Abstract
Existing clinical outcome assessments (COAs) have limitations in capturing treatment effects in autism for those who choose to enrol in clinical research. Some COAs include items that are not relevant or equally important to all autistic people; others are not sufficiently comprehensive. Goal Attainment Scaling (GAS), which measures individual experiences against personalised goals, has been proposed as a more individualised and change-sensitive approach to assess outcomes in clinical studies. We developed a novel GAS measure for use in paediatric autism studies based on qualitative interviews with autistic adolescents, parents and clinicians. Applying a methodologically robust approach, concept elicitation interviews (N = 40) were first conducted to understand and generate a bank of personalised goals, followed by cognitive debriefing interviews (N = 39) to provide feedback on a draft GAS manual. Twenty-five personalised goals were generated for core autistic characteristics (communication, socialisation, restrictive and repetitive behaviours), as well as associated characteristics and impacts. In debriefing interviews, adolescents and parents supported the use of personalised goals and clinicians recommended using such goals in clinical studies and practice. In conclusion, GAS measures may overcome some of the challenges with existing outcome measures and complement future outcome measurement strategies for clinical studies in paediatric autism.
Lay Abstract
Autistic children and adolescents and their parents support the use of personalised goals as a way of measuring the effects of new treatments for those who choose to enrol in clinical research. Health professionals also recommend using personalised goals in both their research and in treatment plans. Establishing personalised goals, a process known as Goal Attainment Scaling (GAS), means identifying goals according to the needs of the individual, as opposed to using standardised outcomes for all participants. Autism is a condition with many different characteristics and degrees of impact, and the tools commonly used to measure treatment effects, known as clinical outcome assessments (COAs), have limitations. Some COAs, for example, include outcomes that are not relevant to all autistic people; others do not include enough outcomes to represent the experience of all autistic people. GAS, which measures individual experiences against personalised goals, may be complementary to existing COAs to provide an individualised or tailor-made tool to evaluate outcomes as a result of an intervention. We developed a GAS measure for researchers/health professionals to use in studies with autistic children and adolescents. Our research was based on a total of 40 interviews with adolescents, parents and clinicians, asking open-ended questions about how children and adolescents experience autism in order to understand and create a bank of potential personalised goals. We then conducted 39 further interviews to ask them for their feedback on the first draft of the measure. In the end, we compiled a list of 25 personalised goals related to core and associated characteristics of autistic children and adolescents, including goals related to communication, socialisation, and restricted and repetitive behaviours. We hope that these findings will help to improve the measurement of meaningful outcomes for autistic children and adolescents in future studies.
Keywords
Introduction
Autism presents particular challenges in terms of assessment, diagnosis and classification, as well as in clinical management and therapeutic intervention (Gosling et al., 2022; Hus & Segal, 2021; Lord et al., 2022). Interventions are often dependent upon life stage, with short-term targeted psychosocial interventions aimed at pre- and school-aged children and their caregivers. Psychopharmacological interventions are more common for adolescents and adults, especially those experiencing co-occurring conditions such as attention deficit/hyperactivity disorder (ADHD), anxiety and aggression (Aishworiya et al., 2022; Lord et al., 2022). Despite these general patterns of intervention, however, researchers still lack basic knowledge about what treatment or assessment strategies in autism are most effective, when and with whom (Lord et al., 2022). There is a clear need for better understanding of the ‘key ingredients’ and mechanisms underlying change (Lord et al., 2022) that more effectively reflect the developmental and dynamic nature of autism (Chen et al., 2022).
Various clinical outcome assessments (COAs) focused on social communication and measures targeting restrictive repetitive behaviours (RRBs) have been used in clinical studies including interventional and non-interventional studies (Anagnostou et al., 2015; Bodfish et al., 2000; Grzadzinski et al., 2021; Scahill et al., 2015). Both types of instruments, however, have limitations. Researchers have argued that existing COAs such as the Vineland-3 (Sparrow et al., 2016) do not sufficiently distinguish between traits of autism and other aspects of neuropsychological functioning (Braconnier & Siper, 2021), while RRBs suffer from a lack of consensus in terms of their classification (Anagnostou et al., 2015). A review of instruments used to measure restricted interests and repetitive behaviours in autistic children and adolescents, for example, found that only 21% instruments were considered appropriate (Scahill et al., 2015); a similar evaluation of instruments used to measure social communication found only 16% of measures were appropriate for use in child and adolescent populations (Anagnostou et al., 2015). Furthermore, a systematic review of the measurement properties of tools used to measure progress and outcomes in autistic children up to the age of 6 years showed ‘patchy’ evidence and limited scope of outcomes, with little evidence to suggest the tools would be good at detecting change in young children (McConachie et al., 2015). In general, psychometric support for most COAs used within autism research is limited, particularly within the target population of autistic individuals, and important properties to support the validity of most measures as clinical trial outcomes (such as sensitivity to change or longitudinal structural invariance) are rarely, if ever, tested (Schiltz et al., 2024).
Regulatory guidance on clinical trial endpoints emphasises the need to align outcome assessments with lived experiences and to reflect what matters most to individuals (FDA, 2023). Personalised endpoints in particular may be of value and are especially important for conditions like autism, which are characterised by heterogeneous traits and impacts. One example of a personalised endpoint is Goal Attainment Scaling (GAS) (Kiresuk & Sherman, 1968; Krasny-Pacini et al., 2013). GAS was first developed as a measurement tool for assessing outcomes in the context of mental health (Kiresuk & Sherman, 1968). Since then, it has been modified and applied as an outcome measure in clinical and research settings across various healthcare disciplines, including rehabilitation, geriatric medicine, community health and drug trials (Cheema et al., 2024; Harpster et al., 2019; Logan et al., 2022; Schlosser, 2004; Turner-Stokes, 2009). Over time, GAS has proven to be an effective method for setting and measuring the achievement of goals both longitudinally and in a broad array of contexts (Turner-Stokes, 2009).
GAS can be approached broadly: for example, when individuals spontaneously create a goal related to a specific concept, or in a more targeted way, by selecting predefined goals from a library. In any case, the basic steps of GAS involve identifying goals, defining the current status (baseline) and determining outcomes on a predetermined scale (i.e. feeling/doing better or worse compared to the previous status). The GAS assessment process also considers both individual and environmental factors by weighting goals according to their importance and difficulty. Finally, GAS involves measuring and reviewing goal attainment by assessing the achieved outcomes and scoring them against the predefined attainment levels (Turner-Stokes, 2009).
The personalised nature of GAS means that the relevance and importance of the measure can be clearly demonstrated. The FDA has highlighted several key advantages to personalised endpoints such as GAS: namely, by being person-focused, they can better reflect how each individual feels or functions, and by isolating single concepts, they are able to exclude items that may not be relevant to all participants (thereby eliminating some of the risk associated with false forms of score inflation/deflation) or mixing affected and unaffected aspects of health within an individual (i.e. when constructing some multi-component endpoints) (FDA, 2023). GAS therefore has the potential to overcome the challenge of heterogeneity in autism by measuring personalised outcomes that are centred on an individual’s abilities, priorities and environment (Lee et al., 2022). Moreover, GAS might likewise address the limitations of existing COAs, which can overemphasise the identification and treatment of symptoms that patients themselves do not associate with distress or regard as impairments to social functioning (Markowitz et al., 2020). In this respect, GAS is in keeping with new and emerging research paradigms that aim to foreground the goals of autistic individuals – including children and adults – themselves (Ashburner et al., 2022; Mottron, 2017; Pellicano et al., 2022).
Components of GAS have been used in autism research within the educational setting (Ruble et al., 2012, 2018, 2022) and the community setting (Lee et al., 2022; McDonald et al., 2023) to assess the extent to which individual goals are achieved as the result of an intervention, and researchers in the adjacent field of intellectual/developmental disability research have published a framework to guide the use of GAS as a methodology for data collection in this area (Shogren et al., 2021). However, implementations of GAS in the autism literature thus far have not been designed for use by clinician-raters in the context of an interventional efficacy study in autism, such as to assess treatment efficacy in the context of large-scale pharmacological or medical device trials (i.e. in which goal levels may be more distal to the intervention mechanisms than in trials of behavioural or psychosocial interventions). Of course, as a standalone endpoint, GAS has its limitations (Urach et al., 2019). When used in tandem with other COAs, however, the FDA has suggested that personalised endpoints such as GAS could inform decision making about the effect of a medical product (FDA, 2023).
The aim of this study is to report on novel efforts undertaken to develop a GAS measure to be used in identifying personalised endpoints for autistic children and adolescents (5–17 years) taking part in future clinical interventional studies such as clinical trials. Further aims of this study include the systematic development of a bank of example goals, a GAS scale and a manual for GAS raters.
Methods
This qualitative study was designed with two waves of interviews: concept elicitation and cognitive debriefing. Across both waves we interviewed three distinct cohorts of participants: (1) autistic adolescents, (2) parents of autistic children and adolescents, and (3) clinicians with experience administering COA in research settings including goal setting in autism. This study received ethical approval from the Institutional Review Board Advarra (ref: Pro00065915).
To ensure relevance for the autism community, an expert panel of autistic individuals, advocates, parents, carers and research clinicians from both academia and industry provided input and oversight throughout the research. Their input included reviewing study materials and providing feedback and input on results and recommendations (Figure 1). This process ensured that the perspectives of all stakeholders were incorporated in the production and refinement of the GAS measure. It is important to note that members of the expert panel were involved in an advisory capacity only and were not study participants.
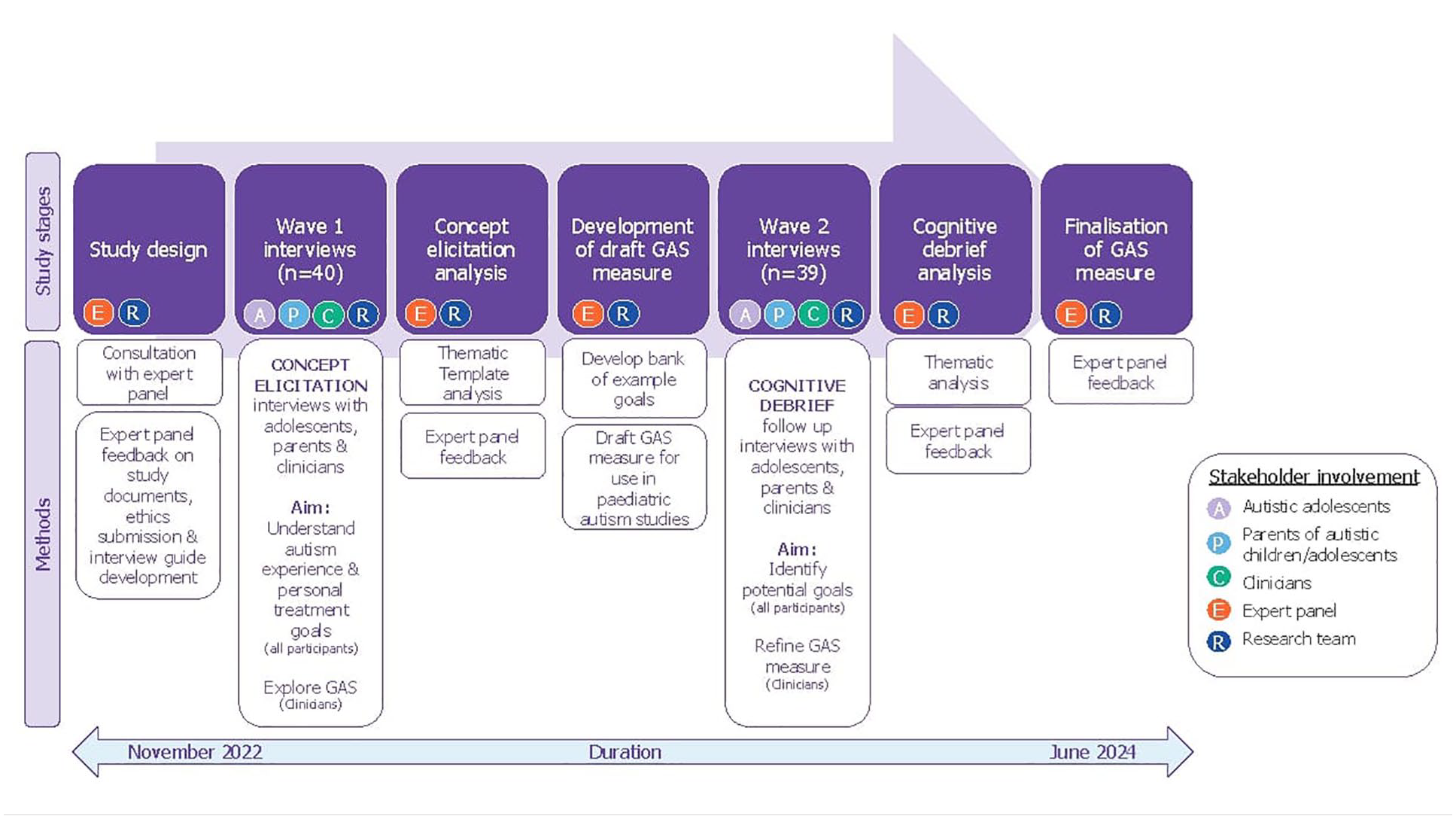
Overview of methods of GAS measure development and stakeholder involvement.
Sample and recruitment
Three participant cohorts, including autistic adolescents (n = 10), parents (n = 20) and clinicians (n = 10) based in the United States and Canada, were recruited using purposive sampling between December 2022 and April 2023, and received compensation in line with fair market value for their participation. Dependent on the group, eligibility criteria included (1) adolescents (12–17 years) with a confirmed diagnoses of autism by a licenced clinician, willing and able to provide written informed assent and parent or guardian written informed consent; (2) parents of an autistic child or adolescents (5–17 years) who had a confirmed diagnosis of autism, no intellectual disability (ID) or a co-occurring diagnosis of mild ID (IQ ⩾ 50); (3) clinicians with PhD, PsyD, or MD qualification, minimum of 5 years’ experience working with autistic children and adolescents or their parents in clinical and/or research settings, with a minimum of 3 years’ experience of administering COAs (e.g. Vineland, clinical global impression scales, personalised goal setting) in clinical and/or research settings. In an effort to ensure representation across the age range of the study, three target sub-group quotas for the parent cohort were set (i.e. 5–7 years (n = 8), 8–11 years (n = 7), 12–17 years (n = 5)) (see Table 2). Note that due to the variation/absence of documented IQ and/or intellectual disability, we were not able to accurately report on this characteristic for all participants.
Interview and analysis procedures
Participants interviewed in wave 1 were followed up in wave 2. Using cohort-specific semi-structured interview guides, 1:1 interviews were conducted in English by experienced researchers at Modus Outcomes using MS Teams. Interview pace and topics were adjusted based on the individuals’ focus and attention span. Audio recordings were anonymised and transcribed verbatim. Transcripts were imported to qualitative analysis software (ATLAS, 2024). Data were analysed using template thematic analysis (Brooks et al., 2015), an approach that is highly flexible allowing for both a priori and in-vivo coding. Preliminary coding was undertaken independently on two initial transcripts by V.H. and B.E. to develop a provisional coding ‘template’. This was applied to remaining transcripts and modified on an iterative basis with input from the Modus Outcomes team, enabling reflexivity in the development of ideas and interpretations of particular phrases. Following coding of all transcripts, the template was refined to improve clarity of codes and remove duplicate codes, jointly done by V.H. and L.B. Themes were organised within a hierarchical structure and mapped to a conceptual model previously developed (McDougall, 2017). This model consisted of n = 3 core domains (Core Characteristics, Associated Characteristics and Impacts), n = 9 subdomains (Communication, Socialisation, Restrictive/repetitive patterns of behaviour, Physical, Cognitive, Emotional/behavioural, Activities of daily living, School/work and Social life). Based on the mapping results, refinements were made, resulting in a new iteration of the conceptual model shown in Figure 2.
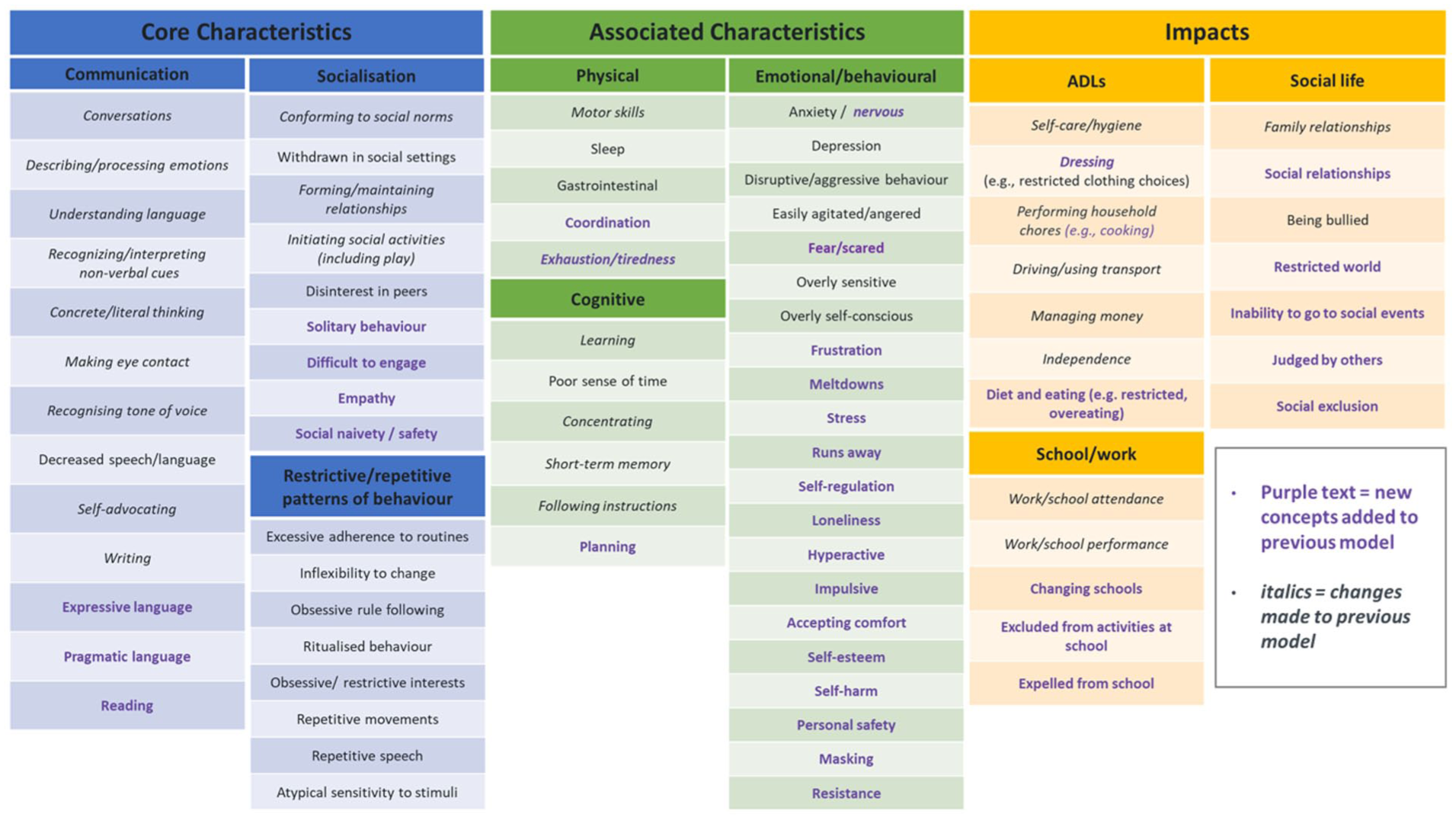
Consolidated conceptual model of the experience of paediatric autism.
Wave 1 concept elicitation interviews, focused on understanding personal treatment goals of autistic children and adolescents, as well as those of their parents, and gaining insights from clinicians. Specifically, participants were asked which concepts of paediatric autism were important to them and to consider three goals they would like to work towards achieving over the duration of a school semester (12 weeks) (Supplemental file 1 and file 2).
Guided by the refined conceptual model, domains and subdomains of importance, and to categorise the goals suggested, these findings supported the generation of a bank of example goals and the development of the draft GAS measure. The bank of example goals are intended to provide GAS raters (researchers/clinicians with experience in autism) with a starting point for discussions with participants about individualised goals they wish to work towards during a clinical interventional study. Ideally, in a study setting, participants will identify three broad goals (either from the bank of examples or from scratch), which would be explored further with GAS raters and defined as specific, measurable, achievable, relevant, time-bound (SMART) goals (Doran, 1981). The GAS methodological framework we adopted, such as, the number of goals individuals set, who determines the goals (i.e. the autistic child/adolescent, their parent or both), how SMART goals are formed, and the goal difficulty and importance rating, were developed during the course of an independent study that was discontinued. The measure was designed to be used by GAS raters setting and reviewing personalised treatment goals in the context of assessing a future clinical intervention in a clinical trial with autistic children and adolescents. Wave 2 interviews focused on cognitive debriefing the bank of example goals with all participants and gaining feedback on the draft GAS manual with clinicians.
Reflexivity
The interviews and analysis were conducted by a multidisciplinary team of researchers with diverse professional backgrounds and perspectives, including psychology, speech and language therapy and clinical practice. Some team members brought direct professional or personal experience to the topic, while others, as relative outsiders, contributed fresh perspectives and helped challenge assumptions during analysis. To enhance reflexivity, we engaged in regular team meetings where we critically reflected on our interpretations and decision-making processes. We also maintained notes and analytic memos to document our reflections and rationale for key methodological choices.
We acknowledge that our individual and collective perspectives inevitably shaped the research, and we offer this statement in the interest of transparency and to support readers in contextualising our findings.
Results
A total of 40 participants were interviewed across both waves (see Table 1). During wave 1, n = 40 interviews took place between December 2022 and April 2023. During wave 2, n = 39 interviews were conducted between November 2023 and January 2024 (due to illness one clinician withdrew from wave 2). Interviews ranged approximately 60–75 min in duration.
Characteristics of sample.
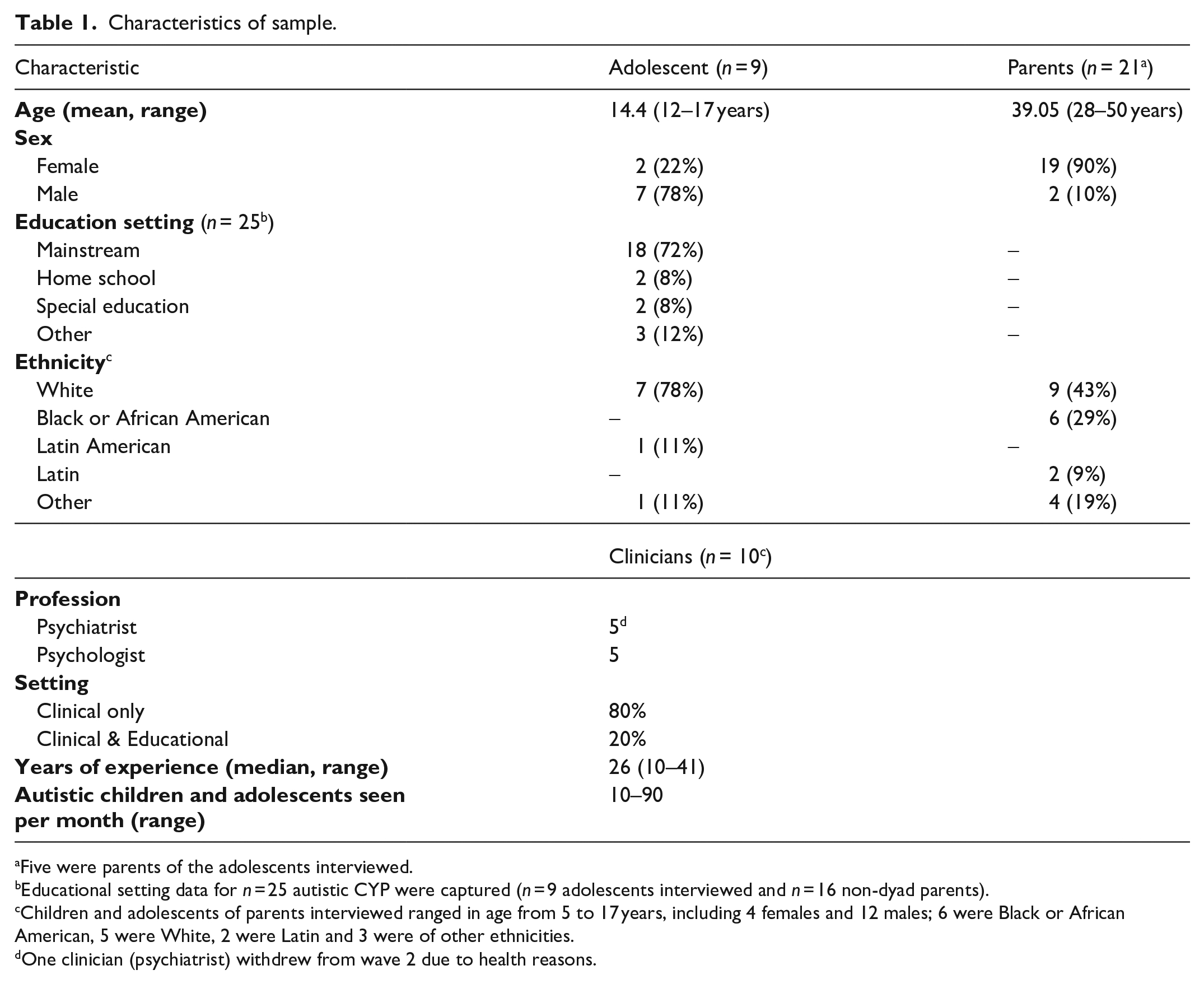
Five were parents of the adolescents interviewed.
Educational setting data for n = 25 autistic CYP were captured (n = 9 adolescents interviewed and n = 16 non-dyad parents).
Children and adolescents of parents interviewed ranged in age from 5 to 17 years, including 4 females and 12 males; 6 were Black or African American, 5 were White, 2 were Latin and 3 were of other ethnicities.
One clinician (psychiatrist) withdrew from wave 2 due to health reasons.
Table 2 provides a breakdown on the clinical characteristics of the study sample.
Breakdown of clinical characteristics of the study sample.
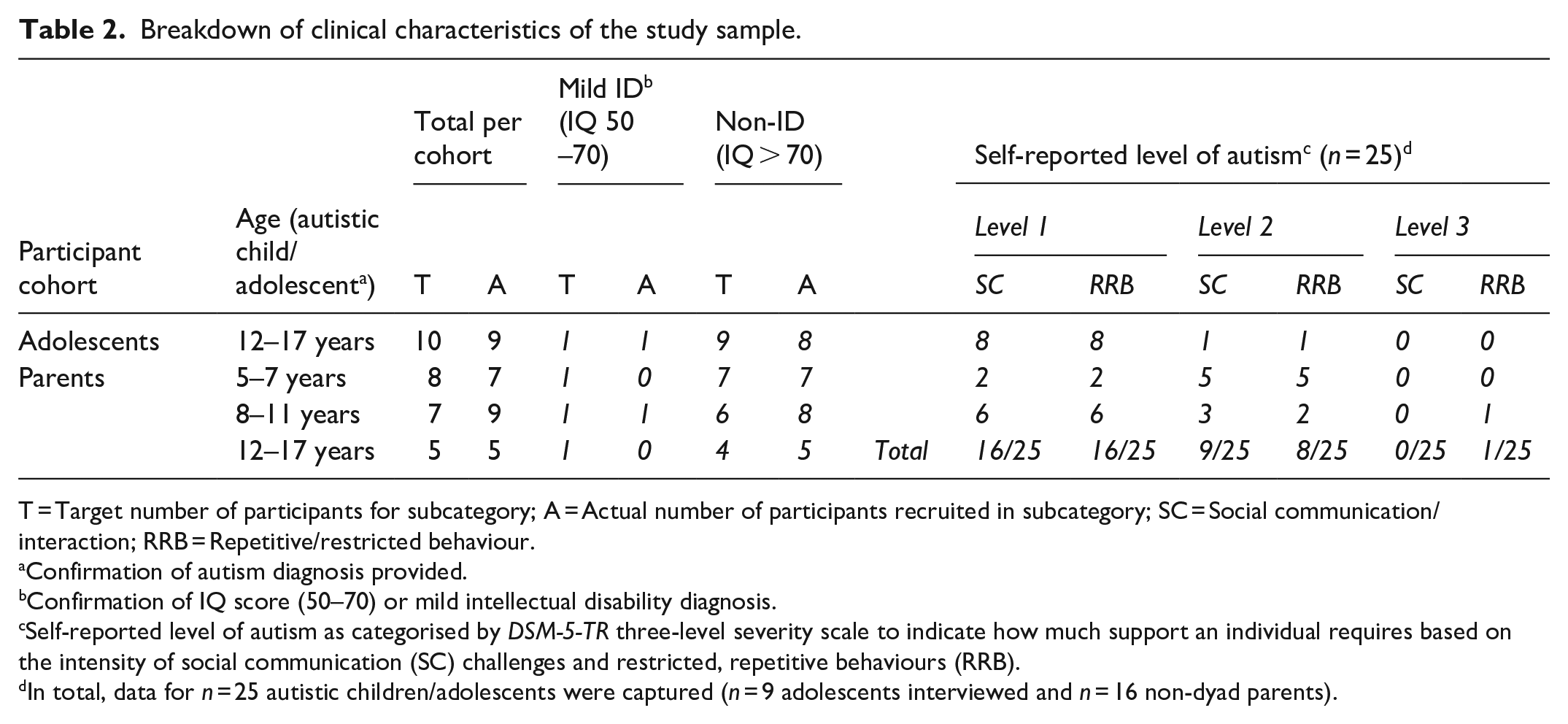
T = Target number of participants for subcategory; A = Actual number of participants recruited in subcategory; SC = Social communication/interaction; RRB = Repetitive/restricted behaviour.
Confirmation of autism diagnosis provided.
Confirmation of IQ score (50–70) or mild intellectual disability diagnosis.
Self-reported level of autism as categorised by DSM-5-TR three-level severity scale to indicate how much support an individual requires based on the intensity of social communication (SC) challenges and restricted, repetitive behaviours (RRB).
In total, data for n = 25 autistic children/adolescents were captured (n = 9 adolescents interviewed and n = 16 non-dyad parents).
We present the findings below of our systematic approach to developing a GAS measure to be used in identifying personalised endpoints for autistic children and adolescents (5–17 years). This involved two waves of interviews aimed at developing; a bank of example goals, a GAS scale and a manual for raters (see Figure 1).
Wave 1 – concept elicitation
Our Thematic Template Analysis expanded on the existing conceptual model (McDougall, 2017), resulting in n = 81 unique concepts spread across the existing subdomains (n = 9) and domains (n = 3). All potential refinements to the model were discussed in detail with the expert panel and resulted in the final conceptual model as shown in Figure 2. Concepts that were raised by ≥ 1 participant are included here for completeness, highlighting the diverse range of experiences described by autistic adolescents, parents and clinicians.
Bank of example goals
Interviews with adolescents uncovered a wide variety of specific goals from improving school grades, to developing culinary skills, and goals based on personal hobbies such as building frog ponds. However, the majority of goals mentioned centred on building independence and learning to do things for oneself:
I don’t have to be doing stuff every single day, but like, increasing the amount of times I’m cooking maybe, make breakfast for myself more and stuff like that. Adolescent (ADOL-CAN-02)
Goals discussed by parents were less varied, focusing on their child learning to become more independent overall, as well as improving their behaviour:
It is very important to reduce his aggression because it might become a habit and he might do that with others. Parent (CG-US-118)
Clinicians frequently highlighted practical goals to build skills towards being more independent:
. . . just environmental awareness that could cause a safety issue, like wandering away from home or walking across the street without paying attention to vehicles, those types of things I would say are pretty urgent . . . Clinician (US-HCP-200)
Explicit and implicit goals were organised across the domains and subdomains of the conceptual model: Core characteristics, Associated characteristics and Impacts. The goals were then categorised according to the three age ranges specified at the outset of the study (5–7 years, 8–11 years, 12–17 years). The set of example goals were discussed further with the expert panel and refined to form a bank of potentially actionable goals to be cognitively debriefed with participants during wave 2 (see Figure 4).
Development of the draft GAS measure for clinical interventional studies in paediatric autism
The research team collaborated with the expert panel to develop a draft GAS measure based on the findings from this study. The measure is structured in three phases; Phase 1: Identifying three personalised goals, Phase 2: Constructing the scale, Phase 3: Assessing and reviewing goal attainment. With interventional studies in mind, a manual was created to guide future GAS raters (who would be required to have extensive clinical experience with autism and the administration of outcome measures in clinical interventional studies) to effectively conduct the goal setting and follow-up assessment with autistic children and adolescents and their parents over the pre-assigned study/treatment period, by
Outlining a step-by-step process for goal setting,
Supporting the GAS rater in facilitating construction of appropriate GAS levels with autistic children and adolescents and their parent (or supporter),
Supporting the GAS rater in facilitating assessment and review of GAS with autistic children and adolescents and their parent (or supporter).
The measure proposes that both the children and adolescents and their parent/supporter are involved in the GAS process. However, the role of ‘primary’ goal setter depends on the child/adolescents age, communication and cognitive ability. Furthermore, the measure recommends GAS raters be flexible in their approach, allowing the child/adolescent as much self-determination as possible when goal setting. However, for younger children the role of ‘primary’ goal setter will always be the parent (or supporter).
The section below describes the three aforementioned phases of the draft measure presented to participants, and outlines the results of the cognitive debriefing in wave 2 interviews.
Wave 2 – cognitive debriefing
Phase 1: identifying three personalised goals
The three domains encompassing the most important concepts of paediatric autism (Core characteristics, Associated characteristics and Impacts) were presented (Figure 3) as an introduction to the GAS measure.
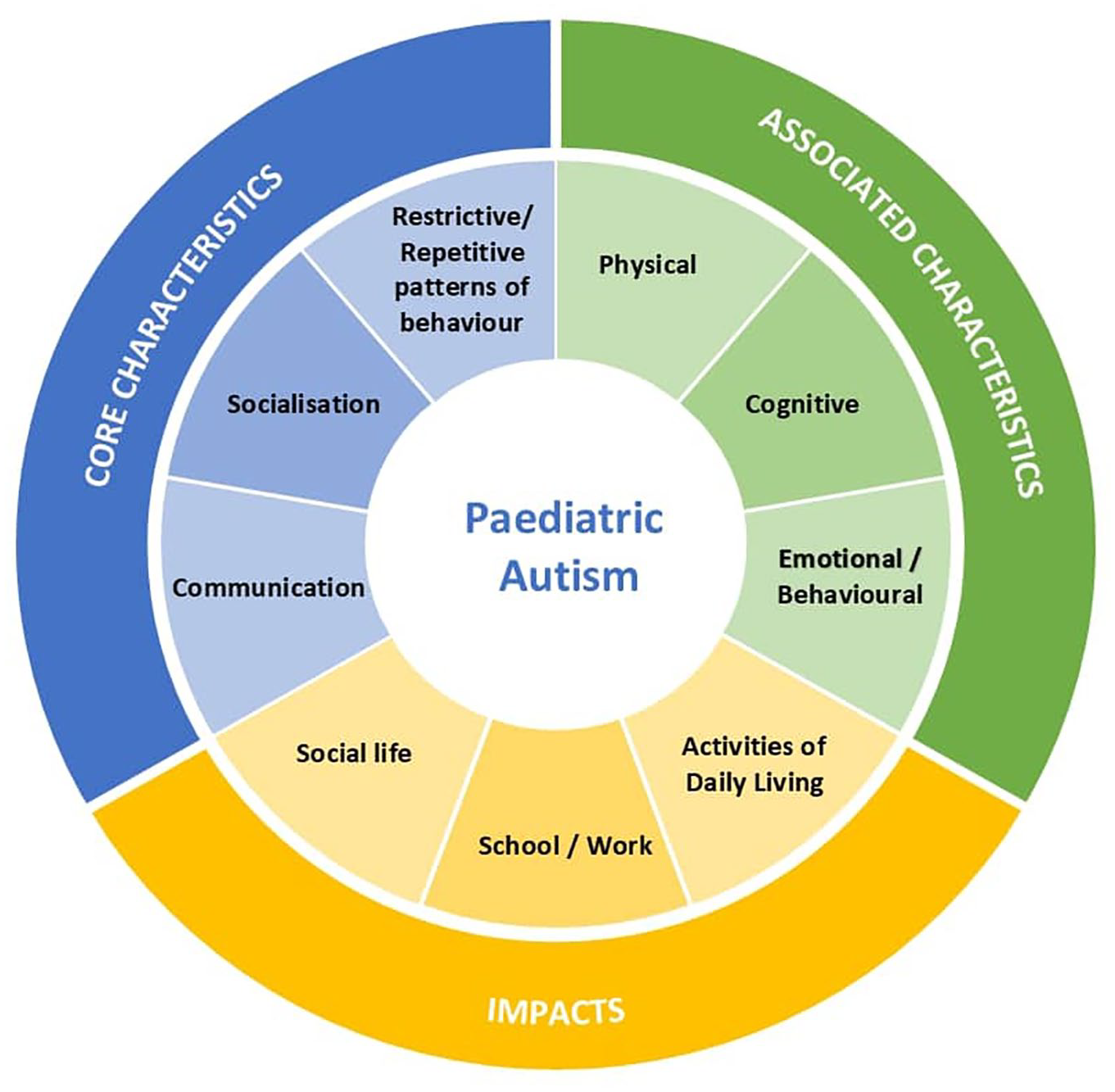
Domains of paediatric autism.
The figure was intended to be a visual aid to stimulate thoughts and discussion on the different areas study participants may choose to set specific goals. Adolescents and parents were asked which areas they considered most important to their experience of autism and if they could create a goal in relation to that area.
All participants spoke positively of the figure, with both parents and clinicians commenting on the clarity of the message being communicated:
Honestly, I was trying to take a screenshot of this diagram . . . It’s extremely relevant, that’s why I want it. It reminds me that these are the areas that autism effects on a daily life, and it really does affect us in every single one of those departments. Parent (CG-CAN-06)
Clinicians confirmed the domains and subdomains aligned with what they typically observe in autistic individuals, noting there were no omissions:
I think this diagram presents nicely as essentially metaphorically a street sign as to where we’re going. And so, you can easily organise your thinking around the three domain areas, and then the titles listed underneath them. So, it seems very clear. Yes, it is something that would be a directive and appropriate. Clinician (US-HCP-203)
Next, participants reflected on the bank of example goals identified during the first wave. Some additional goals were suggested, including communicating with unfamiliar people, spatial awareness and understanding boundaries (personal space), safety/awareness of surroundings, dating (romantic), and feeding. These goals were reviewed with the expert panel, and further refinements to the bank of example goals were made based on the feedback (Figure 4).
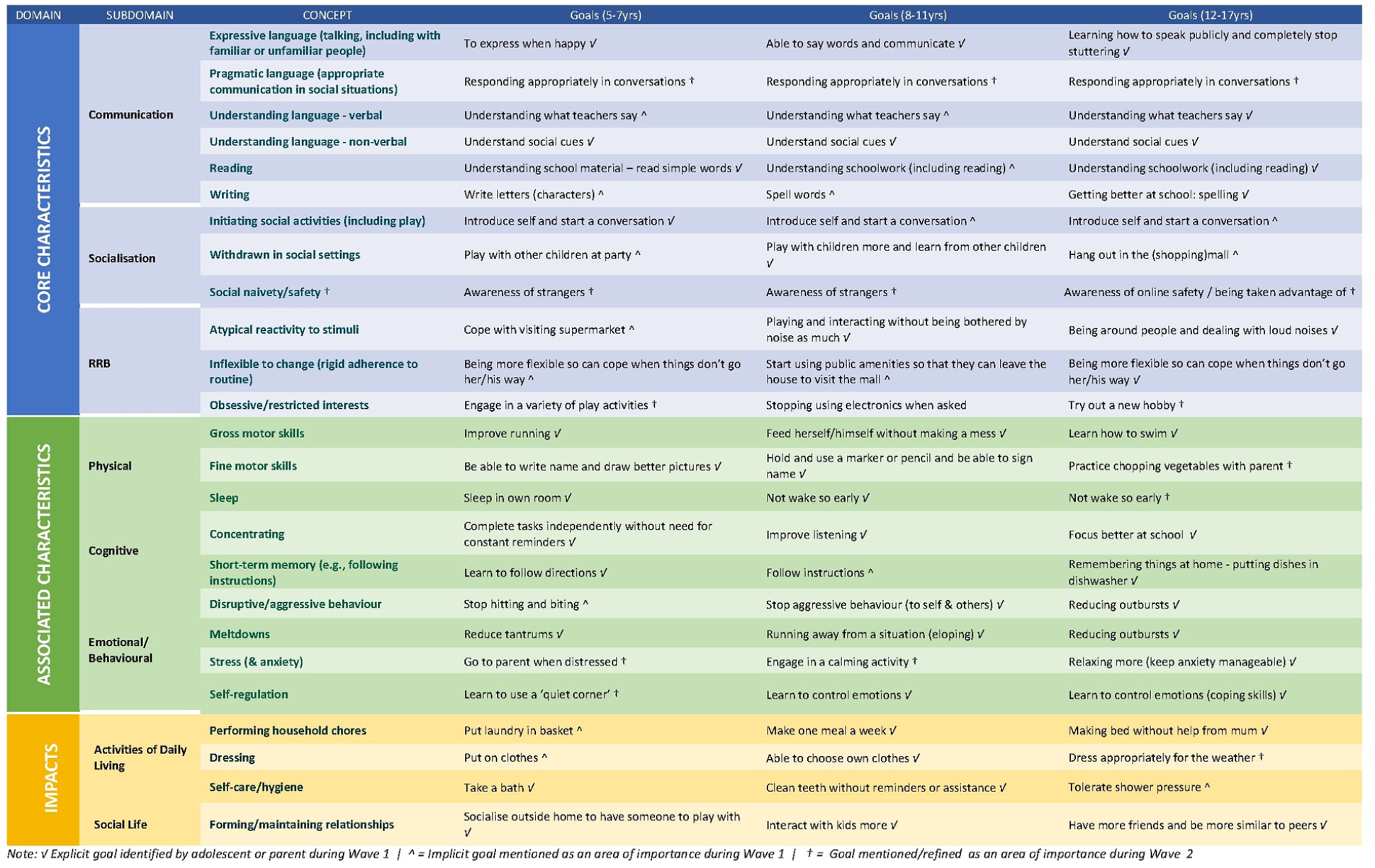
Bank of example goals.
Phase 2: constructing the scale
The first step in constructing the scale involves taking a broad goal and converting it to a SMART goal. For illustrative purposes, an example SMART goal vignette was created and used consistently throughout each step of the scale construction, providing a clear reference point for the GAS rater during the entire explanation of the process. GAS level construction was based on a 5-point scale, ranging from −1 = Worsening, 0 = Baseline, +1 = Mild improvement, +2 = Moderate improvement, +3 = Substantial improvement. In an attempt to arrive at equidistance between each level of the scale, the draft measure proposed that GAS raters construct levels in the following order, 0, +2, +1, +3, −1.
During cognitive debriefing, most clinicians felt that the draft vignette lacked clarity and recommended revisions such as ‘adding the number of reminders required to each level’, to improve it. Clinicians also indicated that while the 5-point scale was sufficient to capture a range of goal attainment levels, readjustment of the levels to include an additional level of worsening would allow the scale to function more appropriately as an endpoint. Some clinicians felt that in a clinical interventional study, the introduction of an intervention could potentially lead to ‘substantial’ worsening over the course of a 12-week period and therefore the scale should be flexible enough to accommodate this. Following consultation with the expert panel, the 5-point GAS level construction was adjusted to −2 = Substantial worsening, −1 = Worsening, 0 = Baseline, + 1 = Improvement, + 2 = Substantial improvement, to reflect the feedback. Figure 5 shows the final example SMART goal and GAS levels included in the measure.
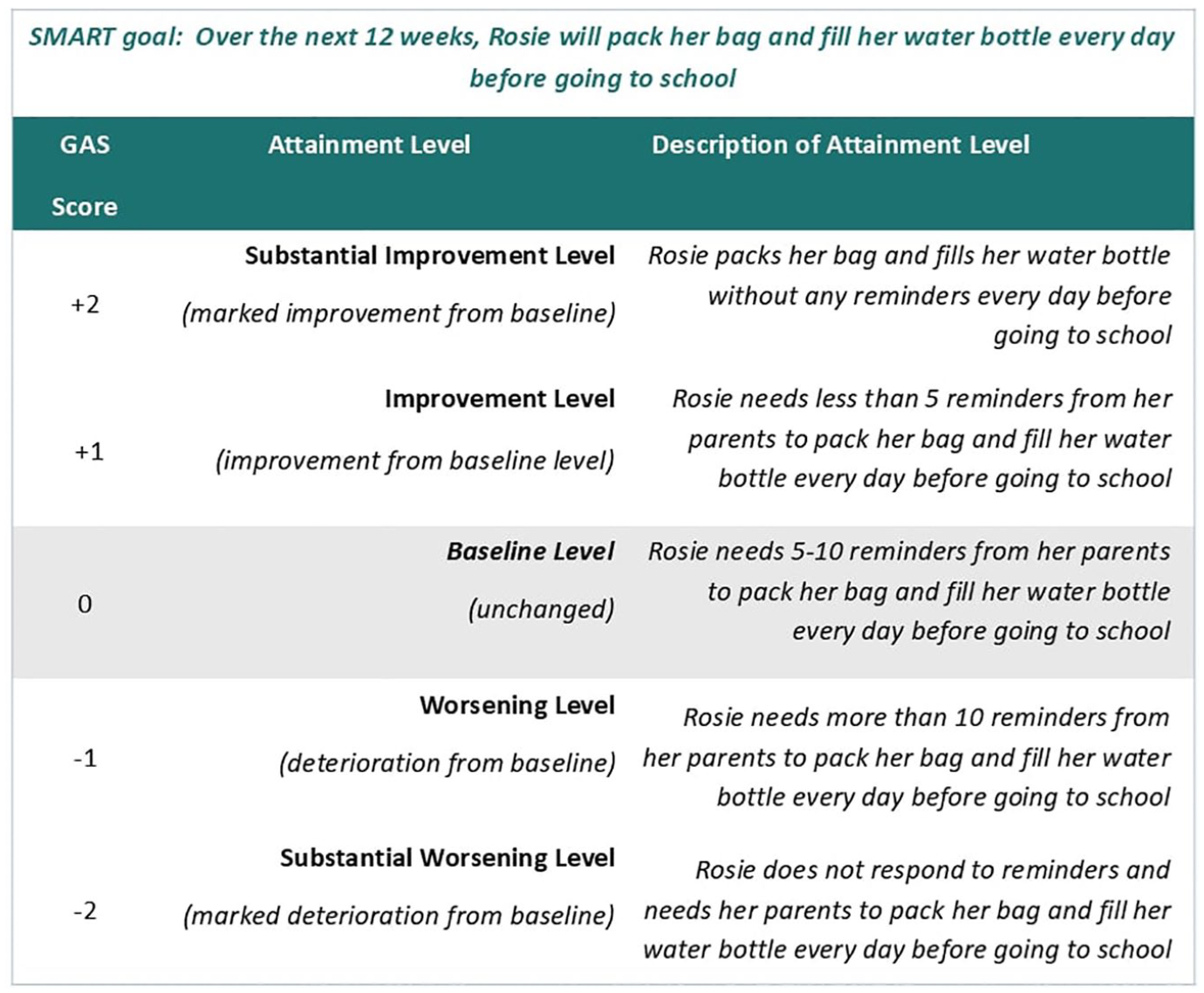
Example vignette and GAS levels for a clinical interventional study.
Weighting goals
A key part of adopting GAS as a personalised endpoint is understanding the relative importance and difficulty of the personalised goals identified. In following the formal process outlined below, adapted from Turner-Stokes (Turner-Stokes, 2009), GAS raters can work with autistic children/adolescents and their parents/supporters to attribute weight to each goal according to its importance and difficulty for the individual (Figure 6). While direct comparison of personalised goals may not be possible, comparing attainment levels of goals that are similarly weighted can provide an indication of the efficacy of an intervention on a group level.
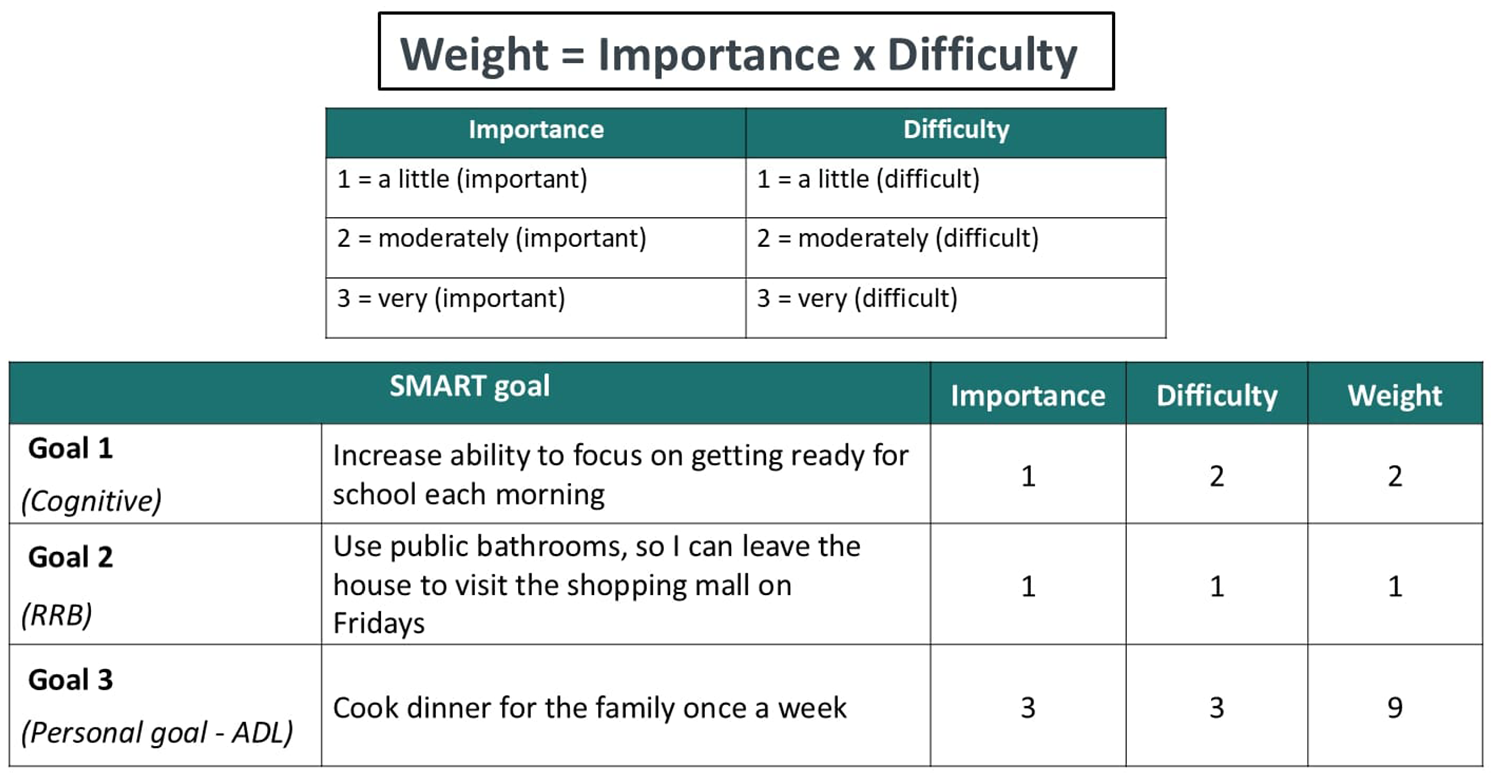
Weighting goals in GAS for a clinical interventional study.
Using the image above, the process of weighting goals was cognitively debriefed with clinicians. The process was found to be easy to follow. While no revisions were suggested, one clinician remarked that it was their first time seeing this method and felt it was something that should be adopted in clinical practice.
Phase 3: assessing and reviewing goal attainment
The final phase of the GAS process is completed during the follow up interview at the end of the study/treatment period. The measure outlines three steps involved in assessing and reviewing goal attainment: (1) assessing current status, (2) confirming status with the child/adolescent or their parent/supporter, (3) documenting the GAS rating. We propose that the voice of the autistic child/adolescent is represented throughout goal attainment assessment and review and created the diagram below (Figure 7), which clearly indicates the roles of active participants in the process. The diagram was presented to clinicians along with a detailed explanation of activities taking place within each task box (detailed guidance is provided separately in the manual).
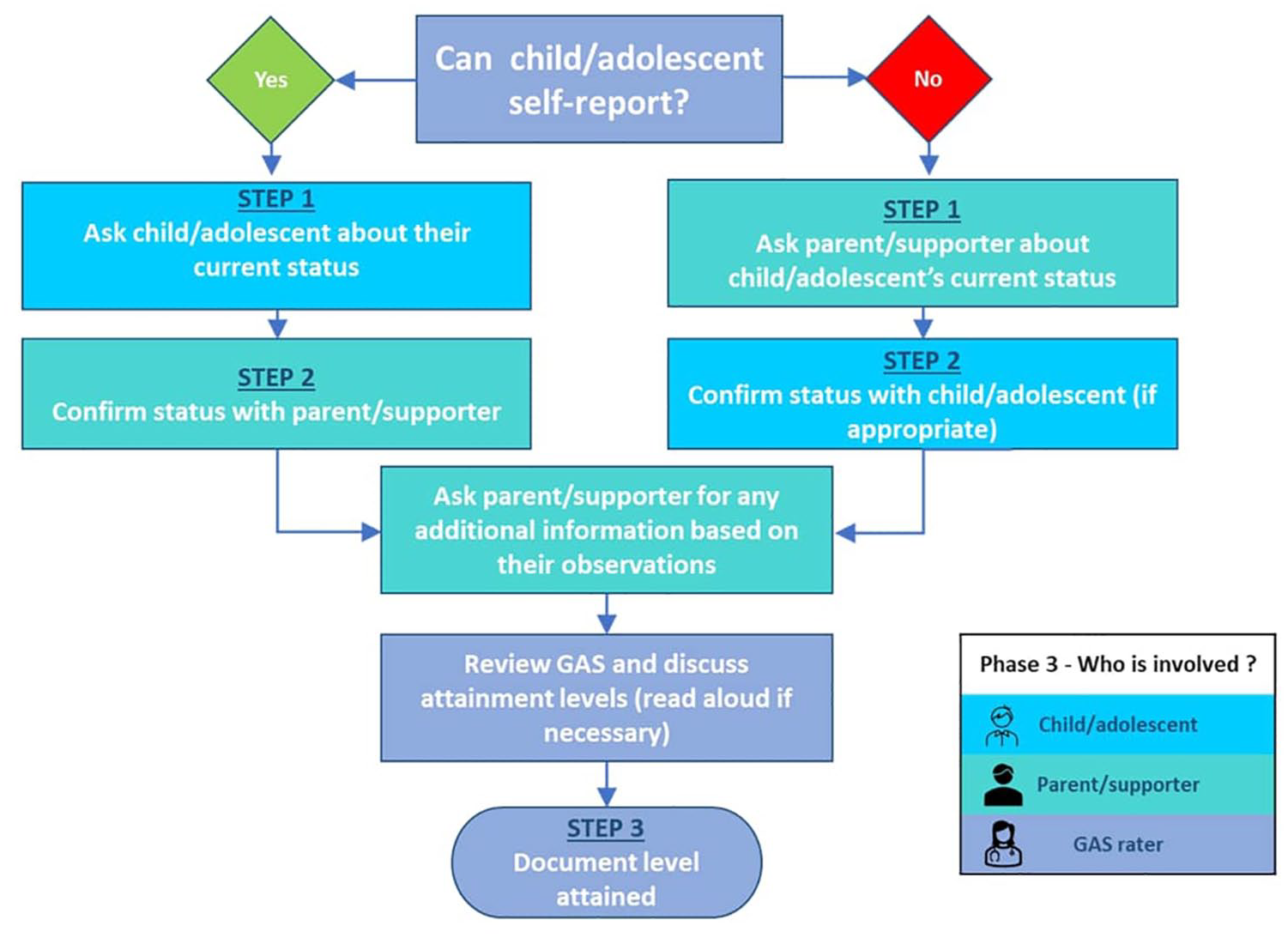
Measuring and reviewing GAS for a clinical interventional study.
Clinicians described the ‘navigation’ through the process as flowing well and ‘making a lot of sense’. One clinician, however, raised the point that the process should allow for jurisdictional variance:
different states [across the United States] have different degrees to which legalities exist for what a child can attest to and not. Clinician (US-HCP-200)
Clinicians also commented on the need for flexibility depending on the individual:
Kids have different levels of insight and different levels of wanting to work with a therapist. So, if they’re new to the process, they don’t trust you or if they’re very, very invested in a certain activity and don’t want to give it up, then you have a problem. Clinician (US-HCP-08)
Some clinicians discussed ‘Confirming status’ as a potentially contentious area:
. . . the kid says, ‘I want to be able to go to the bathroom by myself’, . . . as long as the parent has the good sense not to say something that’s going to upset the child, I think that’s just a point of . . . you could say, this is a goal the parent wants, and the kid might like freak out about it. Clinician (US-HCP-07)
However, clinicians also commented that conflicts such as this are regularly encountered and managed in practice, suggesting the GAS rater should use their experiential judgement resolving discordance between what the autistic child/adolescent and parent/supporter report.
Based on this feedback and further discussion with the expert panel, the measure was revised to include instructions on allowing flexibility, as the ‘primary’ goal setter (and thereby final decision maker) may vary depending on the child/adolescent’s ability to self-report to the level needed to communicate goals. In addition, a section on ‘Other considerations’ was also added to the manual, which includes taking into account the nature of the therapeutic relationship when determining the goal attainment.
Discussion
The GAS manual and bank of example goals described in this paper are underpinned by empirical evidence largely supportive of a conceptual model developed by McDougall (2017). This study has demonstrated the relevance of a GAS approach to adolescents and caregivers and has been developed in line with COA best practice (e.g. FDA PFDD guidance) by starting with qualitative research in autistic children and adolescents aged 5–17 years who were the intended target population for this measure. This measure is intended for researchers interested in using GAS to assess participant treatment priorities across different developmental cohorts (autistic children and adolescents aged 5–17 years), and has the potential for broader use in other age ranges and conditions (Shogren et al., 2021). The framework outlined can be applied to GAS more generally in terms of structure, with the goals/examples updated to reflect context of use specific to autism. Although GAS originated within the context of clinical practice and was subsequently adopted as a research method, the structured approach outlined in this paper also provides the clinical community in the context of interventional studies such as clinical trials with guidance for GAS. This study reports detailed information on the practical implementation of a GAS measure including guidance for GAS raters (the manual) and managing individual self-reporting imprecision (assessing and reviewing process), meeting previous calls to action to ensure rigour and reproducibility (Gaasterland et al., 2019; Krasny-Pacini et al., 2013, 2016; Logan et al., 2022; Steenbeek et al., 2008).
Earlier studies have demonstrated the valuable contribution of Patient and Public Involvement and Engagement (PPIE) in the field of neurodevelopmental and mental health conditions including autism in adolescents, and the paradigmatic ways by which children and adolescents experience their condition both subjectively and within the social context (Fusar-Poli et al., 2024).
Our study prioritised the voice of adolescent participants in several respects. First, we ensured adolescents were active participants and created dedicated interview guides and study materials (with the assistance of the expert panel) that catered to autistic adolescents specifically. We developed the bank of example goals based on goals explicitly mentioned by adolescents; these tended to be quite specific (e.g. ‘building a frog pond’), however, and not necessarily well suited to the outcome of a clinical trial but may be more appropriate in clinical practice settings. We therefore prioritised goals implicitly mentioned by adolescents such as ‘hanging out in the mall’. Goals such as this were discussed as something the adolescent would like to do/improve on, but were not necessarily expressed as a specific goal during the interview. We also introduced goals identified by parents to ensure our coverage of goals was relevant to each of the three age cohorts of the study. Some of the goals are worded from a parents’ perspective (i.e. ‘his or her way’) in acknowledgement of the fact that parents/carers would by necessity have to be involved in identifying goals for young children or more severe or non-verbal adolescents. These steps make our findings truly reflective of the lived experience of autism, thereby enhancing the usability and translatability of the measure developed (Pellicano et al., 2014). The collective contribution of the expert panel of autistic individuals, advocates, parents, carers and research clinicians from both academia and industry brought richer insights and perspectives to each step of the study.
In GAS research, the numerous potential goals in a single study can seem overwhelming, leading researchers to consider having all participants focus on the same goals. However, identical goals should not be the standard for ensuring reliability in GAS across a cohort (Schlosser, 2004). Instead, a bank of evidence-based example goals, such as outlined in this study, provides a helpful starting point from which individualised goals can be generated on the same general problem area (or concept). This echoes the suggestion proposed by Schlosser (2004) in their critical review of GAS in communication disorders and is an approach being trialled elsewhere (Sevinc et al., 2024). We propose GAS raters use the bank of example goals to assist study participants identify three ‘broad’ goals (either selected directly from the examples or created from scratch) to be explored further and developed into SMART goals. While selecting three study goals is common across the literature, unlike this study, other authors in the field of paediatric autism have arrived at goal selection through consultation among adults such as teachers and parents, and excluded the child/adolescent from the process (Ruble et al., 2012, 2018, 2022).
Participant heterogeneity over time, such as childhood developmental changes or natural fluctuations in ability, can likewise pose significant challenges for researchers working to identify personalised endpoints. Managing intra-person variability in core and associated characteristics of autism over time can be difficult. Goals around the most important or difficult domains of autism for a given participant may also change over time, which is a further factor that complicates efforts to measure and evaluate the domains that matter most to participants. These challenges, however, are not insurmountable. Our bank of example goals is categorised according to three specified age ranges, which allows for flexibility in the consideration of goals appropriate to the child/adolescent’s developmental stage.
Other aspects of GAS outlined in this paper share commonalities with existing literature, including, for example, the adoption of a traditional 5-point scale from −2 to + 2 (Logan et al., 2022). Although there is no ‘gold standard’ for baseline level setting, a recent systematic review of 21 RCTs using GAS as an endpoint, found less than half reported any baseline, and of those that did, –1 was most frequently reported, followed by 0 (Cheema et al., 2024). Guided by Logan et al. (2022) review on GAS as an outcome measure in RCTs, and in light of autism not being a regressive condition, we initially opted to set the baseline level at –1, as typically seen in GAS (Kiresuk & Sherman, 1968). However, the feedback from clinicians during wave 2 lead us to readjust the baseline level to 0. In their recent study with autistic youth and adults, Lee et al., also opted to set the baseline at 0, however unlike this study, they adopted a 6-point scale (+1 and +2 dedicated to progress between and the annual goal +3), arguing that a 5-point scale may not have captured the granularity of incremental progress they expected over the 12-month period (Lee et al., 2022). Regardless of where the baseline is set, it is important for scale rigour to ensure goals and attainment levels are established a priori (Turner-Stokes, 2009).
For all its inherent strengths with respect to customisation, researchers utilising GAS nevertheless face challenges when confronted with the overarching need for standardisation in clinical trials. Standardisation in constructing the scale to ensure GAS levels or ratings are truly equidistant can be difficult (Ruble et al., 2018). The comparability of GAS scores across groups and interrater agreement presents additional challenges in the context of a clinical trial that relies on data collected from different sources. However, in this study we have taken pre-emptive steps to mitigate this by standardising the rating process through the development of a robust manual. In accordance with guidance offered by Ruble et al. (2018) we recommend that an independent GAS rater should code each goal for measurability, equidistance, and difficulty. With these considerations in mind, concerns of comparability of GAS descriptions across individuals and groups and the reliability of GAS scores can be overcome using independent GAS raters and blinded observers (determining interrater agreement) (Ruble et al., 2018), as well as case-by-case calculation (Schlosser, 2004). Our proposed formula for weighting goals was adapted from Turner-Stokes (2009). While the weighting process was well received by clinicians, the use of these data during the scoring process is an area for future exploration as there are known benefits and pitfalls of a weighted continuous analysis which warrants further investigation via a dedicated study. Options for future scoring could include raw scores or mean of raw scores, calculating the difference between the initial and attained level, unweighted ordinal analysis and t-scores (Krasny-Pacini et al., 2013).
Despite the strengths of this study, some limitations should be acknowledged in terms of the prospects of using GAS as an endpoint in paediatric studies and the research itself. The key concerns for GAS as a primary endpoint to support regulatory approval and labelling revolve around operationalisation, feasibility, and determining meaningful change (Duke-Margolis, 2017). As with all COAs, validation involves ensuring an instrument is fit-for-purpose, defined as having sufficient validation to support its context of use (FDA, 2022). Decisions about a given COA’s fitness are based on two considerations: the clear description of the Concept of Interest and Context of Use, and sufficient evidence to support the proposed interpretation and use of the COA (FDA, 2022). Gaasterland et al. (2019) propose a conceptual validation framework borrowing from the COSMIN taxonomy (Mokkink et al., 2010) to evaluate GAS measurement properties and suggest validation at both the trial (group) and individual levels. These steps are likely necessary to achieve ‘fit for purpose’ status for FDA approval as a trial primary endpoint. Still, regulatory guidance specific to GAS is lacking, and existing guidelines were largely written from the perspective of patient needs in rehabilitation medicine (Bard-Pondarré et al., 2023; Krasny-Pacini et al., 2016; Turner-Stokes, 2009).
Pragmatic considerations for GAS measures in paediatric clinical interventional studies include the presence of an existing therapeutic relationship, which could potentially compromise the GAS rater’s ability to objectively construct GAS levels and provide an unbiased assessment of attainment. The importance of timeframe considerations in paediatric GAS studies was raised by all participants, including factors such as (1) setting realistic timelines to ensure goals are achievable based on the child/adolescent’s capabilities and the goal complexity; (2) scheduling regular check-in intervals (e.g. weekly and monthly) to assess incremental achievements; and (3) identifying potential barriers to ensure goals remain relevant and attainable. The time of year GAS is set also needs to be considered as changes in schedule due to school holidays and religious holidays/festivals may impact the child/adolescent’s ability to reach a goal. Parents and clinicians also pointed to environmental constraints that could lead to goals becoming more or less achievable depending on the availability of resources, support systems (e.g. parent/supporters), or environmental changes. Personal circumstances such as the child/adolescent’s personal life events, health status, and commitments might likewise impact timelines and achievability of goals. Trial duration may present challenges as well: it may be unreasonable to expect participants to be able to achieve certain personalised goals in trials with shorter duration. GAS measures require raters to be specifically trained, which may translate into a need for greater resources, and ultimately lower practicability.
From a professional perspective, while our study is confined to the perspectives of psychologists and psychiatrists, we acknowledge that outside the context of a clinical interventional setting such as a clinical trial (the context for which this measure was provisionally developed), other disciplines and paraprofessionals provide a larger proportion of services to autistic people. Future researchers considering similar work for a behavioural intervention or for clinical practice should include a wider range of disciplinary professionals (occupational therapists, speech and language therapists, BCBAs, social workers, etc.) as this broader perspective would enhance the development of these scales and ensure suitability for this different context of use.
Finally, our sample was confined to the experience of autism for individuals in the 5 to 17 years age range. The age for self-report for children in the development of paediatric patient-reported outcomes is debated (Arbuckle & Abetz-Webb, 2013; Gale & Carlton, 2023), and in cases of varying ID/IQ it is potentially more complicated. We therefore chose to capture self-reported experiences from adolescents. However, we also acknowledge that autistic children (12 years and below) could potentially have participated in interviews with the support of a parent or caregiver. Future studies should consider the inclusion of members of this younger cohort to better understand their experiences and to improve the applicability of this GAS measure as an endpoint for younger children.
Despite efforts to include children and adolescents with mild ID, due to lack of IQ testing we were met with barriers around clinical confirmation of IQ scores and confirmed diagnosis of mild ID. While not intended to conflate severity of autism symptoms with intellectual disability, as a proxy measure, self-reported Diagnostic and Statistical Manual of Mental Disorders (5th ed., text rev.; DSM-5-TR) levels of autism were collected from parents to provide richer context from which to view our findings. Future research might benefit from a more targeted approach to identifying participants who meet the definition of mild ID (defined as IQ between 50 and 70) and assessing the functioning of GAS in that population. Furthermore, we suggest psychometric validation (e.g. interrater reliability and known groups) as an important avenue for future research to further help establish GAS as a robust clinical outcome measure.
Conclusion
This study has reported the efforts undertaken to develop a GAS measure consisting of a bank of goals and instructions to be used in identifying personalised endpoints for autistic children and adolescents (5–17 years) taking part in a future clinical interventional study such as a clinical trial. While the provisional feasibility of this approach has been demonstrated via this qualitative research, the feasibility of setting and evaluating goals with autistic children/adolescents/parents in the context of an interventional study will need to be further assessed. The measure described in this study provides useful methodological support for researchers considering the use of GAS in paediatric intervention studies in clinical trials in autism and may have utility in real world/clinical practice settings; this, however, is an area for future research. Collaboration with stakeholders including an expert panel consisting of autistic individuals, advocates, parents, carers and research clinicians enhanced the translatability and relevancy of the GAS measure for use in the populations for which it was intended. We suggest future research focusing on the validation of GAS as a primary or secondary endpoint supporting regulatory approval and labelling will help lay the groundwork for success in future paediatric autism studies. Future research would also benefit from the inclusion of a broader cross-section of the diverse range of clinical and other professionals who provide care and services to autistic people.
Supplemental Material
sj-docx-1-aut-10.1177_13623613251349904 – Supplemental material for Development of a Goal Attainment Scale (GAS) outcome measure for clinical interventional studies in paediatric autism
Supplemental material, sj-docx-1-aut-10.1177_13623613251349904 for Development of a Goal Attainment Scale (GAS) outcome measure for clinical interventional studies in paediatric autism by Hannah Staunton, Tammy McIver, Julian Tillmann, Susanne Clinch, Vivienne Hanrahan, Bethany Ewens, Caroline Averius, Alexandra I Barsdorf, Aurelie Baranger, Elizabeth Berry Kravis, Tony Charman, Haraldt Neerland, Alison T Singer, Pamela Ventola, Zackary JK Williams and Louise Barrett in Autism
Footnotes
Acknowledgements
All participants, families and clinicians who participated in the study. Emma Elliot, Jess Mills, Rebecca Rogers, Charlotte Igoe, Jasmin Tinsley, Saba Keshan and Abbie Dunne (Modus Outcomes). Lorraine Murtagh, Thomas Wiese, Philipp Schoenenberger, Marta del Valle Rubido, Stephanie Klupp and Autism CareRing community (Roche). Kari Knox (Ardea Outcomes). Elizabeth Gibbons, Ashley Geiger, Katherine Kirk (formerly COS employees).
Corrections (August 2025):
Article updated online to add the sentence (“Concepts that were…… clinicians”) at the end of first paragraph of Wave 1 – concept elicitation and correct figure 2.
The article has also been updated with grammatical or style corrections since its original publication.
Authors’ Note
Medical writing support for this manuscript was provided by Trais Pearson, PhD, of Modus Outcomes and was funded by Roche Products Ltd.
ORCID iDs
Ethical Considerations
Ethical approval was obtained from the Advarra Institutional Review Board (IRB; Columbia, MD, USA) on 11 October 2022 (Reference: Pro00065915).
Author Contributions
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Roche Products Ltd. funded this study and worked with Modus Outcomes on the design and development of this manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HS, TM and SC are employees and shareholders of Roche Products Limited. CA is an employee and shareholder of F Hoffmann-La Roche Ltd. JT is an employee of F Hoffmann-la Roche. Consulting fees were provided by Roche Products Ltd to Modus Outcomes, Clinical Outcomes Solutions, Rush University and Cogstate. TC has served as a paid consultant to Roche Products Ltd. for this project and receives royalties from Sage Publications and Guilford Publications. ZW has served as a paid consultant to Roche Products Ltd. for this project.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
