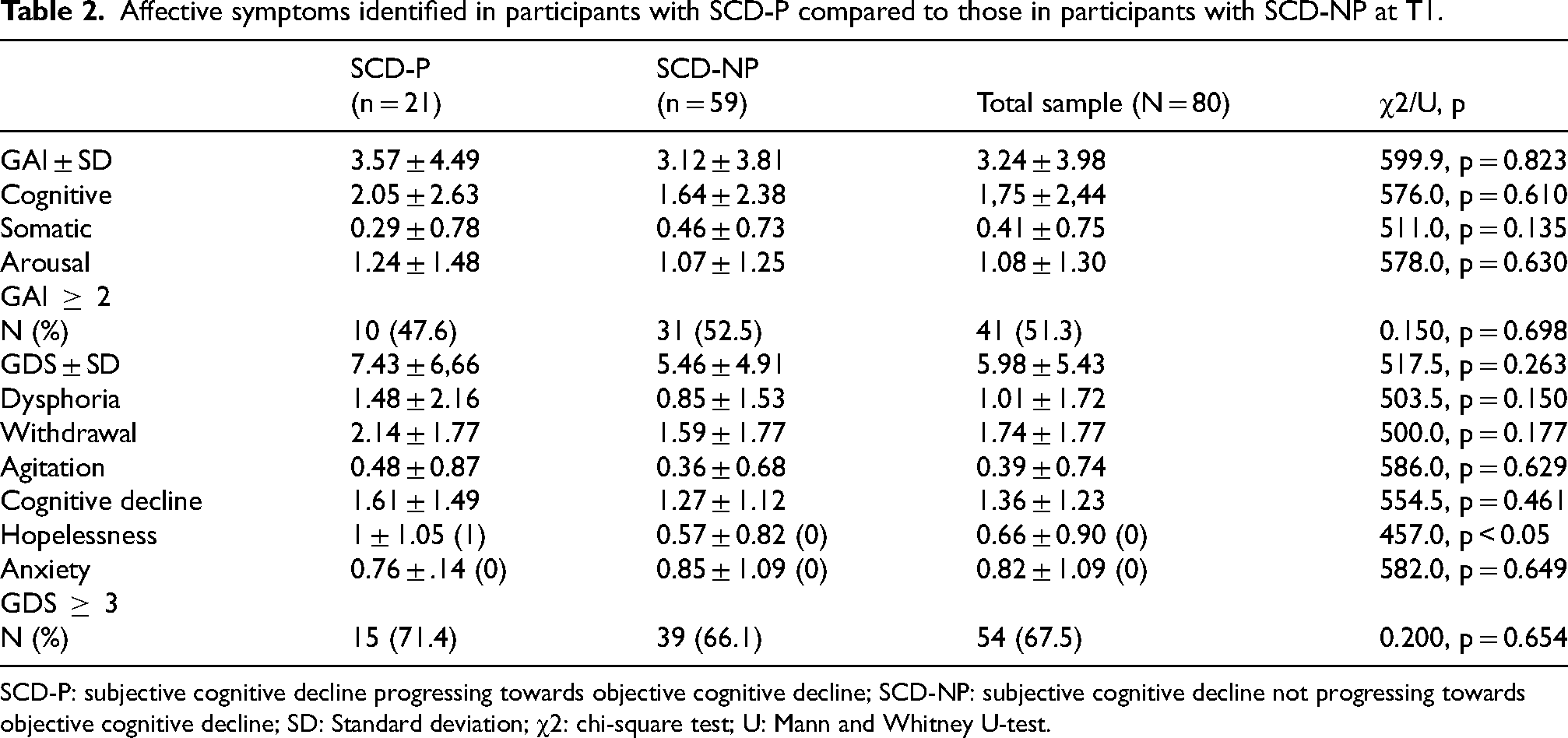
SCD-P: subjective cognitive decline progressing towards objective cognitive decline; SCD-NP: subjective cognitive decline not progressing towards objective cognitive decline; SD: Standard deviation; χ2: chi-square test; U: Mann and Whitney U-test.
Discussion
The overall objective of this study was to characterize in terms of presence, type, and intensity of affective symptoms, individuals with SCD who objectively cognitively declined over a period of 4 years by comparing them to those who had not declined during this same period. The final sample showed that 26% of individuals with SCD developed objective cognitive losses after 4 years. This SCD progression rate was almost identical to that of 27% found in the meta-analysis by Mitchell et al. 8 Overall, our results demonstrated that depressive symptoms of hopelessness were a distinctive feature of SCD-P.
First objective: presence, type, and intensity of depressive symptoms
The first objective of this study was to compare the presence, type, and intensity of depressive symptoms in individuals with SCD-P versus those with SCD-NP. The first hypothesis postulated that depressive symptoms would be significatively more present in people with SCD-P than people with SCD-NP. This hypothesis was rejected. Indeed, 66.1% of SCD-NP individuals compared to 71.4% of SCD-P individuals reported depressive symptoms. This contradicted the study of Ahn and colleagues, 18 implying that reporting depressive symptoms increased the risk of developing objective cognitive losses. However, the evaluation of the presence of depressive symptoms for this latter study was carried out by asking a question taken from the NPI-Q, a scale completed by third parties that simply assesses “feeling sad or saying being depressed.” Given that our study carried out a more exhaustive assessment of depressive symptoms, the probabilities of endorsement on one item among the 30 included in the GDS are higher. Also, unlike the NPI-Q, the GDS is a self-report measure, thus providing direct access to the experience of depressive symptoms, thereby minimizing a bias that can be induced by the perception of a third-party informant. These elements may explain why the results of this study differ from what had been presented in the scientific literature to date.
The second hypothesis concerned the type of depressive symptom. We postulated that hopelessness and dysphoric symptoms would be significantly more present for SCD-P than SCD-NP individuals. This hypothesis was partially confirmed. Hopelessness-type depressive symptoms were significatively more pronounced in the SCD-P group than the SCD-NP group. According to the APA 59 definition, hopelessness is the feeling of not feeling that one's condition will improve or is related to the subjective perception of no longer experiencing positive feelings. This type of symptom is consistent with the results of Thompson and Jones 32 and those of Wang. 33 Furthermore, knowing that SCD sometimes precedes MCI (which precedes AD), it is interesting to note that the experience of hopelessness in elderly people living with AD was a frequent and already documented phenomenon.58,59 Therefore, it appears that hopelessness was an affective experience that united SCD individuals, whose cognition objectively declined over the course of 4 years, along with those living with AD. The SCD-P group thus already had the perception of inexorable cognitive decline. When we looked more precisely at the dimension of hopelessness, four items comprised this dimension that touched on feelings of optimism (item 5), powerlessness (item 10), uselessness (item 17), and despair (item 22). The item showing significant differences between the groups was the item relating to the feeling of powerlessness. Individuals in the SCD-P group would have reported feeling powerless even before declining towards an objective decline. Indeed, powerlessness refers to the state of an individual lacking or believing they lack control or influence over several factors or events that may affect their mental or physical health. 57 Since our finding was correlative, it could not be determined whether the feeling of cognitive decline preceded the experience of powerlessness or whether the feeling of powerlessness preceded the feeling of cognitive decline. The first scenario would be that an individual observing cognitive losses would feel powerless in the face of this decline because they would feel this process as irreversible, leading them towards AD. The second scenario would involve an individual already feeling powerless in the face of their own old age, for example, and would notice more cognitive losses considering these as the inexorable course of the aging process. It is also possible that this same type of individual notices very real cognitive losses that neuropsychological tests fail to measure, however. People with SCD may feel powerless over their cognitive abilities, particularly if they feel their cognitive functions are declining and find themselves unable to stop this process. They may feel like they have no control over their cognitive abilities and their future, which can be a demoralizing experience. Powerlessness can thus lead to hopelessness because people who believe they have no control over their decline experience a bleak outlook on their future. The experience of hopelessness could lead these individuals to make less effort to implement strategies that might delay the onset of objective cognitive decline, which paradoxically would influence the precocity of the decline. Future studies are needed to confirm this speculation. It should be noted that these hypotheses about powerlessness, which represent only one item on the total hopelessness subscale that proved significant, are exploratory given our limited sample size.
Regarding the type of depressive symptoms, dysphoric symptoms, which corresponded to the melancholic symptoms identified by Houde et al., 34 were not found to be significantly different between the groups. The present research analyzed the results of SCD individuals unlike the MCI individuals analyzed by Houde et al., 34 thus it is possible that the type of depressive symptoms could change once the cognitive deficits are objectified. In addition to dysphoric symptoms, symptoms of withdrawal, anxiety, cognitive decline, and agitation did not distinguish our two groups.
Our third hypothesis postulated that the intensity of depressive symptoms would be significantly higher in SCD-P individuals than in SCD-NP individuals. This hypothesis was rejected. We did not find that a greater intensity of self-reported depressive symptoms was manifested in individuals with SCD who developed objective cognitive losses after 4 years, contradicting what several scientific articles have claimed.3,60 A possible explanation for these results may be due to differences in the etiology of this group. For example, the subjective report of a sense of decline in cognitive abilities has also been linked to personality traits like neuroticism. 61 If the impression of decline is explained by stable personality traits throughout the individual's life, they would not be at greater risk of developing objective cognitive losses. Across age groups, depressive symptoms also impact cognition (e.g., McDermott and Ebmeier, 2009). 62 In many cases, cognitive effects resolve when depressive symptoms are treated, and in older adults, this would not necessarily reflect subclinical dementia. Additionally, individuals with a greater intensity of depressive symptoms may be more susceptible to interpretation biases that sensitize them to report more SCD. 40 The lack of significance might also be explained by the relatively small size of the sample compared to other studies carried out in the same field. This could have increased the risk of a type II error.
Second objective: presence, type, and intensity of anxiety symptoms
The second objective of this study was to compare the presence, type, and intensity of anxiety symptoms in individuals with SCD-P. The first hypothesis was that anxiety symptoms would be significantly more present in people with SCD-P than people with SCD-NP. This hypothesis was rejected. Indeed, 52.5% of SCD-NP individuals compared to 47.6% of SCD-P individuals reported anxiety symptoms. This difference did not reach the threshold of significance, contradicting the study of Liew, 20 implying that the report of anxiety symptoms increased the risk of developing objective cognitive losses. In the study by Anh et al., 18 the evaluation of the presence of these symptoms was carried out by asking a single question for the presence of anxiety: “Do you have signs of nervousness, including feeling tense, unable to relax, or short of breath?” Given that our study conducted a more comprehensive assessment of anxiety symptoms, it is likely that individuals recognized themselves in at least one anxiety symptom, making the presence of anxiety symptoms between groups similar, reducing the likelihood of finding a significant difference.
The second hypothesis was that SCD-P individuals would report more somatic anxiety symptoms and arousal than SCD-NP individuals. This hypothesis was also rejected. Regarding anxiety symptoms, no type of anxiety symptom significantly distinguished the groups from each other. The initial postulate was based on Liew's 20 results where, remember, there was only one question that made it possible to distinguish the SCD-P group. Furthermore, the question was relatively general. A parallel was found between the elements contained in this question and the somatic and arousal dimensions identified by Bandari's team (2019) 28 on the GAI. Although it did not prove significant, the study presented here is the only one listed that used the GAI to characterize the experience of anxiety in individuals with SCD-P. This provides a more detailed portrait of this experience than what had been painted earlier.
The third hypothesis was that the intensity of anxiety symptoms would be significantly higher in SCD-P than SCD-NP individuals. This hypothesis was also rejected. For both anxiety and depressive symptoms, the intensity of these symptoms did not differ between the groups. These results showed that the individuals with SCD who developed cognitive losses after 4 years did not present higher levels of anxiety or depressive symptoms than those who did not develop them after the same period. Regarding anxiety symptoms, this contradicted Desai's 40 meta-analysis results. However, looking at the four studies included in that meta-analysis per anxiety symptoms, it was observed that two of the four studies used follow-up periods of more than 10 years,35,63 which makes the generalization of their results to ours unrealistic. Then, regarding the third study, the presence of anxiety was established by a question on the presence of concerns regarding subjective memory loss 64 —a question used to initially identify our SCD group—which also makes their result not clearly applicable to our study. Finally, the fourth study by Liew 20 assessed the intensity of anxiety symptoms by using the question mentioned previously. Also, participants had to evaluate the severity of their symptoms by a Likert scale, which provided a measure of their anxiety about perceived cognitive losses and not a measure of their general experience of anxiety, which might also explain the different results. Also, this lack of significant difference between the groups might be explained by the length of follow-up. Indeed, certain results of other longitudinal studies have led researchers to consider that the intensity of anxiety symptoms becomes important in the progression towards objective cognitive decline when the follow-up is of a longer duration, even more than 10 years. 65
Limitations and perspectives
Some of this study's results might also be explained by its limitations. Firstly, the small sample size may have reduced the power of some of our analyses. It also reduces the generalizability of our results.
Secondly, the time between the two measurement times (4 years) was quite short. It would be interesting to follow our participants longer than 4 years, knowing that it can take up to 15 years between the moment when the first subjective cognitive losses are detected by the individual until the moment when these losses are objectively detectable. 66
Thirdly, in terms of measurement, it would have been interesting to evaluate whether the impression of decline preceded or followed the experience of affective symptoms and whether, depending on the causal chain, there would have been a difference between those declining and those remaining cognitively stable per the type of anxiety and depressive symptoms reported. In retrospect, the SCD group selection criteria could have been more stringent by employing the SCD plus criteria as developed by Jessen et al. 3 These criteria are known to be more precise in identifying individuals with the highest rate of progression towards cognitive deficits. These criteria include the presence of a perceived subjective decline in memory abilities, persistence of this perception for at least 5 years, an age over 60, a report of concern associated with the perceived decline, consultation or seeking medical help for this problem, and that the participant's impression of decline is confirmed by an informant. 3 Unfortunately, the CIMA-Q project began before these criteria were published. However, by not applying these criteria, the present study has provided a more varied portrait of what individuals with SCD look like and still allowed us to draw interesting results regarding affective symptoms. More comprehensive anxiety and depression questionnaires would have served to better detail the types of symptoms. Indeed, the GAI and the GDS were not developed to assess types of symptoms but rather to assess the presence and intensity of affective symptoms.
Finally, in terms of statistical analysis, the fact that we carried out several comparisons may be considered a limitation, but as this area of study has been little developed to date, our results remain interesting for future research. Also, we did not control for the effect of potential covariates (e.g., age, gender, etc.). Given that we had a small sample size, that our groups were of different sizes, and that they did not differ significantly at baseline in terms of age, gender, years of education or intensity of anxiety and depression, we felt justified in not doing so. As age tended to become significant, we re-run the analyses with age as a covariate; results remained not statistically significant.
Conclusions
The general objective of this study was to characterize the anxiety and depressive symptoms of individuals with SCD who developed objective cognitive losses after 4 years. This was accomplished by comparing them to their peers who also presented with SCD but who did not develop objective cognitive losses in the same time interval. Our results demonstrate that the presence and intensity of anxiety and depressive symptoms did not significantly differentiate the groups. There was also no significant difference detected per the type of anxiety symptom reported. However, the depressive symptoms of hopelessness set them apart.
Overall, this study provides avenues for future research. Indeed, it would be relevant for future research to measure hopelessness in other groups of elderly people such as cognitively healthy people as well as those already presenting with MCI or AD. Second, the creation of interventions adapted for this diagnosis should be considered. At the moment, there is no consensus on the best way to care for these individuals, and as determined by the meta-analysis of Bhome et al., 67 the scientific literature currently lacks a high-quality study of effective interventions to treat or support individuals with SCD. Indeed, memory clinic resources are currently focused on treating individuals who already have cognitive deficits. However, treatments targeted for SCD-P individuals could have the potential to slow or at best prevent progression to AD. 67 Regardless of the etiology, individuals with SCD could all benefit from interventions likely to improve cognitive abilities or slow the decline that is perceived in the moment. 68 Indeed, non-pharmacological interventions are in current development. It thus far appears that group interventions involving psychoeducation are effective in improving the general well-being of people with SCD, which may be relevant for individuals at greater risk of experiencing depressive and anxiety symptoms. Our results suggest that it may be beneficial to target hopelessness / powerlessness in non-pharmacological interventions aimed at preventing the progression of people with SCD to MCI or AD. This could include teaching them strategies to improve their memory or, in other words, to improve their sense of self-efficacy. Also, the types of interventions specific to the improvement of metacognitive abilities have seemed to generate more and more interest because deficits in this sphere can contribute to SCD especially for those whose etiology of the disorder is not due to the development of AD. 67 Metternich et al. 69 found that cognitive restructuring reduced subjective memory complaints, while memory training improved objective memory function, which may be more indicated for those with SCD who are at greater risk of developing AD. The results of our study might allow stakeholders to better distinguish these two groups and direct relevant and effective interventions according to their patients’ etiology. Ultimately, this could have a significant impact on AD prevention.
Footnotes
Acknowledgments
The authors would like to thank all members of the CIMA-Q project. Also, for the editing of this article, the authors would like to thank Proof-reading-service.
Author contributions
Beatrice Raymond-Lessard (Conceptualization; Data curation; Formal analysis; Writing – original draft); Bélanger Claude (Supervision; Writing – review & editing); Hudon Carol (Supervision; Writing – review & editing); Grenier Sébastien (Conceptualization; Supervision; Writing – review & editing).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CIMA-Q is a research project funded by the Fonds de recherche du Québec–Santé (FRQS), Pfizer Innovation Program [grant number #27239], the Quebec Network for Research on aging (RQRV; Réseau Québécois de Recherche sur le Vieillissement), a network supported by the FRQS, the Fondation Courtois (NeuroMod project), the Consortium for the Neurodegeneration associated with Aging (CCNA/CCNV; grant CAN #137794), the Canadian Institutes of Health Research (CIHR; grant #154265), and the Foundation Famille Lemaire. Dr Sébastien Grenier is supported by a FRQS Senior salary award.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
