Abstract
Background
In British Columbia, antiretrovirals (ARVs) for HIV treatment (HIV-Tx) and pre-exposure prophylaxis (PrEP) are free-of-charge through publicly-funded Drug Treatment Programs (DTPs). When available, less costly generics are substituted for brand-name ARVs. We describe the incidence and type of product substitution issue (PSI) adverse drug reactions (ADRs) attributed to generic ARVs.
Methods
Cohorts included DTP clients ≥19 years who received generic ARVs for HIV-Tx (abacavir-lamivudine, emtricitabine-tenofovir DF, efavirenz-emtricitabine-tenofovir DF, atazanavir or darunavir between 01 Jun 2017 and 30 Jun 2022) or PrEP (emtricitabine-tenofovir DF, 01 Apr 2018 to 30 Jun 2022). Demographic, ARV and ADR data were extracted from DTP databases and summarized by descriptive statistics. PSI incidence was calculated for each product during the year following brand-to-generic and generic-to-generic transitions (first-year-post-rollout), and compared between generic versions using generalized estimating equations. For context, incidence of any ARV product-related ADR was calculated in the same 1-year periods.
Results
During first-year-post-rollout periods, 5339 HIV-Tx (83% male, median age 52 years) and 8095 PrEP (99% male, median 33 years) clients received generic ARVs, and reported 78 and 23 generic PSIs, respectively. PSI incidence was <1% for most generic ARVs, with mild-moderate symptoms including gastrointestinal upset, headache, dizziness, fatigue/malaise and skin rash. In HIV-Tx clients, the efavirenz-containing product had higher PSI incidence than other ARVs (2.2%, p = .004), due to more neuropsychiatric adverse reactions. Any ADR incidence was stable across measurement periods, and generic PSIs represented less than one third of all product-related ADRs.
Conclusions
Generic substitution of antiretrovirals for HIV-Tx and PrEP was well tolerated, with ≤2% incidence of mild-moderate PSI ADRs.
Keywords
Introduction
Generic medications are less costly alternatives to originator brand products and provide an opportunity for reduced medication acquisition costs for consumers and health-care systems.1–3 Generic alternatives are identical to the original brand product in medicinal ingredients and strength, and have similar non-medicinal ingredients. In Canada, generics must meet the same stringent manufacturing and quality control criteria required of brand-name products, and must demonstrate comparable bioavailability with their brand-name equivalent but are not required to undergo full clinical trial testing. 1
In British Columbia (BC), Canada, antiretroviral (ARV) medications for HIV treatment (HIV-Tx, since 1992) and Pre-Exposure Prophylaxis (PrEP, since 2018) are available through province-wide, publicly funded, BC Centre for Excellence of HIV/AIDS (BC-CfE) Drug Treatment Programs.4,5 Both HIV-Tx and PrEP programs provide ARVs at no cost to treatment-eligible BC residents, with ARV dispensing provided by a centralized pharmacy service.
In alignment with BC Ministry of Health policies, generic ARVs are included in the BC-CfE drug formulary.6–8 At the time of the initial, large-scale formulary transitions to generic ARVs, educational information was distributed to health-care providers and community-based organizations, describing the safety and effectiveness of generic medications.7,9 Information about generic ARVs is also included in BC-CfE drug formulary and treatment guideline updates.8,10 Whenever a new generic product is introduced, ongoing prescriptions are switched to the generic version at the next medication refill and pharmacists inform clients of the substitution with written and/or verbal communication. New prescriptions are automatically dispensed using the generic product.
The BC-CfE Pharmacovigilance service monitors ARV safety in HIV-Tx and PrEP program clients. 11 Routine surveillance is conducted through collection and monitoring of adverse drug reaction (ADR) reports voluntarily submitted by health-care providers. If a person experiences an ADR attributed to a specific generic version of an ARV, this is termed a ‘product substitution issue’ (PSI). 12 In the event of a PSI ADR, the prescriber may request an alternate generic and, if none of the available generic equivalents are tolerated, the brand-name product may be requested. Requests for exceptional provision of non-formulary generics or brand-name ARVs must be accompanied by an ADR report describing the PSI.
The purpose of this study was to characterize the incidence and type of PSIs associated with the most commonly used generic ARV products in BC’s HIV-Tx and PrEP programs. For each studied generic version, we described the incidence, type and timing of reported PSIs during the first year following the generic rollout date, and compared PSI incidence between different generic versions. For context, we conducted similar analyses of the incidence of any ARV product-related ADRs (regardless of manufacturer) in the same 12-month periods and determined the contribution of PSIs to overall product-related ADR reports.
Methods
Data sources and inclusion criteria
Client clinical and demographic data, ARV dispensing information and ADR reports were abstracted from longitudinal, population-based BC-CfE datasets (described elsewhere) 13 and were analyzed separately for HIV-Tx and PrEP clients. The study period extended from the earliest introduction of the studied generics in each program (01 Jun 2017 for HIV-Tx and 01 Apr 2018 for PrEP) until 30 Jun 2022. This time-frame permitted at least 1 year follow-up opportunity after the rollout of each generic product version.
In this study, an ‘ARV product’ refers to a pharmaceutical preparation including one or more ARV ingredients, and a ‘product version’ is a manufacturer-specific version. The BC-CfE formulary includes one ARV product version at a given time, and periodically transitions between versions according to contractual agreements with suppliers. The generic version ‘rollout date’ marks the transition to a different manufacturer’s version and was defined as the first day of the month when the product inventory transition began. If usage of the new generic started near month-end, rollout date was defined as the first of the following month.
For each studied ARV product, HIV-Tx and PrEP clients were included if they received at least a 1-day supply of dispensed product, and were age ≥19 years on the first day of product use within the study period. For analyses of PSI ADR incidence, persons were included if they received at least a 1-day supply of the generic product version within 1 year following the rollout date. For analyses of any ADR, persons were included if they received at least a 1-day supply of the ARV product, regardless of manufacturer, in the specified 1-year period.
We studied the five most-used ARV products having a generic equivalent in BC (details in Supplemental Table S-1). For HIV-Tx clients, these were: The nucleoside(-tide) reverse transcriptase inhibitor (NRTI) combinations abacavir-lamivudine and emtricitabine-tenofovir disoproxil fumarate (tenofovir DF), the non-NRTI (NNRTI) combination efavirenz-emtricitabine-tenofovir DF, and the protease inhibitors (PIs) atazanavir and darunavir. For PrEP clients, emtricitabine-tenofovir DF was the only available generic.
For study purposes, manufacturer names were de-identified, with the originator version described as ‘brand’, and generics identified in chronological order of formulary addition as ‘generic-1’ and ‘generic-2’. Non-formulary generics were collectively termed ‘generic-NF’. Generic-NF ARVs were used ad hoc for managing drug shortages or client-specific needs; therefore they were excluded from generic version-specific PSI analyses, but were included in analyses of any product-related ADRs.
Outcome measures and statistical analyses
To provide a longitudinal overview of ARV product utilization and PSI reporting patterns, monthly product usage and PSI frequency, stratified by product version, were summarized across the entire study period. Longitudinal time arrays of medication possession (dispensing date plus number of days supply) were created. Clients were counted once per product per month, categorized as receiving the product version with the largest days-supply. PSI ADRs were counted in the month they were reported.
The primary outcome was the occurrence of a PSI within the first-year-post-rollout for each generic version. To be classified as a PSI, the ADR report required inclusion of a statement implicating a specific generic version (e.g. nausea following a switch from brand to generic-1). Reports with no generic-specific complaint were recorded as product-related ADRs, but not PSIs. In alignment with Medical Dictionary for Regulatory Activities (MedDRA®) terminology, PSIs were categorized as brand-to-generic or generic-to-generic substitutions, based on the client’s ARV dispensing history.12,14,15 If a generic version-specific intolerance was reported with the client’s first use of an ARV product (e.g. client initiated treatment with generic-1, experienced side-effects, and requested brand-name product), this was considered a variant of brand-to-generic substitution and termed an ‘initial generic’ PSI. This interpretation is consistent MedDRA®, which does not consider the level of authorization of product substitution (prescriber, pharmacy, etc.,) and in our setting, automatic brand-to-generic substitution is dictated by program policies. 7
HIV-Tx and PrEP client characteristics at the start of each first-year-post-rollout period were summarized for each product version using descriptive statistics. A person could contribute data for more than one ARV product or generic version. Categorical variables including biologic sex at birth (male, female, unknown) and prior experience using the ARV product (yes/no) were expressed as proportions. Age was summarized at the start date of each product version use, and overall (at first generic product use in the study period) as median (first-third quartile, Q1–Q3) years. Median (Q1–Q3) follow-up time and time to PSI were calculated from the first product version dispensing date in the measurement period until censoring (earliest of stop date, period end, client moved or died) or PSI report date, respectively.
The PSI analyses were performed for transitions from brand to generic-1 and from generic-1 to generic-2. If the formulary subsequently switched back to generic-1, the distinct first and second generic-1 rollouts were analyzed separately. PSI incidence proportion was calculated as the number of people with a PSI ADR reported in the first year following the generic version rollout-date, divided by the number of people who received at least a 1-day supply of the product version during that year, expressed as a percentage with Wald 95% confidence intervals (95% CI) for binomial proportions. The incidence proportion and 95% CI of ‘any’ product-related ADR (including both PSI and non-PSI ADRs) was evaluated during the same 1-year measurement periods, where period one (P1) aligned with generic-1 first rollout, P2 with generic-2 rollout and P3 with generic-1 second rollout, where applicable.
PSI incidence was compared between generic versions and rollout periods of the same product. We also compared PSI incidence between ARV products (HIV-Tx, generic-1 versions only). Similar comparisons were made for any ADR incidence across measurement periods for each product and between products (comparing P1). These comparisons used generalized estimating equations with binomial distribution, logit link and robust standard errors, and an unstructured correlation matrix to account for repeated measures within individuals. p-value <.05 indicated statistical significance. 16
For descriptive summaries of PSI ADR or any ADR characteristics, the ADR report was the unit of analysis. The type of product substitution (brand-to-generic, generic-to-generic) and post-PSI treatment decision (product version switch, ARV change or discontinuation) were pooled for all PSIs in the HIV-Tx and PrEP cohorts, and expressed as the proportion of total PSIs. Symptoms associated with PSI ADRs and with any ADRs were pooled across all measurement periods for each ARV product. Each ADR report could include multiple symptoms. Symptoms were counted once per symptom category (defined in Supplemental Table S-2), per person per product and expressed as the proportion of reports.
Analyses used SAS 9.4, ©2023 SAS Institute Inc., Cary, NC, USA. Ethical approval was obtained from the University of British Columbia/Providence Healthcare Research Ethics Board (H06-03941).
Results
Characteristics of HIV treatment and pre-exposure prophylaxis program clients who received a generic antiretroviral during the first year following product rollout.
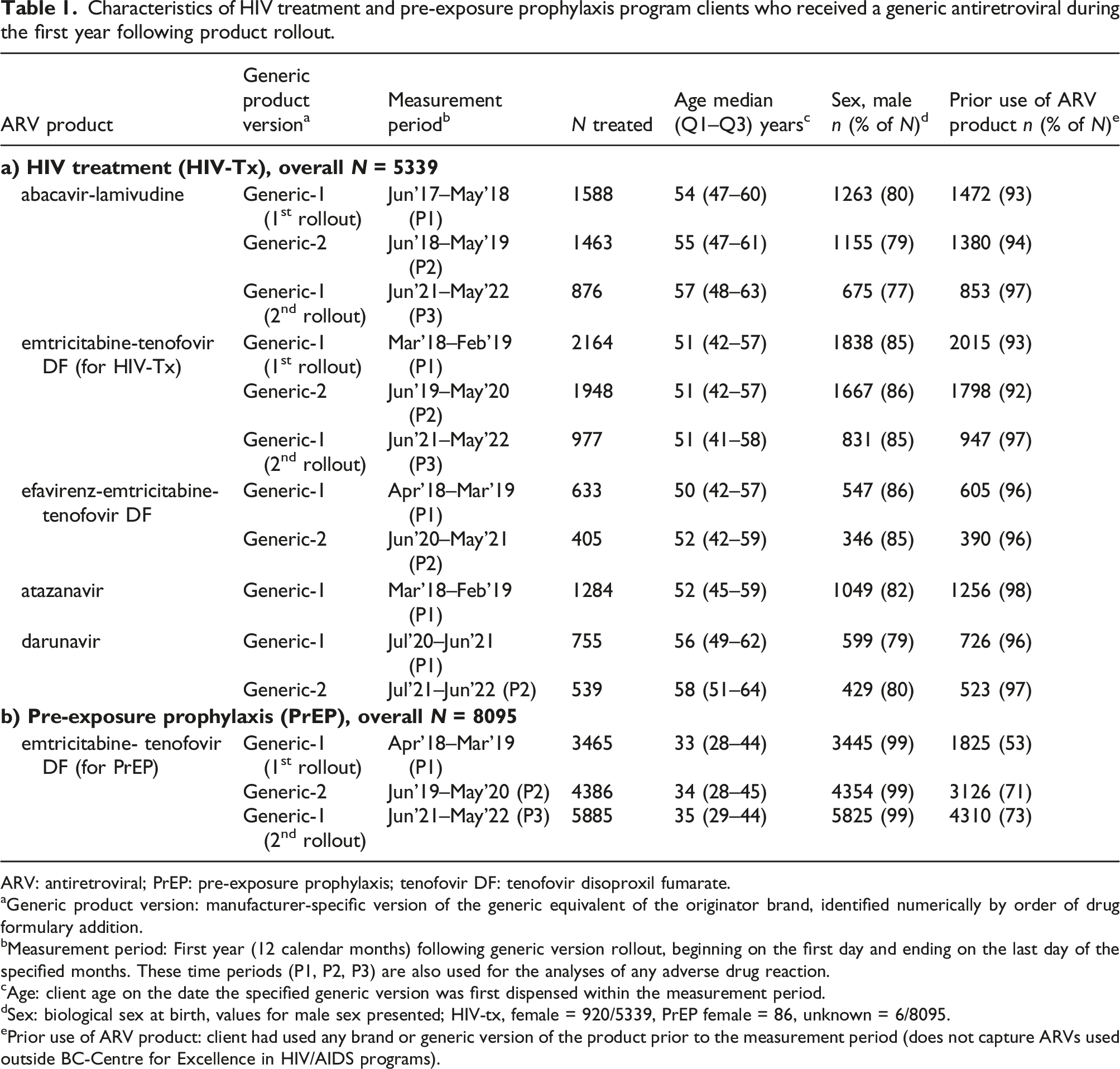
ARV: antiretroviral; PrEP: pre-exposure prophylaxis; tenofovir DF: tenofovir disoproxil fumarate.
aGeneric product version: manufacturer-specific version of the generic equivalent of the originator brand, identified numerically by order of drug formulary addition.
bMeasurement period: First year (12 calendar months) following generic version rollout, beginning on the first day and ending on the last day of the specified months. These time periods (P1, P2, P3) are also used for the analyses of any adverse drug reaction.
cAge: client age on the date the specified generic version was first dispensed within the measurement period.
dSex: biological sex at birth, values for male sex presented; HIV-tx, female = 920/5339, PrEP female = 86, unknown = 6/8095.
ePrior use of ARV product: client had used any brand or generic version of the product prior to the measurement period (does not capture ARVs used outside BC-Centre for Excellence in HIV/AIDS programs).
Figure 1(a) (HIV-Tx) and Figure 1(b) (PrEP) show the longitudinal use of ARV product versions and PSI reporting patterns throughout the study period. For most products, the initial brand-to-generic-1 transition occurred in 2017 or 2018, with generic darunavir being the most recent addition in 2020. Except for atazanavir, all products had two generic versions. Abacavir-lamivudine and emtricitabine-tenofovir DF also had two distinct generic-1 rollouts. In each measurement period, transitions between product versions were near complete within 4 months (Figure 1(a) and (b)). Some abacavir-lamivudine and emtricitabine-tenofovir DF generic transitions were complicated by supply chain disruptions, necessitating temporary switches to brand or generic-NF. (a). Antiretroviral product utilization and generic product substitution issue (PSI) adverse drug reaction (ADR) reports in HIV treatment clients, 1st June 2017 to 30th June 2022. (b). Antiretroviral product utilization and generic product substitution issue (PSI) adverse drug reaction (ADR) reports in pre-exposure prophylaxis (PrEP) clients, 1st April 2018 to 30th June 2022.
The majority (>92%) of HIV-Tx clients had prior experience using the ARV product before receiving a generic version, whereas 53% to 73% of PrEP program clients had known, prior product experience (Table 1). In the HIV-Tx cohort, 1636/5339 (31%) clients contributed data for two or more generic ARV products, and >80% of HIV-Tx clients who received generic-2 or generic-1 s rollout had previously received generic-1 (first rollout), see Supplemental Table S-3. In the PrEP cohort, 2667/4386 (61%) clients received both generic-1 and -2, while only 2059/5885 (35%) of those included in the generic-1 second-rollout period had previously received generic-1 during the first rollout. The difference in HIV-Tx and PrEP client prior product usage and exposure to multiple generic versions is explained by the rapid, ongoing enrolment of new PrEP clients during the study period, compared to a relatively stable cohort of HIV-Tx clients.
Median follow-up time during the first-year-post-rollout periods was approximately 9 months for HIV-Tx and four to 7 months for PrEP clients (Supplemental Table S-4). During these measurement periods, there were 78 generic PSI reports in 71/5339 (1.33%) HIV-Tx clients and 23 PSIs among 21/8095 (0.26%) PrEP clients, representing 78/90 (87%) and 23/33 (70%) of all HIV-Tx and PrEP PSI reports received throughout the entire study period. The median (Q1–Q3) time from generic start to PSI report date was 80 (38–162) days for HIV-Tx and 90 (47–139) days for PrEP clients.
In the HIV-Tx cohort (Figure 1(a), Table S-5), PSI incidence was <1.0% for all generic product versions except efavirenz-emtricitabine-tenofovir DF (for which PSI incidence was 2.2%). Darunavir generic-1 was the only generic with zero PSI reports. In the PrEP cohort, PSI incidence was <0.2% for emtricitabine-tenofovir DF generics in all first-year-post-rollout periods (Figure 1(b), Table S-5). Within each ARV product, there were no statistically significant differences in PSI incidence between generic-1 and -2, or between generic-1 first and second rollouts for either HIV-Tx or PrEP cohorts (all p-values >.05, Supplemental Table S-5).
The between-product comparisons for the HIV-Tx cohort identified efavirenz-emtricitabine-tenofovir DF as having significantly higher PSI incidence (p = .004) versus other ARV generic-1 versions, driven by efavirenz-related neuropsychiatric ADRs.
Figure 2 depicts the incidence of any product-related ADR during the same measurement periods examined for the first-year-post-rollout analyses. Within each product, ADR incidence was stable across measurement periods for both HIV-Tx and PrEP cohorts (all p > .05, Supplemental Table S-6). Comparing incidence of any product-related ADRs across ARV products used for HIV-Tx, ADR incidence was highest for efavirenz-emtricitabine-tenofovir DF and lowest for abacavir-lamivudine (11.9% and 1.9% in P1, respectively, p < .001). For emtricitabine-tenofovir DF, incidence of any ADR was approximately 7% in the HIV-Tx cohort and <1% in the PrEP cohort (Table S-6). In both HIV-Tx and PrEP cohorts, PSIs accounted for 6%–37% of ADRs reported during the initial brand-to generic and generic-to-generic transition periods (Figure 2), with the highest proportion observed for the earliest generic introduction, abacavir-lamivudine. Incidence of any antiretroviral-related adverse drug reaction in HIV treatment and pre-exposure prophylaxis clients during 1 year measurement periods.
Figure 3(a) summarizes the symptoms associated with PSI ADR reports. PSI ADR symptoms were similar across all ARV products in both HIV-Tx and PrEP programs and were predominantly subjective complaints such as gastrointestinal upset (including intolerance of the product taste or smell), headache, dizziness and general fatigue/malaise attributed to the generic substitution. Several, but not all, cases of skin reaction attributed to generic ARVs resolved after switching to another product version, suggestive of a sensitivity to product excipients. Among HIV-Tx clients, the efavirenz-containing product had more central nervous system (CNS) PSIs than other generics, with some reports describing worsening sleep or mood associated with the generic substitution that resolved with switch back to brand-name product. There were no serious (potentially life-threatening or requiring hospital admission) PSI ADRs reported in either cohort. Characteristics of reported antiretroviral generic product substitution issues. (a) Product substitution issue (PSI) adverse drug reaction (ADR) symptoms (b) PSI, type of product substitution (c) Treatment decision post-PSI event.
Symptoms associated with any product-related ADR were consistent with the well established side-effect profiles of these ARVs (Supplemental Figure S-1).17–21 In the HIV-Tx cohort, gastrointestinal side-effects were the most commonly reported ADRs, the efavirenz-containing product had the highest proportion of CNS effects such as sleep and mood disturbances, atazanavir had more hepatic side effects due to benign hyperbilirubinemia, and the tenofovir DF-containing products for HIV-Tx had a relatively a higher proportion of renal and musculoskeletal (osteoporosis) ADRs than other ARVs. In the PrEP cohort, gastrointestinal symptoms, renal impairment and CNS symptoms were among the most common ADRs attributed to emtricitabine-tenofovir DF.
Figure 3(b) and (c) summarize the type of product substitution and ARV treatment decision associated with the 78 HIV-Tx PSIs and 23 PrEP PSIs reported during all first-year-post-rollout measurement periods. In both cohorts, approximately two-thirds of PSI reports involved brand-to-generic substitutions and one-third were related to generic-to-generic switches. Among PrEP clients, 6/4475 (0.13%) people who received a generic product with their initial use of PrEP reported a specific generic-version intolerance. In these cases, an alternate product version was requested in the hope that it would be better tolerated. Following the PSI report, approximately half of HIV-Tx clients switched to the brand version and one quarter to an alternate generic of the product, and 12 clients (15%) changed to a different ARV regimen. This pattern was reversed in PrEP clients, with 15 (65%) switching to an alternate generic and 5 (22%) switching to the brand-name product. A small proportion of HIV-Tx (n = 6, 8%) and PrEP (n = 3, 13%) clients with PSIs temporarily discontinued ARV medications as a result of the generic product intolerance. In all cases, clients subsequently resumed treatment within several weeks (HIV-Tx) or months (PrEP) following the interruption.
Discussion
We studied two large cohorts of 5339 HIV-Tx and 8095 PrEP clients who received generic ARVs during the first year following drug formulary transitions from brand-to-generic or between generic product versions in 2017 to 2022, and found a low incidence, generally <1% of clients, of generic substitution-related ADRs. To our knowledge, this is the largest study to date to focus on the real-world tolerability of generic ARVs, and its strengths include analysis of contemporary, population-based cohorts of both HIV-Tx and PrEP clients in a setting with centralized ARV distribution and ADR collection through an ARV-focused pharmacovigilance service.
Generic ARVs have been used extensively for HIV treatment in low income countries, and there is evidence supporting their safety and effectiveness.22–25 In North American and European health care systems, the potential cost savings from generic ARVs for HIV-Tx and PrEP,2,3,26,27 and clinician and client willingness to consider generic ARVs28–31 have been explored, but there is limited information regarding real-world experiences following implementation of generic ARV substitution in these health care settings.
Several single-clinic studies describe safe and effective de-simplification of single tablet ARV regimens to an equivalent multi-tablet combination including a generic NRTI product.32–34 In Alberta, Canada, 19/257 (7.4%) clients who voluntarily initiated or switched to generic abacavir-lamivudine plus dolutegravir subsequently switched to the brand-name, single tablet equivalent after median 255 days (8.5 months) follow-up. In 2/257, (0.8%) a generic-related ADR was noted as the reason for change (one gastrointestinal upset, one allergic reaction). 33 In France, 4/51 (8.9%) clients who voluntarily de-simplified brand-name abacavir-lamivudine-dolutegravir or emtricitabine-tenofovir DF-rilpivirine switched back to the single tablet regimen within 6 months due to mild gastrointestinal or general symptoms attributed to the generics. 32 The authors noted that 3/4 clients had a history of anxiety, and the relationship between the reported ADR and generic ARVs was uncertain. In Spain, 93 clients were systematically switched from single tablet abacavir-lamivudine-dolutegravir to the equivalent two-tablet regimen. 34 After 6 months follow-up, 2/93 (2%) had a regimen change due to ADRs (worsening migraine, ‘burning mouth’) attributed to the generic abacavir-lamivudine.
Our large cohorts had low PSI ADR rates which were comparable to those seen in the above studies, in a similar follow-up period of approximately 9 months for HIV-Tx and 4–7 months for PrEP. The shorter follow-up for PrEP clients is explained by ongoing enrolment of new clients, and supply chain disruptions that delayed generic transition in some measurement periods. Most PSI reports were received one to 4 months following the generic substitution. The small proportion of PSIs reported outside the one-year-post-rollout measurement periods had similar characteristics as those analysed for the study outcomes (data not shown).
PSIs represented less than one quarter of overall ADR reports in most measurement periods. This proportion was higher (approximately one third of ADR reports) for abacavir-lamivudine; however, this product for HIV-Tx had the lowest overall ADR incidence (2% of persons treated); therefore the absolute number of PSI reports was small. Notably, abacavir-lamivudine was the first large-scale generic ARV rollout in BC and initial anxiety regarding generic ARVs may have exacerbated PSI reporting. Overall, the proportion of ADRs attributed to generic complaints in our study was lower than that observed by others. Jacquot et al. (2014) found that generic product complaints represented half of their pharmacovigilance program’s ADR reports following introduction of a policy requiring an ADR report to support ‘non-substitutable’ brand-name prescriptions. 35
Although the studied NRTI, NNRTI and PI products have different side-effect profiles, symptoms associated with PSI ADRs, were similar across all products, and were generally nonspecific gastrointestinal or general symptoms. In 5/9 cases of worsening neuropsychiatric symptoms attributed to substitution of generic efavirenz, there was prompt symptom resolution after switching to a different product version. A BC-CfE study of untimed efavirenz plasma concentrations before and after brand-to-generic transition (a retrospective analysis of stored samples) found no significant differences in efavirenz levels with brand and generic products. 36 None of our clients with efavirenz PSIs were included in this drug level study, and individualized drug level monitoring was not feasible, therefore possible individual differences in efavirenz absorption affecting generic tolerability cannot be ruled out. Eight of 11 clients with skin rash attributed to generic ARVs subsequently tolerated a different product version, suggestive of a reaction to pharmaceutical excipients. ADRs associated with generic medication excipients have been reported by others, including hypersensitivity reactions attributed to erythrosine, hypromellose, carmine and hydroxypropyl cellulose.37–39
Our study’s results must be interpreted in the context of several limitations. It is not feasible for a pharmacovigilance program to conclusively attribute a suspected ADR to a specific drug, 40 and under-reporting of ADRs is a widely acknowledged issue. 41 On the other hand, the requirement to provide ADR documentation in order to access brand-name medication could motivate reports of generic intolerance for the purpose of obtaining the brand-name product. 35 Studies of generic drug acceptability have observed patient attribution of lower treatment benefit and/or greater side effects with generic versus brand-labelled medication, termed a ‘nocebo’ effect.42,43 Client preferences or psychological discomfort with generics have also been noted as reasons for requesting brand-name drug.31,44 All these factors could influence PSI and overall ADR reporting patterns. We acknowledge that our study’s measurement of PSI incidence may be an underestimate of actual frequency, and that events classified as PSI ADRs may be a hybrid measure of true intolerance of a specific generic version, general ARV-related ADRs or symptoms from unrelated health issues mis-attributed to a particular generic formulation, and ADR reports motivated by a desire for brand-name medication.
In conclusion, ≥98% of clients who received generic ARVs for HIV-Tx or PrEP did not report ADRs attributed to the generic version during the first year following drug formulary transitions. In our experience, key components for successful, large scale, generic ARV substitution include clear communication with prescribers and clients regarding the reasons for, and safety of using generic ARVs, and having structured processes in place to monitor ADR reports and provide alternate generics or brand-name drug when necessary for client safety.
Supplemental Material
Supplemental Material - Adverse drug reactions attributed to generic substitution of antiretroviral medications among HIV treatment and pre-exposure prophylaxis clients in British Columbia, Canada
Supplemental Material for Adverse drug reactions attributed to generic substitution of antiretroviral medications among HIV treatment and pre-exposure prophylaxis clients in British Columbia, Canada by Katherine J Lepik, Olivia L Hunt, Nic Bacani, Lu Wang, Marianne Harris, Junine Toy, Taylor McLinden, Paul Sereda, Linda J Akagi, Erin Ready, Julio SG Montaner and Rolando Barrios in Antiviral Therapy.
Footnotes
Acknowledgements
We gratefully acknowledge the drug treatment program clients and health-care providers, and the technical support provided by Jason Trigg, Irena Filipovic, Daniela Uzelac and Inderpreet Ahuja.
Author’s note
At the time this work was conducted, Olivia Hunt and Taylor McLinden were employed by the BC Centre for Excellence in HIV/AIDS.
Author contributions
All authors contributed to study design, KJL, OLH, NB and LW prepared and analysed the data, KJL prepared the manuscript, and all reviewed the final article.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JSGM’s Treatment as Prevention (TasP®) research, paid to his institution, has received support from the BC Ministry of Health, Health Canada, Canadian Institutes of Health Research, Public Health Agency of Canada, Genome Canada, Genome BC, Vancouver Coastal Health and VGH Foundation. Institutional grants have been provided by Gilead Sciences Inc, Janssen, Merck Sharp & Dohme LLC, and ViiV Healthcare. MH has received honoraria, administered by the institution, for consulting fees and advisory board participation from Gilead Sciences Canada, Merck Canada, and ViiV Healthcare. All other authors declare they have no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
