Abstract
This study aimed to explore the acceptability of a novel device (‘MyShield’): a device used for distraction during clinical procedures. It is a cardboard cuff, designed to fit around the arm, either above the elbow or around the wrist and used to hide the procedure from view. This device was tested in practice, to establish acceptability to children, parents and clinical staff. Fifty-eight children tried ‘MyShield’ during a venepuncture procedure. Feedback from 54 children, 58 parents/carers and 16 clinical staff was collected using surveys and interviews. In 24 cases, observational data were also collected. A large majority of children (94%, n = 51) and parents (96%, n = 56) reported a positive experience when using ‘MyShield’; saying they would likely use it again. Potential of ‘MyShield’ in promoting parent/clinician interaction with the child was highlighted. Data suggests that ‘MyShield’ may be a useful device for children undergoing venepuncture, when used in conjunction with standard care, and subject to individual preferences and choice. Further work is required to establish mechanism of action and whether use of ‘MyShield’ has any impact across a range of short- and long-term outcome measures relating to patient experience and effectiveness.
Background
Children undergo a variety of needle procedures as part of routine medical care (Taddio and McMurty, 2015). Procedures are reported as one of the most frightening and upsetting aspects of medical treatment and hospitalization (Heden et al., 2019; Kleye et al., 2021; Uman et al., 2008). Such procedures, that include venepuncture and venous cannulation, are also a common source of pain for hospitalized children, and are known causes of anxiety, distress and fear (Inal et al., 2012a; Inal et al., 2012b; Orenius et al., 2018; Stevens et al., 2011). Inadequately managed pain can have negative emotional and psychological implications for children and those around them. Quality of a child’s experience during procedures is correlated with anticipatory distress and increased pain during future medical treatment (Campbell et al., 2017); the development of needle phobia (Orenius et al., 2018); increased pain sensitivity; and avoidance of health care as an adult (Palermo et al., 2020). These procedures also have an impact on those surrounding the child, provoking significant distress and anxiety in parents (Brown et al., 2019; Smith et al., 2007), and perceived as challenging by clinicians (Papa et al., 2008).
Implications of improved procedural pain management on the practical dimensions of delivering care, including overall time and success rates for procedures, have been demonstrated (Kennedy et al., 2008). There is evidence that a multi-disciplinary approach using multi-modal interventions improves clinical outcomes and maintains quality of life (Jiminian et al., 2021). When children are prepared and informed about what will happen during a procedure, their overall experience will be better (Bray et al., 2019, 2021). Evidence details a range of both pharmacological and non-pharmacological pain management interventions available and recommended in guidance (Emergency Nurses Association, 2019). Despite this, concerns remain that care provided during procedures can often be inadequate (Eccleston et al., 2021). A recent narrative overview of management during venepuncture places an emphasis on the need to put interventions into everyday practice (Cozzi et al., 2020).
The use of non-pharmacological methods to reduce fear and anxiety associated with procedures are recommended. There is substantial evidence for their effectiveness, with strongest evidence for distraction and hypnosis (Stinson et al., 2008; Uman et al., 2013). Two approaches to distraction are available to use: passive and active. Passive distraction involves diverting the child’s attention away from the source of pain, using auditory or audiovisual methods (such as a toy, a book, flashing lights); or a locally applied vibration, such as BUZZY (Kucuk Alemdar and Yaman Aktas 2019; Moadad et al., 2016); or more recently social robots (Ali et al., 2021; Jibb et al., 2018; Moerman et al., 2018); and/or through engagement with the clinician or parent (McCarthy et al., 2010). In contrast, active distraction encourages the child to do something different, such as play an electronic video game (Inan and Inal, 2018); use a hand-held computer (Crevatin et al., 2016); or use virtual reality goggles (Indovina et al., 2018; Ozkan et al., 2020; Piskorz and Czub, 2017; Wittkopf et al., 2019). Overarching both types of distraction, is a new category of interventions, described as ‘digital technology’, including but not limited to television, DVDs, videos, personal computers or laptops, tablets or iPads, smartphones, mobile apps, video games, virtual reality and humanoid robots (Gates et al., 2020). Effective distraction may not only modify the child’s experience of a painful event itself, it may also buffer memories of the event and have an impact on the child’s response to painful procedures in the future (Cohen et al., 2001; Salmon et al., 2002).
Even randomized controlled studies are yet to identify the most effective distraction method between active and passive interventions and those that are digital or non-digital (Newell et al., 2018). Many of these studies have been judged as low or very low quality (Birnie et al., 2018). This leaves clinical staff unsure of which approach might work best, with which child, under what kind of circumstances.
‘MyShield’ is a more recent addition to the ‘toolbox’ of active distraction devices. It was produced by a product and innovation team K8, working with ConceptoMed, a medtech company (K8 industrialdesign). It is a cardboard cuff, 20 cm by 29 cm (when flat), designed to fit around the arm, either above the elbow or around the wrist and held in place with a sticky pad which attaches the two sides (legs) of the shield under the arm (see Figure 1 for an image of the device). Depending on preference it can be affixed either above or below the tourniquet. The device is used both to hide the procedure from view and engage the child with graphics printed on one side. Children can select their preferred graphics from a range of available designs, which include both single image photographs (of animals), or illustrations of different types of scenes with objects to search for (hidden pictures). Preliminary feedback from an unpublished small-scale study with a Norwegian sample, conducted by the company manufacturing this product, suggests that children would use the device again, if offered in future procedures, and most (77%) had a better experience using ‘Myshield’ when compared to previous experience of the same procedure without the shield (K8 industrialdesign). There is, however, no published and independent evidence in relation to the potential benefit of using this device and no data relating to the use of the device in clinical practice or its acceptability to the target population. An image of the device.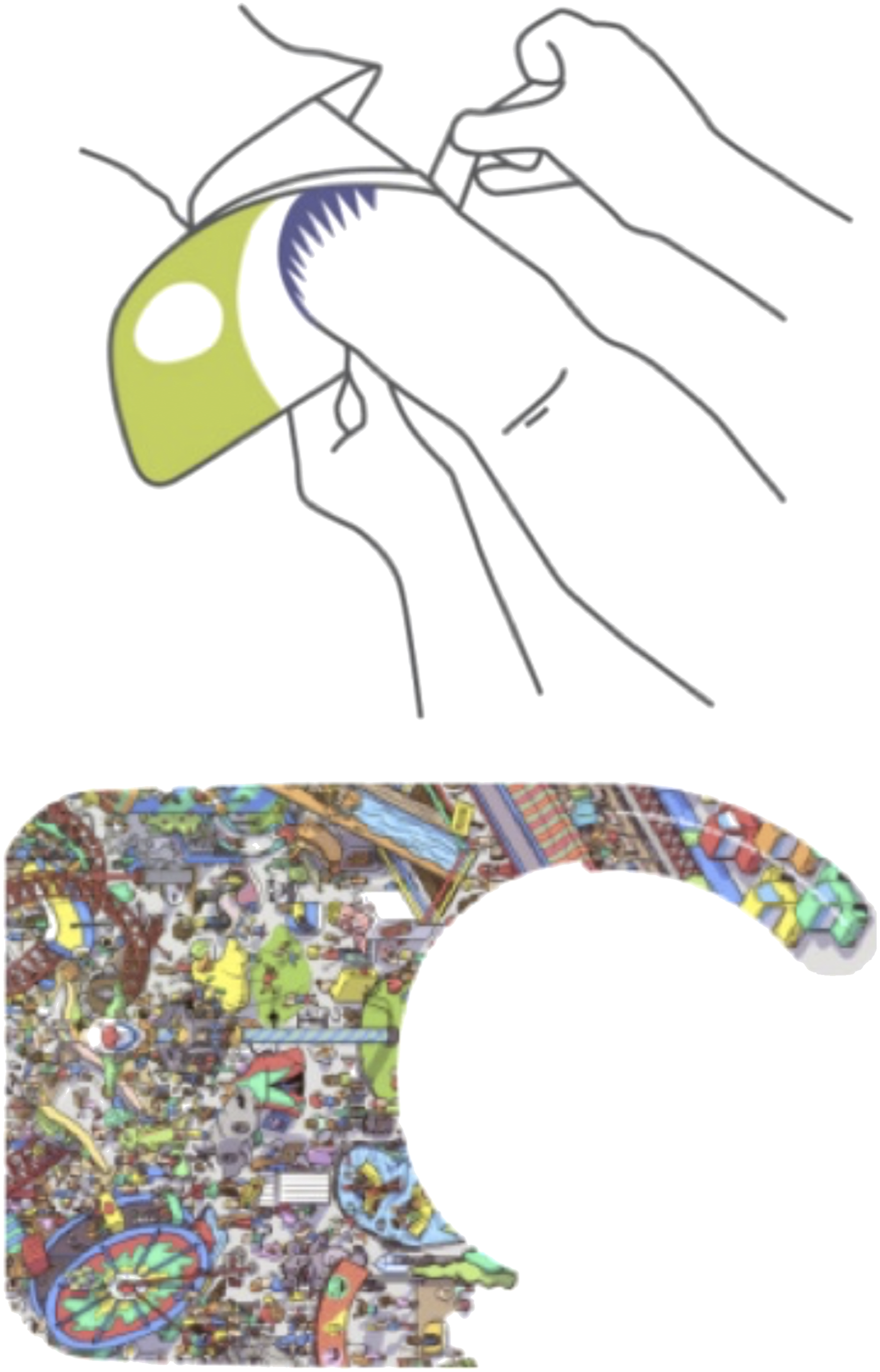
Aim
To understand the practical use of a novel intervention designed to support active distraction of children during venepuncture (‘MyShield’) and the acceptability of the intervention from the child, parent and clinical staff perspectives.
Methods
Study design
This study used a descriptive quantitative and qualitative design (O’Cathain et al., 2015).
Framework
Definitions of the component constructs in the Theoretical Framework of Acceptability (Sekhon et al., 2017).

Setting and sample
Research was conducted in a children’s hospital, in the Greater London Area, in the United Kingdom during 2015. A convenience sample of participants (age range 4–12 years), undergoing non-emergency venepuncture were approached to participate. Children were excluded if they: were unable to complete the data collection surveys in English; had a skin condition that might be exacerbated by use of the device.
In our hospital, venepuncture is undertaken by a range of clinical staff, that includes, phlebotomists, clinic/healthcare assistants, nurses and medical staff. In addition, there is a specialist vascular access facilitator who can be contacted for particularly difficult or challenging venepuncture situations.
Ethical considerations
Our study fulfilled NHS ethical approval was awarded in December 2014 requirements and was awarded in December 2014 (reference: 14/NW/1532). All participants were provided with age appropriate study information and were able to decline participation at any time without comment or penalty. Informed written consent was obtained from parents/caregivers and clinical staff. Assent was obtained from children, whose parents provided consent for them to participate.
Data collection
A lead member of staff was identified for each of the clinical areas involved. They identified and approached potential participants and provided them with information sheets on arrival in clinic/during their stay on the ward. Some parents approached the researcher direct in response to a poster in each of the clinical areas. The researcher followed up on all contacts to answer any questions. Our study involved collecting survey and interview data from children, parents and clinical staff, and observational data of the device being used in practice; corresponding to the elements of the theoretical framework. Parents and children were given separate paper-based pre-procedure surveys eliciting their child’s demographic details and any previous experience of venepuncture.
All participating children completed a pre-procedural survey to assess anticipatory distress and levels of anticipated pain in advance of the procedure. This consisted of two rating scales currently used within our hospital: the faces scale for younger children (age 4–8 years) (Wong and Baker, 1998; Tomlinson et al., 2010); numerical rating scale for older children (aged eight and over) (Castarlenas et al., 2017). One free selection question from a word list was also included to elicit feelings associated with venepuncture. Anticipatory and procedural distress were assessed using a 0–10 scale (0, ‘not at all worried’–10 ‘really worried’). Coping style was elicited using a single item question: ‘Imagine if you scraped your knee or cut your finger. Would you want to look at it while it was being cleaned or someone put a plaster on it, or would you want to look away at something else?’ Children were supported with completion of the survey by the researcher who was able to explain the use of the scale where they were not able to read numbers and written words.
Parents completed three measures pre-procedure: rating their child’s anticipatory anxiety/distress; their own perception of how painful the procedure would be for their child; their own anxiety. All ratings were assessed on a 0–10 scale with 10 being the highest level. Post-procedure surveys included questions relating to their experience of the venepuncture and experience of using the device
Clinical staff either participated in an interview, undertaken by the researcher (AW), or completed a feedback form. Interviews and written feedback both included questions about ease of use, practical considerations, views on design and suggestions for any improvements.
The device was used during the procedure, following manufacturer instructions and as an adjunct to usual practice. Usual practice in this clinic varied somewhat depending on child and parent preferences but all procedures were carried out by a clinic assistant. Parents were usually present unless the child or parent requested otherwise. Where venepuncture proved particularly difficult a second clinic assistant was called upon to assist. In cases where a child had previously been highly anxious, or parents requested support in advance, a play specialist was available to provide specialist input. All families were asked by the person admitting them to clinic if they would like topical anaesthetic such as EMLA cream (lidocaine 2.5% and prilocaine 2.5): only those who requested it received the topical anaesthetic pre-procedure. For a proportion of the procedures the researcher was present to observe, and recorded information about it, including time taken, use of topical anaesthetic, number of attempts at venepuncture, noting also interaction between child, clinician and parent. Some issues which emerged in the first six weeks of the study in relation to practicalities were addressed by the manufacturers in a later, refined version of the device, including size and design features. This involved reducing the length of the ‘legs’ so that they were easier to attach around smaller arms, reducing the total surface area of the shield to minimize the risk of casting a shadow over the arm (some staff expressed concern that it would make it more difficult for them to see the vein), and making it more user friendly by ensuring the ‘key’ for finding things in the pictures was more readily visible.
Data analysis
Descriptive statistics were performed to establish sample characteristics and percentage responses to survey questions. As an acceptability study, inferential statistics relating to the main outcomes of interest was not conducted. Open questions were subjected to content analysis, counting words and phrases used, summarizing and identifying patterns, described as summative content analysis (Hsieh and Shannon, 2005).
Results and findings
Sample characteristics including age, gender and coping style (assessed with a single question – information about you).

Child feedback on use of ‘MyShield’
All participating children had experience of blood being taken. In response to the single item coping style question about whether they would prefer to look or look away whilst an injury was being tended to, 81% (n = 44) of children said they would prefer to look away, 11% (n = 6) said they would prefer to look and 7% (n = 4) said they ‘weren’t’ sure or ‘didn’t mind’. Pre-procedure, median self-reported anxiety and anticipated pain levels were 3 (IQR:4.5) and 3 (IQR:2), respectively. Post-procedure, median self-reported anxiety and pain levels (during the procedure) were 3 (IQR:2) and 2 (IQR:1), respectively.
Children answered two questions about how much they enjoyed using ‘MyShield’ and how easy they found it to use, and one question asked if they would use it again. The majority of children said ‘it was great’ and reported that they were very likely to use it again in future blood tests (Figure 2). Participants selected a range of words to describe their feelings pre-procedure, happy, frightened, worried, brave, calm, bored, tired, interested, upset, scared and post-procedure, happy, calm, worried, bored, interested, brave. Child responses relating to ‘MyShield’ experiences and likelihood of future use.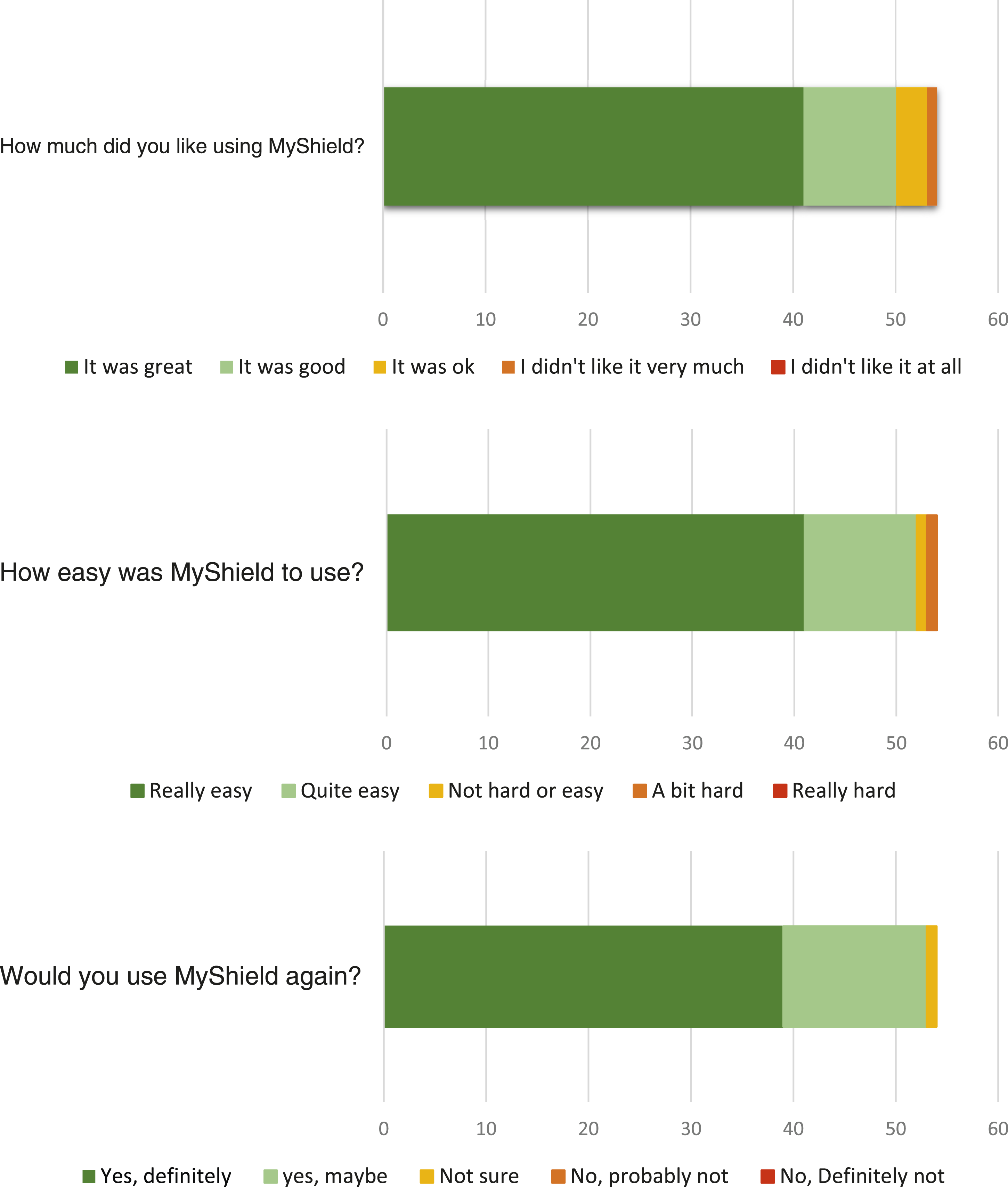
Aspects of ‘MyShield’ that children reported liking included: ‘the pictures’ (61%, n = 33); ‘the colours’ (39%, n = 21); the fact that ‘it hid the needle’ (72%, n = 39); and ‘looking for things in the pictures’ (17%, n = 9). However, a small minority of children (2%, n = 1), did not like the fact that it hid the needle. When asked to compare the experience of having blood taken using ‘MyShield’ with experiences of having blood taken without it, 94% (n = 51) of children reported that ‘MyShield’ made the venepuncture procedure ‘better’; 5% reported that it was ‘just the same’; and 1% (n = 1) reported that ‘MyShield’ made the experience ‘worse’. ‘MyShield’ was described as helpful: ‘It is very good, I want to use it again’; ‘blood tests are better with MyShield’. Despite this, some improvements were highlighted by children: ‘add some more animals and put glitter on it’; ‘add in characters from Frozen, or Disney’.
Parent feedback on use of ‘MyShield’
Parents’ median score was 8 (IQR: 2) on the question of how much their child liked using ‘MyShield’, on a scale of 0 (‘Didn’t like it at all’) to 10 (‘Really liked it’). Their median score was 9 (IQR: 2) (on a scale from 0: ‘Not at all likely’ to 10 ‘Definitely’) when asked how likely their child was to use ‘MyShield’ again. Pre-procedure, the median score for parent reported levels of the child’s distress about the procedure was 6 (IQR:2.25) and anticipated pain was 3 (IQR:2). Post-procedure, median parent reported levels of the child’s distress and pain during the procedure were 5 (IQR:3) and 3 (IQR:3), respectively. Median parent reported levels of their own anxiety about their child’s procedure were 2 (IQR:3) pre-procedure and 2 (IQR:2.25) after the procedure. As previously noted, these figures should be interpreted with caution given the design and purpose of the study.
Ninety-six percent (n = 56) of parents whose children tried using ‘MyShield’ reported that it made their child’s experience of having blood taken ‘better’. Four percent of parents (n = 2) reported that their child’s experience was ‘just the same’, but none reported that ‘MyShield’ made it ‘worse’. Some parents gave feedback through open comment boxes in the surveys. They suggested that some of the designs, which featured a single photograph of an animal, ‘don’t really give enough to talk about’, although others said that their child really enjoyed using them because they loved animals and therefore found it engaging. One area for improvement was to: ‘add more pictures to select, or have flashcards to swap in and out rather than a permanent picture’.
One parent suggested that introducing the device to children with enough time before the procedure was important: ‘It takes a few minutes to get engrossed, giving [it] to the child earlier is better, the search game needs more time to be effective’.
Some parents highlighted practical issues such as finding it hard to attach to the arm, with the potential for it being: ‘fiddly for the nurse with the tourniquet’, or the ‘key highlighting pictures for the child to search for being difficult to see’.
One parent noted that their child responded to being given a choice of designs: ‘My child was interested in looking and selecting a “MyShield” for the procedure’.
Parents also commented on the benefits of not seeing the needle going in, describing it as ‘very good distraction’.
Whether the intervention offers anything more to some children than what they already use was highlighted by a parent: ‘He currently looks at an Ipad (plays a computer game) when having bloods taken and this has proved a very good distraction device. He did look at the “MyShield” during the procedure and engaged with it - I am unsure whether he would choose the “MyShield” over the Ipad for future blood tests’.
Observational data and clinical staff feedback The researcher observed 24 cases. Topical anaesthetic was used in 63% (n = 15) of these cases, with an average time of 4.5 min taken for the procedure (measured from the time the child sat down with the clinic assistant to the time the procedure was complete and they were ready to leave the procedure room). In 80% of cases (n = 19), the child attempted to look behind the device as soon as they felt the sensation of topical anaesthetic being applied to their skin or the clinical assistant touched the arm to find a vein. Parents were often involved in the procedure by the clinic assistant who would suggest the child sat on the parent’s lap, or sat next to them and spoke to their parent for the duration of the procedure. Those using ‘MyShield’ often had parental involvement in searching for the images or talking about the image. Clinical staff feedback primarily related to practical, individual and contextual considerations in using ‘MyShield’ and their views on its optimal use in practice. Many staff suggested that ‘MyShield’ would be particularly useful in clinical contexts where there are limited resources available for children undergoing procedures. This included non-specialist children’s services, or contexts where there is unlikely to be play specialist input or availability of non-pharmacological distraction ‘tools’. Where play specialists are available prior to and during procedures ‘MyShield’ may not offer any advantage over the methods they use currently, and clinical staff suggested that children who are particularly anxious about having blood tests would require additional input. Clinical staff noted that due to the fact that it is single use and disposable, ‘MyShield’ could be particularly useful in cases where strict infection control procedures are implemented. In these situations, it would have a distinct advantage over other commonly used distraction devices such as books or toys. They also highlighted its potential for use with procedures other than venepuncture, such as small wound dressings. Some practical issues raised were related to its optimal use, with staff suggesting that it needed to be fixed high on the arm so that it did not interfere with placement of a tourniquet, and consideration given to lighting since there was potential for the device to cast a shadow over the venepuncture site. None of the shields broke or slipped off during use, or interfered with the procedure itself once it was being used. All children who agreed to have the shield on during a procedure kept it on once it was placed. In relation to ‘MyShield’ graphics, staff commented that the devices with ‘busier’ pictures promoted more active involvement and provided the greatest potential for effective distraction. Clinical staff reinforced need for engagement between staff involved in procedures, parents and children, and more complex designs were seen as potential ‘tools’ to facilitate this engagement. One member of clinical staff commented that usual practice with distraction was to engage the child’s attention away from the site of the procedure, and suggested that this device would not necessarily achieve this aim, since the distraction device was situated at the location of the venepuncture. Although none of the shields failed to obscure the view of the needle, one member of staff noted that the device would not shield all aspects of the procedure from view, and for children who are very afraid or anxious about needle procedures, highly needle phobic, any of the equipment associated with venepuncture had the potential for causing distress – and this would need to be considered.
Discussion
The primary aim of this study was to establish the acceptability of ‘MyShield’ to children, parents and clinical staff. In addition, we sought preliminary feedback on its impact on patient experience. Key findings revealed the vast majority of children and parents reported that ‘MyShield’ made the experience of venepuncture better, were likely to use it again and found it easy to use. Clinical staff found it to be highly acceptable for use as an adjunct to usual care though not as a replacement for specialist input where required.
Clinical staff and parents both noted that designs with greater potential for active involvement and engagement from the child were more likely to successfully distract children during procedures. This is consistent with some of the existing evidence on effective distractors in these settings, which suggests they are most effective when more active cognitive processing and more motor responses are required (Dahlquist et al., 2002; Duff et al., 2012). The greater the interactiveness, the more superior the distraction technique (Birnie et al., 2014). Gates et al. (2020) have reaffirmed the view that distraction appears to have a positive effect on both pain and distress, but digital technology does not seem to confer an obvious advantage over non-digital distractors. Feedback from clinical staff in our study was consistent with this; they also suggested that the designs which facilitated engagement between staff or parents and the child were more likely to be effective. Also, a device such as ‘MyShield’, which does not require additional input from specialist staff, might be particularly useful in settings with few resources available to distract or engage children, such as in the community.
Further research is required to establish who might benefit most from using ‘Myshield’, but our preliminary data suggests that those most likely to express an interest in trying the device are children who would prefer to look away from a procedure and those who do not already have established routines for dealing with blood tests. Bernard et al. (2004) found that children’s reported preferences did not correlate with their actual behaviour during a venepuncture procedure and suggest possible interpretations: children may have difficulty identifying their own coping strategy, or the question asked may not reflect different approaches at different developmental stages. However, children’s expressed preference for looking or not looking at what is happening to them should always be given due consideration (Bisogni et al., 2014). Their active participation and control in selecting distraction techniques, as one element of choice in how a procedure is done, may be critical as a coping strategy (Nilsson et al., 2011) and as a way of reducing feelings of uncertainty and helplessness during medical procedures (Duff, 2003).
It is not possible, from the current study, to conclude what the precise mechanism of action of ‘MyShield’ actually is. For example, potential improvement in patient experience would manifest itself by its use as a distraction device, by concealing the needle, an interaction or both or another mechanism altogether. It is, however, likely that both developmental and individual characteristics of the child, and the degree to which it is used interactively or not, will be critical in understanding how its effectiveness varies in different situations and with different individuals. Our study presents only preliminary data on the acceptability of the intervention to those involved in venepuncture procedure and its potential impact on patient experience. It resonates with other studies in highlighting context and preferences as important indicators for choice of distraction to use (Gates et al., 2020). However, it differs to many studies examining the effectiveness of non-pharmacological interventions by asking children directly why they liked the device and what suggestions they had for its improvement.
Strengths and limitations
Results of this study should be considered in the light of the following limitations. First, the local context in which we tried the device was a specialist children’s hospital with a dedicated play specialist service and training for staff in pharmacological and non-pharmacological techniques for pain management. Standard practices for the management of pain and distress during blood tests were in place, including play specialist involvement and use of distraction ‘tools’. This, along with an experienced patient population, may explain the relatively low recruitment to the study and high refusal rate. Future work would benefit from the inclusion of children from a range of clinical settings.
Second, our sample was predominantly attending outpatients, accessing specialist services and was therefore likely to be experienced in having medical procedures. Many parents and children declined participation because they did not think it would be helpful; primarily either because children were happy to have their blood taken without any intervention, or because the child was highly anxious and had an established routine for coping with procedures. This limits generalizability of our conclusions in relation to the issues that children undergoing these procedures for the first time, and their parents and clinical staff, may face. Future work should include children with varying experience of having blood taken.
Third, this study did not include any randomization. This was a study to examine acceptability; our focus was therefore on assessment of the practicality of the intervention ‘MyShield’. This allowed us to explore any uncertainties regarding the device and to learn more about the use of the intervention in practice, before a full trial might be embarked upon. ‘MyShield’ was found to be useful to some children and clinical staff did not report it being a barrier or cause any delay to procedures significantly.
Finally, a strength of our study was to use qualitative and quantitative methods of data collection to examine both intervention content and delivery from the perspective of all stakeholders; we learnt from children, their parents and clinical staff, and this combination of perspectives should feature in other acceptability studies.
Implications for practice
Clinical staff require a ‘toolbox’ containing different kinds of distraction for use during venepuncture, as we know ‘one size never fits all’. When making a choice of which technique might be the best fit, consideration should be given to the following: is this the right patient, right time, right context, right place. In responding to these prompts, clinical staff will need knowledge of all techniques in the ‘toolbox’. This small study has shown that ‘MyShield’ was easy to use, the experience for the majority was a positive one, and therefore could be used in every-day clinical practice.
Conclusion
This preliminary data suggests that ‘MyShield’ was acceptable. Further work, that includes control data, is clearly required to confirm if it is a useful device for children undergoing venepuncture, when used in conjunction with standard care, subject to individual preferences and choice. This would also help to establish the precise mechanism of action of ‘MyShield’, and reveal trends that might be important such as age of the child, and previous experience of venepuncture. The findings here suggest that ‘MyShield’ is a ‘good fit’ for deliverers and receivers of the intervention, it might therefore find a place in the ‘toolbox’ of interventions available to clinical staff, for children and their families to select from.
Footnotes
Acknowledgements
We would like to acknowledge clinical staff, patients and families who took part in this study, including: Zoe Wilks (Head of Nursing, Outpatients department), the Outpatients nursing team and clinic assistants, Rosa Clayden and Jenny Dyer (play specialists), the nursing team and healthcare assistants at the Gastroenterology Suite and nursing staff and healthcare assistants in the Clinical Research Facility. Thanks also go to Jo Wray for a final proof read, and support with presentation of data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This acceptability study was funded by Conceptomed SA and supported in part by the NIHR Great Ormond Street Hospital Biomedical Research Centre. The authors received funding and materials from the company manufacturing the device (Conceptomed SA). This funding was used on researcher time and the materials received consisted of ‘MyShield’ for use in the study. The research team developed the protocol independently.
Ethical approval
Ethical approval was awarded from NRES Committee North West Liverpool Central (Project ID-164,750).
