Abstract
Objectives
To determine seroprevalence, seroconversion, and mother-to-child transmission (MTCT) rates for dual and triplex infections of human immunodeficiency virus (HIV), hepatitis B virus (HBV), and hepatitis C virus (HCV) among pregnant women.
Methods
A multicentre prospective cohort study was conducted in six randomly selected tertiary hospitals from six geopolitical zones of Nigeria. Consenting participants were tested at recruitment for triplex infections and followed-up till delivery. Retests were performed at delivery for those who tested negative for all three infections/positive for only one. Polymerase chain reaction was used for validation while rapid test kits were employed for initial screening.
Results
Of the 2775 participants recruited, 13 (0.47%; 95% CI: 0.25%–0.80%) and 4 (0.14%; 95% CI: 0.04%–0.37%) were seropositive for dual and triplex infections, respectively. Dual infections revealed seroprevalences of 0.22% for HIV-HBV (6/2775; 95% CI: 0.08%–0.47%), 0.14% for HIV-HCV (4/2775; 95% CI: 0.04%–0.37%), and 0.11% for HBV-HCV (3/2775; 95% CI: 0.02%–0.32%). Multivariable analysis highlighted significant associations between HIV/HBV co-infection and religion (adjusted odds ratio (aOR): 0.068, 95% CI: 0.006–0.757) and house ownership (aOR): 1.65 × 10–9, 95% CI: 1.60 × 10–9–1.70 × 10−9). Continuing our follow-up until delivery for 2403 initial participants, 2386 did not have dual or triplex infections at the start. Upon retesting at delivery, three of these women were seropositive for a dual infection of HIV and HBV, giving a seroconversion rate of 0.12% (95% CI: 0.03% to 0.37%). MTCT rate stood at 0% at 6-week post-delivery.
Conclusion
We observed a relatively low seroprevalence and seroconversion rates for dual and triplex infections of HIV, HBV, and HCV among pregnant women in Nigeria and no MTCT.
Introduction
Hepatitis B virus (HBV), hepatitis C virus (HCV), and human immunodeficiency virus (HIV), along with their dual or triplex combinations, continues to be a significant global public health concern, primarily impacting women. 1 In 2022, there were about 39 million HIV-positive individuals in the world. 2 Of these, 53% were women and girls, 37.5 million were adults, and 1.5 million were children under 15 years. 2 Currently, 257 million individuals are afflicted with chronic hepatitis B, and another 71 million with chronic hepatitis C. Should the current rate of spread remain unchanged, it is forecasted that both hepatitis B and C will lead to 20 million deaths from 2020 to 2030. 3
There is evidence that co-infections with HIV, HBV, and HCV are common in sub-Saharan Africa. 4 A recent study in Northwest Ethiopia reported that the co-infection rate of HBV and HCV among HIV-positive pregnant women was 0.4% for each. 4 According to another recent study conducted in South Africa, 12.2% of those living with HIV/AIDS are also co-infected with HBV. 5 The high rate of co-infections of HBV or HCV among HIV-positive population could be attributed to common routes of transmission such as sharing of sharps, sexual intercourse or mother-to-child transmission. Concerning dual and triple infections during pregnancy, a recent study by Tesfaye et al. revealed that a history of abortion (AOR = 11.028, 95% CI = 1.671–72.776, p = .013) and prior blood transfusion (AOR = 11.298, 95% CI = 1.066–119.777, p = .044) were significantly linked to an increased risk of HBV infection. 4 However, multiple sexual partners (AOR = 18.819, 95% CI = 1.074–329.680, p = .045) and a history of abortion (AOR = 12.550, 95% CI = 1.174–134.202, p = .036) were the only significant predictors of HCV and HIV infections, respectively. 4 Compared to any of these single infections, coinfection can result in more rapid disease progression, a variety of liver-related disorders, dysfunction of non-hepatic organs, and ultimately death.5–9 Antiviral drug adverse effects, such as hepatotoxicity, drug resistance, and a lack of necessary responses, make treating coinfected individuals more difficult. 5 Conversely, coinfected persons need to receive various pharmacological treatments concurrently, for the treatment of HIV in addition to HBV and/or HCV.5–7
UNAIDS and WHO warn that there has been uneven progress in reducing new HIV, HBV, and HCV infections, increasing access to treatment, and ending AIDS and chronic hepatitis B and C-related deaths.3,4,10 This is because many vulnerable people and populations have been left behind, despite the availability of an expanding range of effective HIV and hepatitis prevention tools and methods and a massive scale-up of HIV and hepatitis B and C screening and treatment in recent years.3,4,10 Persistent financial constraints, along with stigma, discrimination, and social marginalisation, continue to hinder HIV and hepatitis B and C screening, treatment, and prevention efforts. These challenges remain significant barriers to addressing dual and triple infections effectively. Sustained commitment and ongoing epidemiological research are essential to strengthening global responses and ensuring the success of screening and treatment programmes. 2 There is currently a paucity of information regarding the seroprevalence, seroconversion, and transmission of dual and triplex infections from mother-to child during pregnancy in Nigeria. While some small-scale research has been done on dual and triplex infections in Nigerian cities, it has only been done at the state or city level and with a small sample size.11–13
However, a significant gap in large-scale research is needed to determine the nationwide seroprevalence, seroconversion, and risk factors associated with dual and triplex infections. Furthermore, prior studies on the seroprevalence and mother-to-child transmission of triplex infections during pregnancy were inconsistent in the period for assessments and methods of assay for HIV and HBV viruses. Therefore, the authors suggested conducting more extensive and detailed prospective studies on dual and triple infections, given their clinical and epidemiological importance in co-infected individuals. They also highlighted the need to utilise Polymerase Chain Reaction (PCR) technology for accurate diagnosis.14,15 Therefore, this study focuses on assessing the seroprevalence, seroconversion rates, and mother-to-child transmission rates of dual and triplex infections of HIV, hepatitis B, and hepatitis C viruses among pregnant women in Nigeria.
Methods
Study design and setting
This prospective cohort study was carried out between December 2020 and December 2021 at six tertiary hospitals each from a region of the countries six geopolitical zones. Apart from the lead institution for the TETFund National Research Fund 2019 (Nnamdi Azikiwe University Teaching Hospital, Nnewi (South- East), the other five institutions were randomly selected and included: University of Abuja Teaching Hospital, Gwagwalada (North-Central), University of Port Harcourt Teaching Hospital, Port Harcourt (South-South), Obafemi Awolowo University Teaching Hospital Complex, Ile-Ife (South-West), University of Maiduguri Teaching Hospital, Maiduguri (North East) and Aminu Kano Teaching Hospital, Kano (North West) from which we got representative participants. The full description of the study protocol 16 as well as findings from the pilot study had been published elsewhere.17,18
Participants
The study participants were drawn from pregnant women receiving antenatal care (ANC) in the selected hospitals. These women were categorised into two groups based on their HIV, HCV, and HBV status. The exposed group included pregnant women with at least two infections of HIV, HCV, or HBV, while the unexposed group comprised pregnant women either without any of these infections or with only one infection. Both groups of pregnant women and their infant pairs were monitored prospectively through the delivery period and up to 6 weeks post-delivery.
Inclusion and exclusion criteria
Women who attended their first ANC visit at the six tertiary hospitals during the study period and consented to participate were included. However, those who were critically ill and unable to continue follow-up visits, as well as those who declined participation, were excluded. Additionally, pregnant women with an indeterminate HIV serostatus or uncertain gestational age were not included in the study.
Sample size determination
The sample size for this study was calculated using Cochran’s formula for sample size determination, as outlined by Noordzij et al., 19 which is applicable when the population exceeds 10,000, as mentioned in the published protocol. 16 The formula used was N = Z2αPQ/d2. Here, ‘Z’ represents the standard normal deviation corresponding to a 95% confidence interval, ‘P’ denotes the issue prevalence (which, in this case, is the prevalence of health facility delivery rate among pregnant women in Nigeria, identified as 38.0% based on a recent study by Adedokun and Uthman utilising data from the 2013 Nigeria Demographic and Health Survey involving 20,192 women who had given birth within 5 years of the survey 20 ), ‘Q’ equals 1-P, and ‘d’ is the desired margin of error set at 0.05. The final figure was adjusted to accommodate a potential 10% attrition or nonresponse rate, resulting in a non-inferiority sample size of 363, which was then rounded up to 400. Consequently, a minimum of 400 pregnant women were enlisted from each of the six hospitals chosen, and a total of 2775 women were enrolled.
Sampling techniques
A tertiary hospital from each of Nigeria’s five geopolitical zones, in addition to the lead institution in the South-East geopolitical zone of the country, was selected through a simple random sampling method employing a lottery system. Every eligible pregnant woman who provided informed consent was enrolled progressively until each selected hospital’s allocated sample size was achieved.
Data collection procedure and quality assurance
A validated, pre-tested, structured, and interviewer-administered questionnaire was used to collect the data. The information was gathered using an interviewer-administered, structured, and standardized questionnaire. Individual interviews using a standardized questionnaire were used to gather clinical and sociodemographic data: numerous sexual partners, age, ethnicity, marital status, education, gravidity, and parity.
The tool was initially created in English, then translated into the mother tongues of Igbo, Yoruba, and Hausa to avoid communication problems, and lastly translated back into English to guarantee consistency with the assistance of linguists and public health specialists. Experts in infectious diseases, paediatricians, midwives, and obstetricians participated in face and content validity testing. Following delivery, information on the outcome variables – such as whether the newborn was born alive or stillborn, and other pertinent data were gathered from the mother and the infant. Additional information was gathered using a checklist from the pregnant women’s integrated prenatal, labour, delivery, and postnatal care records after delivery. Under the direction of the obstetricians who served as study site coordinators, data was gathered by skilled research assistants.
Supervisors and data collectors received training. Throughout the data-collecting period, daily supervision was provided by the supervisors, the lead investigator, or site coordinators. At the health facility where the pregnant women received their ANC follow-up, appropriate health information was given to them regarding the significance of consistent ANC attendance and, if feasible, institutional delivery.
Study variables
Operational definitions of study outcomes
The study’s outcome variables were the mother-to-child transmission rates, seroconversion rate, and seroprevalence rate. Seroprevalence for dual or triplex infections refers to the number of pregnant women who, at the start of the trial, tested positive for at least two illnesses – HCV, HBV, or HIV. Seroconversion for infections with multiple or triplex pregnant women who tested negative for any dual or triplex infection at the start of the study and tested positive for at least two or triplex infections during labour or delivery. When newborns exposed to at least two infections of HIV, HBV, or HCV become seropositive for at least two infections of HIV, HBV, or HCV at birth or within 6 weeks of life, it is implied that MTCT has occurred.
Exposure variable
The status of HIV, HBV, and HCV was taken into account as an exposure variable. As per the World Health Organisation, an individual who tests positive for HIV and has a second HIV antibody test or a positive virological test for HIV or its components confirmed by a second virological test acquired from a different determination is considered to be HIV positive. 16 As part of the body’s normal immune response to infection, the body routinely develops antibodies to HBsAg. 16 Hepatitis C virus infection is said to occur when a reactive or positive HCV antibody test is obtained. During the initial ANC follow-up, HIV, HBV, and HCV testing and diagnosis were carried out at the ANC clinics following Nigerian testing and counselling guidelines.
Independent variables
The independent variables include the study participants’ sociodemographic obstetric and medical factors.
Outcome ascertainment
Rigorous measures were employed during the data collection to ensure accurate outcome ascertainment. The gestational ages of the individuals were determined using the last menstrual period (LMP) and, when available, an ultrasound conducted before 20 weeks of gestation. We calculated the difference in days between the gestational age as determined by early ultrasound (before 20 weeks) and the date of the last menstrual period for pregnant women with a known LMP. If the discrepancy was less than 7 days, the LMP was utilised as the gestational age. Conversely, if the gestational age discrepancy exceeded 7 days, the age indicated by the ultrasound was adopted. In cases where an ultrasound before 20 weeks was not performed, the LMP was exclusively used to determine the gestational age.
Detection of dual and triplex infections
For serological testing, approximately 10 mL of peripheral blood were collected from each participant by trained medical professionals or laboratory scientists. The blood samples were briefly tested for HBV antigen and antibodies against HIV and HCV. Participants who initially tested negative for HIV, hepatitis B, and C viruses at the baseline were re-screened during labour or delivery.
For HIV testing, the Alere Determine HIV-1/2 test kit (Alere Medical Co. Ltd., Matsudo, Japan) was employed in a series, adhering to the Nigerian National HIV testing guidelines. In cases of positive results, confirmation was sought using the Uni-Gold Recombigen® HIV-1/2 (Trinity Biotech, Ireland) assay, followed by the HIV1/2 STAT-PAK (Chembio Diagnostic Systems, Inc., USA), as a tie breaker when the first two tests showed discordance results. According to the manufacturer, the HBsAg and anti-HCV were tested using the ELISA kit from LabACON (Hangzhou Biotest Biotech Company, Ltd., China), which boasts a 99.0% specificity and a 99.9% sensitivity. Each kit included controls to ensure accuracy. All procedures strictly followed the manufacturer’s guidelines, ensuring precise and reliable outcomes reporting.
Determination of viral load for HIV, hepatitis B, and C viruses
The Roche Cobas CTM/CAP real-time PCR technology was utilised to measure the viral loads of HIV, HBV, and HCV. After being separated at the collection sites, all blood samples designated for viral load testing and newborn status evaluation were stored in a freezer at minus 25°. These samples were then batched and transported under cold chain conditions to the Molecular Virology Laboratory at Nnamdi Azikiwe University Teaching Hospital (NAUTH) in Nnewi, Anambra State, Nigeria, and to other participating hospitals for analysis.
Diagnosis of infant HIV, HBV, and HCV
The HIV status of infants exposed to the virus was determined following the latest Nigerian National PMTCT (Prevention of Mother-To-Child Transmission) Guidelines. 21 All newborns exposed to HIV, HBV, and HCV were screened using DNA PCR (Polymerase Chain Reaction). Dried blood samples (DBS) were collected from the infants at birth and again at 6 weeks old. These samples were then analysed using PCR at the Molecular Virology Laboratories to diagnose infections.
Data management and statistical analysis
Initially, the collected data were cleaned, coded, and entered into a Microsoft Excel spreadsheet. Subsequently, the data were transferred to SPSS version 23.0 (IBM, Armonk, NY, USA) for comprehensive analysis. The normality of continuous variables was evaluated using the Kolmogorov–Smirnov test. Continuous data were presented as means and standard deviations, while categorical data were expressed as frequencies and percentages. The independent sample t-test was utilised to compare continuous data with a normal distribution. Descriptive statistics, including frequencies, percentages, means, and standard deviations, were used to outline the characteristics of the study participants.
Depending on data suitability, categorical variables were analysed and compared based on participants’ exposure status using Pearson’s chi-square test or Fisher’s exact probability method. A generalised regression analysis was conducted to assess the influence of risk factors on the incidence of dual and triplex infections, revealing the likelihood ratios of predictor variables. For the multinomial regression analysis, variables were selected based on their significance in the bivariate analysis (p-value <.05) or their clinical relevance. This approach ensured that key predictors were appropriately considered in the multivariable model. The model’s goodness-of-fit was assessed using Pearson’s goodness-of-fit criterion, with a probability value greater than 0.05 indicating an acceptable model fit. Within the multivariable model, variables with a p-value less than 0.05 were considered statistically significant, maintaining a 95% confidence interval.
Ethics statement
The study was approved by the National Health Research Ethics Committee, Nigeria, on 23 January 2020, with ethical approval number NHREC/01/01/2007–23/01/2020. Written informed consent was obtained from all participants before their enrolment in the study, ensuring that they fully understood the study’s objectives, procedures, potential risks, and benefits. Participants were assured that their participation was entirely voluntary and that they had the right to withdraw from the study at any time without any consequences. To maintain confidentiality, all collected data were anonymised and securely stored, with access restricted to authorised research personnel only. Unique identification codes were used instead of personal identifiers to ensure privacy. For participants who tested positive for HIV, HBV, or HCV during the study, appropriate counselling and referral to designated healthcare facilities were provided in accordance with national treatment guidelines. Treatment costs for positive cases were not covered by the study, but participants were directed to available government and non-governmental programmes that provide free or subsidised treatment and care. The study was conducted in strict compliance with the ethical principles outlined in the Declaration of Helsinki and relevant national guidelines on human research ethics.
Results
Cohort profiles
In this study, 2775 pregnant women were enrolled in the antenatal clinics (ANC) of six tertiary hospitals across Nigeria’s six geopolitical zones. The distribution of participants was as follows: 400 from Nnamdi Azikiwe University Teaching Hospital, Nnewi (South-East); 529 from University of Abuja Teaching Hospital, Gwagwalada (North-Central); 445 from University of Port Harcourt Teaching Hospital, Port Harcourt (South-South); 401 from Obafemi Awolowo University Teaching Hospital Complex, Ile-Ife (South-West); 500 from University of Maiduguri Teaching Hospital, Maiduguri (North East); and 500 from Aminu Kano Teaching Hospital, Kano (North West).
These participants were monitored from their initial ANC visit through 6 weeks post-childbirth. Of the initial 2775, 2403 women (86.59%; 95% CI, 83.17% to 90.13%) were followed up until delivery. Among the 2403 followed, 2386 were initially free from dual or triplex infections at enrolment. However, at repeat testing during delivery, 3 of these 2386 participants tested seropositive for a dual infection of HIV and HBV, resulting in a seroconversion rate of 0.13% (95% CI, 0.03% to 0.36%). There were no recorded cases of seroconversion for HIV-HCV, HBV-HCV, or triplex infections among the participants.
Sociodemographic characteristics of the study participants
Sociodemographic distribution of respondents.

ANC: antenatal care; HIV: human immuno deficiency virus; HBV: hepatitis B virus; HCV: hepatitis C virus; NHIS: national health insurance scheme; 1 Nigeria Naira: 0.0011 United State Dollar.
Dual and triplex infections of HIV, HBV and HCV
Prevalence of HIV, HBV, HCV, single, double, and triplex infections, during pregnancy, labour, at birth and at 6 weeks post-delivery.

HIV: human immuno deficiency virus; HBV: hepatitis B virus; HCV: hepatitis C virus.
Seroconversion rates for dual and triplex infections
Table 2 and Figure 1 show that 2403 of the 2775 participants (86.59%; 95% CI, 83.17% to 90.13%) were monitored until delivery. Among these 2403 women, 2386 were initially free from dual or triplex infections at enrolment. However, upon retesting during delivery, three of these 2386 individuals tested seropositive for a dual infection of HIV and HBV, resulting in a seroconversion rate of 0.12% (95% CI, 0.026% to 0.367%). There were no cases of seroconversion for HIV-HCV, HBV-HCV, or triplex infections among the participants. Flow chart of the study participants. pos + ve: positive; neg−ve: negative; HBV: hepatitis B virus; MTCT: mother-to-child transmission.
Mother-to-child transmission of dual and triplex infections
Table 2 outlines the MTCT rates for dual and triplex infections of HIV, HBV, and HCV. Among the mother-infant pairs that were positive for either dual or triplex infections, 20 mothers (17 with dual and triplex infections followed until labour or delivery, and three who seroconverted) had live births, resulting in 22 babies exposed to HIV, HBV, and HCV, which included two sets of twins. At birth, 2 of these 22 infants tested positive for both HIV and HBV, leading to a mother-to-child transmission (MTCT) rate for HIV-HBV at birth of 9.09% (95% CI: 7.785% to 10.442%). Notably, these two dual-positive infants were born to women who experienced seroconversion (as detailed in Figure 1). Subsequently, 19 infants (86.36%; 95% CI: 82.524 to 90.336%) were followed until 6 weeks post-birth. At this point, none of the infants, including those initially testing positive, were found to be HBV positive upon retesting. Thus, the MTCT rate for HBV at 6 weeks was determined to be 0.0%.
Risk factors for HIV-HBV dual infections
Regression analysis showing chi-square and significance levels of likelihood ratios of predictor variables.

ANC: antenatal care; HIV: human immuno deficiency virus; HBV: hepatitis B virus; HCV: hepatitis C virus; X2: Chisquare; predictor variable with p < .05 were put in a second table. Significant of p-values in bold.
Multinomial regression analysis for HIV and HBV dual infections.
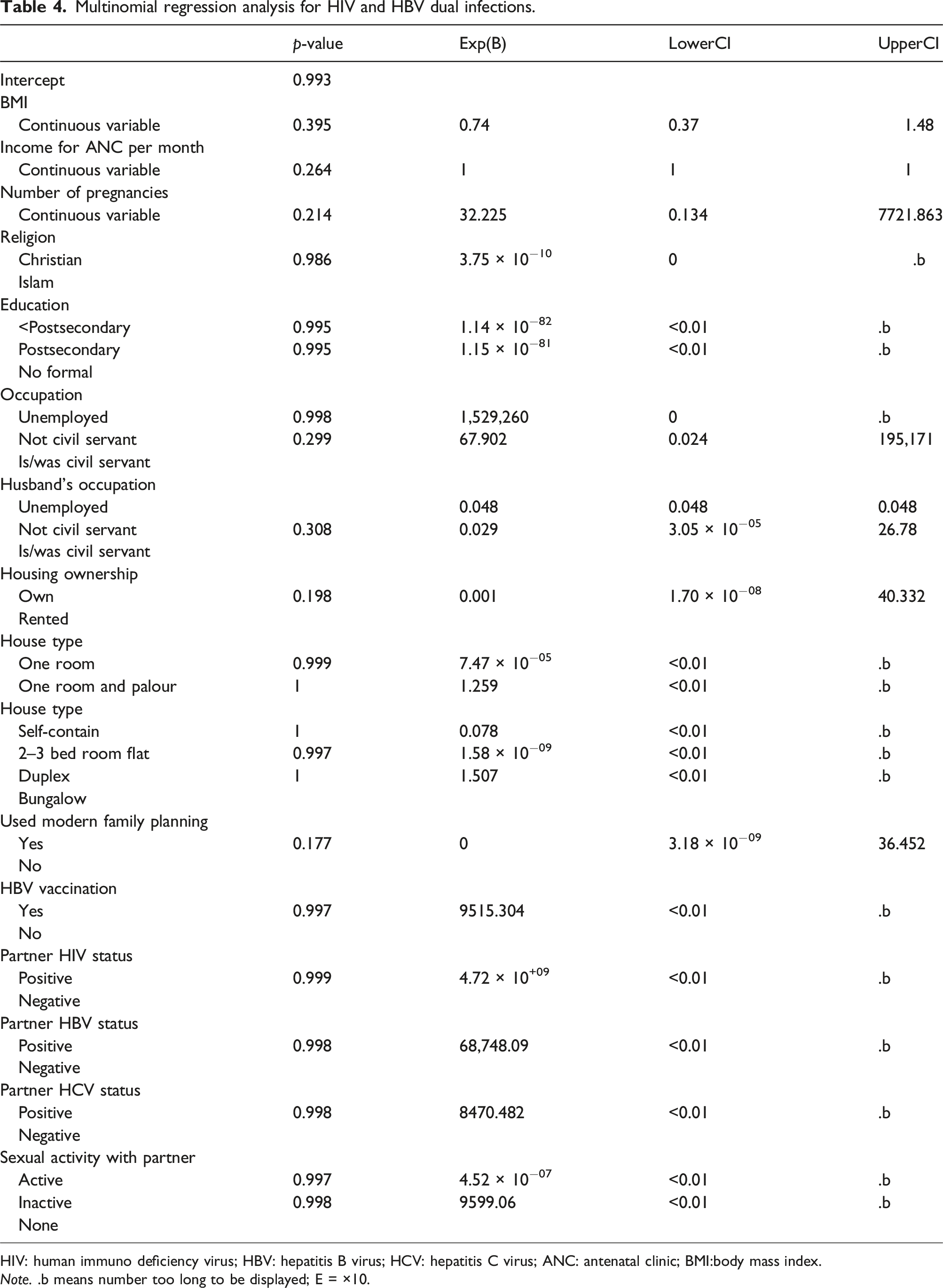
HIV: human immuno deficiency virus; HBV: hepatitis B virus; HCV: hepatitis C virus; ANC: antenatal clinic; BMI:body mass index.
Note. .b means number too long to be displayed; E = ×10.
Multinomial regression table for HIV and HBV dual infections based on likelihood ratio.

E = ×10. Signifiacnt of p-values in bold.
Risk factors for HIV-HCV dual infections
Multinomial regression analysis for HIV and HCV dual infections.

BMI: body mass index; HIV: human immuno deficiency virus; HBV: hepatitis B virus; HCV: hepatitis C virus.
Note. No model fitted based on the likelihood ratio p-values; b means number too long to be displayed; E = ×10.
Risk factors for HBV-HCV dual infections
Multinomial regression table for HBV & HCV dual infections.

ANC: antenatal care HIV: human immuno deficiency virus; HBV: hepatitis B virus; HCV: hepatitis C virus; BMI: body mass index.
Note. No model fitted based on the likelihood ratio p-values; b means number too long to be displayed; E = ×10.
Discussion
The impetus for this study stemmed from the absence of comprehensive national and hospital-based data on the seroprevalence, seroconversion, and mother-to-child transmission of triplex infections during pregnancy in Nigeria. Key findings of the study included a seroprevalence of 0.22% for HIV-HBV, 0.14% for HIV-HCV, 0.11% for HBV-HCV, and 0.14% for triplex infections. A notable correlation was observed between the occurrence of HIV/HBV co-infection and factors such as the individuals’ property ownership and religious affiliation. The study also found that the seroconversion rate for HIV-HBV dual infections was 0.12%, and remarkably, the mother-to-child transmission rate for these infections was 0% at 6 weeks post-birth, indicating no transmission from the mothers to their newborns during this period.
The frequency of dual infection with HIV and HBV among pregnant women in Nigeria has been documented in several studies and reviews.12–15 In this study, the rate of HIV/HBV coinfection was 0.22%. Comparatively, higher rates of HIV-HBV coinfection have been reported in Angola, 22 Northwest Ethiopia, 4 Dar es Salaam, Tanzania, 23 Addis Ababa, Ethiopia, 24 and Rwanda, 25 with prevalence rates of 0.4%, 0.4%, 2.8%, 3.4%, and 4.1%, respectively. While differences in prevalence may be attributed to variations in healthcare infrastructure, screening programmes, and access to preventive measures, direct comparative studies evaluating awareness and public health initiatives between Nigeria and these East African countries and Angola are limited.6,7,11–15,17 However, Nigeria has implemented several national policies and programmes focused on viral hepatitis and HIV prevention, including routine screening during antenatal care, increased access to antiretroviral therapy, and integration of HBV vaccination into childhood immunisation programmes.11–15,17 Additionally, factors such as population differences, genetic susceptibility, cultural practices, and healthcare-seeking behaviour may contribute to the observed variations in HIV-HBV coinfection rates across regions. 4 Also, it could be due to the high proportion of women who are educated in the current study and they may have a tendency to observe safe practices that could prevent dual and triplex infections. Further research is needed to comprehensively assess the impact of public health interventions on infection prevalence in different settings.
There was a strong association between women’s simultaneous HIV/HBV infections and their house property ownership and religion. The tendency is that persons who own property may be of higher socioeconomic status and HIV, HBV, and HCV are conditions that are more associated with lower socio economic status. In a previous study in Uganda, HIV/HBV co-infections were independently associated with having lived in an internally displaced persons’ camp and having shared housing with HBV-infected people during childhood. 26 Another previous Ugandan study did not find any correlation between HIV/HBV co-infection and religion. 27 Our findings have clinical significance, and more research focused on HIV-HBV cohorts is required to see whether changing one’s lifestyle might enhance these associations.
Although research on pregnant individuals with HIV/HCV co-infection remains relatively limited, several studies have been conducted in Nigeria and other parts of Africa, providing valuable insights into the prevalence, risk factors, and potential maternal and neonatal complications associated with co-infection.6,7,11–15,17,28 However, there is still a need for more comprehensive studies focussing on longitudinal pregnancy outcomes, access to treatment, and the impact of co-infection on both maternal and neonatal health. In this study, 0.14% of participants were identified as co-infected with HIV/HCV, reinforcing the need for continued surveillance and targeted interventions to mitigate potential adverse effects. In contrast, a study conducted in France reported an HIV/HCV dual infection prevalence of 1.7% (95% CI: 1.3–2.1) among pregnant women within the HIV-positive cohort. 29 This prevalence remains higher than the 0.14% observed in our study, highlighting potential regional differences in risk factors, healthcare access, and harm reduction strategies.13,29 The variation may be influenced by differences in intravenous drug use rates, blood transfusion practices, and HCV screening policies between France and Nigeria.3,12,13,29 Nevertheless, the study’s binomial regression analysis highlighted that factors such as parity, religion, house ownership, and the use of family planning methods significantly influenced the prevalence of HIV/HCV dual infection among women. However, an adjusted analysis could not be conducted due to the absence of a model based on the likelihood ratio p-values.
Previous reports on the prevalence of HBV-HCV dual infections among pregnant women in Nigeria remain limited, with many studies being regionally focused and lacking national representation.12,13,30–33 While some studies12,13,30 have involved relatively small sample sizes (e.g. 102 participants), others, such as those with 2439 pregnant women,31,33 were more comparable to our study in scale. However, a key limitation of these studies was the lack of comparisons to a reference group, limiting their ability to establish broader epidemiological trends and risk factor associations.30,31 Additionally, only a handful of national studies are conducted in hospital settings. In Ethiopia, the prevalence rates for pregnancy-related HBV-HCV infections were reported as 1.1% in one study 24 and 0.0% in a more recent one. 32 While our results support these earlier findings,24,32 it is worth noting that differences in regional practices and testing methods may contribute to variations in the reported prevalence rates.
The HBV and HCV statuses were ascertained in the current investigation using PCR. Some authorities may argue that the use of PCR alone could cause an underestimation of the percentage of HBV or HCV status since the presence of HBsAg or an HCV antibody test alone may indicate a temporary infection rather than a transformative infection where the HBV or HCV genome has integrated into the human genome. However, according to the 2020 revision of the World Health Organisation’s HCV categorisation, verified molecular methods like PCR should ideally be used to confirm the existence of HCV.3,14,17 PCR rules out the false positives including those who continue to have circulating antibodies but no active disease. These antibodies are actually evidence of previous disease or immunity. Our data which uses the gold standard of the PCR are giving the actual true values while other studies which may have used only antibody detection may actually have inflated values. This is because PCR was used for confirmatory testing, while initial screening was performed using rapid test kits.
Regression analysis revealed that several factors, such as employment, home ownership, use of family planning methods, and HBV vaccination status, appeared to affect the women’s HBV-HCV dual positive status. However, no model was fitted based on the likelihood ratio p-values, so we could not perform an adjusted analysis.
There is little research on triplex infections during pregnancy in Nigeria,14,33,34 as most of the information was derived from non-obstetric population data. But a recent meta-analysis of triplex infection in Nigeria by Eleje et al. found that the pooled prevalence of triplex infection in pregnancy was 0.03% (95% CI: 0.02–0.04%), with a higher prevalence in the HIV-positive group (0.08%) than in the general obstetric group (0.00%). 14 Furthermore, compared to certain other nations, this study’s overall prevalence of triplex infection during pregnancy was lower. For example, in Ethiopia, 24 it was 0%, while in a systematic review conducted in Africa, 35 it was 0.7% in the non-obstetric population.
In this study, we observed a seroconversion rate of 0.12%. Before this research, there was a lack of detailed data on the seroconversion rates for dual and triplex infections during pregnancy in Nigeria, which is one of the aims addressed by our investigation. The significance of our findings lies in the possibility that widespread antiretroviral therapy, routine HBV vaccinations, and comprehensive prenatal care could help maintain low seroconversion rates for dual and triplex infections. The observed minimal seroconversion rate in Nigeria might also be attributed to the adoption of universal precautions, safer sex practices, extensive use of antiretroviral drugs, and regular immunization.12–14,18 However, to provide a comparative perspective, studies conducted in other African countries, such as Ethiopia and Rwanda, have reported higher rates of HBV and HCV seroconversion among pregnant women.4,25 Similarly, studies in Angola and Tanzania have documented greater HIV-HBV co-infection rates compared to our findings.22,23 These differences may reflect variations in healthcare infrastructure, public health interventions, and screening coverage. Further comparative studies are needed to assess the long-term impact of preventive measures on seroconversion rates across different settings.
There is a notable lack of comprehensive data on the MTCT rates for dual and triplex infections of HIV, HBV, and HCV in the obstetric population. While MTCT is a recognized concern in obstetrics, indicating potential shortcomings in PMTCT programs for HIV, HBV, and HCV, the infants in this study exhibited zero MTCT rates for these infections at 6 weeks of age. Our findings align with those from a recent Nigerian meta-analysis aiming to assess MTCT rates of hepatitis B, hepatitis C, and HIV among pregnant women with one, two, or all three infections. 15 Additionally, the review highlighted a lack of data on MTCT rates of hepatitis C in mono-infections, as well as the combined mother-to-child transmission rates of HIV, hepatitis B, and hepatitis C among mother-infant pairs with dual or triplex infections in Nigeria. 15
The mother-to-child transmission (MTCT) rate for HBV in this study was 0%. This finding aligned with a Chinese study where only 2 out of 1544 subjects experienced MTCT of HBV. 36 To provide a more contextually relevant justification, national data from Nigeria would be ideal. However, limited studies specifically report the MTCT rate of HBV in Nigeria. Some reports suggest that Nigeria has made progress in hepatitis B vaccination coverage, with increasing uptake of the birth dose and routine immunization.18,37,38 Nevertheless, further studies are needed to evaluate the effectiveness of these preventive measures in eliminating MTCT of HBV in the Nigerian population. If national data remain unavailable, the observed 0% MTCT rate in this study should be interpreted cautiously, considering potential factors such as sample size and healthcare interventions received by participants. Nevertheless, the discrepancy in infant test results may be attributed to passive transfer of maternal antibodies at birth, which waned over time.
The discussion persists on how strengthening interventions for maternal and infant HIV, HBV, and HCV during pregnancy could impact Nigeria’s progress toward achieving the WHO 2030 target for global hepatitis elimination. This discussion is distinct from our study’s focus, which was confined to examining the epidemiology of dual and triplex infections during pregnancy, including their prevalence, seroconversion rates, and mother-to-child transmission (MTCT) rates, without considering the non-obstetric population. Additionally, there remains a notable gap in knowledge about the prevalence of triplex infections in this vital demographic despite concerns that it may impact pregnancy outcomes and increase the risk of perinatal transmission. 28
Even without the non-obstetric population, we were nevertheless able to detect a significant trend in the elimination of HIV, HBV, and HCV during pregnancy thanks to the design of our study. The implementation of the PMTCT package, the WHO ‘treat all approach’, birth dose hepatitis B vaccination, and the Sustainable Development Goals (SDG) of ending HIV/AIDS and hepatitis B by 2030 are just a few of the significant efforts made over the last two to three decades to make ART, HBV vaccination, and related interventions accessible and affordable for all people living with HIV/AIDS and HBV.39–41
Since antiretroviral therapy and HBV antivirals effectively suppress the HIV and HBV viral load and eventually reduce the likelihood of unfavourable birth outcomes among HIV and HBV-positive women to a certain extent, these therapies have, in turn, improved the health and welfare of women living with HIV and HBV.42,43 We do not fully understand why women with dual or triplex infections have a better prognosis in terms of zero MTCT rates. In addition to a better response to treatment, younger age, comprehensive knowledge of MTCT, a lower presentation stage, and male partner involvement in sustaining interventions for preventing MTCT are proposed as factors for these better prognosis.44,45
Strengths and limitations of the study
The study has several strengths. Our study’s primary strength is its evaluation of MTCT, seroconversion, and seroprevalence rates of triplex infections at the national population level across an observational period of up to 6 weeks after birth. The study’s external validity is reinforced by the fact that it was a multisite, nationwide, data-based investigation that enabled us to extrapolate the findings to state and federal levels. This study represents the most comprehensive investigation of dual and triplex infections among pregnant Nigerian women to date, yielding robust findings. Additionally, the inclusion of the husband’s results allowed for an assessment of potential household transmission dynamics. Certain limitations to our study are worthy of being discussed. This study has a restriction about institutional settings, whereby only pregnant women who underwent ANC follow-up were included in the analysis, notwithstanding its strengths. For several reasons, some expectant women in the community did not receive PMTCT services in addition to ANC follow-up. Regretfully, the study’s findings may be reported too little if pregnant women who did not receive PMTCT services or ANC follow-up were excluded.
Conclusion
Pregnant women in Nigeria showed comparatively low rates of seroprevalence and seroconversion for dual and triplex infections of HIV, HBV, and HCV, but low or nonexistent rates of MTCT. Considering this is necessary while offering maternal health services, especially prenatal care and delivery services, which are primarily for women who have dual and triplex infections. Current efforts at preventing increasing rates of HIV/HBV/HCV dual or triplex infections in Nigeria among pregnant populations should be sustained with further enhancement of policies and practices for prevention of MTCT.
Footnotes
Authors’ note
GUE presented the preliminary Result of this work as an oral presentation at the State General Meeting/Scientific Conference/4th Late Emeritus Prof FA Nwakor Memorial Lecture organised by Nigerian Medical Association Conference, Anambra State Branch held in Awka, Anambra State, Nigeria on 23 July 2022.
Acknowledgments
We are grateful to the six tertiary hospitals for allowing us to conduct this research. We want to thank data collectors and supervisors for their dedication and cooperation throughout the data collection process. We would also like to thank everyone who participated in the tool validation and language translation process. Finally, we would like to express our appreciation to the study participants without whom the study could not be realised.
ORCID iDs
Statements and declarations
Author contributions
GUE is the principal investigator. GUE and OCE conceived the study. Data assessment was performed by HAU, POF, OML, IIM, GOA, AR, CUO, MTC, RCC, NNI, CHN, SOK, CNO, SNC, CEU, ICO, AA, ROE, HCU, CHJ, SOI, BAA, UIA, UCO, EAE, OEI, ODO, POA, CPC, HIS, FEA, AIN, SAO, OSU, CCO, IKN, AAO, EOU, SIN, ICA, DCI, EPI, URE, NPO, COE, IAY, CCN, AEA, KCA, CGO, OKN, KCN, RKY, IAE, MOI, EAO, CCN, and JII. Calculations and data interpretation were performed by GUE, CHN, CGC, LIE, CSG, and MDE. Statistical analysis was performed by CGC and MDE. CGC, LIE, MDE, and CSG prepared tables and figures. The first draft of the paper was written by GUE and EPI, CUO EOU, AOU, JII, and COE critically revised the paper. All authors reviewed and edited the final draft. All authors critically reviewed the article, gave final approval of the version to be published, agreed on the journal to which the article has been submitted, and agreed to be accountable for all aspects of the work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the TETFund National Research Fund 2019 (Grant number TETFund/DR&D/CE/NRF/STI/33).
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
We can share the data if there are reasonable requests. Data can be shared by the corresponding author.
