Abstract
Angiotensin-converting enzyme 2 (ACE2) is the receptor that enables SARS-CoV-2 to invade host cells. Previous studies have reported that reducing ACE2 expression may have an anti-SARS-CoV-2 effect. In this study, we constructed a pGL4.10-F2-ACE2 vector with double luciferase genes (firefly and Renilla luciferase) under the control of the ACE2 promoter and used it to screen compounds from Chinese traditional medicinal herbs (CTMHs) that can inhibit ACE2 transcription in human cells. We transfected HEK293T cells with pGL4.10-F2-ACE2 and treated them with CTMH compounds and then measured fluorescence to evaluate the indirect inhibition of ACE2 transcription. Out of 37 compounds tested, andrographolide demonstrated a dose-dependent inhibition of ACE2 transcription. We further confirmed by RT-qPCR and Western blot assays that andrographolide also reduced ACE2 expression in BEAS-2B cells in a dose-dependent manner. Moreover, pseudovirus infection assays in BEAS-2B cells demonstrated that andrographolide can inhibit SARS-CoV-2 infection in a dose-dependent manner. These results suggest that andrographolide has potential anti-SARS-CoV-2 activity and could be a candidate drug for COVID-19 prevention and treatment.
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a highly transmissible coronavirus that has caused a global pandemic of coronavirus disease 2019 (COVID-19) since its emergence in December 2019. 1 As of March 2023, SARS-CoV-2 has infected more than 676 million people and killed over 6.8 million worldwide (Johns Hopkins Coronavirus Resource Center: https://coronavirus.jhu.edu). SARS-CoV-2 can cause fever, severe respiratory illness, and various types of pneumonia. Although SARS-CoV-2 is less pathogenic than SARS-CoV, it can still induce flu-like symptoms and severe infections in some cases. Most drugs currently used for the treatment of COVID-19 are associated with adverse reactions. Regrettably, there are no antiviral drugs available on the market that are both effective and safe. 2
The binding of the spike protein of SARS-CoV-2 virus to ACE2 is crucial for cellular invasion by the new coronavirus.3,4 Therefore, interfering with ACE2 expression and blocking SARS-CoV-2 infection hold significant promise for the prevention and treatment of COVID-19. Despite many drugs targeting spike–ACE2 interactions for COVID-19 treatment, satisfactory results have not been achieved. Traditional medicinal plants have played a major role in the treatment of COVID-19.
5
Andrographis paniculata (Burm. f.) Wall. ex Nees. is an herb in the family of Acanthaceae, also known as King of bitters, Indian Echinacea, clinically used to prevent and treat respiratory tract infections, intestinal typhoid, acute jaundice, and pneumonia.
6
Andrographolide is one of the main active components of Andrographis paniculata (Figure 1), which belongs to the diterpene lactones, bitter in taste and cold-natured, mainly used to clear heat and detoxify swelling. Further, it was found to be effective against multiple viral infections like dengue,
7
swine flu,
8
hepatitis C,
9
influenza,
10
Epstein–Barr virus (EBV),
11
and herpes simplex virus 1(HSV-1)
12
in previous experimental studies. Andrographolide was recently predicted in silico to have a potent anti-SARS-CoV-2 activity through specific targeting of the host ACE2 receptor and viral factors, that is, RNA-dependent RNA polymerase, main protease, 3-CL protease, PL protease, and spike protein.
13
Recently, Shi et al. applied an enzyme-based assay to demonstrate an inhibitory effect of andrographolide against SARS-CoV-2 main protease (Mpro).
14
Structures of andrographolide.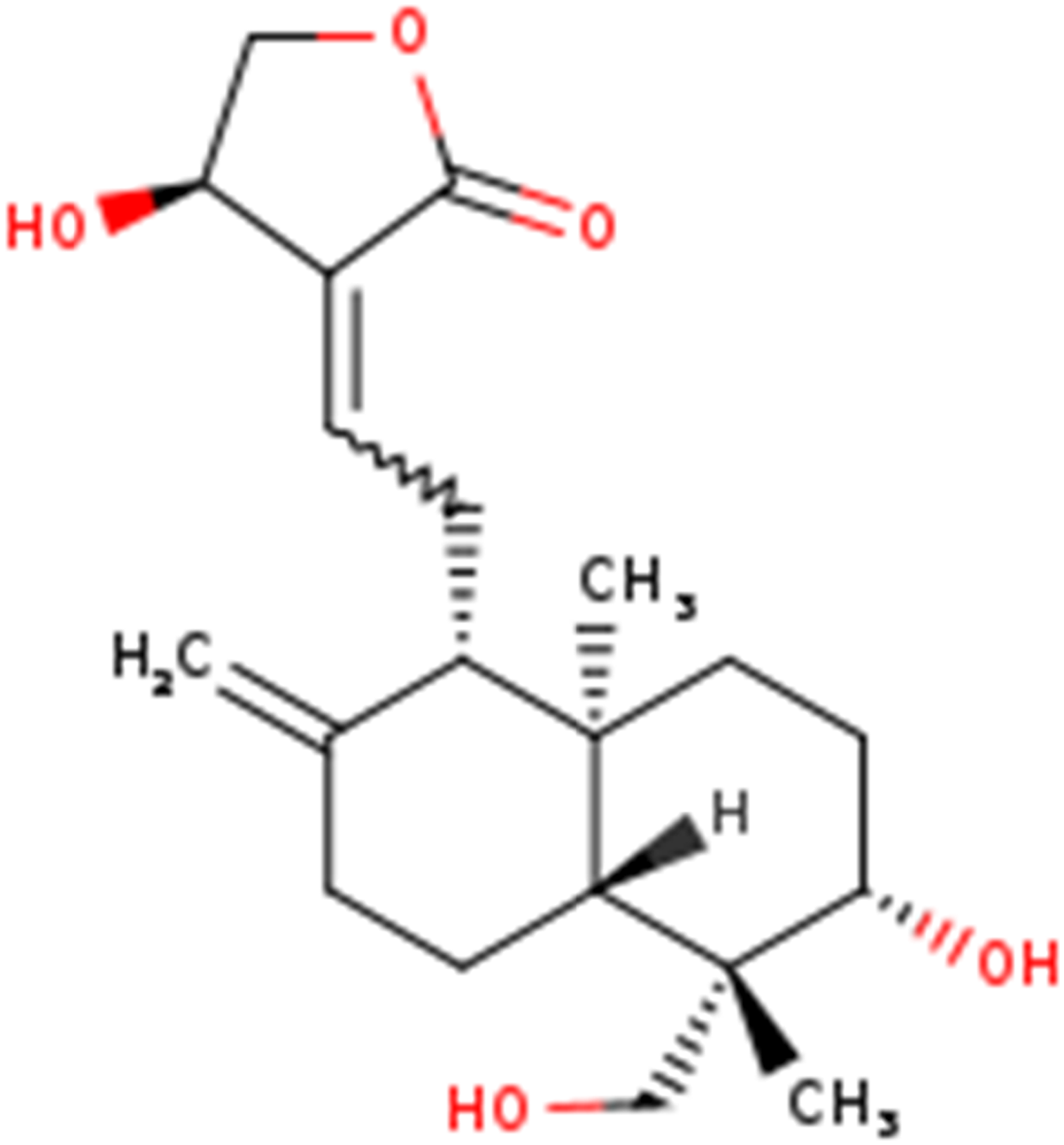
In this study, we investigated the regulatory effect of andrographolide on the expression of ACE2, the receptor for SARS-CoV-2, using a drug screening system based on pGL4.10-F2-ACE2. We also analysed the inhibitory effect of andrographolide on ACE2 expression and confirmed its antiviral activity against SARS-CoV-2 using a pseudovirus assay. This study will provide innovative ideas and methods for screening andrographolide and its derivatives for the treatment of COVID-19 and elucidating their mechanism of action.
Materials and methods
Materials and reagents
The human embryonic kidney epithelial cell line HEK293T and the bronchial epithelial cell line BEAS-2B were obtained from the China Typical Culture Collection (Wuhan, China). Andrographolide was purchased from Sigma-Aldrich (St. Louis, MO, USA). The plasmid extraction kit, agarose gel DNA extraction kit, and cleaning kit were purchased from Axygen (Tewksbury, MA, USA). The pGEM-T vector and E1960 fluorescence detection kit were purchased from Promega (Madison, WI, USA). The Cell Counting Kit-8 cell proliferation-viability assay kit was purchased from Beyotime Biotechnology (Shanghai, China). Our laboratory constructed pGL4.10-F2 based on pGL4.10, which contains firefly luciferase and Renilla luciferase genes and can be used for dual fluorescence reporter gene detection. 15 Pseudovirus-SARS-CoV-2 (B.1.617.2) was purchased from General Biology (Anhui, China), which encapsulates RNA sequences of green fluorescent protein (GFP) and luciferase. Infection efficiency can be determined by observing GFP expression and detecting luciferase activity. 16
pGL4.10-F2-ACE2 vector construction
Specific primers for human ACE2 were designed based on the sequence of the human ACE2 promoter (GenBank ID: AY217547). The sequences of the forward and reverse primers were 5′-AACCCTCGAGGGCAAAGTCATGTATTTGGA-3′ (XhoI site is underlined) and 5′-GAGCTAAGCTTCGTCCCCTGTG-3′(HindIII site is underlined), respectively. ACE2 promoter fragment was amplified by PCR using human genomic DNA as the template and was digested with XhoI and HindIII restriction endonucleases. The digested fragment was then ligated with pGL4.10-F2 plasmid that had been cut with XhoI and HindIII to generate a recombinant vector pGL4.10-F2-ACE2, in which the expression of the firefly luciferase reporter gene was driven by the ACE2 promoter. 15
Luciferase reporter assays
HEK293T cells were inoculated in 6-well plates at 3.6 × 105 cells/well using cell culture medium containing 10% (v/v%) fetal bovine serum at 5% CO2 and 37°C. When the cells covered 80% area of the well, pGL4.10-F2 and pGL4.10-F2-ACE2 were wrapped with liposomes and transfected into HEK293T cells. After 6 h of transfection, the cell culture medium was replaced with fresh medium containing 10% fetal bovine serum (FBS) by adding a certain concentration of andrographolide (0, 10, 20, 40, 60, and 80 μmol/L) and incubated for 24 h. The cells were collected and subjected to fluorescent assays using the Dual-Luciferase Reporter Assay System (Promega) and Cytation 5 Multi-Mode Reader Fluorescence Validation (BioTek, Vermont, United States). This experiment was repeated two times with three biological replicates in each experiment.
CCK-8
Cell suspensions of 100 μl containing 5 × 104 cells/mL were inoculated into a single well of 96-well plates. After 24 h of incubation, andrographolide was added to each well to different final concentrations (10, 20, 40, 60, 80, 100, and 160 μmol/L). After further incubation for 24 h, CCK-8 was added to each well and incubated for another hour before the colorimetric assay. The absorbance OD450 value of each well was measured after the addition of CCK-8. Cell survival rate (%) of each experimental group was calculated = OD (test - blank wells)/OD (control - blank wells) × 100%. The experiment was repeated three times with five parallel wells each time.
Assays of ACE2 expression in BEAS-2B cells
RNA extraction was performed from BEAS-2B cells treated with andrographolide at the aforementioned concentrations for 24 h. The extracted RNA was used as the template for reverse transcription with random primers. The qPCR assays for ACE-2 gene transcription were carried out using the following primer pairs: ACE2-F: 5′-CAT TGG AGC AAG TGT TGG ATC TT-3′; ACE2-R: 5′-GAG CTA ATG CAT GCC ATT CTC A-3′. The internal control, β-actin, was amplified with the following primer pairs: β-actin-F: 5′-GTC ACC AAC TGG GAC GAC AT-3; β-actin-R: 5′-GCA CAG CCT GGA TAG CAA CG-3′. The qPCR was performed using the Power Up™SYBR™ Green Master Mix kit. The 2−ΔΔCt relative quantification method was used to analyse the relative expression of ACE2.
BEAS-2B cells after incubated for 24 h with andrographolide of the aforementioned concentrations were harvested, and the total cellular protein of the cells was extracted for Western blot assays.
Pseudovirus infection
BEAS-2B cells were cultured in 96-well cell culture plates to cover 40% of the well’s area and incubated with different concentrations of andrographolide (0, 10, 20, and 40 μmol/L) for 6 h. Pseudovirus (10 μL/well) was added to the wells and co-incubated with the cells for 8 h and then the medium was replaced by fresh medium and re-incubated for 48 h. The green fluorescence of the cells was observed using a fluorescence microscope. Luciferase assays were conducted using the Luciferase Reporter Assay System. 20 µL of Luciferase Assay Reagent was added to each well to measure the activity of firefly luciferase. Results were presented as the ratio of luminescence from the experimental reporter to that of the control reporter and were calculated and normalized relative to control wells. Each well was subjected to three replicates for analysis.
Drug property prediction
The ADME parameters, pharmacokinetic properties, drug-likeness, and medicinal chemistry friendliness of the compound were analysed using SwissADM. 17
Statistical processing
Data were analysed by one-way ANOVA using GraphPad Prism 8 statistical software, and p < .05 was selected to indicate that the differences were statistically significant.
Results
Construction of a drug screening platform targeting the ACE2 promoter
After the pGL4.10-F2-ACE2 recombinant vector was constructed and verified by double enzyme digestion (Figure 2(A)), the target fragment was inserted into the pGL4.10-F2 vector. Sequencing results further confirmed that the cloned ACE2 promoter gene met the experimental requirements and did not contain any insertion or point mutations. The pGL4.10-F2-ACE2 vector was successfully constructed and transfected HEK293T cells (Figure 2(B)). The promoter activity was confirmed by Dual-Luciferase Reporter Assay System (Figure 2(C)). This drug screening platform targeting the ACE2 promoter was successfully constructed. Using this platform, we screened 37 common Chinese herbal monomers and found that the andrographolide had an inhibitory effect on the ACE2 promoter activity (Supplemental S1). As the drug concentration increases, andrographolide demonstrates a significant decrease in luciferase activity. This suggests that the drug is capable of reducing the expression of the ACE2 promoter (Figure 2(D)). We selected this drug for further validation. Construction of a drug screening platform targeting the ACE2 promoter. (A) Gel electrophoresis of pGL4.10-F2-ACE2 was carried out using double restriction enzyme digestion. Lane 1 shows the pGL4.10-F2-ACE2 recombinant, Lane 2 shows the double restriction enzyme digestion product of pGL4.10-F2-ACE2, and M denotes the marker. (B) Vector image of pGL4.10-F2-ACE2. (C)Validation of constructed ACE2 promoter reporter gene drug screening method. Compared with the pGL4.10-F2-ACE2 vector, the activity of the dual-luciferase backbone pGL4.10-F2 was significantly lower. **p < .01. (D) Detection of dual luciferase activity after the action of different concentrations of andrographolide. Compared with the blank group: *p < .05 and **p < .01.
Effect of the CCK-8 assay on the activity of andrographolide on cells
The results showed that the cell viability showed a decreasing trend with the increase of the drug concentration of andrographolide, as shown in Figure 3. Compared with 0 μmol/L, the survival rate of HEK293T cells under the effect of andrographolide for 24 h was greater than 80% when the concentration of andrographolide was lower than 80 μmol/L (Figure 3(A)). The survival rate of BEAS-2B cells was over 80% after 24 h of exposure to andrographolide at concentrations up to 40 μmol/L, compared with the 0 μmol/L (Figure 3(B)). Effect of andrographolide on cell viability. Cell viability of HEK293T cells (A) and BEAS-2B cells (B) was measured after 24 h treatment with different concentrations of andrographolide. Compared with the blank group: *p < 0 .05 and **p < 0 .01.
The inhibitory effect on ACE2 expression in BEAS-2B cells by andrographolide
The transcription level of ACE2 mRNA was observed to decrease in a dose-dependent manner with increasing drug concentration. At a concentration of 40 μmol/L of andrographolide, the expression of the ACE2 gene was reduced by approximately 50%, as demonstrated in Figure 4(A). Western blot analysis revealed a dose-dependent inhibitory effect of andrographolide on ACE2 protein expression, with a similar 50% reduction observed at a concentration of 40 μmol/L of andrographolide (Figure 4(C)). Andrographolide downregulates ACE2 expression at mRNA and protein levels. ACE2 mRNA (A) and protein (B and C) expression was evaluated using RT-qPCR and Western blotting after treatment with andrographolide. Compared with the blank group: **p < 0 .01.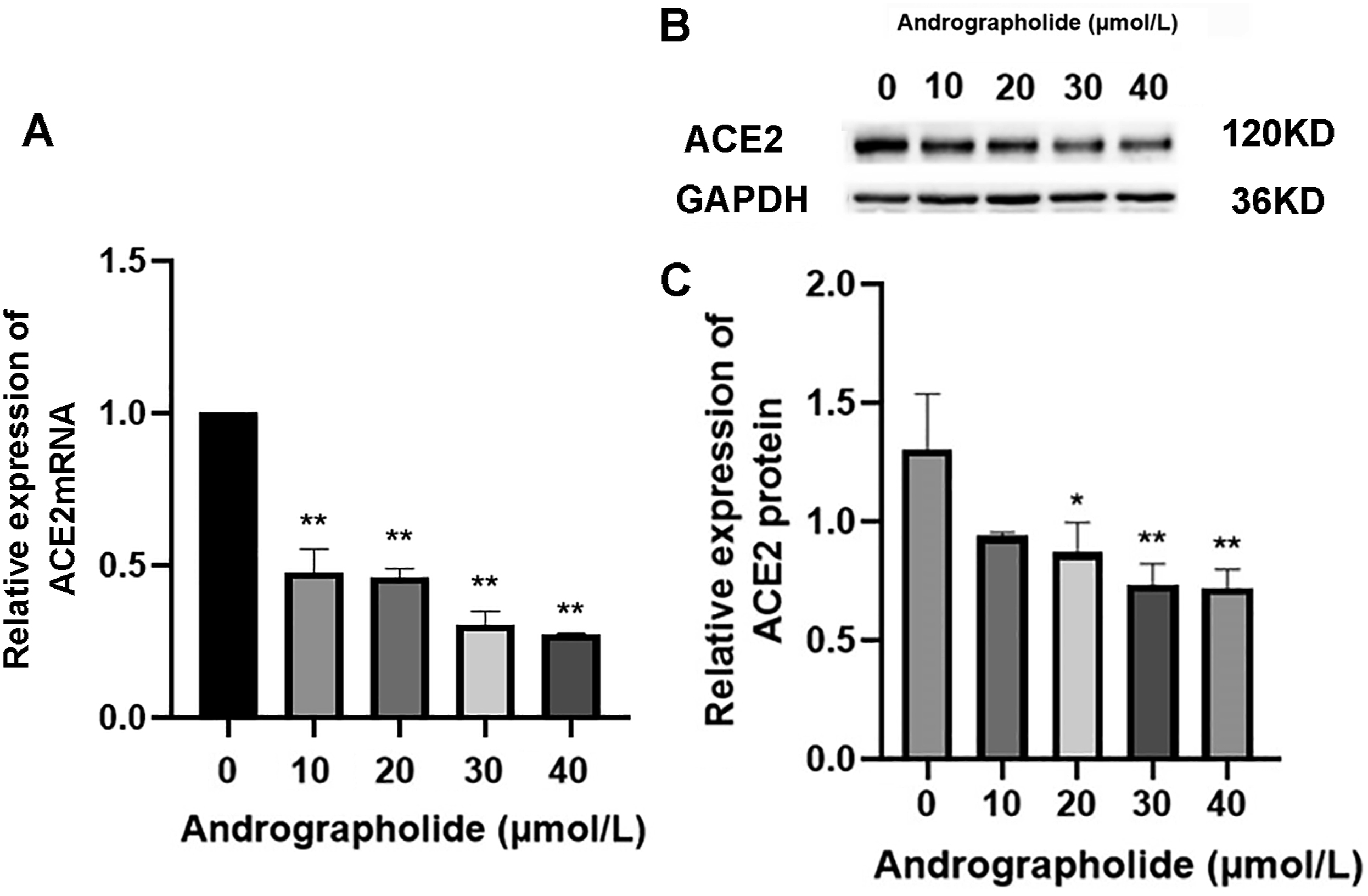
Inhibition of SARS-CoV-2 pseudovirus-infection by andrographolide
Compared with the control group, BEAS-2B cells infected with SARS-CoV-2 pseudovirus exhibited a significant increase in relative luciferase activity and fluorescence intensity, indicating successful pseudovirus infection and activity. However, the addition of varying concentrations of andrographolide resulted in a significant reduction in dual luciferase activity, suggesting the inhibitory effects of andrographolide on pseudovirus infection in cells. These experimental results are illustrated in Figure 5(A) and (B). Furthermore, the predicted drug properties of andrographolide demonstrated favourable drug-like properties, as depicted in Figure 5(C). Andrographolide inhibits SARS-CoV-2 pseudovirus infection and has favourable drug properties. (A) The luciferase activities of the reporter genes in each group were measured using the luciferase assay kit after infection with SARS-CoV-2 pseudovirus and treatment with different concentrations of andrographolide. Compared with the blank group: *p < 0 .05 and **p < 0 .01. (B) Fluorescence photos of SARS-CoV-2 pseudovirus cells infected with different concentrations of andrographolide (100×). (C) Andrographolide drug property prediction using SwissADME web tool. The Bioavailability Radar displays the drug-likeness of a molecule. The pink area represents the optimal range for each property. Six physicochemical properties are taken into account: lipophilicity, size, polarity, solubility, flexibility, and saturation.
Discussion
ACE2 is a key receptor for SARS-CoV-2 to enter human cells. Therefore, finding drugs or compounds that can inhibit the function of ACE2 may become one of the strategies to treat COVID-19. 18 Lobeline, a nicotine receptor agonist, has been shown to reduce the expression and activity of ACE2. Research indicates that lobeline may alleviate lung damage and inflammation from COVID-19 by modulating the ACE2 and angiotensin-II balance. 19 Studies show that glucocorticoids can alleviate lung damage and inflammation in COVID-19 by inhibiting ACE2 expression. 20 Furthermore, studies indicate that ursodeoxycholic acid can inhibit ACE2 expression and activity, potentially preventing viral entry into human cells. 21
Traditional Chinese medicine has been used in the treatment of COVID-19 in China and has shown promising results in improving symptoms, shortening the duration of illness, and reducing the need for intensive care. 22 Some traditional Chinese medicines have been found to have potential anti-ACE2 activity, including baicalin, hesperetin, and glycyrrhizic acid.23,24 Baicalin and hesperetin are flavonoids extracted from Scutellaria baicalensis and Citrus aurantium, respectively, while glycyrrhizic acid is derived from the root of Glycyrrhiza glabra. These compounds have been shown to inhibit ACE2 expression and may help prevent SARS-CoV-2 infection by reducing the number of available receptors for the virus to bind to.25,26 A recent study reported that andrographolide and baicalin synergistically inhibited coronavirus by downregulating ACE2 protein level. 27 However, more research is needed to confirm their efficacy and safety for the treatment of COVID-19.
This study utilized dual-luciferase reporter gene technology to construct a recombinant vector, PGL-4.10-F2-ACE2, which was employed to screen active ingredients targeting ACE2 gene expression. The human ACE2 promoter controlled the expression of firefly luciferase in the vector. The activity of the ACE2 promoter was evaluated by calculating the ratio of firefly luciferase activity to Renilla luciferase activity in 293T cells transiently transfected with the recombinant vector. Using this system, we screened one active ingredient, andrographolide, from 37 traditional Chinese medicine active molecules with an inhibitory effect on ACE2 expression. Andrographolide is often used in clinical practice in combination with antibiotics for the treatment of inflammation and viral diseases. 26 In recent years, additional functions of andrographolide have been discovered, including antipyretic, hepatoprotective, hypoglycemic, immunoregulatory, anti-cardiovascular and cerebrovascular diseases, and neuroprotective effects.28–30 We further confirmed the anti-COVID-19 virus infection effect of andrographolide using qPCR, Western blot, and pseudovirus infection experiments.
Pseudovirus refers to a type of retrovirus that can integrate the membrane glycoprotein of another virus of a different species, thus forming a membrane of an exogenous virus while maintaining the genome characteristics of the retrovirus itself. 31 We used the pseudovirus of SARS-COV-2, which contains the spike glycoprotein (B.1.617.2) mutation protein of the novel coronavirus on the surface of the membrane, and the RNA sequence of green fluorescent protein (GFP) and luciferase was encapsulated inside the virus. The inhibitory effect of andrographolide on the activity of the ACE2 promoter in HEK293T-ACE2 cells infected with the pseudovirus-SARS-COV-2 was reflected by the ratio of dual-luciferase activity, which reduced the efficiency of pseudovirus-SARS-COV-2 infection.
Real-time fluorescent quantitative PCR and Western blot were used to detect the effect of andrographolide on the expression of ACE2 in BEAS-2B cells. This study demonstrates that andrographolide can target the regulation of ACE2 expression to inhibit SARS-CoV-2 infection, thereby exerting its role in the prevention and treatment of COVID-19. The potential value of andrographolide for clinical prevention and treatment of COVID-19 has been revealed.
Conclusion
In this study, a sensitive and rapid drug screening system was established. The screening demonstrated the effect of andrographolide in targeting and regulating ACE2 expression to inhibit SARS-CoV-2 infection. Our results suggest that andrographolide may be effective in combating COVID-19 by modulating ACE2 receptors through multiple active ingredients, core targets, and pathways, ultimately leading to the inhibition of ACE2 promoter activity. Our findings highlight the potential clinical utility of andrographolide in the treatment of COVID-19.
Supplemental Material
Supplemental Material - Andrographolide suppresses SARS-CoV-2 infection by downregulating ACE2 expression: A mechanistic study
Supplemental Material for Andrographolide suppresses SARS-CoV-2 infection by downregulating ACE2 expression: A mechanistic study by Qing Li, Hongmei Lu, Yongdui Ruan, Yuxuan Geng, Zuguo Zhao, Ying Liu, Long Feng, and Wentao Guo in Antiviral Therapy.
Footnotes
Author Contributions
YG and HL participated in the experiments, collated data, visualized data, and performed statistical analysis. YR and LF reviewed the manuscript and provided funding. DL was responsible for research oversight. GZ and YL provided suggestions and ideas for manuscript writing. LF and WG contributed to research execution, management, and coordination. All authors contributed to the article and approved the final manuscript version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received funding support from several sources, including the Guangdong Provincial Medical Science and Technology Research Fund Project (A2021440), the Guangdong Provincial Bureau of Traditional Chinese Medicine Scientific Research Project (20211220), the Social Science Development Project of Dongguan City (20211800905542), the Guangdong Medical University Scientific Research Project (GDMUQ2021005), and the Discipline Construction Project of Guangdong Medical University (4SG21229GDGFY01).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
