Abstract
Background
Substantial body composition alterations have been reported after starting combined antiretroviral therapy (cART). We characterized a cohort of chronically infected and virologically suppressed (VL < 50 copies/ml) men (≥50 years old) living with HIV (MLWH) who were switched to integrase inhibitors (INSTI), and compared their body composition parameters and proinflammatory/endocrine profiles to age-matched MLWH on integrase inhibitor free (non-INSTI) regimens, taking into account neighborhood-level measures of socioeconomic status (SES). In addition, we used previously published HIV-seronegative men of the same age as controls.
Methods
We used dual energy X-ray absorptiometry to quantify body composition parameters, and measured plasma proinflammatory/endocrine markers in 56 MLWH. We compared body composition to a publicly available dataset of 450 HIV-seronegative men of similar age. Within the MLWH group, body composition and plasma proinflammatory/endocrine markers were compared between individuals on INSTI and non-INSTI regimens, accounting for SES.
Results
Men living with HIV tended to have a greater android/gynoid ratio compared to HIV-seronegative men (
Conclusions
Among cART-experienced MLWH, those receiving INSTI-containing regimens had modestly lower adiposity compared to non-INSTI MLWH, although these differences were explained by SES.
Introduction
Obesity and HIV are both disproportionately represented in lower income communities.
1
Rates of HIV infection among the urban poor are more than twenty times that of non-urban and/or individuals living above the federal poverty line.
2
Poor
Obesity has been associated with inflammation, insulin resistance, an increased risk of type 2 diabetes, cardiovascular disease (CVD), 6 and overall mortality. 7 Obesity poses a particular risk for people living with HIV (PLWH) as adipose tissue can also serve as an HIV reservoir and impair the efficacy of combination antiretroviral therapy (cART). 6 Although body mass index (BMI) is typically used to identify obese individuals, dual energy X-ray absorptiometry (DXA) provides detailed quantitative information about body composition and fat distribution. 8
Weight gain after cART initiation is common, with differences potentially due to the specific regimen. 6 Integrase inhibitors (INSTIs) have been associated with greater weight gain when administered to cART-naïve individuals6,8 and have been associated with new-onset diabetes mellitus. 9 The impact of switching to INSTI on whole-body and regional adiposity in people with prolonged exposure to cART remains unclear.10–12 Reports have shown an increase in adipose tissue upon switching from a non-INSTI regimen to an INSTI regimen 10 ; however, this increase was only seen within certain groups when the sample was subdivided by race, sex, and age. 11 Notably, although there is a known association with socioeconomic status and body composition, 13 as well as a known increased risk of HIV diagnosis for those of lower socioeconomic status,1,2 we are not aware of any study that combines HIV status, cART regimen, socioeconomic status, and body composition.
Given the unclear effects of INSTI on whole-body and regional adiposity, we measured body composition and adipose distribution in a cohort of chronically infected older (≥50 years old) men living with HIV (MLWH) who were virologically suppressed (<50 copies/mL) on cART and compared these parameters to a publicly available cohort of age-matched men without HIV. We evaluated for potential differences in body composition, proinflammatory cytokines, and hormones between MLWH who were receiving INSTI-based cART compared to non-INSTI-based cART. In particular, we focused on the effects of ADI, a measure of socioeconomic status, on body composition.
Methods
Participants were recruited from the Infectious Disease Clinic at Washington University in St. Louis and the WUSTL AIDS Clinical Trial Unit. All individuals were male, sedentary (self-reported exercising for less than 2 h per week in the 3 months prior to evaluation), ≥50 years old, HIV-positive, taking cART, and virologically suppressed. Participants self-reported their cART regimen, as well as previous medication history. All MLWH on INSTI (
Whole-body DXA scans were performed on all participants using a GE-Healthcare Lunar I scanner (Wauwatosa, WI). These scans quantified height, weight, total body fat percentage, fat mass, total mass, trunk fat, leg fat, and android/gynoid ratio using Encore Software (version 16). Fat mass index (FMI), total body fat percentage, and trunk-to-leg fat ratio were calculated. 7
Participant addresses were geolocated via the Google Maps API. Geocoordinates were then mapped to census tract via tigris. 14 The 2019 Area Deprivation Index based averaged on American Community Survey data from 2015 to 2019 was downloaded from the University of Wisconsin’s Neighborhood Atlas 15 and matched to participant census tracts.
Data from HIV uninfected males of similar age were downloaded from a publicly available dataset. 7 Total body fat percentage, FMI, android/gynoid ratio, and trunk-to-leg fat ratio were available. Published participant data was stratified by age group (either 50–59 years old or 60–69 years old), but actual participant ages were not available. Measures of socioeconomic status were also not available for this cohort. Other than selection on age group, no matching was performed.
Regarding markers of endocrine function and circulating proinflammatory markers, we collected standard blood chemistries (see Supplemental Table 1). We computed the Homeostatic Model Assessment for Insulin Resistance (HOMA-IR). 16
Statistical analysis
Demographic variables were compared using Kruskal–Wallis for continuous and Fisher’s exact test for categorical variables. A series of linear models were used to test for differences by HIV status or INSTI usage after correcting for age. For equations (1a) and (1b), the following variables were included as response variables: total body fat percentage, FMI, android/gynoid ratio, and trunk-to-leg fat ratio. Rather than using age as a continuous variable, participants were classified as either age 50–59 years old or ≥60 years old due to previously described limitations in data availability for HIV-seronegative controls.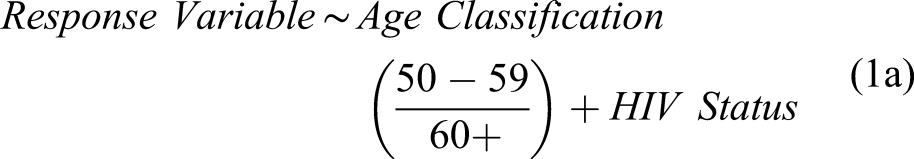
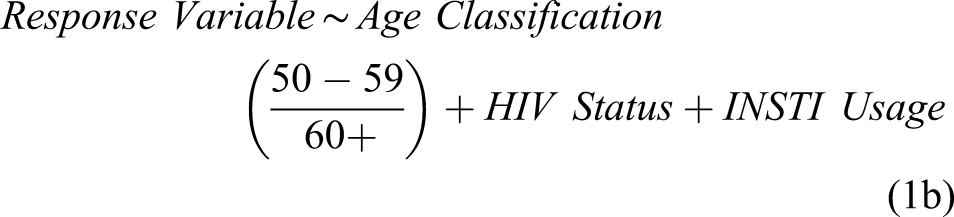
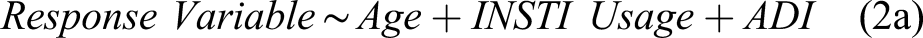
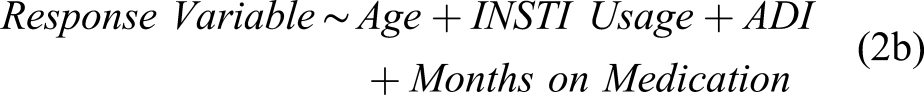
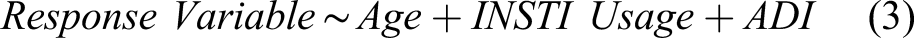
Results
Participant characteristics: Demographic characteristics for men living with HIV (MLWH) who were on integrase inhibitors (INSTIs) or not on INSTIs and comparable HIV-seronegative men. We present mean (standard deviation) for continuous measures and count (percentage) for proportional values. When values existed for all three groups,

*Body mass index (BMI). **Homeostatic model assessment for insulin resistance (HOMA-IR). ***combination antiretroviral therapy (cART).
MLWH versus HIV-negative men
Body composition characteristics between MLWH and seronegative controls: MLWH had significantly higher android/gynoid ratio compared to HIV-seronegative males. INSTI users had significantly lower markers of body fat (fat mass index, android/gynoid ratio and trunk-to-leg ratio) than all other participants. These results are associated with equations 1a and 1b.

INSTI versus non-INSTI MLWH
Body composition characteristics between men on INSTI and non-INSTI regimens: Differences in fat mass index and trunk-to-leg ratio were no longer observed once we control for socioeconomic status, as measured by Area Deprivation Index (ADI). Controlling for months on medication further eliminates observed differences in body composition; however, very few samples were available for analyses including time on medication. These results are associated with equations (2a) and (2b).

Discussion
We evaluated changes in body composition and adipose tissue distribution after switching of cART regimens in MLWH. The current analyses were performed within a cohort of older MLWH who have a long average duration of infection, consistent with the aging demographics of MLWH in the United States. 18 We also considered a neighborhood-level measure of poverty and opportunity, 15 finding relatively high levels of deprivation in our cohort, consistent with previous trends linking poverty and HIV positivity. 1 Consistent with previous work, we observed a higher android/gynoid ratio for cART-experienced MLWH compared to HIV-seronegative men.6,8 These observed differences aligned with prior studies despite demographic differences in cohorts (∼90% NHW for the HIV-seronegative controls, 64% Black for the MLWH).
Although some studies have observed weight gain in INSTI users, most were conducted within cART-naïve groups. 8 Body composition information for INSTI users who were not cART-naïve when switching is more equivocal. Within these studies, the general composition of American-based cohorts used was 80% male and between 30% and 50% African American or Black. 8 The cohort we present here is 100% male and 64% Black. Because race is frequently used as a proxy for many extenuating factors, 19 we instead focused on three possible explanations regarding potential effects of INSTI usage on weight gain: metabolic differences (as measured by the plasma biomarkers), cART experience (as measured by time on medication), and/or socioeconomic status (as measured by ADI).
After controlling for differences in age, we saw no relationships between INSTI use and any of the plasma biomarkers, in models that either included or excluded ADI and/or time on medication. Thus, we do not attribute observed body composition differences in measured hormonal or proinflammatory measures. We further did not observe any relationship between time on medication and body composition. However, this brings us to a major limitation of the study. We relied on participant self-reported data. We did not have time on INSTI distinct from time on all medications, meaning we were unable to assess length of time on either regimen as a relevant covariate. Further work in a cohort with more complete medical history is necessary to understand the importance of duration of infection, time on medication, and time on previous regimen prior to switching. Within the confines of the available data in this study, we observed no differences in body composition by time on cART.
We observed that INSTI users had a trend level association with lower total body fat percentage (
Of the factors considered, then, we conclude that our observed body composition differences are largely driven by sampling effects. Social determinants of health and the environment in which study participants live can have a substantial impact on study findings. Our findings highlight the importance of including sociocultural context of participants in analyses.
Supplemental Material
Supplemental Material - Socioeconomic status largely explains integrase inhibitors-related body composition differences in chronically infected men living with HIV
Supplemental Material for Socioeconomic status largely explains integrase inhibitors-related body composition differences in chronically infected men living with HIV by Julie K. Wisch, Sarah A. Cooley, Kevin E. Yarasheski, W. Todd Cade, Dominic N. Reeds, Brittany Nelson, Ruth Alemu5, Tricia H. Burdo, and Beau M. Ances in Antiviral Therapy
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kevin E. Yarasheski is currently employed by and holds equity in C2N Diagnostics, LLC unrelated to this work. Tricia H. Burdo is a member of the scientific advisory board and holds equity in Excision BioTherapeutics, Inc. unrelated to this work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Health (NIH) grants R01NR012907 (BA), R01NR012657 (BA), R01NR014449 (BA), R01DA054009 (BA and TB), P30DK056341 (KEY, DNR), P30DK020579 (KEY), and P41GM103422 (KEY). This work was also supported by the generous support of the Barnes-Jewish Hospital; the Washington University Institute of Clinical and Translational Sciences Foundation (UL1 TR000448); and the Hope Center for Neurological Disorders
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
