Abstract
In endemic areas, hepatitis C virus (HCV)/hepatitis B virus (HBV) coinfection is common, and patients with coinfection have a higher risk of developing liver disease such as hepatocellular carcinoma, liver fibrosis and cirrhosis. In such cases, HCV predominates, and HBV replication is suppressed by HCV. HCV core proteins and interferons that are activated by HCV are responsible for the suppression of HBV. Immunosuppression is also seen in patients with HCV and HBV coinfections. A decrease in HCV-neutralizing antibody response and circulation of Th1-like Tfh cells is observed in patients with HCV and HBV coinfection. Both viruses interacted in the liver, and treatment of HCV/HBV coinfection is genotype-based and complex due to the interaction of both viruses. In HCV-dominant cases, direct-acting antiviral drugs and peg interferon plus ribavirin are used for the treatment, with continuous monitoring of AST and ALT. HBV-dominant cases are less common and are treated with peg interferon and nucleoside nucleotide analogues with monitoring of AST and ALT. The SVR rate in HCV-HBV coinfection is higher than that in monoinfection when treated with direct-acting antiviral drugs. But there is a risk of reactivation of HBV during and after therapy. The rate of reactivation is lower in patients treated with direct-acting antiviral drugs as compared to those treated with peg interferon plus ribavirin. Biomarkers of HBV such as HBcrAg, HBV DNA and HBVpg RNA are not effective in the prediction of HBV reactivation; only the hepatitis B surface antigen titre can be used as a biomarker for HBV reactivation. HCV can also be reactive, but this is found in very rare cases in which HBV is present and is treated first.
Introduction
Hepatitis C and B were shown to be the most dominant causes of hepatitis in several groups, including the public, children, healthcare workers and individuals with acute and chronic liver disorders. 1 The annual mortality rate for those suffering from HBV-related diseases is close to 0.9 million. The HCV affects about 71 million people globally, is one of the prominent causes of chronic hepatitis, and kills 0.4 million people every year. The hepatitis C and B viruses affect around 300 million people worldwide. 2 They typically coexist among patients in highly endemic areas or among those at great risk of infection, like those who inject drugs or homosexual men, due to shared means of epidemiological and transmission characteristics. The frequency of HCV-HBV dual infection ranges between 1% and 15% globally, depending on research populations and geographic areas. Hepatitis C and B viruses are the two most prevalent causes of liver disease in the whole globe. Individuals who have both HCV and HBV are more probable to develop cirrhosis, have more severe liver disease, and are at a higher risk of developing hepatocellular carcinoma. 3 HCV-HBV coinfection might occur through parenteral viral transmission. HBV and HCV have significantly different life cycles, even though they like to multiply in hepatocytes. Unlike HCV, which only replicates in the cytoplasm of hepatocytes, while a DNA virus called HBV replicates in the nucleus. 4 They may interact in coinfected cells and elicit unique viral expression and serologic patterns because they both include RNA replicate intermediates. HBV-HCV co infection is more difficult than HCV or HBV monoinfection. Coinfection occurs when two or more reproducing organisms coexist in the same host. People who have simultaneous HCV and HBV infections are more likely than those who have only had one infection of either virus to develop hepatocellular carcinoma and liver cirrhosis. 5 Patients who have hepatitis C and B need vigilant monitoring and potent antiviral drugs. Coinfection with HBV and HCV is not rare in people with cirrhosis or chronic hepatitis, as well as in patients with high-risk behaviours. The frequency of HCV/HBV coinfection in HBsAg-positive persons is estimated to be between 5% and 20% and between 2% and 10% in HCV-positive patients. 6 Furthermore, in 31.1–61.1% of people with chronic HCV hepatitis, the occurrence of anti-HBc in the absence of HBsAg is considered serological proof of past HBV exposure. Patients with coinfection may display a wide variety of HCV and HBV virological patterns, with the great majority of studies in Western populations revealing a preponderance of HCV replication over HBV replication. When HBV/HCV coinfected people are tracked over time, their virological patterns often reveal dynamic characteristics. 7 So HBsAg carriers who also had HCV infection had lower levels of HBV viraemia, HBsAg in their blood and HBsAg in their livers than HBV-infected people who only had HBV.
Global epidemiology of HCV-HBV coinfection
HCV and HBV coinfection is a significant public health concern worldwide. The prevalence of HCV-HBV coinfection varies across different regions. 8 High rates of coinfection are found in countries with a high prevalence of both HCV and HBV, particularly in certain parts of sub-Saharan Africa and East Asia. HCV-HBV coinfection can lead to more severe liver disease, increased risk of liver cirrhosis, hepatocellular carcinoma (liver cancer), and a higher mortality rate compared to monoinfection with either virus. 9 Injection drug use, unsafe medical practices (e.g. contaminated needles) and unprotected sexual activity are common risk factors for acquiring both HCV and HBV infections, increasing the likelihood of coinfection. Coinfection may be under diagnosed due to the absence of specific symptoms and limited access to screening. Testing for both HCV and HBV is essential to identify coinfected individuals. 10 Vaccines are available for preventing HBV infection, but no vaccine currently exists for HCV. Prevention strategies focus on harm reduction approaches, such as safe injection practices and promoting condom use. Antiviral therapies are available for both HCV and HBV, although treatment outcomes can vary depending on the individual’s condition. The prevalence of HCV-HBV coinfection in Sub-Saharan Africa region is estimated to range from 2% to 40%, with variations observed between countries and populations within countries. 11 Coinfection rates in East Asian countries, such as China and Vietnam, have been reported to range from 3% to 20%, again with variations depending on specific populations and geographic areas. 12 Data on HCV-HBV coinfection in Europe is limited. However, studies have indicated prevalence estimates ranging from 3% to 10% in certain countries, including France, Italy and Greece. Limited data is available for coinfection rates in North America. In the United States, studies have suggested prevalence estimates between 3% and 15% among certain populations, such as people who inject drugs. HCV-HBV coinfection prevalence in Latin American countries varies, with estimates ranging from 2% to 30% in different populations. 12 Countries like Brazil, Mexico and Argentina have reported higher prevalence rates. Although the latter limits may soon be lifted due to widespread patient access to low-cost generic drugs, there are still a number of challenges that threaten the WHO aim of eradicating HBV and HCV by 2030. The most crucial is that there are no screening programs for viral hepatitis in the general population throughout the world, making reliable estimates of the disease’s worldwide impact unattainable. 13 In reality, only 9% of the 240 million people globally with chronic HBV infections and 20% of the 70 million people with chronic hepatitis C have been diagnosed. A second hurdle is the low incidence of HBV and HCV cures in comparison to the annual number of new infections. 13 Only 10% of the infected population is receiving or has received interferon-based therapies in the HBV scenario, and treatment-related prevention-of-transmission strategies, which are regarded as strategically important in the fight against viral hepatitis, are only nearly fully implemented in the context of transmission from mother to child. 14 Unfortunately, damage reduction policies are not covered. In the HCV arena, antiviral have effectively cured 10% of the infected population globally, including individuals who previously received interferon-based regimens. However, there is a need to scale up anti-HCV therapy globally because, according to an epidemiological study conducted in 91 representative nations, in order to eliminate HCV by 2030, there must be 1.7 million cases of infection per year and a cumulative number of cured patients and end-stage HCV-related deaths that exceeds the number of newly infected by 7% each year. 14 Currently, a parallel decline in HCC incidence due to antiviral treatment is expected to occur at a somewhat moderate rate. This is because just a few nations are on track to eliminate HCV as a public health problem by 2030.
Natural course of chronic hepatitis B and C coinfection
Co-dominant, HBV-dominant or HCV-dominant patterns result from coinfection, none of which is replicative. Over time, the serologic profiles could change. In a coinfection where HCV predominates, HCV replicates vigorously while inhibiting HBV replication. 15 Some people with HCV dominance could also be hiding an HBV infection. Serum antibodies are commonly positive in occult HBV infection, as was previously mentioned. However, all serum indicators are negative in 20% of patients. Rare coinfections with HBV predominance have little to no active HBV or HCV replication. Both viruses sometimes have positive serologies but negative PCR results, suggesting that they are not actively replicating. Over time, this condition may progress to aggressive infections. 16 During a 10-year longitudinal study, Weigand et al. tracked the serologies of 85 coinfected people. Each serologic pattern was shown to be somewhat common (46.9% HCV dominant, 17.9% co-dominant, 21.1% neither replicative and 14.1% HBV dominant). This evidence obviously shows that HCV predominates, and it is consistent with the previously described tendency toward HBV suppression. The non-replicative group, on the other hand, seems to constitute a significant proportion. The lack of antiviral treatment control before enrolment might have been a weakness of this study. In a second longitudinal study, Raimondo et al. 17 found comparable frequencies for each serologic pattern: 22% codominant, 47% HCV dominant, 13% HBV dominant and 14% neither replicative. This research monitored the serologies every 2 months for a year and found that 32% of patients had substantial variations in their level of viral and viraemia dominance pattern. This study did not look at long-term changes that lasted longer than a year. The authors did not discuss the treatment significance of these variances. In contrast to this study, Weigand et al. did not follow serologies throughout preset time periods in their previous research. Therefore, comparing differences over a long period of time is difficult. Rodriguez-Inigo et al. examined liver biopsy samples from six individuals who had occult HBV and chronic HCV infections. 18 It was discovered that 11.9% of cells were only infected with HBV, 41.9% were infected with HCV and nearly half (47% were infected with both. 19 Individuals with coinfection may have a range of HCV and HBV virological patterns, although the bulk of research in Westerners reveals that HCV replication outnumbers HBV replication in most patients. When HCV/HBV coinfected people are followed over time, their virological profiles tend to be dynamic. HBV viraemia, blood HBsAg levels and intrahepatic HBsAg levels were all lower in HBsAg carriers with concomitant HCV infection compared to HBV monoinfected individuals. 20
When using direct-acting antiviral (DAA) medicine, patients with chronic hepatitis C who are also co-infected with HCV and HBV are at risk for HBV reactivation.

In coinfection, HCV neutralizing antibodies and Tfh cell distribution levels
Tfh cells are a specialized CD4+ T-cell lineage essential for T cell-dependent antibody manufacturing because they promote B cell maturation and selection of high-affinity antibodies in germinal centres. It was recently discovered that circulating Tfh (cTfh) cells, which share a phenotype and function with germinal centre Tfh cells, promote plasma cell and B-cell maturation as well as antibody production.
32
It was observed that HBV coinfection with HCV changed the location of cTfh cells and lowered HCV nAb responses when compared to HCV monoinfection. Like GC Tfh cells, cTfh cells contain morphological and functional characteristics that might elicit an immune response. In HCV monoinfection, there is a relationship between the circulating Tfh1 subset and the nAb responses to HCV.
33
This is consistent with previous research demonstrating that CXCR3-biased cTfh subsets are associated with high avidity and broad nAb responses following influenza vaccination and in HIV-1 controllers. HBV coinfection lowered the prevalence of the Tfh1 subgroup and hindered HCV nAb responses relative to HCV infection alone.
34
Furthermore, HBV coinfection changed cTfh cell differentiation toward Tfh2 cell development, which was shown to be adversely related to HCV nAbreactions in HBV/HCV coinfection. Tfh cell growth is caused by persistently elevated antigen levels during infection or immunization. During chronic HBV infection, significant amounts of soluble HBsAg build up in the blood, which may control how the host’s immune system reacts to infections.
35
This study raises the possibility that HBsAg is responsible for changing the makeup of cTfh cells, which influences the HCV nAb response. If HBsAg controls Tfh cell responses directly, further investigation is required. Although the evidence is mixed, it appears that HBV and HCV coinfection resulted in viral reciprocal interactions. The order of the infections, as well as the stage of the illness, may influence whether and how these viruses interact.
36
It was also observed that HBV replication predominates during HCV/HBV coinfection in this cohort, which may help explain why HCV/HBV coinfection varies from HCV monoinfection in the distribution of cTfh subsets. Therefore, a more thorough and accurate knowledge of the mechanisms underlying the decline in HCV antibody responses brought on by HBV coinfection would result from research on the function and development of HCV glycoprotein-specific B cells and Tfh cells in HCV/HBV coinfection.
37
Finally, study had founded that the Tfh1 subgroup is connected to HCV nAb responses. HCV nAb responses, which are related to serum HBsAg, are inhibited, and cTfh cell distribution is decreased because of HCV/HBV coinfection (Figure 1). An illustration of the three hypothesized methods by which HCV inhibits HBV replication HBV polymerase is first made inactive by a complex between the HCV core protein and it. Furthermore, miRNA 122 promotes HCV replication while suppressing HBV replication. Last but not least, HCV activates the IFN gene to create IFN, which inhibits HBV viral replication.
7
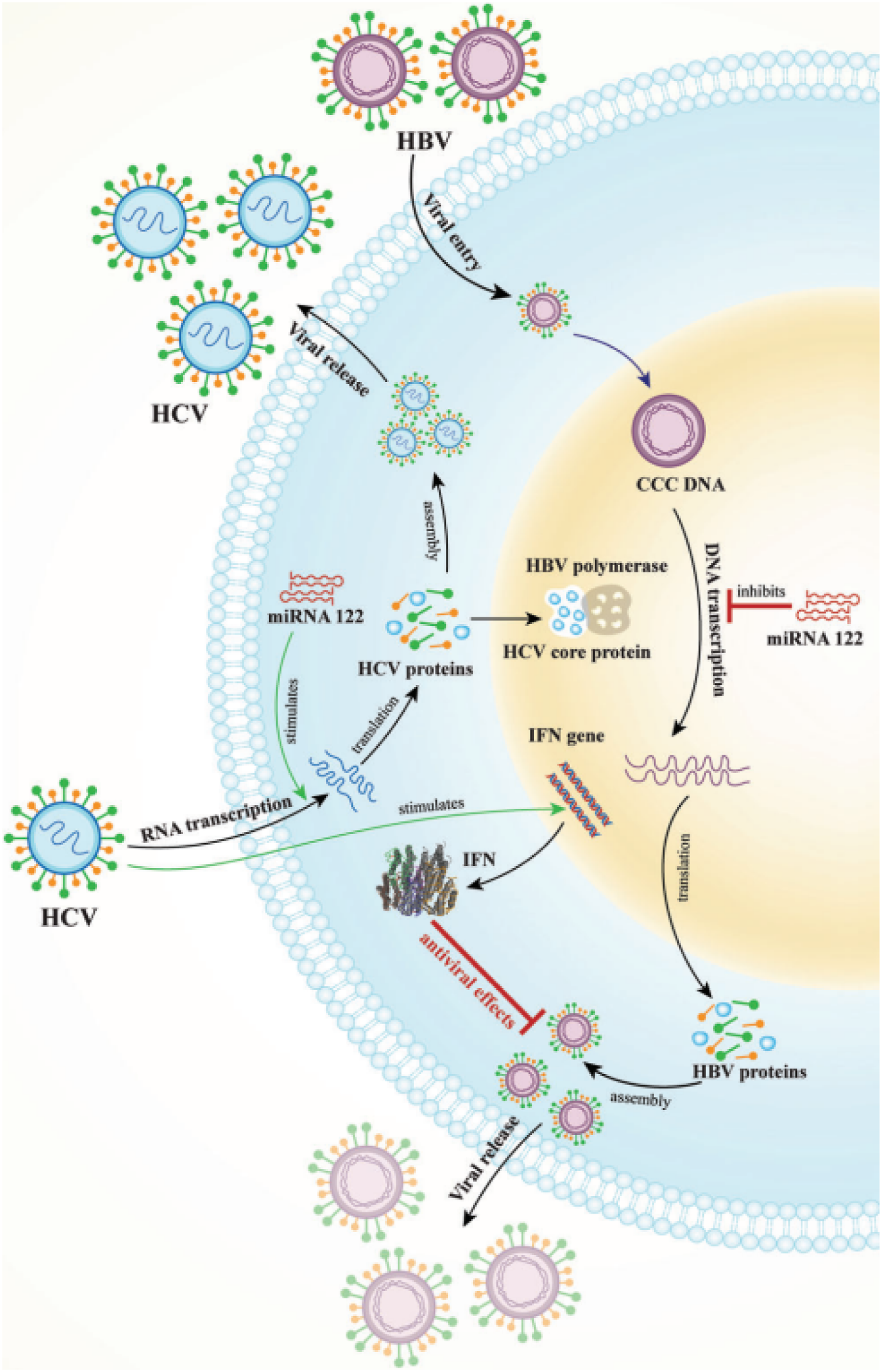
HBV/HCV coinfection with HBV and HCV predominance or superinfection
HCV and HBV infections may occur in two ways. Viral co-transmission is plausible since HCV and HBV share several transmission pathways, including intravenous drug use, blood transfusion and vertical transfer. 38 HCV-HBV coinfection, however, may happen when a patient already has a long-term infection with one virus and acquires the other. The most frequent reason for coinfections is superinfection, and HCV is more frequent than HBV superinfection. 2 One virus often predominates over the other in clinical situations. When both HCV and HBV are present in a cell, dominance occurs when one viral genome is suppressed by the other. The replication of the non-dominant virus is hindered, while the replication of the dominant virus is faster. When there is codominance, HBV and HCV reproduce roughly equally. 15 Virus serologies differ depending on whether the coinfection is contemporaneous or superinfectious. Because anti-HCV and HCV RNA are usually present in serum, HCV infection is almost always visible. HBV infection, on the other hand, might be either visible or hidden. 39
Either HCV or HBV can predominate. The timing of each infection seems to be critical for one virus’s supremacy over another. While the majority of patients from Europe and the United States had HBV superinfection at CHC, those in places where HBV infection is prevalent, such as Asia-Pacific nations, may have been born with HBV infection and developed HCV superinfection later in life. Most patients, however, have HBV replication suppression. Because HBV replication was diminished, the majority of HCV/HBV coinfected individuals (56%) exhibited HCV dominance.
40
The bulk of these infections in Greece, like in most countries with moderate endemicity for HBV infection, were caused by horizontal transmission during early childhood; therefore, people were infected with HBV for many years before getting HCV.
41
According to Chen et al.,
42
HBV dominance comes from a smaller section of hepatocytes that are uninfected by HBV and so ‘ready’ for HCV superinfection in people who acquired HCV infection after HBV infection. Coinfected individuals must have extensive serological and virological testing before starting antiviral therapy. IFN-based therapy has been shown to be secure and capable of achieving SVR rates that are equivalent to those anticipated with HCV monoinfection, hence reducing liver-related side effects as shown in Figure 2.43,44 There were 25 people in the group who had received treatment. Most patients (63.9%) received IFN-based treatments.
45
A long-term follow-up investigation on HBV superinfection in CHC patients was carried out in Italy by Sagnelli et al.
46
During acute hepatitis B, all patients had undetectable HCV RNA, even though more than 90% of patients eradicated HBsAg. Following HBV superinfection, several case studies demonstrated that HBV had a dominant role in HCV clearance. Because the T-cell response is expected to target HBV, eliminating HCV is favourable in HBV superinfection. As a result, it is unknown what effect NUC treatment had on this patient’s HCV and HBV clearance.
47
Furthermore, data show that acute HBV superinfection promotes HCV and HBsAg clearance; in eight untreated HBV superinfection patients, it established a 75% HBsAg clearance rate and a 50% spontaneous HCV eradication rate. Research found that the HCV core protein inhibits HBV replication, and that HCV may also directly affect HBV production.
48
In the presence of HCV structural genes, these researchers reported a milder, up to 20-fold decrease in HBV particle production and a moderate, 2–4-fold decrease in HBV antigen and HBV mRNA expression. When the researchers compared these data to the HCV core protein in the nucleus, they found that when the HCV core protein was cut down to 122 N-terminal amino acids,
49
it didn’t lose any of its suppressive power. Their results hint at a distinct but minor overall effect on HBV transcription as well as an additional, more substantial influence on HBV morphogenesis or secretion. The interferon lamda 3-4 (IFNL) gene, IFN 1, and two loci on chromosome 19q13 are represented, and these regions include important (SNPs) single nucleotide polymorphisms implicated in the elimination of HCV.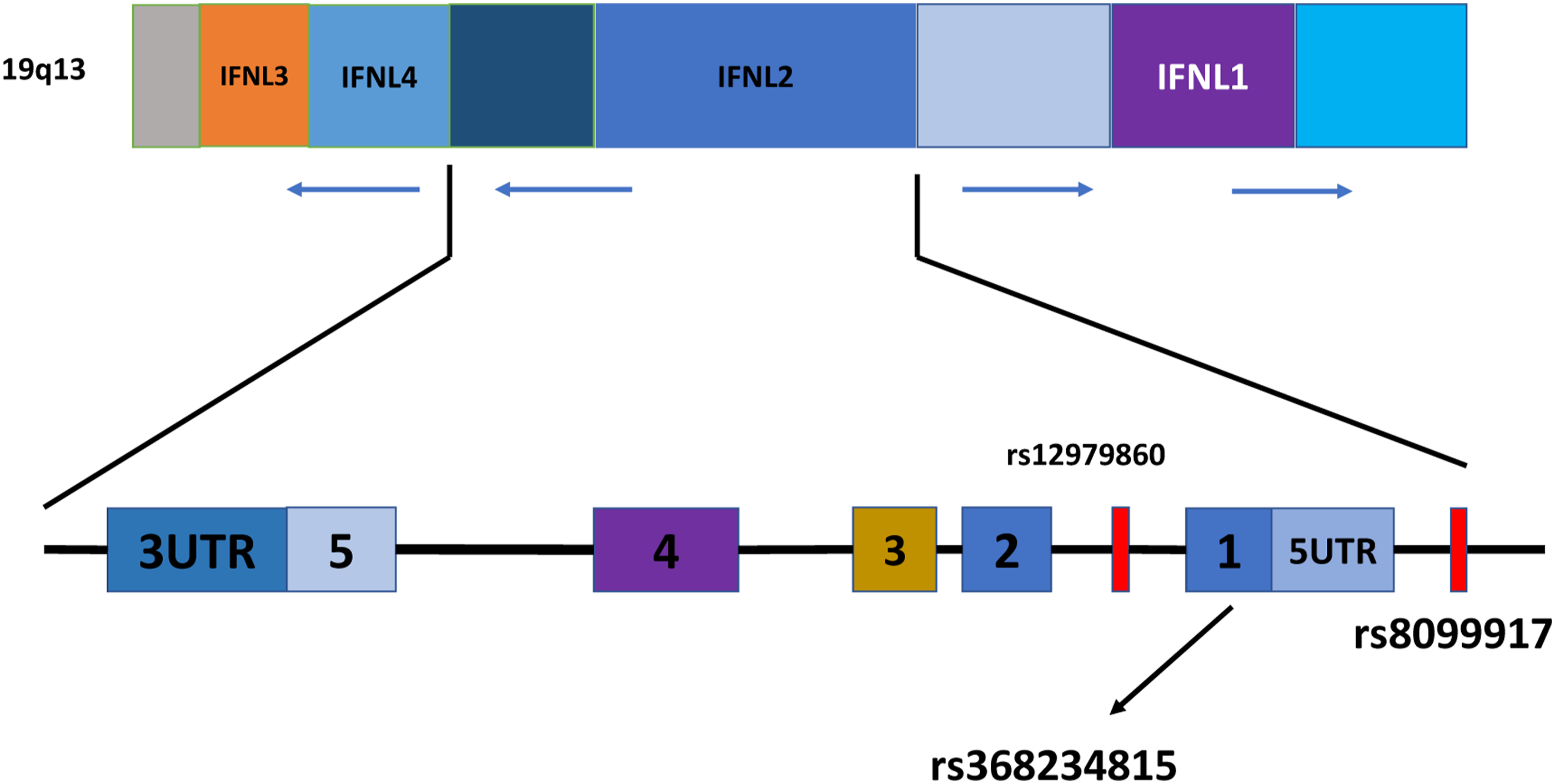
Coinfection and viral interactions
According to clinical research, people with HCV infection who also have occult HBV infection have more liver damage and a greater risk of developing HCC and cirrhosis. Furthermore, it is possible that HCV may obstruct HBV replication to the point where it is hard to detect whether an infection is present. 50 In contrast, in vitro studies show that the HCV ‘core’ protein has an important inhibitory effect on HBV replication. All forms of immunosuppression may trigger HBV reactivation and the reappearance of the overt serologic pattern because HBV DNA remains in the liver of patients with occult HBV infection, both as a component of the free episome and host genome, because it can still generate proteins and complete virions. 51 Although the hepatitis C and B viruses are both hepatotropic, meaning that their primary site of reproduction is the liver, their immune responses to both acute and chronic infection differ dramatically. If both viruses are present in the same liver, the situation might get much worse. A recent study looked at the distribution of cTfh (T follicular helper cells) and the generation of HCV-neutralizing antibodies in relation to co-existing HBV. 52 Individuals with HCV monoinfection (n = 84) and HCV/HBV coinfection (n = 77) participated in the study. In the HCV/HBV coinfection group, the frequency of circulating Th1-like Tfh cells reduced and HCV neutralizing antibody responses were significantly lower than in the HCV monoinfection group. 53 HCV coinfection reduced HBV replication in coinfected cell cultures and humanized mice. In vitro inhibition of interferon signalling diminished HBV suppression. After being reduced in vivo by HCV superinfection, HBV viraemia reappeared after the eradication of HCV by DAA therapy, which was also associated with a diminished hepatic interferon response. Overall, findings hint at a reduced hepatic interferon response after HCV clearance as the reason for HBV reactivation. 37 Another study looked at potential HBV reactivation routes in individuals with HCV/HBV coinfection after HCV was eliminated with DAA therapy. Murai et al. 54 investigated HCV and HBV interference in primary human hepatocytes and the activation of the RIG-I-like helicase pathway in HBV/HCV coinfection. They discovered that HCV infection reduced HBV replication and activated the RLH pathway. DAA-mediated HCV eradication increased HBV replication while decreasing the RLH pathway in mice. These findings helped to explain why HBV replication increased after HCV elimination, most likely due to RLH pathway down-regulation. To summarize, recent evidence indicates that HCV and HBV interact both in vivo and in vitro.
The CD81/NTCP/Mir122/HepG2 cell line supports HBV/HCV infection and replication as well as a suitable response to IFN therapy, according to research. In the coinfection model, all evaluated DAA drugs – including LDV, DCV, IFN, SOF and therapy – significantly elevated HBV markers in comparison to the untreated group. The HBV monoinfection study group had lower levels of secreted HBsAg and HBeAg than the control group, except for HBeAg in the DCV treatment. 55 According to the findings of luciferase experiment, DCV may increase HBV transcription. Furthermore, SOF inhibited HBV replication in HBV monoinfection, a mechanism that requires further investigation. It is yet unknown why HBV levels increase in people who also have HCV when they get DAA medication. Individuals with HBV/HCV coinfection showed a lower hepatic IFN response following DAA treatment, according to Cheng et al. 53 They hypothesized that the innate immune response played a role in the interaction of HBV and HCV. According to their findings, IFN efficiently reduces HBV replication in both HCV/HBV coinfection and HBV monoinfection, although in dosage studies, HBV may rebound more during coinfection than during HBV monoinfection. We observed that when HBV and HCV were coinfected, ISGs, including ISG15 and TRIM5, were significantly elevated. These findings imply that the interaction between HCV and HBV involves innate immunity. The dominance of one virus over the other seems to be time dependent. Although most patients from the United States and Europe had HBV superinfection at CHC, those in high-risk locations, such as Asia-Pacific nations, may have been born with HBV infection, with HCV subsequently arising as a superinfection. However, most individuals have HBV replication suppression. These findings indicate that HBV replication was suppressed in most of our HCV/HBV coinfected individuals (56%), resulting in HCV dominance. However, a considerable proportion of our patients had active HCV/HBV infection (25%) or acute HBV superinfection (44%), showing that HBV played an important role in our group. Greece has been linked with mild HBV infection endemicity; nonetheless, during the previous 20 years, a significant number of immigrants who tested positive for HBsAg or HBeAg altering the epidemiology of HBV infection.
The paediatric population and HCV/HBV coinfection
Although the preceding studies focused on coinfected adults, coinfected children have a comparable tendency toward more severe liver disease. A study of 10 children aged 5–17 with a coinfection and chronic hepatitis found that the coinfected group had higher necroinflammation. A more substantial difference was seen when comparing children with HCV monoinfection to those with HBV monoinfection. 56 Despite being helpful for understanding the onset and course of liver disease in kids with both disorders, it is challenging to interpret this information in terms of potential therapy implications. The FDA has granted authorization for the use of DAAs (direct-acting antivirals), more particularly sofosbuvir/ledipasvir and ribavirin (RBV)/sofosbuvir, to treat HCV in teen-agers between the ages of 12 and 17. However, younger children cannot take these medications. The severity of these disorders in children has grown, drawing attention to a possible advantage of expanding DAA use. 57 However, given current treatment guidelines, research on children under the age of 12 and those aged 12–17 should be undertaken separately since their impacts on therapy are diverse. Licensed HBV medications, like HCV medications, have age and child restrictions, with children having stricter restrictions. The use of defovir and tenofovir is permitted in children less than 12 years of age, lamivudine and entecavir is permitted in patients older than 2 years and PegIFN (Pegylated Interferon) is permitted in individuals older than 12 months. 58 Children with HCV and HBV coinfections will very certainly be treated with the previously indicated HCV and HBV therapies. There are no specific recommendations for the care of children who are also affected.
Treatment
Peg interferon alfa and RBV were shown to eradicate HCV in patients with HCV/HBV coinfection. Patients with decompensated cirrhosis of the liver or other problems may not be able to follow this plan. 45 Prior to the discovery of DAA (direct-acting antiviral)-based anti-HCV drugs, it was difficult to characterize the optimal treatment strategies for coinfected patients who were contraindicated for IFN-based therapy. 59 Not only is DAA-based therapy safer than IFN-based therapy, but it is also much more effective, improving the rate of HCV clearance in individuals with monoinfection. 60 For patients with coinfections, DAA-based therapy offers the potential to bridge the therapeutic gap. A recent Taiwanese multicenter study discovered that the sustained virologic response (SVR) rate for coinfected patients with HCV genotype 1 or 2 infection who received 12 weeks of ledipasvir/sofosbuvir therapy was 100%. 61 Patients who have had pegIFN or DAA-based HCV treatment may experience HBV reactivation. Study findings showed that throughout the 12-week DAA medication phase and the 108-week post-treatment monitoring period, 73% of patients had HBV reactivation episodes. 12% of those who had HBV reactivation also needed anti-HBV medication due to clinical hepatitis activity. The risk of HBV reactivation was greatest during therapy. 62 The mechanism of HBV reactivation after HCV treatment is still a fascinating unsolved mystery. Most recent study found that inhibiting IFN signalling in vitro lowered HBV suppression. 63 Clinically, HBV viraemia returned following HCV clearance with DAA treatment and was related to a decreased hepatic interferon response after being initially suppressed by HCV superinfection. 64 Patients with HCV/HBV coinfection who had low pre-treatment HBsAg titres had a significantly greater chance of clearing the HBsAg molecule within a year of starting DAA therapy; patients with higher pre-treatment HBsAg titres were more likely to experience HBV reactivation and, in some cases, liver failure. All cirrhotic patients must get anti-HBV NUC prophylaxis, and those with higher baseline HBsAg levels are advised to do so. 53
Treatment with RBV and PegIFN
The efficiency and duration of the HCV SVR obtained with RBV and pegIFN alfa treatment were presumably good and unaffected by coinfection with HBV. 65 Patients with HCV/HBV coinfection may benefit from pegIFN-based treatment to both treat their HBV infection and manage their chronic HCV infection. 30 The yearly rate of HBsAg seroclearance over the course of a 5-year follow-up after therapy was 5.5%. HBsAg seroclearance was a favourable result for 36% of the patients in different research that examined the outcomes of 192 HBV/HCV coinfected patients following anti-HCV treatment. 66 The likelihood was 5.7 out of 100. Furthermore, the result was independently predicted by a pre-treatment HBV DNA level of 299.9 IU/mL. We believe that therapy will ultimately lower the long-term risk of HCC development and liver-related death in individuals with HCV and HBV coinfection. The researchers investigated national databases for Taiwan and carried out a case-control investigation, finding that coinfected individuals receiving pegIFN and RBV therapy had a lower probability of developing HCC and passing away from liver disease. 67 This is supported by data from prospective research comparing the use of PegIFN and RBV in the treatment of HCV coinfection versus HCV monoinfection. Furthermore, this research found that 25% of individuals with concurrent infections continued to seroclear the hepatitis B surface antigen following therapy. 68 Liu et al. discovered that after PegIFN/RBV treatment, HCV sustained virological response was similar in coinfected patients (73% vs. 77%). In comparison to the previous research, only 11.2% of patients with coinfection had eradicated the hepatitis B surface antigen. These two trials together provided evidence for the notion that RBV/PegIFN is equally effective for treating HCV and only slightly effective for treating HBV in those who are coinfected. 45 According to different research, PegIFN-based drugs may have a negligible antiviral impact on HBV and HCV. Anti-HCV drugs might make the effects of the infection better. In addition, treatment reduced the risk of HCC by 34%. The results did not address the potential for HBV reactivation. The mortality decrease seen in therapy groups may be partially or completely offset by the higher mortality brought on by reactivation. 43 Treatment of HCV-GT1/HBV dual-infected patients who had previously failed to respond to PEG-IFN or RBV with boceprevir proved successful, particularly in cases of non-cirrhotic recurrence. Although safety is still a concern, effectiveness was only adequate in previous null responders and/or cirrhotic individuals.
Treatment with DAA
The DAA treatment, which has established to be a simple, timesaving, highly effective and well-tolerated HCV treatment option, raises two issues. First, if DAA-based therapy outperforms pegIFN with RBV in eradicating HCV RNA; second, whether coinfection with HBV affects the efficacy of DAA-based therapy in the treatment of HCV patients. To discover answers to these questions, a multi-centre study was conducted in Taiwan on patients with HBV and HCV coinfections. There were 111 people in all, with 61% having genotype 1 HCV, 39% having genotype 2 and 16% having compensated cirrhosis. HCV SVR was obtained in all 108 individuals. The SVR remained steady for 108 weeks after the DAA therapy was completed. 62 Individuals with HCV and HBV coinfections reacted extremely well to a 12-week ledipasvir/sofosbuvir DAA combination, according to reported data. A concurrent HBV infection would have no effect on the therapeutic response. In contrast to the response to HCV treatment, the implications of a co-existing HBV infection remained unclear. 69 DAA therapy plus NUC prophylaxis for HBV/HCV coinfected patients with cirrhosis, even HBV DNA undetectable at baseline to prevent potential risk of HBV flare-related hepatic failure and mortality. 70 Four of the six patients had cirrhosis, and three had liver failure because of reactivation. Their findings clearly demonstrated that, in coinfected individuals, anti-HBV NUC prophylaxis should be considered prior to initiating HCV DAA. 71 The 45 coinfected subjects in this Italian study exhibited an HCV continued virological response of 93.9%, even though six of them showed symptoms of HBV reactivation, necessitating further nucleoside analogue treatment. However, it also provided a variety of genotype-based DAA regimens. Inferences are thus restricted to DAAs as a class rather than drugs or therapeutic modalities. DAAs have little impact on HBV but have a sustained virological response of more than 90% for HCV. 72
Current treatment for HCV/HBV coinfection
It is crucial to determine the dominant virus, treat that infection and keep a watch out for the reactivation of the non-dominant virus before creating a treatment strategy for persons with HCV/HBV coinfection. For this reason, the replication status of both viruses should be extensively assessed in individuals with HCV and HBV coinfection. 73 Testing should include HBs antibodies and antigen, HBe antibodies and antigen, HCV RNA levels and HBV DNA levels. Testing for anti-HBc antibodies is necessary to rule out a prior HBV infection. International and national hepatology organizations advise treating HCV/HBV coinfected people with HCV dominance as monoinfected patients since there is no appreciable difference in the rate of SVR between HCV/HBV monoinfected and HCV coinfected individuals. 74 Blood ALT levels and hepatitis B viral indicators must be closely watched both during and after DAA therapy due to the possibility of HBV reactivation after HCV eradication. There are some discrepancies in the therapy of HBV-r amongst the various hepatologic organizational recommendations for the care of HCV/HBV coinfected individuals, even though they all provide similar indications for antiviral medicine. 75
The EASL recommends that coinfected individuals who meet the conditions for HBV treatment get antiviral medication in the form of a nucleoside or nucleotide analogue (NA) in accordance with accepted guidelines. Those who test positive for HBsAg but do not satisfy the criteria for HBV treatment should receive NA prophylaxis for a minimum of 12 weeks after the initiation of DAA medication. If HBV medication is stopped, patients should be monitored periodically. Any individual with a history of HBV infection should undergo routine ALT testing as shown in Figure 3. If ALT activity rises, HBs antigen and HBV DNA should be tested; if either is detected, therapy with NA should commence. During and after DAA therapy, careful monitoring for HBV reactivation is required; however, the frequency and duration of post-therapy surveillance are not advised.
76
Before beginning DAA medication, HBsAg-positive patients should have their HBV DNA levels checked. Prophylactic antiviral medication should be started in HBs antigen-positive patients who do not meet the requirements for HBV treatment and have low or undetectable HBV DNA levels. Twelve weeks should pass following the conclusion of DAA therapy before stopping prophylaxis. The Asian Pacific Association for the Study of the Liver recommends routine HBsAg testing before starting treatment in those with persistent HCV infection.
77
People with HBs who have severe liver fibrosis, cirrhosis or a history of HCC and who don’t meet the requirements for HBV therapy are told to take NA medication. For patients without these conditions, close monitoring is recommended throughout DAA therapy and for 24 weeks after treatment is finished. Since there is no way to predict how long the prophylaxis will last, prophylactic NA medication should be started prior to beginning DAA treatment for the safety of HBs antigen-positive patients who are receiving it.
78
Routine baseline HBV DNA testing or the use of NA as an HBV reactivation preventative in HBs-negative coinfected individuals who have previously had HBV infection is not recommended. If HBV-r is found, NA should be started, but only if HBsAg, HCV RNA and HBV DNA liver function tests reveal abnormalities during and after DAA therapy. Few national recommendations exist for managing HBV reactivation. For HBsAg-positive patients, prophylactic NA medication is advised up to week 12 after the conclusion of DAA therapy. Polish or Korean regulations make no mention of this time frame.
79
People who have previously been infected with HBV should have their ALT activity constantly monitored and tested for HBsAg and HBV DNA if it rises. The proper anti-HBV therapy with NA must be started as soon as HBV-r is confirmed. An approach to managing HBV-HCV coinfection.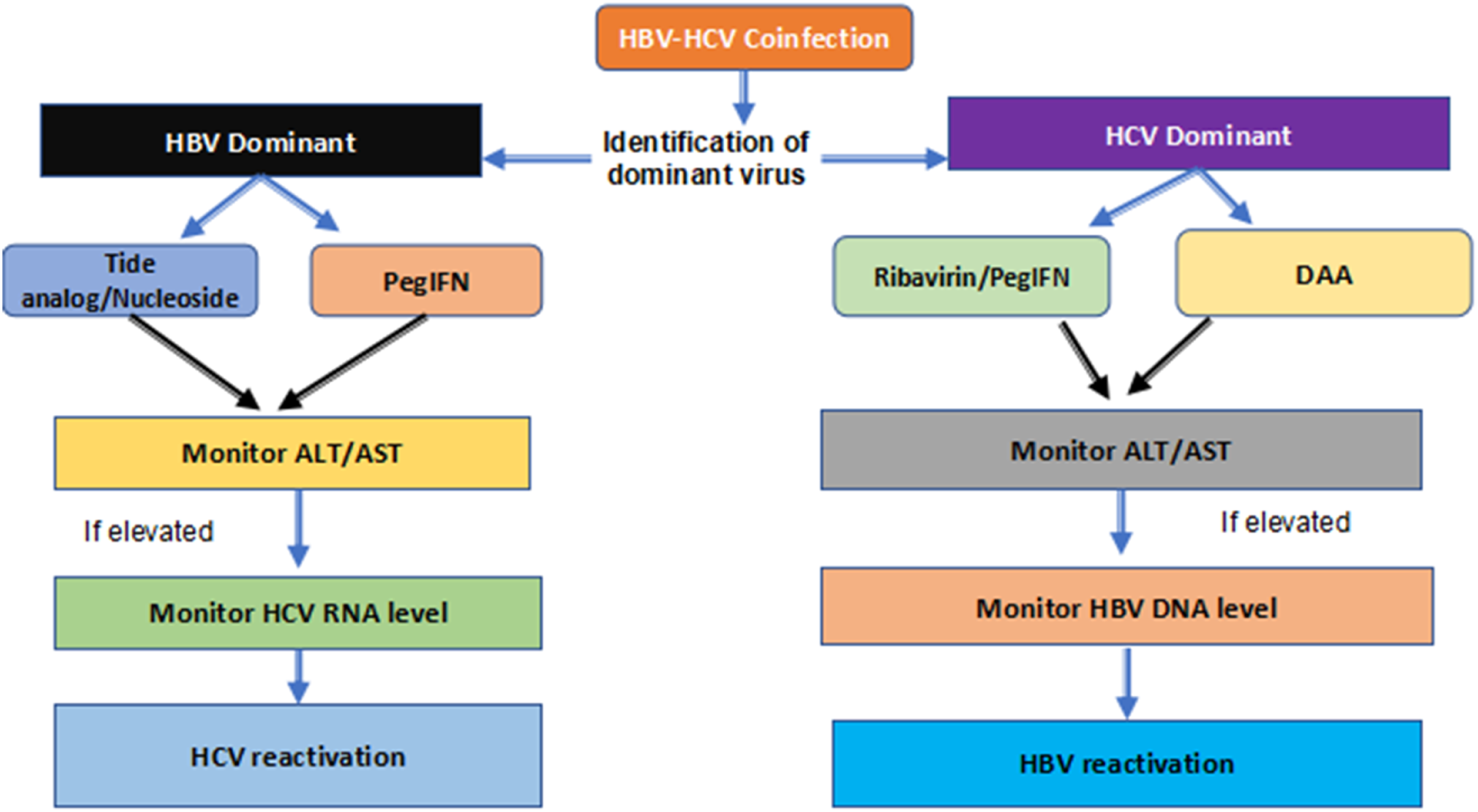
SVR rate in HCV/HBV coinfected individuals treated with DAA
Research comparing the prevalence of HCC in coinfections and monoinfections.

Threat of additional liver diseases
According to most current hospital-based cohort research, the development of liver disease is adversely affected by HCV/HBV coinfection. In comparison to 111 people with HBV monoinfection during a 10-year period, those with coinfections had a hazard ratio of 3.6 for HCC and 2.5 for cirrhosis. Several studies have used the fibrosis 4 parameters index to figure out how people with chronic hepatitis C are doing and what stage their liver fibrosis is at. A research study examined the efficacy of FIB-4 in 153 non-cirrhotic patients with chronic HBV/HCV coinfection; 56 patients got pegIFN/RBV therapy, while 96 patients received no treatment. 88 The FIB-4 score and the frequency of HCC and LC were also factors that the researchers intended to investigate. 29 In the treated groups, a high baseline FIB-4 index was associated with an increased risk of developing LC and HCC; the FIB-4 index fell only in the treated group that had SVR. According to these findings, coinfected people with higher pre-treatment FIB-4 levels were more likely to suffer liver damage and needed rigorous monitoring following HCV therapy. Butt A and his coworkers also investigated the probable function of FIB-4. 80 In 115 HBV/HCV coinfected people treated with DAA, the FIB-4 score-measured liver fibrosis was worse and the SVR rate was lower. Overt HBV infection has a negative influence on the clinical outcomes of chronic HCV infection, as evidenced by positive blood HBsAg. Research showed that among patients with HCV and HBV coinfection, effective HCV eradication results in long-term benefits, with a reduction in risk of 50.2% in serious liver-related illnesses, 52% in the development of HCC and 71% in instances of hepatic decompensation. Furthermore, compared to patients with HCV/HBV coinfection, or HCV monoinfection those who obtained HCV SVR with IFN-based treatment had a decreased risk of significant liver consequences. Additionally, it was shown that patients with HBV and HCV coinfections were more likely to have serious liver-related problems than HCV infected people who did not respond to IFN-based treatment. 89 According to the study’s findings, the probability of liver-related side effects after interferon-based treatment did not vary between those with HCV/HBV coinfection and those with HCV monoinfection, either with or without HCV SVR. The use of anti-HBV medications for patients with hepatitis B infection, the fact that interferon is helpful against both HCV and HBV, and the fact that most HCV/HBV coinfected individuals had inactive HBV infections could all be factors contributing to the lack of difference. 90 Patients with a dormant HBV infection or those who have HBV that has been suppressed have a very low chance of developing serious liver problems.
HBV reactivation after IFN and RBV therapy
A concentration of HBV DNA higher than 100 IU/mL or a fresh appearance of HBV DNA at a concentration higher than two logs higher than the baseline level is considered an HBV reactivation. Depending on the criteria employed, HBV reactivation may occur in more than 50% of HBsAg-positive patients getting DAA treatment for CHC. HBV reactivation, on the other hand, is extremely rare in people who have had their HBV hepatitis cured. 17 However, according to recently released data, HBV reactivation were common in CHB and CHC patients, but there were no clinical flare-ups, and none of the patients needed HBV antiviral treatment. It seems that HBV reactivation may happen significantly sooner following DAA treatment than it would after IFN-based therapy. 19 The US Food and Drug Administration subsequently issued a notice on the possibility of HBV reactivation and suggested that all patients receiving DDA therapy undergo testing for HBV and be closely monitored. If active CHB or ‘occult’ HBV infection is detected, it is recommended to provide concomitant NUC treatment. Patients taking DAAs who are anti-HBc-positive or HBsAg negative should be monitored and tested for HBV reactivation only if ALT levels are high, according to more recent EASL recommendations for the management of hepatitis B. 20
Studies have shown that clinical reactivation symptoms are not necessarily correlated with a significant increase in HBV replication. A retrospective review of Department of Veterans Affairs data found that eight out of thirty coinfected veterans treated with DAAs had HBV reactivation, as indicated by an increase in HBV DNA levels of more than 1000 IU/mL relative to their pre-treatment baseline. Six of the eight veterans whose HBV was reactivated had higher levels of alanine aminotransferase, which is a sign of a clinical HBV flare. 48 According to a random study, the percentage of reactivation was the same (26.6%) among 139 individuals who had the same symptoms. However, only three individuals (2.2%) were found to have severe flare symptoms. HBV in the serum might be a factor in post-treatment reactivation. In a trial of 45 coinfected patients taking DAAs, the probability of reactivation was much lower in covert HBV infection than in overt infection. The total reactivation rate in this research was 2.2%. Some theories propose that immune escape mutations facilitate HBV reactivation. No reactivating controls must be used to identify the specific viral alterations related to HBV reactivation because of the intrinsic diversity of HBV in persons and groups. 72
HBV reactivation after DAA therapy
Those taking DAAs experienced HBV reactivation more rapidly than those getting IFN-based treatments, which is important to note. Additionally, due to HBV reactivation, individuals who got DAA rather than IFN-based treatment had a greater chance of developing hepatitis. 91 The re-emergence of serum HBV DNA in HBV/HCV coinfected patients in whom HBV DNA was undetectable at baseline has been linked to the increase in ‘replicative space’ for HBV in hepatocytes after HCV clearance; however, this relationship may also be influenced by the effects of IFN on the mechanism of innate antiviral immunity. DAAs seem to have no direct impact on HBV replication or the innate antiviral immune response, unlike IFN, despite their unrivalled speed and potency in inhibiting HCV replication. 92 In the setting of chronic HCV/HBV coinfection, these innovative regimens provide a possibility to evaluate viral interference as a replication regulator in vivo. DAAs’ ability to treat individuals with severe liver disease, who are more likely to have an undetected HBV infection, exacerbates the problem. 93 Recent anecdotal data shows that people with chronic HCV hepatitis and overt or occult HBV coinfection may experience a resurgence of serum HBV DNA after HCV suppression or clearance during the early stages of DAA treatment. 40 Scientist observed HBV virological markers for 108 weeks following the end of therapy to better understand the temporal profile and risk of HBV reactivation. Of the 111 individuals, clinical reactivation occurred in 10 cases. It should be kept in mind that clinical reactivation might happen later; four participants had it between trial weeks 12 and 48. According to our research, most HCV/HBV coinfected patients who take HCV DAAs may experience HBV reactivation. Only a tiny percentage of those with HBV virologic reactivation had symptoms and needed to be treated for the virus. It should be mentioned that, up to 3 months after the end of medication, clinical reactivation may take place. In HBsAg-positive patients using DAAs, other studies in addition to ours have shown a risk of HBV reactivation ranging from 25% to 87.5%. These results conclusively show that coinfected individuals have a greater risk of HBV reactivation. As shown in clinical study, routine blood HBV DNA monitoring and prompt anti-HBV NUC treatment in response to HBV reactivation may reduce the prevalence of severe hepatitis activity. 94
Increased risk for HBV reactivation
The chance of HBV recurrence increases to 75% when the four individuals whose HBV was previously stably suppressed by NUC therapy are excluded. In the absence of the immune-modulating effects of DAAs, the same innate and acquired immune response mechanisms that kept HBV in balance before HCV eradication are likely to be able to handle the relatively modest increase in HBV replicative drive that occurs after HCV eradication without causing a cytolytic flare. Due to the forced introduction of NUCs in response to a reported increase in HBV DNA, it is difficult to tell whether the reactivation would have been clinically mild and self-contained or if it would have led to a significant hepatitis flare. 95 Even in those with ‘resolved’ HBV infection, using immunosuppressive medications and/or chemotherapy increases the chance of HBV reactivation. 96 In a meta-analysis of patients with cured HBV infection who were getting chemotherapy for haematologic malignancies, reactivation happened in 14% of those with anti-HBc antibodies and 5% of those with both anti-HBc and anti-Hepatitis B surface antigen (anti-HBs) antibodies. 97 In 24.8% of patients with HBV-related HCC who had radiation, HBV reactivation took place. According to a thorough study of anticancer therapies in patients, most medicines used to treat solid tumours may cause HBV reactivation; as a result, HBV testing should be done before to starting pharmacological therapy. 98 Additionally, patients receiving treatment for autoimmune diseases or transplant recipients have had HBV reactivation. Similar HBV reactivation cases have been testified in HIV-positive people starting or stopping antiretroviral therapy.
Lesser risk of HBV reactivation
A method for finding HBV DNA in blood that is very sensitive showed that three out of thirty-seven anti-HBc-positive people had signs of low-level HBV replication during or after taking DAA. The probability of HBV reactivation was lower in the other group, which comprised individuals who had already been exposed to the virus and who could also be occult carriers. 99 The rise in HBV DNA was so low that it could not be detected by a typical commercial test, it self-remitted without the need for NUCs; and it was not linked to an ALT flare or a diminished efficacy of DAAs on HCV, even though this discovery supports the hypothesis that HBV and HCV infections interact. Even though two out of three HBsAg negative, anti-HBc positive patients had extreme HBV infection reactivation after DAAs, none of the patients in this group showed concurrent immunosuppressive symptoms. Among the risk variables were HIV infection and a prior Burkitt’s lymphoma that was maybe treated with monoclonal antibodies. It is possible to conclude that when administering DAAs to HCV patients who are also immune compromised, such as those who have had organ transplants or who have haematological conditions.
Prevention of HBV reactivation
Up until recently, the mainstay of HBV reactivation prevention has been preventive antiviral medication. Preoperative antiviral treatment, however, may lower the incidence of reactivation after rehepatectomy in patients with HBV-related HCC. 100 Like this, HBV reactivation in patients with HCC who had hepatic resection but had undetectable HBV DNA in their blood was linked to the absence of antiviral therapy. Lamivudine substantially decreased chemotherapy-related HBV flares in breast cancer patients, according to a new meta-analysis. 101 Surprisingly, lamivudine with adefovir dipivoxil was more effective at preventing reactivation in Chinese patients than lamivudine alone. Tenofovir and entecavir may be the best medications for avoiding HBV reactivation in chemotherapy patients, according to a network meta-analysis. Therefore, it is crucial to identify any chronic or occult HBV in HBV/HCV coinfected individuals who are thinking about receiving DAA treatment. 102 Anti-HBc is not a good proxy for occult HBV infection because occult infections are often seronegative, especially in people with HIV. The most effective antiviral treatment for HBV reactivation significantly reduces viral replication while clearly preventing the development of drug resistance. Although tenofovir may seem like the best option, it is not always accessible, especially in communities with few resources. 103
Biomarkers for HBV reactivations
Biomarkers for HBV reactivation refers to specific laboratory markers that can indicate the reactivation of HBV in individuals who have a history of HBV infection. Reactivation occurs when the virus becomes active again after a period of inactivity or low viral replication. Biomarkers commonly used to monitor HBV reactivation include: HBsAg is a protein on the surface of the HBV. Its presence indicates active HBV infection. 104 An increase or reappearance of HBsAg levels may suggest HBV reactivation. HBeAg is another viral protein that indicates active viral replication. The loss or reappearance of HBeAg, along with changes in other serological markers, can indicate HBV reactivation. Measuring HBV DNA levels in the blood provides information about viral replication. 105 An increase in HBV DNA levels, particularly after a period of undetectable or low levels, can be indicative of reactivation. ALT is an enzyme released into the bloodstream when liver cells are damaged or destroyed. Elevated ALT levels can suggest ongoing liver inflammation, which may be associated with HBV reactivation. 106 It’s important to note that the interpretation of these biomarkers should consider the individual’s medical history, the specific context of their treatment (e.g. immunosuppressive therapy) and the established cutoffs or thresholds for reactivation defined by clinical guidelines. There were no hepatitis flare-ups in HBV/HCV coinfected individuals who did not take NA medication; however, there was a risk of HBVr (22.5%) following DAA treatment. The P12 blood levels of HBcrAg and HBV DNA were higher in patients who did not get NA therapy than in those who did. From baseline to P12, HBsAg levels in individuals with HBVr were higher than those in those without HBVr. A baseline HBsAg level of 8 IU/mL may also be used to rule out the threat of HBVr. HBVr was not predicted by other HBV biomarkers, with HBcrAg, HBV DNA or HBV pgRNA. 105 46 people with HBV/HCV coinfection received DAAs. All patients successfully completed the 12th week of post-DAA therapy. The expected HBVr indicators were checked after DAA therapy. Without receiving NA therapy, seven patients (23%) acquired HBVr without experiencing a flare-up of hepatitis. From baseline to P12, individuals with HBVr showed higher HBsAg titres than those without HBVr. The specificity, sensitivity, positive negative value and positive predictive value for predicting HBVr were, respectively, 85.7%, 75.0%, 50% and 94.7% for a baseline HBsAg titre higher than 20 IU/mL. The patient did not have HBV if the baseline HBsAg titre was less than 8 IU/ml. 104 Another study compared the blood levels of cytokines and chemokines in 25 patients with HCV/HBV coinfection before and after DAA therapy. Twenty individuals exhibited considerable virologic reactivation, compared to six patients who had minor clinical reactivation. There was no statistically significant difference in baseline and week-4 blood levels of cytokines and chemokines between individuals with and without virologic reactivation. 107 Serum HBV DNA levels were the only factor associated with viral reactivation at baseline. Individuals with clinical reactivation exhibited greater baseline TNF-alpha, lower week-4 IFN-gamma and a substantial reduction in CCL2 and TNF-alpha following DAA medication, indicating a potential role for host cytokines and chemokines in the onset of HBV clinical reactivation. Patients treated with DAAs for their coinfected HBV and HCV exhibited increased pre-treatment TNF-alpha levels, decreased IFN-gamma levels, decreased CCL2 and TNF-alpha levels, and limited clinical HBV reactivation. These cytokines and chemokines could be used to find early HBV reactivation after DAA therapy. 108
Biomarker for HCV reactivation
In comparison, “HCV reactivation” refers to the resurgence of HCV replication in individuals who have a history of HCV infection. Biomarkers used to monitor HCV reactivation include: Similar to HBV DNA, measuring HCV RNA levels in the blood helps assess viral replication. An increase or reappearance of HCV RNA can indicate HCV reactivation. Elevated ALT levels may suggest liver inflammation and can be a sign of HCV reactivation. 109 Monitoring these biomarkers, along with clinical assessment and follow-up, is crucial in identifying HBV or HCV reactivation promptly and guiding appropriate management strategies. It’s worth mentioning that these biomarkers are general indicators and should be evaluated in the context of each patient’s individual case. Consultation with healthcare professionals and adherence to clinical guidelines is important for accurate interpretation and appropriate management of HBV or HCV reactivation. After receiving first-line therapy, there have been a few sporadic instances of HCV reactivation. But this happens far less often. An increase in HCV RNA of more than 1 log IU/mL above the basal level is considered HCV reactivation. It often results in no clinical indications, in contrast to HBV reactivation. In a study of coinfected cirrhotic patients receiving 18 months of nucleotide treatment, 12.5% of instances of HCV reactivation were seen. 61 96% of the patients in this group were HBV-free. The researchers were unable to determine the predominant virus in each instance prior to beginning therapy since there were only 24 participants in this tiny investigation. This might affect both the result and the rate of HCV reactivation. In general, HCV reactivation is uncommon since it is often dominant and rapidly treated. 103
Revised international guidelines/guidance for the management of patients with HCV and HBV coinfection
According to EASL recommendations, people with HBV/HCV coinfection may get the same anti-HCV treatments as those with HCV monoinfection. The EASL 2017 HBV Guidelines should be followed for treating patients with coinfections who meet the HBV treatment requirements. For coinfected individuals who are HBsAg-positive before beginning DAA medication and for at least the first 12 weeks after beginning DAA therapy, NUC prophylaxis is advised. Once HBV medication has been stopped, HBV activity should be monitored. 64 The AASLD practice guideline offers several suggestions for NUC prevention in those who are coinfected. The AASLD HBV recommendations state that blood HBV DNA and ALT levels should be used to determine HBV therapy for those with HBV monoinfection. DNA levels of HBV should be checked every 4–8 weeks while taking DAA and then every 3 months thereafter in individuals who have not had NUC therapy before starting DAA. 110 For HBsAg and HBV DNA testing, only individuals who have ALT flares while taking DAA are qualified. Recent APASL recommendations state that these coinfected individuals should have their HBV reactivation regularly monitored for 24 weeks after treatment. Since it is anticipated that HCV DAA won’t have a direct or immunomodulatory influence on HBV replication, it is recommended that the status of HBV infection be evaluated often if IFN-free regimens are utilized. 111 If anti-HBV treatment is clinically necessary, it must start right away. It is not apparent, despite EASL recommendations, whether prophylactic NUC is better than the therapeutic strategy for HBV reactivation.
Conclusion
HCV/HBV coinfection is common in endemic locations and among people at risk for illnesses that are transmitted parenterally. It is somewhat known how the two viruses interact with one another in the liver. In addition to being of academic significance, these results will contribute to the development of more effective antiviral medicines and methods to prevent HBV reactivation after HCV cure by DAA. Despite its rarity, HCV-HBV coinfection might aggravate liver disease and raise the likelihood of side consequences such as HCC. Due to the interplay of the two viruses and the possibility for either virus to reactivate when an antiviral treatment is only effective against one of the viruses, coinfections are difficult to treat. While treating the HCV-HBV coinfection, it is vital to be cognizant of the possibility of reactivation, particularly as DAAs become more widely accessible. Before initiating HCV treatment, HBV testing should be conducted. Continuous monitoring for HBV DNA replication in people who are battling other illnesses is also critical. There are few paediatric therapies available, and general data on children with coinfections is scarce. Children thus have considerably different challenges than adults in addressing coinfection.
Footnotes
Author contributions
Quratulain Maqsood and Aleena Sumrin: Conceptualization, Data analysis and curation, Validation, Writing - original draft, review and editing. Maryam Iqbal and Saima Younas: Data analysis and curation, Validation, Writing - review and editing. Nazim Hussain: Validation, Writing - review and editing. Muhammada Mahnoor and Abdul Wajid: Software, Supervision, Project administration, Validation, Visualization, Writing - original draft, Writing - review and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
