Abstract
Background
JNJ-4964 is a TLR7 agonist, which, via a type I interferon (IFN)–dependent mechanism, may enhance host immunity suppressed by persistent exposure to hepatitis B antigens in chronic hepatitis B.
Methods
PK and PD data were pooled from 2 studies involving 90 participants (n = 74 JNJ-4964, dose range 0.2–1.8 mg; n = 16 placebo) in a fasted state. Food effects on PK were studied in 24 participants (1.2 or 1.25 mg). A population PK model and PK/PD models were developed to characterize the effect of JNJ-4964 plasma levels on the time course of IFN-α, IFN-γ–inducible protein 10 (IP-10 or CXCL10), IFN-stimulated gene 15 (ISG15), neopterin and lymphocytes following single and weekly dosing in healthy adults. Covariate effects, circadian rhythms and negative feedback were incorporated in the models.
Results
A 3-compartment linear PK model with transit absorption adequately described JNJ-4964 PK. Bioavailability was 44.2% in fed state relative to fasted conditions. Indirect response models with maximum effect (Emax) stimulation on production rate constant (kin) described IFN-α, IP-10, ISG15 and neopterin, while a precursor-dependent indirect response model with inhibitory effect described the transient lymphocyte reduction. Emax, EC50 and γ (steepness) estimates varied according to PD markers, with EC50 displaying substantial between-subject variability. Female and Asian race exhibited lower EC50, suggesting higher responsiveness.
Conclusions
PK/PD models well characterized the time course of immune system markers in healthy adults. Our results supported sex and race as covariates on JNJ-4964 responsiveness, as well as circadian rhythms and negative feedback as homeostatic mechanisms that are relevant in TLR7-induced type I IFN responses.
Introduction
Toll-like receptor 7 (TLR7) is a member of the pathogen-recognition receptor family that activates the host’s innate and adaptive immune responses. TLR7 is located within the endosomes of plasmacytoid dendritic cells (pDCs) and B cells. 1 TLR7 activation results in the induction of type I interferon (IFN) responses, proinflammatory cytokines and chemokines that can directly or indirectly stimulate hepatitis B virus (HBV)-specific antiviral effects. 2 An example of a direct effect would be IFN-α inhibiting pregenomic RNA encapsidation, enhancing covalently closed circular DNA (cccDNA) degradation, and causing epigenetic modification of cccDNA transcription. 3 Indirect mechanisms include the expansion of natural killer (NK) cells and HBV-specific T-cell response.4,5 Oral administration of a TLR7 agonist has another potential benefit of triggering the gut-associated lymphoid tissues with resulting innate immune activation at the liver, and thus avoiding the undesirable systemic adverse effects of IFN-based therapies.2,6
Several oral TLR7 agonists have been investigated in clinical studies as a promising strategy for treatment of chronic hepatitis B virus (CHB) infection. 7 For example, vesatolimod (also known as GS-9620) pharmacokinetics (PK) and pharmacodynamics (PD) has been studied in healthy adults, 8 as well as CHB,9,10 hepatitis C virus, 11 and human immunodeficiency virus (HIV) patients.12,13 A double prodrug TLR7 agonist, RO7020531, has also been studied in healthy adults14,15 and CHB patients.16,17 High between-subject variability was observed in terms of the fold-change increase in the TLR7 PD markers, such as IFN-α, IFN-γ–inducible protein 10 (IP-10), neopterin and IFN-stimulated gene 15 (ISG15), indicating variable sensitivities.8–11,14 In addition, there was insufficient understanding of the extent of stimulation needed to elicit appreciable clinical efficacy towards HBV DNA decline and/or hepatitis B surface antigen (HBsAg) reduction when administered alone or in combination with nucleos(t)ide analogues.9,10 Meanwhile, higher doses are associated with adverse events (AEs), such as transient flu-like symptoms and lymphocyte reduction.8,14
JNJ-64794964 (JNJ-4964; formerly AL-034 or TQ-A3334) is an oral TLR7 agonist with potent anti-HBV activity in vitro, inducing antiviral IFNs, proinflammatory and anti-inflammatory cytokines and chemokines in human whole blood cells and peripheral blood mononuclear cells, 18 and HBsAg seroconversion in preclinical studies. 19 Two phase 1 clinical trials NCT03285620 (Study 1, also known as AL-034-1201, sponsored by Janssen Biopharma Inc) 20 and ChiCTR1800015418 (Study 2, sponsored by Chia Tai-Tianqing Pharmaceutical Group Co., Ltd.) 21 have been conducted in healthy adults to evaluate the safety, tolerability, PK and PD of JNJ-4964. In these studies, single doses of 0.2–1.8 mg resulted in dose-dependent transient induction of cytokines and chemokines and transient reduction in lymphocytes, consistent with TLR7-induced type I interferon responses.2,22 Weekly doses of 1.25 mg resulted in induction that returned to baseline before administration of the subsequent dose, and the response after the last dose was not enhanced nor reduced compared to after the first dose. 20 In a separate analysis of data from Study 1, transcriptomic and phenotypic immune cell changes in peripheral blood after JNJ-4964 dosing showed increased expression of cell-surface activation molecules on NK cells and B cells. 23
The work presented here focused on the development of mathematical models to characterize the population PK/PD effects of JNJ-4964 on serum IFN-α, IP-10, neopterin, expression of ISG15 fold change, and lymphocytes; and (2) to explore the effects of covariates (weight, sex and race [Asian vs non-Asian]). In addition, physiological mechanisms, such as feedback regulation of the type I IFN responses24,25 and circadian rhythms,26,27 were also tested in the models. These models can serve as a simulation tool to optimize the study designs of future investigation of HBV treatment strategy involving a TLR7 agonist toward achieving a functional cure of HBV.
Methods
Study design
Study 1 included a total of 42 participants (32 on JNJ-4964 and 10 on placebo). In the first phase, 8–10 participants were randomized per cohort and received a single oral dose of either JNJ-4964 0.2 mg (n = 6), 0.6 mg (n = 6), 1.25 mg (n = 8), 1.8 mg (n = 6), or placebo (n = 2/dose cohort) under fasted conditions. Following a ≥6-week wash-out period, participants receiving JNJ-4964 1.25 mg under fasted conditions were given 1.25 mg (n = 7) or placebo (n = 1) under fed conditions (a standard meal consumed before dosing). In the second phase, 1 cohort of 8 participants received doses of JNJ-4964 1.25 mg (n = 6) or matching placebo (n = 2) in fasted conditions once weekly for 4 weeks.
Study 2 included a total of 48 participants (42 on JNJ-4964 and 6 on placebo). In the single-ascending dose phase, participants were randomized to receive a single oral dose of either JNJ-4964 0.2 mg (n = 2), 0.5 mg (n = 8), 1.0 mg (n = 8), 1.8 mg (n = 8), or placebo (n = 2/dose cohort except the 0.2-mg cohort) under fasted conditions. In the food effect cohort, 16 participants were randomized to receive the 1.2 mg dose either fasted on Day 1 and with food on Day 15, or with food on Day 1 and fasted on Day 15.
Sampling schedules for collections of PK, IFN-α, IP-10, neopterin and ISG15 for both studies are provided in Table S1 in the Supplementary Information.
Assays
Plasma concentration of JNJ-4964 was evaluated using a validated liquid chromatography with tandem mass spectrometry method (lower limit of quantification [LLOQ] 1 pg/mL). IFN-α was assessed by an enzyme-linked immunosorbent assay (ELISA; LLOQ was 12.5 pg/mL for Study 1 and 1.491 pg/mL for Study 2). IP-10 was analysed by the appropriate Luminex assays (LLOQ was 25 pg/mL). Neopterin was analysed by ELISA, and ISG15 expression fold change was analysed using quantitative polymerase chain reaction.
Population PK/PD analyses
Population PK/PD analyses were conducted using nonlinear mixed-effects models implemented using NONMEM version 7.4.3 (ICON Development Solutions, Ellicott City, MD), with a first-order conditional estimation with interaction (FOCEI) method in ADVAN6. 28 Random effects for interindividual variability in parameters were considered and assumed to be log-normally distributed with covariance matrix Ω.
Observed data were fitted after natural-logarithmic transformation on both sides. 29 Residual unexplained variability was entered in an additive manner: ln(Yobs,ij) = ln(Ypred,ij) + ε, where Yobs,ij and Ypred,ij are observed and predicted concentrations, respectively, of a participant i at time j, and ε is residual unexplained variability, assumed normally distributed with mean 0 and variance σ2. Concentrations below their respective LLOQ were incorporated into the likelihood calculation using the M3 method. 30 As a model evaluation step, diagnostic plots were assessed and visual predictive checks based on 100 simulation replicates were performed.
The schematic of the PK/PD model is shown in Figure 1. A brief description is provided below, while the model equation details can be found in the Supplementary Information. JNJ-4964 PK/PD model diagram. 
A dose-proportional 3-compartment PK model with transit compartment absorption
31
was used to describe the PK of JNJ-4964 after oral administration. The effect of food on PK was modelled as a relative bioavailability term for the fed state. The model-predicted plasma concentration time course based on the individual PK parameter estimates was used to drive the stimulation or inhibition Emax-type function where
To account for the TLR7 signal transduction delay, 3 transit compartments32,33 were used and the rate constant was estimated as
Simulation
The population PK/PD models with the interindividual variability were subsequently used for clinical trial simulation to inform the dose-PD response and maximum concentration (Cmax)-PD response relationship. PD response was defined as an IFN-α, IP-10, ISG15, and neopterin fold change ≥2, and a lymphocyte nadir of ≤0.65 × 109 cells/L. The simulation included a total of 5000 participants, with a 1:1:1:1 ratio of male non-Asian, male Asian, female non-Asian and female Asian.
Ethics
All participants in both studies provided informed consent. Both studies adhered to the International Council for Harmonisation guidelines on Good Clinical Practice and Declaration of Helsinki principles.
Results
Participants and data
Baseline demographics.
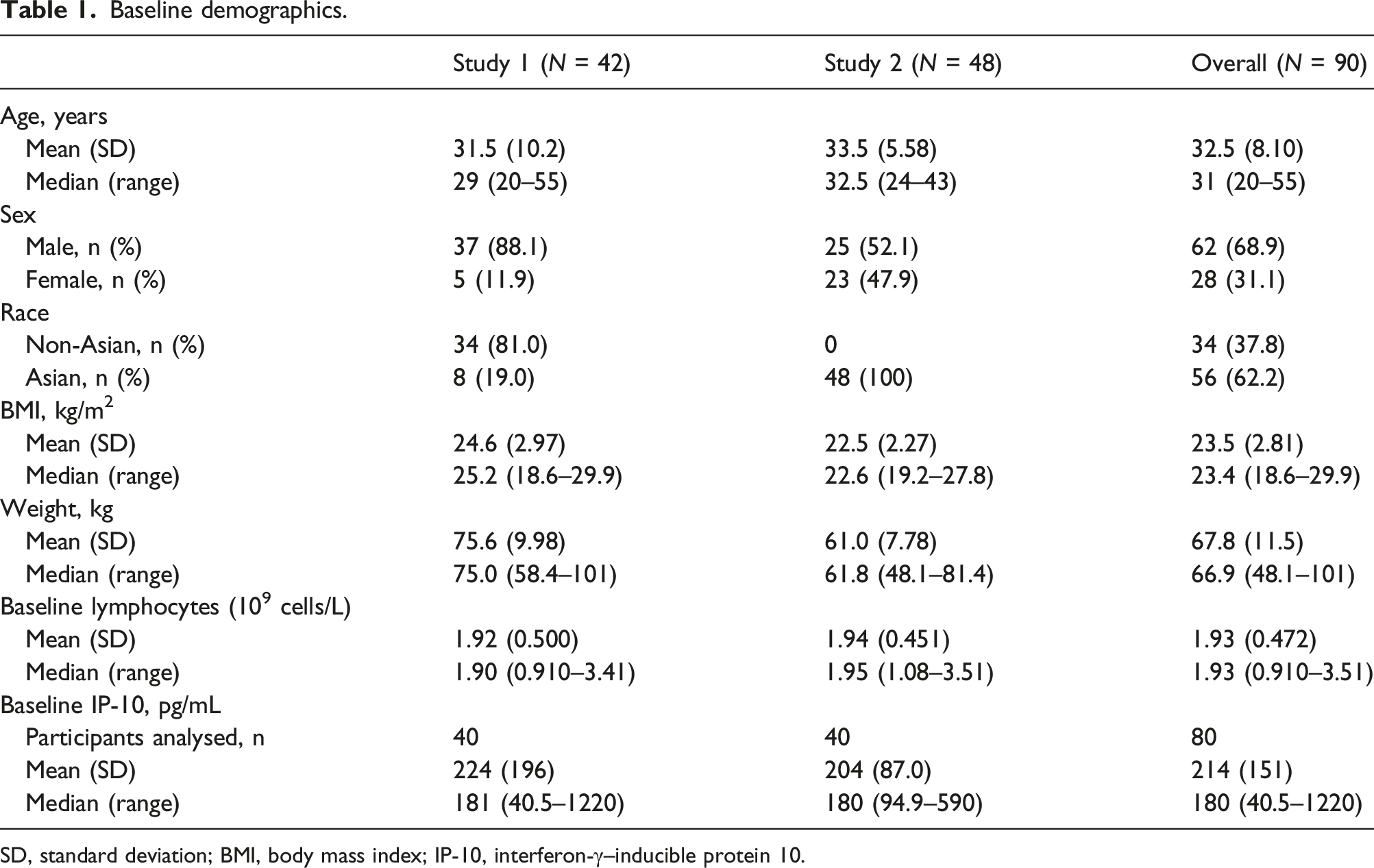
SD, standard deviation; BMI, body mass index; IP-10, interferon-γ–inducible protein 10.
The number of participants and observations of the PK/PD data included in the analyses were summarized in Table S2 in the Supplementary Information. All available lymphocyte data from 90 participants were included in the analysis. Elevated baseline IP-10, neopterin and ISG15 were observed for 2 placebo participants, one of whom also had elevated baseline IFN-α; these levels declined over time. Because their PD profiles were not consistent with the assumption of steady-state baseline in the absence of drug, these 2 placebo participants were excluded from the IFN-α, IP-10, neopterin and ISG15 analyses.
Study 2 did not have neopterin measurements. In addition, another 8 participants from the Study 2 food effect cohort who were administered JNJ-4964 with food on Day 1 and in fasted condition on Day 15 did not have IFN-α, IP-10, or ISG15 samples. Therefore, the number of participants for population PK/PD modelling of lymphocytes, IFN-α, IP-10, neopterin and ISG15 were 90, 80, 80, 40 and 80, respectively.
All models successfully reached convergence criteria based on the FOCEI method in NONMEM. The population PK and most of the PK/PD parameter estimates had reasonable relative standard errors, as presented in Table S3 in the Supplementary Information. The negative feedback rate constant ktol had higher uncertainty. The visual predictive check plots assessment shown in Figure 2 showed no bias, and the median and 5th and 95th percentile of the simulation (100 replicates) were in good agreement with the distribution of the observed data. Time course of model-predicted and observed data stratified by study and dose cohort. (A) Plasma concentration of JNJ-4964. (B) Lymphocytes. (C) IFN-α. (D) IP-10. (E) Neopterin. (F) ISG15 fold change. Observed (black lines and circles) are overlaid with median (red) and 5th and 95th percentiles (purple) of simulations (n = 100 replicates). IFN, interferon; IP-10, interferon-γ–inducible protein 10; ISG15, interferon-stimulated gene 15; JNJ-4964, JNJ-64794964; QW, once weekly.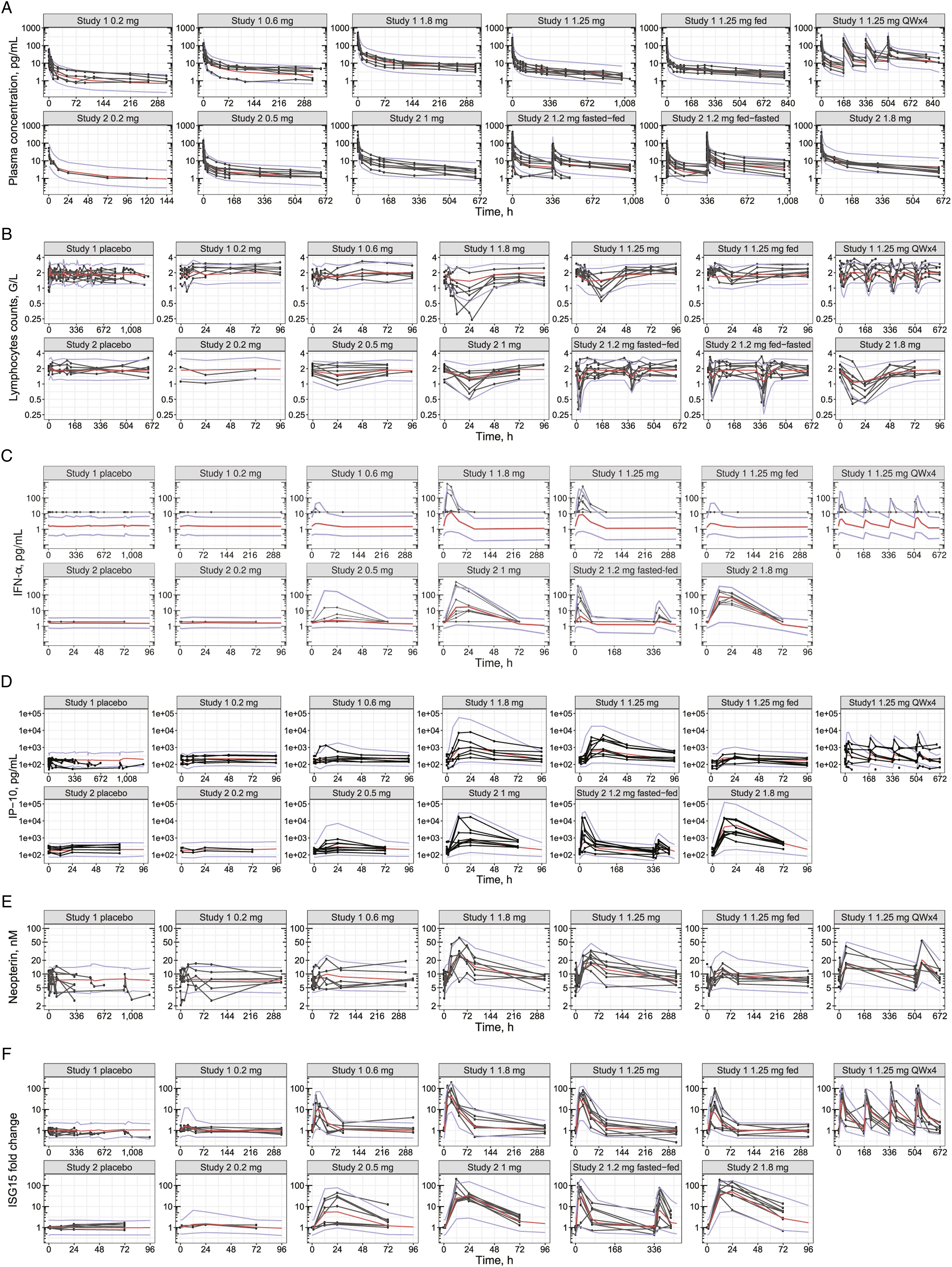
The PK of JNJ-4964 was found to be dose proportional for the 0.2–1.8-mg dose range evaluated in these studies. The median time to maximum plasma concentration (tmax) was 0.5 h, followed by a rapid initial distribution phase and a long and variable terminal elimination half-life (t1/2 term; median range 150–591 h), as obtained from non-compartmental analyses (NCA). 20 In Study 1, despite the wash-out period of ≥6 weeks following JNJ-4964 1.25 mg (fasted), predose plasma concentrations (Cpredose) were quantifiable for all participants (mean [standard deviation] Cpredose 1.85 [0.597] pg/mL). 20 This meant comparison of non-compartmental PK parameters between the second fed dose versus the first fasted dose would be confounded by the residual drug level in the system from the first fasted dose. Similarly, in Study 2, the fed versus fasted dose was only 2 weeks apart. On the other hand, the population PK model allowed for a better estimation of the true food effect by pooling the data from the 2 studies and appropriately accounting for the residual drug levels that were carried over prior to the second drug administration.
The 3-compartment PK model in Figure 1 described the PK of JNJ-4964 well. Interindividual variability was estimated on the volume of distribution (VC) and relative bioavailability (F). The bioavailability of JNJ-4964 in the fed condition was estimated to be 44.2% compared to the same dose under fasted conditions. Most compounds with high solubility and low permeability (Biopharmaceutics Classification System III), such as JNJ-4964, have negative food effects. 38 However, there was no universal rule and the mechanisms underlying the food effect can be different. 39 Neither sex, race, nor weight were significant covariates on the PK parameters. Based on the typical 3-compartmental parameters of the JNJ-4964 PK model in this analysis, the apparent terminal half-life was 628 h. The large variability in the observed terminal half-life (150–591 h) and the discrepancy between the observed and the model-predicted terminal half-life (median [5th–95th percentiles] of 627 [614–676] hours) could be attributed to the dependency of the NCA-based half-life on the linear regression with lack of observations at later time points. 40
Simulation
Based on these models, simulation was performed to illustrate JNJ-4964 dose- and concentration-response relationships for the relevant PD response (Figure 3 and Table S4 in the Supplementary Information). Among the explored PD effects, the predicted order of greatest to least responsiveness was fold change ≥2 ISG15, IP-10, neopterin, IFN-α and lastly nadir lymphocytes ≤0.65 G/L. In addition, across sex and race categories, the predicted order of greatest to least responsiveness was female Asian, female non-Asian, male Asian, and lastly male non-Asian. Model-based dose–response (A) and Cmax-response (B) relationship following a single fasted dose of JNJ-4964. Responses include maximum fold change ≥2 of ISG15 (purple), IP-10 (red), neopterin (orange), IFN-α (green) and nadir lymphocyte ≤0.65 G/L (blue). Overall population was simulated as 1:1:1:1 for male non-Asian, male Asian, female non-Asian and female Asian, respectively. Error bars at the bottom indicate the median, 5th–95th percentiles of the JNJ-4964 Cmax in the simulation (Table S4 in the Supplementary Information). Cmax, maximum concentration; IFN, interferon; IP-10, interferon-γ–inducible protein 10; ISG15, interferon-stimulated gene 15; JNJ-4964, JNJ-64794964.
In addition to relating JNJ-4964 Cmax with the PD effects, in these clinical studies a positive relationship was observed between increases of systemic IFN-α and the appearance of pyrexia AEs and low lymphocyte count (Figure S1 in the Supplementary Information). In particular, pyrexia AEs were observed alongside IFN-α levels ≥100 pg/mL: 9/15 (60%) of participants with a maximum IFN-α concentration ≥100 pg/mL had a pyrexia AE versus 0/66 (0%) in cases where the maximum IFN-α concentration was <100 pg/mL. While the models do not predict the pyrexia AE, the observations from the clinical studies stated previously could inform the target setting for the PK/PD modelling work.
To illustrate the sex and race effects, Figures 4A and 4B show the subgroup Cmax-response relationship for maximum IP-10 fold change ≥2 and maximum IFN-α ≥100 pg/mL, respectively. Additional Cmax-response relationship plots for maximum ISG15, neopterin fold change ≥2 and nadir lymphocytes ≤0.65 G/L are provided in Figure S2 in the Supplementary Information. Model-based Cmax-response relationship following a single fasted dose of JNJ-4964, stratified by male non-Asian (purple), male Asian (teal), female non-Asian (green) and female Asian (red) for maximum IP-10 fold change ≥2 (A) and maximum IFN-α ≥100 pg/mL (B). Cmax, maximum concentration; IFN, interferon; IP-10, interferon-γ–inducible protein 10; JNJ-4964, JNJ-64794964.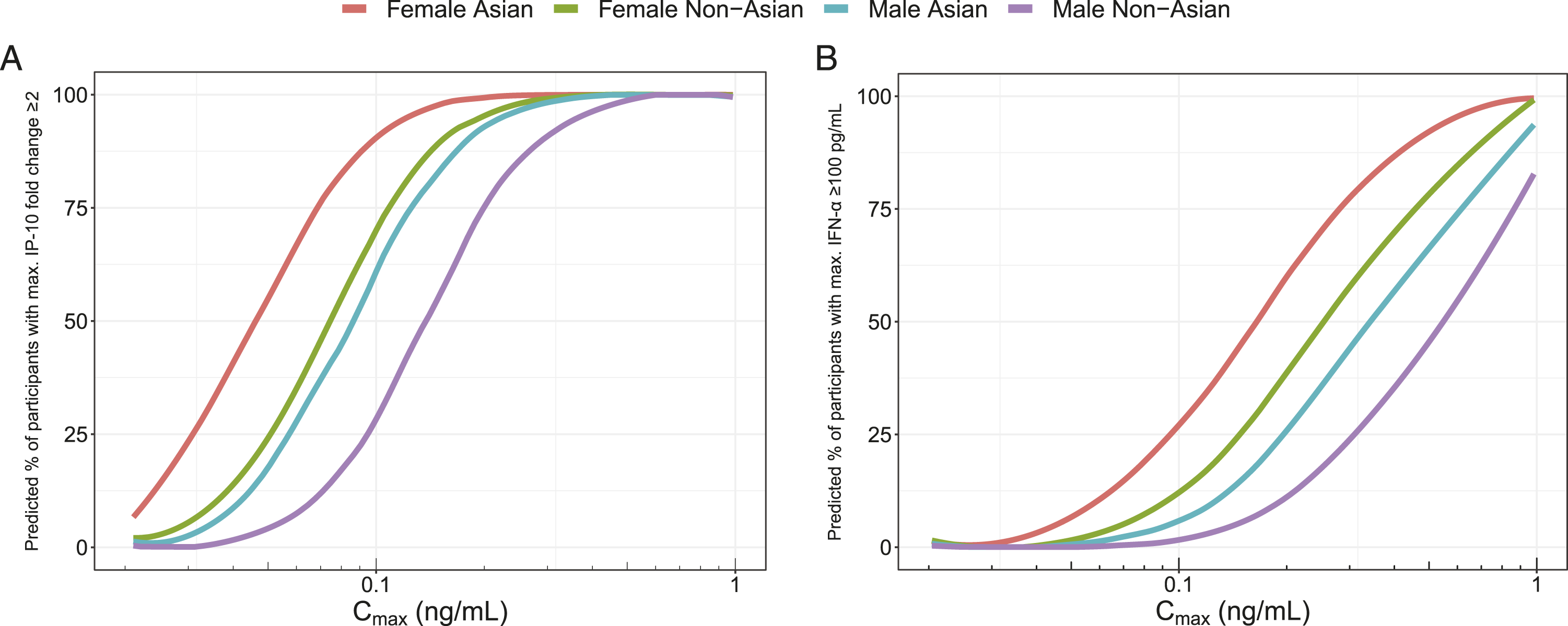
Discussion
The pooled clinical data from the 2 studies presented in this analysis supported characterization of a sigmoid Emax stimulation function for the PK/PD effects of JNJ-4964 on IFN-α, IP-10, neopterin, ISG15 and lymphocytes. The estimates for Emax, EC50 and γ vary among the PK/PD models, indicating a varying degree of induction across these PD markers. 6 This possibility is supported by observations for the TLR7 agonist vesatolimod (GS-9620) in HIV controllers: IFN-α levels after dosing with vesatolimod were relatively low compared to increases in IP-10 and downstream cellular activation in CD69+CD56dim and CD69+CD56bright NK cells. 13
The components of negative feedback, sex and race effect and circadian variation were incorporated when the data allowed. Consequently, negative feedback was not included for lymphocytes since the inclusion of the precursor-mediated indirect response did not allow identification of a negative feedback. The race effect on EC50 was also not included for neopterin given that Study 2 did not have neopterin data and Study 1 had limited sample size (8 Asians out of the 42 participants). Along the same vein, the circadian rhythm was estimated as allowed by more intensely sampled PD data, such as in the case of lymphocytes and IP-10.
The PK/PD properties of JNJ-4964 in cynomolgus monkeys were also previously assessed, focusing on IP-10 and ISG15. 41 The models for cynomolgus monkeys were used as a starting point for the current analyses. The PK was also described using a 3-compartmental model with transit compartment absorption, but with a saturable pre-systemic elimination process to account for the greater than dose-proportional increase in exposure with the dose range of 0.05–15 mg/kg. The PD data were described using an indirect response model with signal transduction; however, the stimulation function was a power function instead of an Emax-type equation due to a dose range of 0.05–0.6 mg/kg for the PD data. Because of these differences, the PK/PD parameters could not be directly compared between cynomolgus monkey and human data. Interestingly, the observed individual maximum induction of IP-10 was similar between cynomolgus monkeys and human data, which was approximately 10 ng/mL.
The γ parameter indicates the steepness of the JNJ-4964 concentration-effect curve. Our models estimated γ as 1.27, 5.21, 4.11, 3.01 and 3.02 for lymphocytes, IFN-α, IP-10, neopterin and ISG15, respectively. The high γ in all markers besides lymphocytes suggests underlying positive feedback mechanisms associated with TRL7 agonism, such as those mediated by interferon regulatory factor (IRF)-7 and stimulator of interferon genes, or TLR7 autoinduction.42–44 On the other hand, simultaneously, there can be negative regulators playing roles in maintaining homeostasis, such as TRIM35, SOCS and USP18.24,25 The existence of negative feedback mechanisms was supported by the fact that the model fit improved. The observed data suggested that after type I IFN responses, as measured by IFN-α, IP-10, neopterin and ISG15, reached their respective peaks, these responses returned to baseline more quickly than predicted by a model without negative feedback.
The effect of sex and race was introduced through a proportionality factor on the JNJ-4964 EC50 parameter for female in reference to male sex and for Asian in reference to non-Asian race, respectively. The effect of female sex on EC50 was estimated between 50% and 74% (0.526, 0.503, 0.559, 0.634 and 0.736 for lymphocytes, IFN-α, IP-10, neopterin and ISG15, respectively). The lower EC50 in females compared to males indicates higher responsiveness, which can be related to many possible hypotheses: 1) TLR7 is an X-linked gene that escapes the X-chromosome inactivation; therefore, females have a double dosage of the TLR7 gene compared to males; 45 2) females have higher basal IRF5 expression, which impacts the production of IFN-α; 46 3) females have a higher percentage of IFN-α–producing pDCs;46–48 4) females have higher expression of IFN-α/β receptor subunit 2 on pDCs; 48 and 5) the effect of sex hormone oestrogens through oestrogen receptor signalling to enhance the pDC responses upon TLR7 stimulation. 47
The effect of Asian race on JNJ-4964 EC50 was estimated between 60% and 75% (0.678, 0.659, 0.637 and 0.724 for lymphocytes, IFN-α, IP-10 and ISG15, respectively). This higher responsiveness in Asian compared to non-Asian participants may be related to the association between TLR7 gene variants and TLR7 expression level. In particular, TLR7 single nucleotide polymorphism (SNP) rs3853839 G allele was associated with higher expression of TLR7 compared to the C allele,49,50 and G allele has a higher frequency in Asian compared to Caucasian individuals (0.777 vs 0.168, respectively, based on data from the 1000 Genomes Project). 50
In the flow cytometry peripheral blood mononuclear cell analyses from participants of Study 1 single-ascending dose cohorts, 23 increased CD69 expression on NK cells (a phenotypic marker for NK cell activation) was observed from a JNJ-4964 Cmax of ∼80 pg/mL. However, the largest percent change from baseline CD69 expression on NK cells was not observed in those participants with the highest JNJ-4964 Cmax, but rather in those participants with the strongest PD responses (i.e., IFN-αmax levels ≥100 pg/mL). In addition, increased CD86 expression (costimulatory molecule, upregulated upon activation) on B cells was also explored, but there was no significant correlation between increase in CD86 expression on B cells and JNJ-4964 Cmax. However, consistent with the increased CD69 expression on NK cells, increased frequency of B cells expressing CD86 was observed in those participants showing the strongest PD responses (i.e., IFN-αmax levels ≥100 pg/mL). This suggests that it is not PK exposure only that results in downstream immune cell activation. The modelling results indicate variability on PD responsiveness, with sex and race as covariates on EC50, while sex and race are not covariates for PK. Given that JNJ-4964–induced cytokine responses (e.g., IFN-α) correlate with downstream immune cell activation, simulation based on the PK/PD models (for example, Figures 3 and 4) can be useful to inform future clinical studies.
Conclusion
The combined dataset of the two phase 1 studies has made possible the development of a robust set of population PK and PK/PD models for JNJ-4964 effects on IFN-α, IP-10, ISG15, neopterin and lymphocytes in healthy participants. Compared to the data from each study alone, the larger sample size and more balanced distribution of sex and race in the combined dataset enabled better characterization of the PK/PD model parameters and covariate effects. JNJ-4964 PK is dose proportional up to 1.8 mg, with bioavailability estimated as 44.2% in the fed versus fasted condition. No sex, race, weight, body mass index, or SNP effect (TLR7 rs3853839, TLR7 rs179008, IL28B rs12979860, IL28B rs8099917) was identified on PK. While no covariates were identified for the PK model, sex and race were significant covariates on EC50 of the PD markers. Female and Asian participants had higher responsiveness to JNJ-4964, likely related to TLR7 gene X-chromosome non-inactivation and a higher frequency of TLR7 SNP rs3853839 G allele than C allele, respectively. The modelling analyses supported circadian rhythms and negative feedback as homeostatic mechanisms that are relevant in TLR7-induced type I IFN responses. Weekly dosing frequency did not result in any changes in response (e.g., no tachyphylaxis) and can be explored in future clinical development.
Supplemental Material
Supplemental material - Population pharmacokinetic/pharmacodynamic models of JNJ-64794964, a toll-like receptor 7 agonist, in healthy adult participants
Supplemental material for Population pharmacokinetic/pharmacodynamic models of JNJ-64794964, a toll-like receptor 7 agonist, in healthy adult participants by Liviawati S Wu, Yue Hu, Edward J Gane, Leen Slaets, An De Creus, Yanhua Ding, Junqi Niu, Christian Schwabe, Nele Goeyvaerts, Zhongnan Xu, Dandan Huo, Marianne Tuefferd, Inge Verbrugge, Pieter Van Remoortere, Ullrich Schwertschlag and Joris Vandenbossche in Antiviral Therapy
Footnotes
Acknowledgements
We thank the study investigators, site staff and volunteers who participated in this study. Writing and editorial support for the development of this manuscript were provided by Kurt Kunz, MD, MPH (Lumanity Communications Inc.), and were funded by Janssen Pharmaceuticals.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LSW, LS, ADC, NG, MT, PVR and JV are employees of Janssen Pharmaceuticals and Johnson & Johnson stockholders. US is an independent trial consultant. JD, JN and YH did not declare any conflicts of interest related to the study. ZX and DH are employees of Chia Tai-Tianqing Pharmaceutical Group Co., Ltd. EJG has been an advisor and/or speaker for AbbVie, Gilead Sciences, Janssen, Novartis, Roche and Merck. CS has advised for Johnson & Johnson and Vir Biotechnology.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Janssen Pharmaceuticals.
Data accessibility statement
The data sharing policy of Janssen Pharmaceutical Companies of Johnson & Johnson is available at https://www.janssen.com/clinical-trials/transparency. As noted on this site, requests for access to the study data can be submitted through Yale Open Data Access (YODA) Project site at ![]() .
.
Supplemental material
Supplemental material for this article is available online.
