Abstract
Alterations in the development of attention control and learning have been associated with autism and can be measured using the ‘antisaccade task’, which assesses a child’s ability to make an oculomotor response away from a distracting stimulus, and learn to instead anticipate a later reward. We aimed to assess these cognitive processes using portable eye-tracking in an understudied population of pre-school children with and without a diagnosis of autism spectrum disorder in community settings in New Delhi, India. The eye-tracking antisaccade task was presented to children in three groups (n = 104) (children with a clinical diagnosis of autism spectrum disorder or intellectual disability and children meeting developmental milestones). In accordance with findings from high-income, laboratory-based environments, children learnt to anticipate looks towards a reward, as well as inhibit eye-movements towards a distractor stimulus. We also provide novel evidence that while differences in inhibition responses might be applicable to multiple developmental conditions, a reduced learning to anticipate looks towards a target in this age group may be specific to autism. This eye-tracking task may, therefore, have the potential to identify and assess autism specific traits across development, and be used in longitudinal research studies such as investigating response to intervention in low-resource settings.
Lay abstract
The development of cognitive processes, such as attention control and learning, has been suggested to be altered in children with a diagnosis of autism spectrum disorder. However, nearly all of our understanding of the development of these cognitive processes comes from studies with school-aged or older children in high-income countries, and from research conducted in a controlled laboratory environment, thereby restricting the potential generalisability of results and away from the majority of the world’s population. We need to expand our research to investigate abilities beyond these limited settings. We address shortcomings in the literature by (1) studying attention control and learning in an understudied population of children in a low- and middle-income country setting in India, (2) focusing research on a critical younger age group of children and (3) using portable eye-tracking technology that can be taken into communities and healthcare settings to increase the accessibility of research in hard-to-reach populations. Our results provide novel evidence on differences in attention control and learning responses in groups of children with and without a diagnosis of autism spectrum disorder. We show that learning responses in children that we assessed through a portable eye-tracking task, called the ‘antisaccade task’, may be specific to autism. This suggests that the methods we use may have the potential to identify and assess autism-specific traits across development, and be used in research in low-resource settings.
Keywords
Introduction
Attention control plays a crucial role during child development, by filtering information from the environment. It influences multiple levels from perception to learning and memory, and comprises one of the core foundational domains for executive function (Astle & Scerif, 2009; Hendry et al., 2016; Scerif, 2010). It is important for later social adjustment as well as for academic performance (Papageorgiou et al., 2014; Rueda et al., 2010). The development of these domains is generally understood to be susceptible to environmental influences, including socioeconomic factors (Amso & Scerif, 2015). Alterations in attention control have also been associated with a range of developmental conditions, including autism spectrum disorder (ASD; Bedford et al., 2012; Elsabbagh, Fernandes, et al., 2013; Hendry et al., 2018; Johnson, 2012; Wass et al., 2015).
Across infancy and early childhood, and as the visual system develops, attention control abilities develop – specifically the ability to flexibly shift and inhibit attention. This ability has been measured using visual attention tasks, for example, the antisaccade task. The antisaccade task measures the endogenous process of inhibiting automatic attention by measuring whether a participant can suppress a saccade to a distractor (known as a ‘prosaccade’) and also their learning to shift attention and make an anticipatory saccade in the opposite direction (an ‘antisaccade’). When traditionally used in adult populations, the antisaccade task uses verbal instructions (Guitton et al., 1985; Munoz & Everling, 2004); however, in its implementation for infants and children (Portugal et al., 2021), participants are trained to learn to look in the opposite direction to the distractor by presenting a delayed target stimulus and reinforcing this response with an animated reward stimulus. Therefore, over the course of the antisaccade task, children learn to inhibit orienting their gaze to a distracting irrelevant visual stimulus (prosaccade) (Nakagawa & Sukigara, 2007) and also to make faster and anticipatory looks in the opposite direction towards a ‘reward’ stimulus presented instead (antisaccade). The prosaccades are thought to reflect exogenous processing, whereas the voluntary antisaccades reflect endogenous processing (Amso & Scerif, 2015; Scerif et al., 2005). From as early as 4 months of age, infants are able to learn to selectively inhibit prosaccades (Johnson, 1995), but learning to produce antisaccades takes longer to develop, and there is ongoing improvement in the frequency of antisaccades through childhood (Scerif et al., 2005).
The inhibition of prosaccades and the generation of antisaccades have been measured in both neurotypical children and children with a diagnosis of a neurodevelopmental disorder. Scerif et al. (2005) found that toddlers with Fragile X Syndrome (FXS) showed reduced learning to inhibit looks towards the distractor, compared to the comparison group. This may reflect a reduction in the active process of inhibition in the FXS group, but may also suggest reduced learning that the distractor is irrelevant (and thereby demonstrate a reduced drive to look towards it). In this study, the FXS group made the same number of antisaccades as the comparison group, with neither group demonstrating a significant increase in number of antisaccades made across the task. Therefore, Scerif et al. (2005) proposed that, while FXS toddlers were able to anticipate looks to the target, they were unable to use this learning to modify behaviour and inhibit prosaccades in the same way as the comparison group. Yet, these findings are also compatible with these children being more efficient in their oculomotor control, since they can both orient to the distractor and, in anticipation, to the target stimulus.
Moreover, a similar reduced decrease in prosaccades has been reported in autistic participants (Mosconi et al., 2009). Mosconi et al. (2009) utilised the antisaccade task to examine inhibitory control in autistic adolescents, and found an association between a reduced ability to inhibit prosaccades and ASD-related repetitive behaviours. Authors hypothesised that reduced inhibitory control, mediated by alterations in frontostriatal systems that support the ability to voluntarily suppress context-inappropriate responses (Rubia et al., 2007), may contribute to repetitive behaviours observed in autism.
This reduced inhibitory control, and particularly prepotent inhibitory control – that is, the ability to suppress or inhibit a response – in autistic compared to non-autistic groups has been widely reported. It was found to be one of the most replicated findings in a meta-analysis of attention in autism (Geurts et al., 2014). Age significantly influences performance (Geurts et al., 2014), but the majority of studies have focused on late childhood from approximately 7 years and into early adolescence, with limited evidence from early childhood. Early childhood represents a highly dynamic stage of brain development, with brain plasticity and the ability to adapt to environmental circumstances being at its peak; it is also a window during which behaviours associated with neurodevelopmental disorders may become apparent (Lockwood Estrin & Bhavnani, 2020; Wetherby et al., 2004; Zwaigenbaum et al., 2013). Furthermore, there is accumulating evidence and hypotheses suggesting that later emerging autism traits may reflect adaptations to the social world and environment, rather than be a marker of initial atypicality (Johnson, 2017). There is, therefore, a need for further research in younger age groups to more fully understand trajectories of development.
The majority of our current knowledge of brain development results from samples in high-income countries (HICs), and from studies conducted in highly controlled laboratory-based environments, thereby restricting the potential generalisability of results and away from the majority of the world’s population. There is, therefore, a need to expand our research of neurodevelopment to investigate abilities in children beyond these limited settings, such as in low- and middle-income countries (LMICs). This may also be important because, from the limited evidence that currently exists, we know that there is a strong association between poverty and other socioeconomic factors, and the development of children’s executive functioning (Farah, 2017; Noble et al., 2005, 2007; Noble, Farah, & McCandliss, 2006; Noble, Wolmetz, et al., 2006). However, this work has mainly focused on broad-based cognitive performance, and lacks in specificity regarding the underlying cognitive systems, such as attention control (Noble et al., 2007). This need to include more diverse samples when investigating neurodevelopment further increases when investigating neurodevelopmental disorders because the majority of children reside in LMIC settings, with an estimated 2 million children with ASD between the age 2–9 years living in India alone (Arora et al., 2018). Moreover, the majority of our current understanding of autism comes from western countries, where sociocultural norms may differ compared to other contexts, thereby affecting the process of identifying, referring and providing services for individuals with ASD (de Leeuw et al., 2020; Durkin et al., 2015; Rice & Lee, 2017), and also potentially the manifestation of autistic traits (Freeth et al., 2013; Matson et al., 2011). Therefore, recently, there has been a call for greater diversity and a global perspective in autism research (de Leeuw et al., 2020; de Vries, 2016; Durkin et al., 2015). By diversifying our samples, we will allow for greater understanding of developmental trajectories in children, and of how this may be impacted by sociocultural and socioeconomic environments.
In this study, we aim to address these gaps in the literature and advance our understanding of autism by (1) investigating attention control and learning in a population of children in an under-researched LMIC setting, (2) focusing research on a critical young age group of pre-school children and (3) using portable eye-tracking technology that can be taken into communities and healthcare settings to increase the accessibility of research in hard-to-reach populations.
We examined attention control and learning as assessed by the antisaccade task in children aged 3–5 years with and without a diagnosis of ASD in New Delhi, India. By using eye-tracking technology to measure visual attention, we can objectively and accurately capture the gaze of a child as they engage in a screen-based task. Eye-tracking is a particularly useful tool for studies with young children and children with reduced verbal abilities, as tasks can be conducted without the need for verbal instructions (Karatekin, 2007; Richmond & Nelson, 2009). We include analyses unique to revealing aspects of performance on this task not before studied, by using generalised estimating equation (GEE) to assess learning. We hypothesise that we would observe learning across the task by a decrease in prosaccades (learning to inhibit prosaccades), and an increase in the number of antisaccades made (learning to anticipate looks to the target) as children learn the contingency between distractor and target stimuli and modify their behaviour accordingly. We also hypothesise that the decrease in prosaccades will be reduced in the ASD group compared to the comparison groups, while there may not be differences in the number of antisaccades.
Methods
Participants
Participants between the ages of 3 and 5 years were recruited from Delhi/NCR regions. Children were recruited into three groups: (1) children with no existing clinical diagnosis or developmental delay (comparison group); (2) children with a clinical diagnosis of ASD; and (3) children with a clinical diagnosis of intellectual disability (ID), but without a diagnosis of ASD. Socio-demographic information was taken for each participant to calculate their Kuppuswamy’s Socioeconomic Status (SES) score (details below). From a total of 115 participants recruited and consented to the study, 11 were excluded due to insufficient eye-tracker data being collected (n = 1 child refused to partake in the whole eye-tracking assessment (ID group); for n = 10 assessments children did not complete the task, and therefore insufficient data were collected (n = 7 ID; n = 3 ASD) – detailed criteria below). This left 104 participants for data analysis (n = 32 in the comparison group; n = 46 in the ASD group; n = 26 in the ID group).
Recruitment
Children in the clinical groups were recruited from the Child Neurology Outpatient Department and case registries of the All India Institute of Medical Sciences (AIIMS), New Delhi. Children in the comparison group were recruited from communities in the New Delhi region, through contacts established by existing child development studies. Our recruitment strategy was based on previously reported strategies for these three groups in this setting (Dubey et al., 2021).
Community involvement
For community testing, a community mobiliser at each site location assisted the research team in recruitment and in explaining the study’s objectives and methods to potential participants. This community mobiliser was someone who was a local resident in the community where the study was being conducted.
Inclusion/exclusion criteria
The inclusion criteria for recruitment were, for the comparison group, age-appropriate attainment of developmental milestones and no clinical diagnoses of neurodevelopmental conditions (e.g. ID, attention-deficit/hyperactivity disorder (ADHD)), mental health or physical health (see below) conditions. The ASD and ID group had a clinical diagnosis using Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-V) diagnostic criteria by an experienced local qualified psychologist or paediatrician working at AIIMS. For the ASD group, the clinical diagnosis of ASD was endorsed by the AIIMS-Modified-INCLEN Diagnostic Tool (INDT)-ASD (Arora et al., 2018; Gulati et al., 2019; Juneja et al., 2014) by trained researchers on the project. For participants where the AIIMS-Modified-INDT-ASD assessment conducted by the researcher on the project did not indicate high scores in the ASD group (⩾5, as indicated in the AIIMS-Modified-INDT-ASD diagnostic criteria), the research team discussed the case with the clinician who had given the original ASD diagnosis and who was working with the family, until consensus was reached with the research and clinical team regarding the participant’s group allocation. Exclusion criteria for all participants included hearing, visual or physical impairment that may interfere with their ability to provide responses to the eye-tracking protocol. All experimental procedures were reviewed and approved by Institutional Ethics Committees.
Parent/guardians questionnaires
Parents/guardians were asked to complete a set of questionnaires relating to child and family demographics. A researcher administered each question verbally to ensure there was no difficulty with literacy skills that would affect the respondent’s ability to complete the questionnaires. A Kuppuswamy score was calculated from the father’s education level, occupation and family income; this scale has been used in research for Indian urban populations to assess SES (Sharma, 2017). A binary variable of ‘lower’ or ‘higher SES’ was created for analysis, where ‘lower SES’ was defined by Kuppuswamy’s category of ‘Lower middle’ or ‘Upper lower’, and ‘higher SES’ was defined by Kuppuswamy’s category of ‘Upper’ or ‘Upper middle’. This variable was used to investigate the impact of SES status in a sub-group analysis.
Developmental measure
Developmental measures and autism traits were assessed in all participants by a local psychologist who was part of the research team. These assessments were conducted on the same day as the eye-tracking assessment where possible, or within 1 month. The Developmental Profile–3 (DP-3) was used to assess child development, and to confirm that developmental milestones had been attained in accordance with age in the comparison group. The DP-3 is designed to assess development and functioning across five areas: physical, adaptive, social-emotional, cognitive and communication, through both parental-report and the assessor’s observation of child behaviour; it is used routinely in clinical practice and for research purposes in this setting.
Autism measure
The INDT-ASD (Juneja et al., 2014) and the AIIMS-Modified-INDT-ASD Tool (Gulati et al., 2019) have been specifically developed for an Indian paediatric population for clinical assessment of ASD, and have demonstrated high sensitivity in screening and diagnosis of ASD. It shows a high validity against Diagnostic and Statistical Manual of Mental Disorders (4th ed., text rev.; DSM-4-TR) diagnoses and Childhood Autism Rating Scale scores as well as with DSM-5 (Vats et al., 2018). Autism traits in this study were therefore assessed using the AIIMS-Modified-INDT-ASD Tool; this tool was administered by an experienced psychologist and trained researcher on the project. The tool has two sections comprising of 28 questions for seven items (three items for Social Interaction and Communication and four items for Restrictive and Repetitive Behaviours); representing domains of DSM-5 criteria for ASD diagnosis (see the study by Gulati et al., 2019 for details). For each item, the assessor has a trichotomous endorsement choice (‘yes’, ‘no’, ‘unsure/not applicable’), which is based on parent report as well as the assessor’s observation of the child. Each item is then given a score of ‘1’ for ‘Yes’ or ‘0’ for ‘No’ or ‘unsure/not applicable’; higher scores reflected more autism symptoms (Gulati et al., 2019; Juneja et al., 2014). A total score of ⩾5 is used clinically to diagnose ASD using the AIIMS-Modified-INDT-ASD Tool.
Assessment procedure
Assessments took place within a community centre or within a healthcare facility. For the comparison group, community centres were found within a short distance of participants’ homes to minimise burden of travel for participants and to enhance the accessibility of this research (Lockwood Estrin et al., 2022). For children in the clinical groups, assessments took place in a room in the Child Neurology Department at AIIMS. They were scheduled on the same day as their outpatient appointment to minimise disruption for participants and to increase accessibility for participation. In both settings, the assessment took place in a small single room with limited public access. Following informed consent, the child sat either on a mat on the floor or on their parent’s lap, in front of the monitor and eye-tracker, which was placed on a small table. Children were instructed to look at the screen. The parent or guardian present during the assessment typically sat next to the child. Due to the well-known challenges of engaging young children during neurodevelopmental assessments, and especially in non-lab-based environments (Lockwood Estrin et al., 2022), if the child became agitated or disinterested in the task, the parent encouraged them to look at the screen before the antisaccade trial began (i.e. at the gaze-contingent fixation point appearing on the screen before the start of each antisaccade trial, see details below). Breaks were provided whenever the child required to minimise fatigue. For their participation, small toys were given to the child following the assessment.
Eye-tracking equipment and set-up
Eye-tracking data were acquired from a Tobii X2-60 (Tobii AB, Sweden), at a sampling rate of 60 Hz. Tasks were presented on a monitor connected to a Macbook Pro computer (Apple Inc., USA), using a custom-written stimulus presentation framework (Task Engine, sites.google.com/site/taskenginedoc/), running in MATLAB using Psychtoolbox 3 (Brainard, 1997; Kleiner et al., 2007). At the start of the assessment, participants were positioned in front of the monitor, with online feedback providing information to optimally position the participant as close as possible to the centre of the eye-tracker head box to maximise data quality. Following this positioning, an automatic five-point calibration was performed, for which the child sat and watched five points on the monitor appear consecutively – calibration proceeded automatically until accuracy and precision criteria were met for at least one eye on each of five calibration points. This process typically took under 5 min. Following calibration, a battery of eye-tracking tasks was conducted – this battery was closely based on those used in multiple longitudinal studies across the United Kingdom and Europe (Bedford et al., 2012; Elsabbagh, Fernandes, et al., 2013; Loth et al., 2017); the battery included eye-tracking tasks – not presented in this article – such as the gap-overlap task (Bedford et al., 2012; Elsabbagh, Fernandes, et al., 2013) and the pop-out task (Elsabbagh, Gliga, et al., 2013). Eye-tracking tasks were interspaced to maintain the child’s interest and thereby to minimise data loss.
Antisaccade task
The antisaccade task was shown to the child as part of a battery of eye-tracking tasks (see above). At the start of each task or trial, a gaze-contingent fixation point was presented to the child to draw their attention to the centre of the screen; the trial only progressed when the child looked at the fixation point. The antisaccade task was presented in a set of four blocks, interspaced between other eye-tracking tasks to ensure the child did not lose interest. At the beginning of each trial, a gaze-contingent fixation stimulus was presented in the centre of the screen; when gaze fell upon this stimulus, the trial started. This allowed the child to control the pace of the trials to suit their attention level. When the trial started, a distractor stimulus (a black circle) appeared for 200 ms on one side of the screen. After 600 ms, a target stimulus (a red circle) was presented on the opposite side of the screen. When the child looked at the target, an animation of an animal accompanied by the appropriate animal sound replaced it (the reward; Figure 1). Following this, the next fixation point was shown. The trial terminated when the child looked at the reward, or after 2.5 s, whichever was sooner. There were 12 trials presented for each of the four blocks; the distractor and target did not change sides to enable learning across the block. All elements of the task, including the reward stimuli, remained the same across the four blocks of trials. If the participant became bored or fussy, or if they did not look at the fixation, the experimenter could skip the current trial and move on to the next.
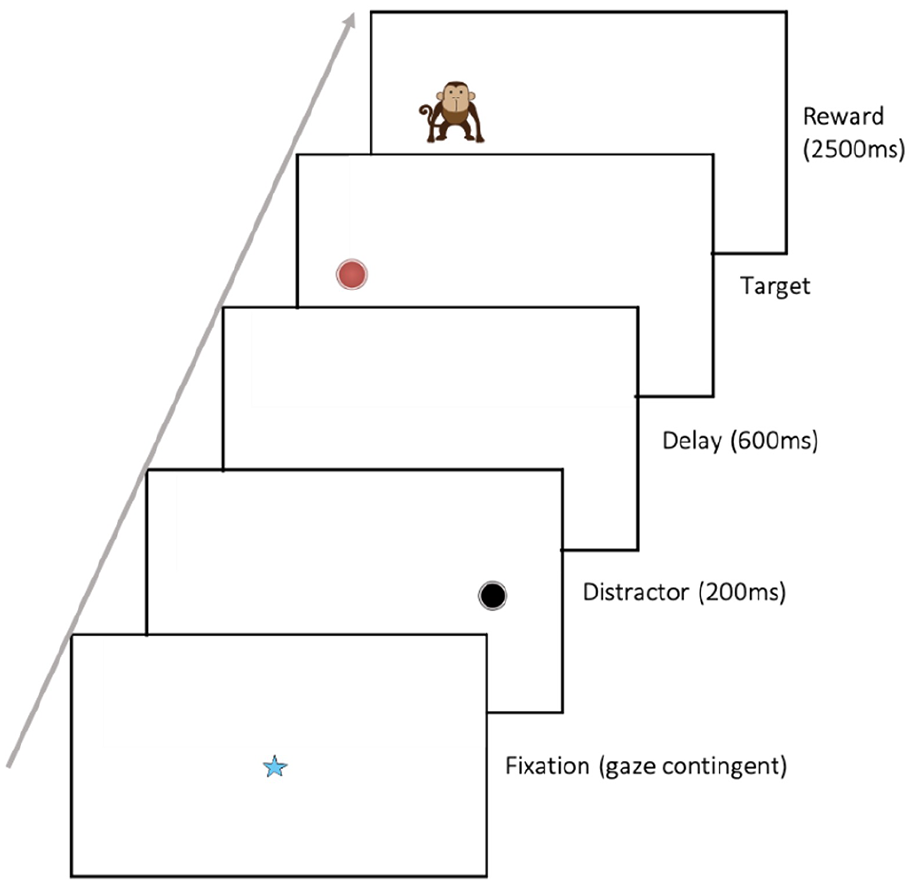
Antisaccade stimulus sequence. Schematic figure* of stimulus sequence for experimental trials in the antisaccade task. Every trial started with the central fixation stimulus onset. When the trial started, a distractor stimulus (a black circle) appeared for 200 ms on one side of the screen. After 600 ms, a target stimulus (a red circle) was presented on the opposite side of the screen. When the child looked at the target, an animation of an animal accompanied by the appropriate animal sound replaced it (the reward).
Data analysis
Looks and reaction times towards stimuli were measured offline. First, each trial was analysed for inclusion; trials were excluded from analysis if: the child did not look at the central fixation point (and if the experimenter skipped this trial) or did not look at either the distractor or target stimulus; if the child looked at the target in under 100 ms of the target appearing (considered too short for a reactive saccade, see below); if the child took longer than 2.3 s (from start of trial) to look at the target stimulus, as this would indicate child not engaging with the task; if the child’s eye gaze on the screen monitor was not detected for >50% of the trial time (from central stimulus presentation to reward appearing). Second, participant data were validated for inclusion: if the participant had five or fewer valid trials, the child was excluded from further data analysis. As outlined in the participants section above, 11 children were excluded from analysis using these criteria (n = 3 excluded due to > 50% missing proportion of eye-gaze data acquired; n = 8 excluded due to having too few valid trials). Data from 104 participants were therefore analysed. Data were transferred to STATA14 (StataCorp. 2015) for statistical analysis.
Calculating prosaccades and antisaccades
For each valid trial, it was identified whether the participant looked at the peripheral distractor stimulus (prosaccade), and whether the child made an anticipatory look to the target (antisaccade); the prosaccades or antisaccades were calculated offline using STATA14. A prosaccade was defined as present (i.e. coded as ‘1’) if the participant looked at the peripheral distractor stimulus in a valid trial. An antisaccade was defined (coded as ‘1’) when the child looked towards the target stimulus before its onset, or shortly after (up to 100 ms post-target onset, as in other studies using antisaccade tasks; Scerif et al., 2005; and adults: Guitton et al., 1985).
Statistical analysis
To analyse the number of antisaccades and prosaccades across trials, and between groups, GEE analyses were conducted. The number of antisaccades or number of prosaccades were entered into the GEE model as the dependent variable, with trial number, block and group entered as independent variables, along with their interaction terms. GEE models analyses were built with a binomial distribution, a logit link function, an exchangeable correlation matrix and a robust estimator (as per Vernetti et al., 2018). Learning in this task may not only result in more antisaccades (anticipatory saccades) but also in faster saccades to the target. We, therefore, also examined the reaction time of the child’s look towards the target in a separate GEE model (see Supplementary Material A).
Results
The age, sex and demographics of participants included in the analysis can be seen in Table 1 (also see ‘Socio-economic scores’ section below for details); participant autism trait scores (as assessed using INDT-ASD) across groups can also be seen in Table 1. There were no differences in the participant’s age at assessment between groups (F = 0.78; p = 0.459). For the majority of participants, mothers were the primary caregiver (87.13%).
Group characteristics and demographic information.
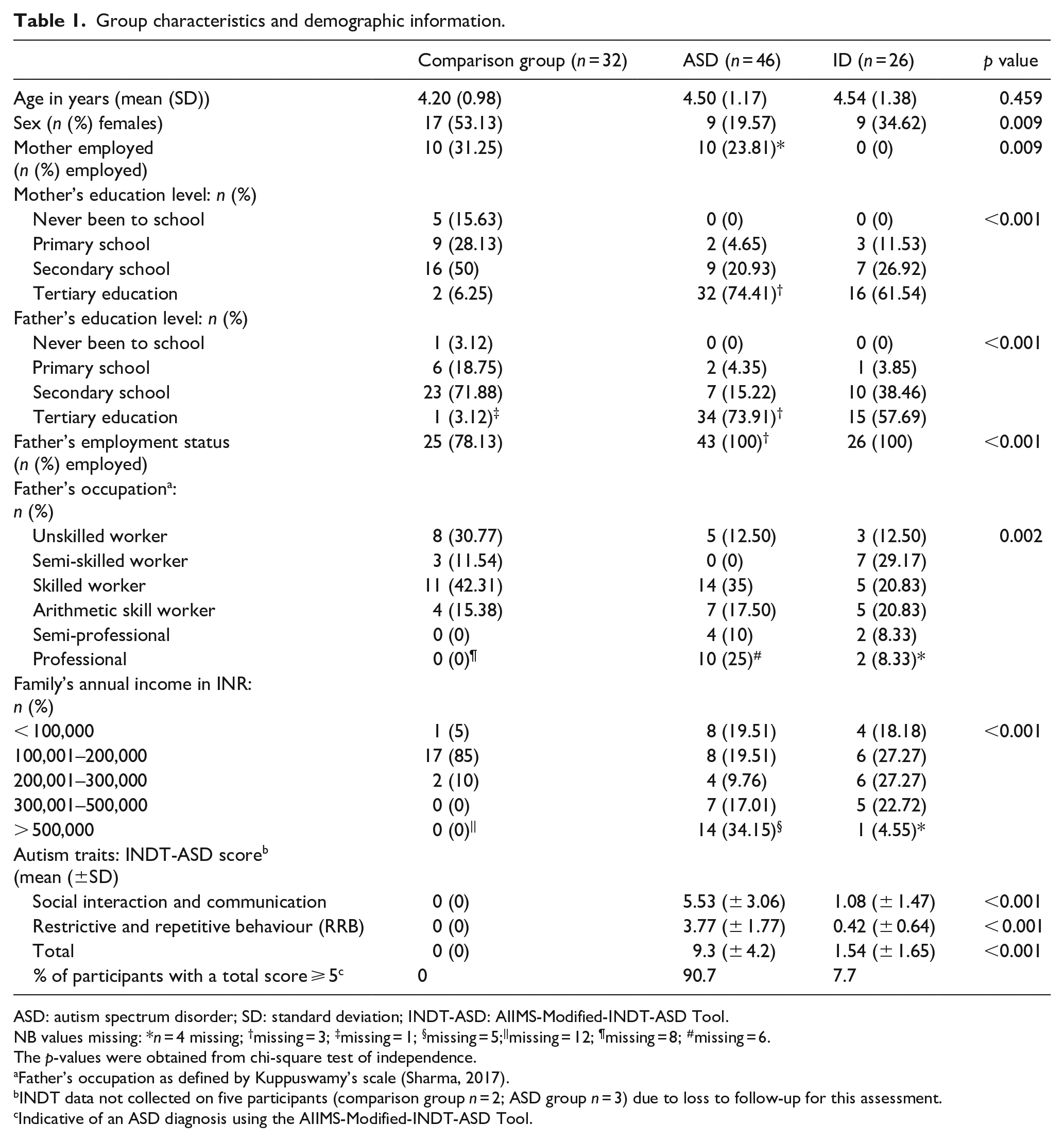
ASD: autism spectrum disorder; SD: standard deviation; INDT-ASD: AIIMS-Modified-INDT-ASD Tool.
NB values missing: *n = 4 missing; †missing = 3; ‡missing = 1; §missing = 5;||missing = 12; ¶missing = 8; #missing = 6.
The p-values were obtained from chi-square test of independence.
Father’s occupation as defined by Kuppuswamy’s scale (Sharma, 2017).
INDT data not collected on five participants (comparison group n = 2; ASD group n = 3) due to loss to follow-up for this assessment.
Indicative of an ASD diagnosis using the AIIMS-Modified-INDT-ASD Tool.
Antisaccades and prosaccades
Figure 2 shows the proportion of prosaccades (Figure 2(a)) and antisaccades (Figure 2(b)) made across valid trials in block 1 compared to block 4, by group. The mean number of valid trails across participants was 31.61 (±10.5). The ID group had significantly less valid trials than the other groups (comparison group: 35.66 (±11.5); ASD group: 31.20 (±11.4); ID group 26.96 (±12.9); F(2,109) = 3.68, p = 0.028). As we were investigating learning across the task, the analysis focused on the slope (trial effect) rather than absolute group differences in number of prosaccades/antisaccades.
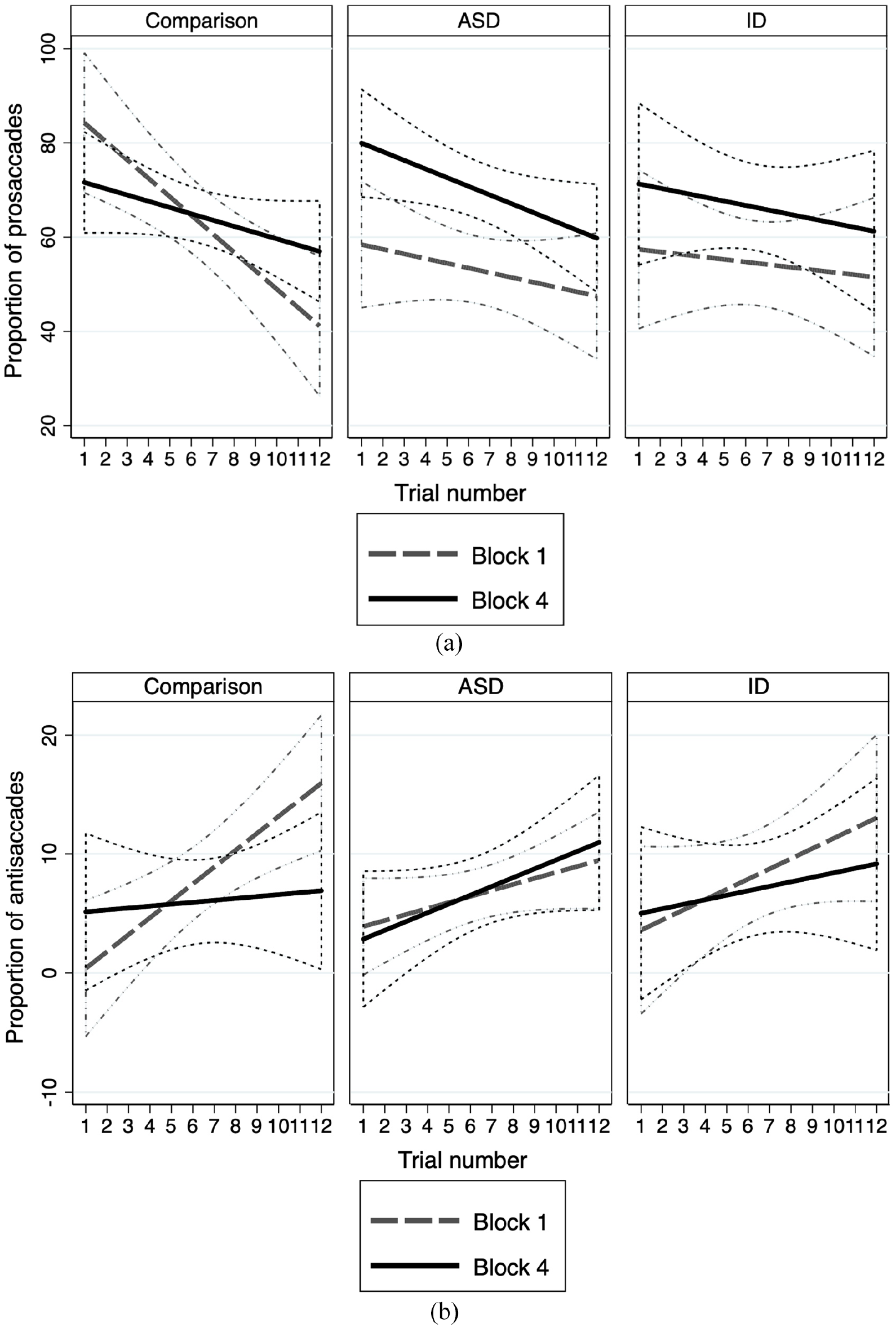
(a) Proportion of prosaccades across trials in block 1 compared to block 4 by group. It shows proportion of prosaccades made by group in block 1 compared to block 4. The comparison group shows a greater rate of reduction in prosaccades in the first block compared to ASD and ID groups. In block 4, all groups show reduced rates of reduction of prosaccades; this reduction is less in the clinical groups compared to the comparison group. (b) Proportion of antisaccades produced across trials in block 1 compared to block 4 by group. It shows proportion of antisaccades made by group in block 1 compared to block 4. The comparison group shows a greater rate of production of antisaccades in the first block compared to ASD and ID groups. In block 4, all groups except the ASD group show reduced rates of production of antisaccades compared to block 1. In block 4 the ASD group continued to learn to produce antisaccades at a greater rate compared to the comparison group.
Prosaccade analysis
If a prosaccade was made in a trial, it was coded as 1, if no prosaccade was made, this was coded as 0. Participants made prosaccades in 60.9% of trials overall (the comparison group made a prosaccade in 63.7% of trials, ASD group in 62.1% of trials, and ID group in 53.8% of trials). To evaluate changes in prosaccades over the course of the task, the number of prosaccades were entered into a GEE analysis as the dependent variable. No effect of age was observed when included in the model as an independent variable, and was therefore not included in the full GEE model. The clinical groups made fewer prosaccades from the start of the task than the comparison group, that is, the intercept was lower (ASD: coefficient = −1.784, p < 0.001, 95% confidence interval (CI) = [–2.71, –0.96]; ID: coefficient = −1.99, p = 0.001, 95% CI = [–3.14, –0.84]). We found an overall reduction in the number of prosaccades made across trials in all groups (coefficient = −0.240, p < 0.001, 95% CI = [–0.32, –0.15]). This decrease was found to be reduced in the clinical groups compared to the comparison group (i.e. the ASD and ID groups showed less inhibition of looks to the distractor across trials, compared to the comparison group) (ASD: coefficient = 0.178, p = 0.001, 95% CI = [0.07, 0.29]; ID: coefficient 0.209, p = 0.001, 95% CI = [0.08, 0.33]).
A reduction in the number of prosaccades made was also observed across blocks (coefficient = −0.394, p < 0.001, 95% CI = [–0.62, –0.18]). This effect across blocks was reduced in the clinical groups compared to the comparison group (i.e. a reduced inhibition of prosaccades in the clinical groups compared to comparison group was observed across blocks (ASD: coefficient = 0.671, p < 0.001, 95% CI = [0.34, 0.99]; ID: coefficient 0.508, p = 0.014, 95% CI = [0.10, 0.91]).
Finally, we found that the decrease in number of prosaccades made across trials (within blocks), reduced across blocks (trial × blocks, coefficient 0.052, p < 0.001, 95% CI = [0.03, 0.08]). This effect was also found to differ between groups, with a greater reduction of the trial effect across blocks (i.e. clinical groups carried on decreasing the amount of prosaccades in the later blocks) found in the clinical groups compared to the comparison group (ASD: coefficient = −0.068, p < 0.001, 95% CI = [–0.11, –0.03]; ID: coefficient = −0.068, p = 0.02, 95% CI = [–0.11, –0.02]).
We followed up this block effect with additional analyses conducted in each block: a reduction in the number of prosaccades made across trials was found in block 1 (coefficient = −0.174, p = 0.003, 95% CI = [–0.25, –0.21]) and block 2 (coefficient = −0.203, p < 0.001, 95% CI = [–0.21, –0.12]), but not in block 3 or 4. A positive group × trial effect was observed in both clinical groups compared to the comparison group in block 1 (ASD: coefficient = 0.126, p = 0.003, 95% CI = [0.04, 0.21]; ID: coefficient = 0.143, p = 0.008, 95% CI = [0.04, 0.25]) and 3 (ASD: coefficient = −0.108, p = 0.036, 95% CI = [–0.21, –0.01]), with a non-significant trend observed in block 2 (coefficient = 0.083, p = 0.081, 95% CI = [–0.01, 0.18]); no trend was observed in block 4. As block 4 demonstrated no significant trial effect, we conducted an additional sensitivity analysis excluding block 4 from the GEE model. This sensitivity analysis yielded similar results, but with larger effect sizes observed than in the full model (see Supplementary Material B for sensitivity analysis results).
In summary, both clinical groups showed a lower number of prosaccades than the comparison group from the start of the task, and decreased their number less within and across blocks. However the trial × block effect demonstrated that clinical groups continued to learn to inhibit their prosaccades through to the later blocks, compared to the comparison group who demonstrated reduced learning in later compared to earlier blocks.
Antisaccade analysis
If an antisaccade was made in a trial, it was coded as 1; if no antisaccade was made, this was coded as 0. Participants made antisaccades in 8.2% of trials overall (comparison group made antisaccades in 6.6% of trials, ASD group in 8.6% of trials and ID group in 10.1% of trials). To evaluate how antisaccades varied across the course of the task: the number of antisaccades was entered into a GEE analysis as the dependent variable. There was a significant effect of age on number of antisaccades (coefficient = −0.254, p = 0.023, 95% CI = [–0.47, –0.04]); therefore, age was kept in all analyses. There was no age × group effect. There was no significant difference in the number of antisaccades made from the start of the task between groups, that is, no difference in the group intercepts (ASD: p = 0.164, 95% CI = [–0.55, 3.26]; ID: p = 0.099, 95% CI = [–0.29, 3.33]).
As predicted, we observed an overall increase in the number of antisaccades made across trials (coefficient = 0.291, p < 0.001, 95% CI = [0.17, 0.41]) and across blocks (coefficient = 0.431, p = 0.041, 95% CI = [0.02, 0.84]). This increase in the number of antisaccades made across trials (slope) was reduced in the ASD group compared to the comparison group (i.e. ASD group shows a flatter, but still positive slope, compared to comparison group across trials; group × trial coefficient = −0.178, p = 0.047, 95% CI = [–0.35, –0.002]). The ID group showed no significant difference in slope compared to either group.
We found that the increase in number of antisaccades made within blocks, differed across blocks, with a reduced slope observed in later blocks (trial × block coefficient = −0.061, p < 0.002, 95% CI = [–0.10, –0.02]). This block × trial effect was found to differ between groups, and found to be less evident, with the slope (i.e. number of antisaccades increasing across trials) reducing less between blocks for the ASD group compared to the comparison group (coefficient = 0.073, p < 0.023, 95% CI = [0.01, –0.14]). No difference was found with the ID group.
We followed up this block effect with additional analyses conducted in each block: an increase in the number of antisaccades across trials was found (block 1: coefficient = 0.213, p < 0.001, 95% CI = [0.12, 0.31]; block 2: coefficient = 0.191, p = 0.012, 95% CI = [0.04, 0.34]; block 3, a trend was found: coefficient = 0.117, p = 0.059, 95% CI = [–0.004, 0.24]; no effect found in block 4). A trend towards a group × trial effect was also found in block 1 (ASD group showed a smaller increase in number of antisaccades made across trials compared to comparison group (block 1: coefficient = −0.140, p = 0.057, (95% CI = [–0.28, 0.004]); this effect was not found in the other blocks – perhaps due to the small numbers of antisaccades made in each block individually. As with the prosaccade analysis described above, we conducted a sensitivity analysis excluding block 4 from the GEE antisaccade model due to its lack of trial effect. This sensitivity analysis yielded similar results (see Supplementary Material B for sensitivity analysis results).
In summary, all groups showed similar numbers of antisaccades produced at the start of the task; however, the ASD group did not increase the amount of antisaccades at the same rate as that of the comparison group either across trials or blocks. The trial × block effect suggested that the ASD group continued to learn to produce antisaccades in the later blocks at a greater rate compared to the comparison group. This effect was not true for the ID group.
Reaction time to look at fixation stimuli
As the number of prosaccades differed between groups from the start of the task, we wanted to investigate whether this difference was driven by group levels of attention to the screen. Therefore, as a proxy measure of child attention to the screen, we calculated the average time taken for a child to look towards the fixation stimuli presented throughout the battery of eye-tracking tasks. We used a Kruskal–Wallis H to test this measure of attention between groups, and found a statistically significant difference between the three groups (χ2(2) = 39.31, p = 0.0001); post hoc Mann–Whitney tests revealed significant differences between each group (comparison group vs ID: z = –5.359, p < 001; comparison group vs ASD: z = –5.584, p < 0.001; ASD vs ID: z = –6.255, p < 0.001).
We tested whether this attention measure was predictive of antisaccade production in children, or of the ability to inhibit prosaccades. No significant relationship was observed between the proportion of antisaccades and reaction time to the fixation stimuli. A significant negative relationship was found between the proportion of prosaccades and reaction time to stimuli (coefficient = −0.01, p = 0.044, 95% CI = [–0.01, –0.001]), but with a small effect.
RRBI and social interaction and communication scores
To evaluate the association between RRBIs or social interaction and communication (see Table 1) and our dependent variables (antisaccades and prosaccades), we included the INDT score in place of the group variable in a separate GEE analysis. No significant effect on the number of prosaccades made was observed with the RRBI or social communication scores. There was less of an increase in the number of antisaccades made across the trials (i.e. slope) with increased RRBI scores (coefficient = −0.074, p = 0.003, 95% CI = [–0.12, –0.03]). When looking at the block × trial effect, the increase in antisaccades between blocks was also weaker when RRBI scores were higher (coefficient = 0.029, p = 0.016, 95% CI = [0.01, 0.05]). No effect on the number of antisaccades made was observed with the social interaction and communication scores.
Socioeconomic scores
The comparison group had a lower income level and maternal education than the ASD and ID groups (Table 1); for example, 74.4% of mothers in the ASD group and 61.5% of mothers in the ID group had an education level higher than secondary school, but only 6.3% of mothers in the comparison group were educated to this level. Moreover, the derived variable on SES (Kuppuswamy’s SES Scale, see Methods section) illustrated that 17 participants (100%; but n = 15 missing data) in the comparison group were from a lower SES status, compared to 13 (34.21%; n missing = 8) in the ASD group and 10 (47.62%; n missing = 5) in the ID group (p = 0.001); however, there was a large amount of missing data (n missing = 28). Therefore, as there were significant differences found in SES between groups, a sub-group analysis was conducted, where we sought to investigate whether the relationships between prosaccades or antisaccades and trial, blocks and groups were sensitive to the SES status of participants. No significant main effect and no significant interaction effects on group, trial or block were identified for prosaccades or antisaccades, when using the Kuppuswamy’s SES Scale or maternal education as a measure of SES; maternal education is known to be a core dimension of SES, showing strong associations with child cognitive development (Harding et al., 2015; Reardon, 2011; see Supplementary Material C).
Discussion
This is the first study to investigate attention control and learning using portable eye-tracking technology in pre-school children in a community LMIC setting in New Delhi, India. In accordance with findings from high-income, laboratory-based environments (Scerif et al., 2005), we found that young pre-school children learn to inhibit prosaccades and increase the number of antisaccades made over the course of the task. This result is indicative of young children learning to control their visual attention by learning to inhibit their eye-movement towards a distractor, as well as learning to anticipate a look towards the rewarded target stimulus. Few such studies have focused on these early childhood years, despite age significantly influencing attention control (Geurts et al., 2014; Klein, 2001); the majority of studies have focused on children who were aged between 8 and 15 years (Geurts et al., 2014). Perhaps this paucity is primarily due to the well-known difficulties of engaging young children’s attention during assessment, thereby impacting data collection in this age group. The first study to implement the antisaccade task in toddlers found that the number of antisaccades produced increased between 8 and 38 months, highlighting pre-school age as a key period of its development (Scerif et al., 2005). Interestingly, Scerif et al. (2005) found that typically developing toddlers made antisaccades in 14.0% of trials overall, compared to our older age group, where children aged 3–5 years made fewer antisaccades (in 6.6% of trials overall). A large study of participants in an older age group, aged between 5 years to late adulthood, demonstrated that the greatest changes in production of antisaccades occurred between 5 and 15 years, but the youngest 5-year-old group showed the greatest distribution of responses (Munoz et al., 1998), further highlighting the developmental component of this task. Our results add to this body of literature, specifically by closing the gap in the evidence between ages 3 and 5 years. Moreover, we found a significant effect of age on production of antisaccades, but not prosaccades, in this age group, and demonstrate a reduced number of antisaccades in older pre-school children than younger children. It is possible, considering the known association between SES and child development (Farah, 2017), that the lower SES of this population interacted with age of these children during these critical years, thereby impacting the trajectory of development seen in high-income samples. Although our study did not fully assess this interaction directly (see Limitations section), this is an important avenue for future research.
We investigated attention control between three groups of children and found that both clinical (ASD and ID) groups showed a reduced ability to inhibit prosaccades across the task compared to the comparison group. We also observed that the ASD group, but not the ID group, demonstrated further difficulty in learning to anticipate looks towards the target, that is, did not increase the amount of antisaccades at the same rate as that of the comparison group from the start of the task. In comparison, previous results in toddlers with FXS showed that compared to the comparison group they did not learn to inhibit looks towards the distractor (prosaccades) over the course of the antisaccade task, although they did make the same number of antisaccades (Scerif et al., 2005). Taken together, our results and comparison with similar studies on young children with developmental disorders, suggests that while difficulties in visual inhibition responses might be applicable to syndromic ASD, the difficulty in learning to produce antisaccades in this age group may be more specific to ASD. This finding may reflect reduced learning in the ASD group of the reward rule, that is, to anticipate looks towards the target. Alternatively, it is also possible that this would be a consequence of the reduced ability to inhibit looking at the distractor, given that a moderate correlation was found between prosaccades and antisaccades (see Supplementary Material D). However, the differential group results between the prosaccade and antisaccade analysis more strongly suggest that there are two underlying mechanisms involved: first, in learning to inhibit the distractor; and second, in learning the reward rule. Previous studies in slightly older children have shown that groups of children with ASD demonstrate more difficulty learning an abstract rule than children with developmental delay (Jones et al., 2013). This study also found that rule learning was facilitated by providing children with concrete reinforcement, with authors suggesting therefore that children with ASD had an underlying difficulty in forming conceptual understanding of rules. The antisaccade task, as administered in this study relied on the child learning the reward rule without any verbal instruction. It is, therefore, possible that the ASD group’s difficulty to produce antisaccades results from a difficulty in forming an understanding of the rules of the task. Furthermore, it has been postulated that rules that appear to lack a clear rational or which are of an arbitrary nature leads to observed difficulties for autistic children (Bíró & Russell, 2001); it is possible that the reward rule of the antisaccade task falls into this category, thereby explaining this group’s difficulty in learning to look at the target compared to the comparison group.
However, interestingly, once learnt, the ASD group continued to apply the reward rule across the task for longer than the comparison group. Here our results suggested that while the ASD group may have a reduced rate of learning in the shorter term (i.e. results demonstrated a reduced slope within blocks for ASD compared to the comparison group), the learning process continued for a longer time than the comparison group (across blocks the ASD group continued to learn at a greater rate than the comparison group), although did not reach the same level as the control over the course of the trial. This finding may also be in keeping with the study by Jones et al. (2013), where children, once they had learned the rule, were able to apply it consistently, suggesting that the ASD group had difficulty in learning abstract rules, rather than applying them once learnt.
Furthermore, it is important to note that the mechanisms between producing prosaccades and antisaccades differ, with prosaccades being a reactive response to a distractor stimulus, and antisaccades being an anticipatory response. Previous studies have found a typical, or in some studies a superior, reactive performance in autistic individuals, but mechanisms underlying this are poorly understood (for review: studies by Kaldy et al., 2016; Mottron et al., 2006). One study found that superior visual search strategies in infants predicted an ASD diagnosis at 3 years, and that this effect was specific to ASD compared to other developmental disorders (ADHD) or mental health outcomes (e.g. anxiety symptoms; Cheung et al., 2018). On the contrary, difficulties in anticipation and prediction have been demonstrated in children with ASD (Senju et al., 2010). It has been argued that prediction can be considered a form of hypothesis testing, and it has been demonstrated that infants stop making anticipatory looks if there is no hypothesis to test in simple deterministic situations, that is, where there are no hypotheses to test (Téglás & Bonatti, 2016). Therefore, we cannot rule out the possibility that the ASD group may have learned the rule faster than other groups, and as such, have no hypothesis to test, and therefore do not anticipate the deterministic outcome. However, our results demonstrating increased learning by the ASD group in later blocks argues against this hypothesis, in favour of the abstract rule account outlined above.
In terms of measuring children’s gaze towards the screen, we found differences between the three groups; the clinical groups showing greater time taken to look towards the fixation stimuli than the comparison group, suggesting reduced orienting towards the screen. It is possible that this reduced orienting to the task influenced results between groups. This points to an overall need to adjust experimental calibration parameters, such as time given for participants to understand the task for different groups to ensure that they begin the task with an equal level of understanding. However, as we found no statistical association between proportion of antisaccades and reaction time to the fixation stimuli, and only a small effect of reaction time on the proportion of prosaccades made, we can be confident in our results presented.
Restricted and repetitive behaviours (RRBIs) are a hallmark of ASD, and when investigating the specific effects of autism traits on the production of antisaccades across trials, we found that the RRBI score appeared to be a driving effect; with higher RRBI scores associated with reduced learning to produce antisaccades. We found no effect of RRBI scores on the inhibition of prosaccades. These results again suggest different mechanisms driving the reduced ability to inhibit looks at the distractor, compared to learning the reward rule. Such difficulties in rule learning have previously been associated with restrictive and repetitive behaviours, but particularly the ability to shift rules such as during a card sorting task (Reed et al., 2013). While the antisaccade task does not involve shifting rules, it is possible that initial learning to inhibit looking towards an irrelevant stimulus combined with an additional requirement to learn the reward rule might necessitate a cognitive flexibility that proves more difficult for the ASD group compared to comparison groups in pre-school-aged children. Literature examining executive processes in autistic individuals has indicated particular difficulties in cognitive flexibility (Ozonoff, 1995), and which has previously been related to restricted and repetitive symptoms of autism (Lopez et al., 2005). However, our results in pre-school children appear in contrast with results from autistic adolescents, where inhibitory control, as measured by the antisaccade task by number of prosaccade errors, has been associated with RRBI scores (Mosconi et al., 2009), highlighting the age dependency of these responses. While the mechanisms linking inhibitory control deficits and RRBs remain unclear, Mosconi et al. (2009) highlighted the altered role of the frontostriatal system, which develops chiefly through adolescence (Darki & Klingberg, 2015), in ASD group difficulties of suppression of the behavioural response and its association with RRBIs.
One limitation of this study was the high proportion of missing data regarding SES of participants in each group; this limited the analysis to only a small sub-group of participants to assess the effect of SES on attention control. As our study did not aim to assess the interaction between SES and child development between groups, cohorts were not designed to be matched on SES; however, further investigation of SES and potential impact on attention control, with a cohort recruited specifically to answer this question, would be interesting in this age group. Moreover, the lower SES found in our comparison group might have led to an underestimation of the differences found between groups, and so a sample matched by SES may elucidate further group differences. A further limitation is that the setting of the eye-tracking assessment differed for the clinical (clinical setting at a hospital) compared to the comparison group (community centres close to participant’s houses). However, both settings were comparable in terms of distractions to the child by family members (in both settings, family members were present during the assessment and the assessment took place in a small single room with limited public access). The unfamiliarity of the clinic setting to the child compared to a local community centre might also have impacted the child’s engagement with the task. Further studies are therefore needed to investigate the impact of setting on child engagement in these tasks. One further potential limitation of this study was that, while the participants in the ID group were recruited based upon expert local clinical assessment using DSM-5 diagnostic criteria (see Methods section), we did not include a standardised intelligence quotient (IQ) assessment in the research protocol; however, multiple domains of development (including cognitive) were assessed in all children using DP-3, which is an assessment routinely used in clinical and research studies within this setting. Finally, we also found that, for all groups, there was a reduced engagement in the task by the fourth antisaccade block of trials; this may point to the fact that eye-tracking battery was relatively long, and shorted batteries may be beneficial for children participating in future studies.
One strength of this study is that we used a case–control study design to directly compare groups of children with a clinical diagnosis of ID (without ASD), ASD and a comparison group of children meeting their developmental milestones. IQ has also been found to significantly impact inhibitory control performance (Geurts et al., 2014), and ID, defined as an IQ below 70, is often diagnosed in autistic individuals (Baio et al., 2018). Clinical symptoms also can overlap between ASD and ID, leading to potential diagnostic confusion (Pedersen et al., 2017). However, prior ASD research has mostly been limited in their study design to differentiate between ASD and ID; the majority of studies investigate autistic individuals without ID while statistically controlling for IQ. However, it is important to include individuals in research with all levels of intellectual ability, especially when aiming to assess behaviours specific to ASD (Thurm et al., 2019). This may be particularly important in LMIC settings where the majority of children with diagnosis of ASD also have ID, because these children are more likely to be bought into healthcare systems due to additional difficulties (Daley, 2004).
Conclusion
We provide novel evidence that observed difficulties in learning to inhibit visual attention responses might be applicable to multiple developmental disorders, but the difficulty to learn to produce antisaccades in this age group may be specific to ASD. We also find evidence of reduced initial rates of learning in the ASD group, but continued learning over the course of the task, at a greater rate than the comparison group. This eye-tracking task may therefore have the potential to identify and assess autism specific traits across development, and be used in longitudinal research studies such as investigating risk factors for development or response to intervention in low-resource settings. Previously, such investigation has almost exclusively been conducted in western, high-income settings, and in controlled lab-based environments that are inaccessible to the majority of children. Because majority of the world’s autistic population lives in LMICs, a global perspective on ASD is of urgent importance to improve our understanding of the behavioural expression of ASD, which is likely to be culturally and contextually biased (de Leeuw et al., 2020; de Vries, 2016; Durkin et al., 2015). Moreover, when research is restricted to HICs, this has reduced the generalisability of our understanding of brain development (Shen et al., 2021) by excluding populations where the majority of children with developmental disorders reside. Our study therefore represents an important step to address these shortfalls in the literature by assessing visual attention and learning in early childhood using portable eye-tracking technology in a group of young children with and without ASD living in an LMIC setting in India.
Supplemental Material
sj-docx-1-aut-10.1177_13623613221149541 – Supplemental material for Attention control in autism: Eye-tracking findings from pre-school children in a low- and middle-income country setting
Supplemental material, sj-docx-1-aut-10.1177_13623613221149541 for Attention control in autism: Eye-tracking findings from pre-school children in a low- and middle-income country setting by Georgia Lockwood Estrin, Luke Mason, Rashi Arora, Supriya Bhavnani, Jayashree Dasgupta, Sheffali Gulati, Teodora Gliga and Mark H Johnson in Autism
Footnotes
Acknowledgements
The authors would like to thank all participants and their families for taking part in this research. We also thank Professor Vikram Patel and Dr Gauri Divan for their guidance with this research and study design.
Author contributions
G.L.E., L.M., S.B., J.D., S.G., T.G. and M.H.J. contributed to the study conception and design. Material preparation and data collection were performed by G.L.E., L.M., S.G., R.A., S.B. and J.D. Data processing and analysis was performed by G.L.E. and L.M., and advised by T.G. and M.H.J. The first draft of the article was written by G.L.E. All authors commented on article drafts, and read, revised and approved the final article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All experimental procedures were reviewed and approved by the Institutional Ethics Committees of Sangath (approval number: GD_2018_39) and the Department of Psychological Sciences, Birkbeck, University of London (approval number 171897), and AIIMS Institutional Ethics Committee (approval number RP-37/2018, RP-49/2019).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Wellcome Trust (204706/Z/16/Z). For the purpose of open access, the author has applied a CC BY public copyright licence to any author-accepted manuscript version arising from this submission.
Data access statement
Due to the nature of this research, participants of this study did not agree for their data to be shared publicly, so supporting data are not available.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
