Abstract
Background
Few pharmacokinetic data were reported on dispersible tablets despite their increasing use. One hundred fifty HIV-infected children receiving lamivudine were enrolled in the MONOD ANRS 12,206 trial. Three galenic forms were administered: liquid formulation, tablet form and dispersible scored tablet.
Method
HIV-infected children <4 years old were enrolled in the MONOD ANRS 12,206 trial designed to assess the simplification of a successful 12-months lopinavir-based antiretroviral treatment with efavirenz. Lamivudine plasma concentrations were analysed using nonlinear mixed effects modelling approach.
Results
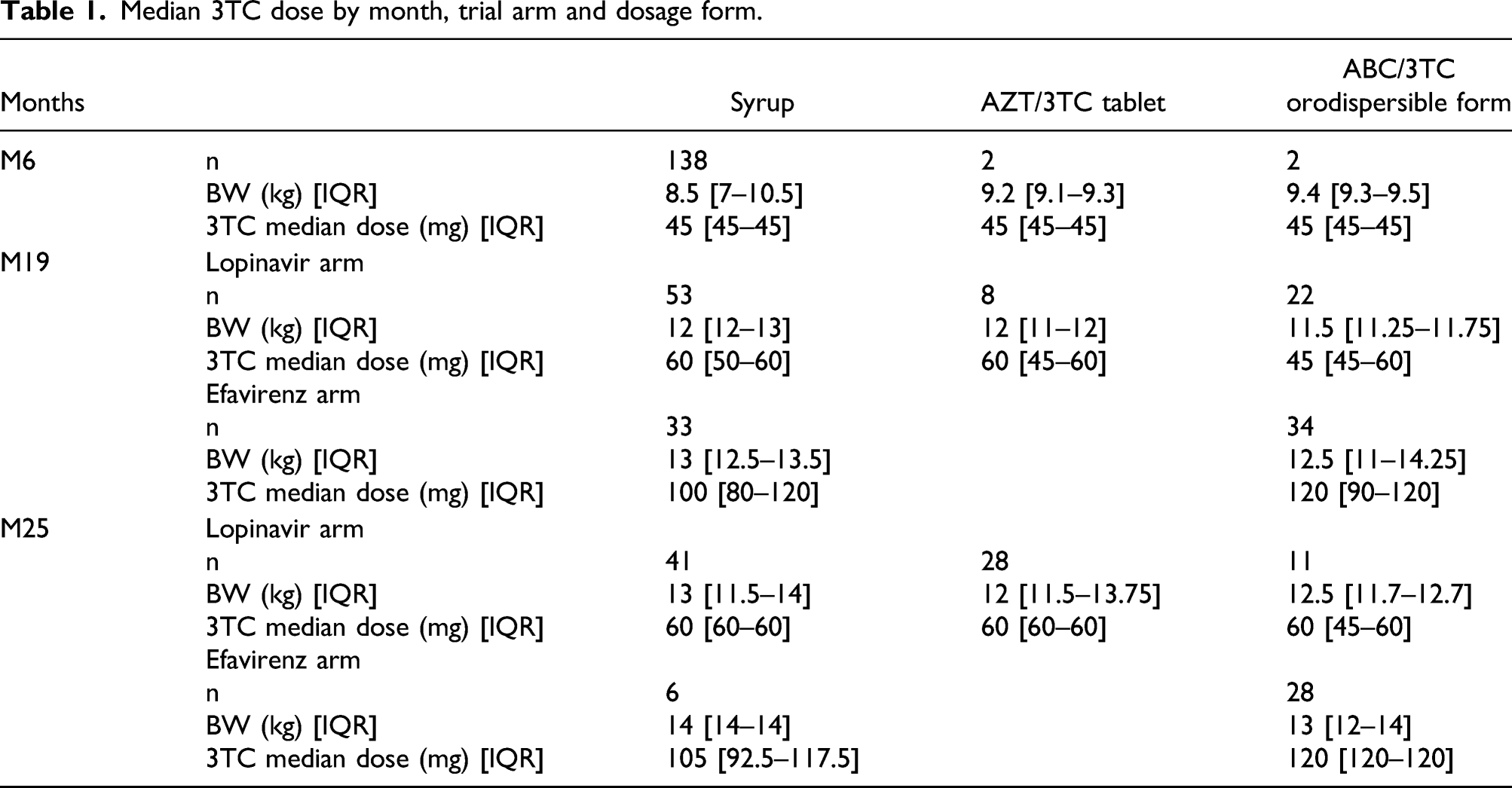
One hundred and fifty children (age: 2.5 years (1.9–3.2), weight 11.1 (9.5–12.5) kg (median (IQR)) were included in this study. Over the study period, 79 received only the syrup form, 29 children switched from syrup form to tablet 3TC/AZT form, 36 from syrup to the orodispersible ABC/3TC form and two from the 3TC/AZT form to the orodispersible ABC/3TC form. The 630 lamivudine concentrations were best described by a two-compartment model allometrically scaled. Galenic form had no significant effect on 3TC pharmacokinetic.
Conclusion
This trial provided an opportunity to compare three galenic forms (liquid formulation, tablet form and dispersible scored tablet) of lamivudine in the target population of young HIV–1-infected children. Galenic form had no significant effect on lamivudine pharmacokinetics.
Introduction
Immediate initiation of combined antiretroviral therapy (cART) in all children living with HIV under 2 years of age regardless of their immune or clinical status has been recommended by the WHO since 2008. 1 However, the availability of drugs in appropriate formulations remains limited for infants and toddlers. 2 Development of orodispersible fixed dose combinations (FDCs) has improved adherence, but little is known about the evaluation of these galenic forms.
Lamivudine (3TC) is used in combination with highly active antiretroviral therapy for children living with HIV, available in solid and liquid forms, in single entity and fixed dose combination products. However, little information is given on the dosage forms used. Contradictory results were reported in studies comparing solid and liquid formulations, from no difference to an increase of 55% of the exposure to 3TC.3–5 This study allows to compare three different galenic forms of lamivudine.
Methods
The ANRS 12,206 MONOD randomised trial (see group composition in appendix 1) was aimed to assess a simplified once daily cART based on efavirenz versus a twice daily regimen based on lopinavir/ritonavir among West African children living with HIV and virologically suppressed after a 12-month cART based on LPV/r initiated before the age of two. 6

Therapeutic education was given systematically by the assistant pharmacist and the social worker when the drugs were given to families. Three galenic forms were subsequently used over the MONOD trial duration: first, a liquid formulation (lamivudine 10 mg/mL); then, a tablet form (3TC/zidovudine [AZT] 30 mg/60 mg Avocomb Kid®); last, a dispersible, scored tablet (Abacavir [ABC]/[3TC] 60 mg/30 mg produced by Cipla®). The dosage used using syrup formulation was 10 mg/mL (4 mg/kg every 12 h in the lopinavir group or 8 mg/kg every 24 h in the efavirenz group), fitting with WHO weight-bands (M6). Then, tablets or orodispersible fixed dose formulation tablets for ABC-3TC using WHO weight-bands dosing was done to substitute for the syrup formulations at M19 and M25. In order to come as close as possible to the dosage in mg/kg, the tablets were given either whole or in half a tablet.
Study design and procedures were already published. 7 For the MONOD non-inferiority trial, the statistical parameter of interest was the difference in viral success rate 12 months after the switch, defined as the rate in the LPV arm (control) minus the rate in the EFV arm, using a chi-square test. We aimed to obtain a viral success of at least 76% at 12 months post-switch. We pre-specified that the margin of the 95% confidence interval of the difference in the primary outcome between the two arms less than 14% would meet our criteria for non-inferiority. Based on our anticipated enrolment of 146 children with 73 children per arm, we expected an 80% power to detect this difference. 7
The pharmacological data analyses (time, concentration) have been done with a population approach using the MONOLIX software.
Pharmacokinetic specific blood sample were scheduled during the initial cohort phase of the trial at visit M6 and after that during the randomised simplification phase at visit M19 and visit M25. The pharmacokinetic parameters (PK) of each antiretroviral drug received by children have been measured by one or two blood samples per child during each visit drawn at two random points in time after the drug intake. The times of last drug intakes were recorded precisely to allow an estimation of different pharmacokinetic parameters. Different time samples were defined as follows: - T0, just before taking the drugs; - T1, 1/2 h at 1:30 after taking; - T2, 2:30–35 h after taking - T3, about 8 h after taking.
Plasma 3TC concentrations were determined according to a validated method. After a simple protein precipitation of 10 μL of plasma with methanol, the chromatographic separation was performed using an UPLC coupled to a mass spectrometric detection MS/MS (Xevo TQ-S from Waters). The analytical column was an Acquity BEH phenyl (1.7 μm, 2.1*50 mm). The range of quantification was 1–3000 ng/mL. The nonlinear mixed effect modelling programme MONOLIX 2019R2, with SAEM algorithm and left censoring of concentrations below the LOQ was used to analyse the data. One- or two-compartment structural models, multiplicative, additive or combined residual variability, and exponential inter-subject variability on each parameter were tested. An allometric model was added to represent physiological evolution: P i = PSTD × (BW i /BWSTD)PWR, where PSTD is the standard value of parameter for a patient with the standard body weight value and P i and BW i are the parameter and body weight of the ith individual, allowed to standardise the parameters estimates for a median weight. From the data, the PWR (power) exponents could be estimated. However, these were typically 0.75 for clearance parameters and 1 for volumes of distribution parameters according to the theory of the allometric scale. Four covariates have been tested: sex, age, body-weight and galenic forms. All of the covariates were tested via an upward model building. A covariate was selected if (i) its effect was biologically plausible, (ii) it produced a minimum decrease of 6.63 U (chi-square test, 1 df, p < 0.01) in the objective function value (OFV) and (iii) it produced a reduction in the variability of the pharmacokinetic parameter, assessed by the associated inter-subject variability. The most significant covariate of all the covariates tested was added in an intermediate model. In this intermediate model, all other covariates were tested and the most significant one was selected. This process was repeated until no more covariate was significant. Models were evaluated thanks to diagnostic graphics and prediction corrected visual predictive check.
Results
One hundred and fifty children (77 females and 73 males) and 630 plasma concentrations were available for pharmacokinetic evaluation. Median [IQR] age was 2.5 years [1.9–3.2] and 11.1 [9.5–12.5] kgs for body-weight. The median lamivudine dose per day administered was 100 mg, for doses ranging from 50 to 300 mg. Since children were included in the lamivudine study prior to the lopinavir/efavirenz switch, 142 children received twice daily and 58 received once daily. Indeed, some children received a twice daily dose first and then a once daily dose if they were randomised to the efavirenz arm.
Median 3TC dose by month, trial arm and dosage form.
Population pharmacokinetic parameters of lamivudine.

aThe typical parameters represent a patient weighing 70 kg according to an allometric model: (typical value) (typical parameter) (body weight/70)PWR, where PWR 0.75 for the CL and Q terms and 1 for the V c and V p terms. K a , absorption rate constant; CL/F, apparent elimination clearance, V c /F apparent central volume of distribution; V p /F, apparent peripheral volume of distribution; Q/F, inter-compartmental clearance; residual variability estimates; and, interindividual variability estimates.
bRSE %, relative standard error (standard error of estimate/estimate 100).

Prediction-corrected visual-predictive check for lamivudine twice daily (left) and once daily (right). Grey areas represent 95% CIs of 5th, 50th and 95th simulated percentiles. Lines are empirical (observed) 5th, 50th and 95th percentiles. Circles are the non-censored, cross the censored observations. Black circles are observations from the syrup form, green circles from the tablet form and red circles from the orodispersible form.
Median (95% CI) value obtained from the model with an estimated bioavailability value for each of the three galenic forms.

Discussion
A two-compartment model with first order absorption and elimination best described lamivudine pharmacokinetics, as in previous studies in adults and children.3,8–11 The apparent elimination clearance (CL/F= 33 L/h/70 kg) was consistent with previous adult studies,13,14 and the effect of body-weight, here in allometric scale, was also added on lamivudine apparent clearances and volumes of distribution.3,4,9,11,12,15,16 This population model allowed to investigate the effect of three galenic forms on 3TC pharmacokinetic. Indeed, there are few pharmacokinetic data on new galenic forms, especially orodispersible forms. In order to expand access to treatment for these children, appropriate drug formulations are required, particularly the simpler dose combination mini- or scored tablets preferred by caregivers and children as young as three years of age. 16 Solid formulations also reduce cost and promote adherence as compared to solutions, which often require refrigeration, have a short shelf-life, often require clean water (which is not available everywhere), are not easy to store or transport, and are often complicated for health-care workers to prescribe and for caregivers to administer. 17 The MONOD trial provided a unique opportunity to compare three galenic forms (liquid formulation, tablet form and dispersible scored tablet) of lamivudine in the target population of young HIV–1-infected children who were ready to change from liquid formulation to solid formulations of lamivudine. We showed that the three galenic forms were not statistically different in terms of pharmacokinetic properties. These results can be extrapolated to younger and lighter children because the model is an allometric model. Thus, if we know the weight of each child, this model can be estimated; however, it should be confirmed by prospective data. For both forms, apparent parameters were increased by less than 10% compared to syrup and the effect was not significant. Even if the study was not designed for the comparison of galenic forms and does not allow to have a significant coefficient, the value of the effect is very low and makes it possible to conclude that there will be no clinical impact. These results are consistent with those of Bouazza et al. 3 and Piana et al. 4 who reported there were no differences in the pharmacokinetics of lamivudine after administration of the solid or liquid dosage form.18,20 In contrast, Vanprapar et al. 19 and Kassirye et al. 5 have observed higher 3TC exposure in children with the dispersible tablets versus oral liquid. These studies, including our current study, are population PK studies, in which it is more difficult to identify a difference in absorption. Increased absorption could be an explanation for the higher exposure of the dispersible tablet formulation versus oral liquid in two other studies with full PK curves (as Cmax was increased with 55 and 59%). Finally, an explanation for this difference found in these two studies could be the way of splitting the tablets which is not necessarily very reproducible.
In conclusion, the three dosage forms (syrup, tablet, orodispersible tablet) had no influence on the pharmacokinetics of lamivudine. Our results provide reassuring added value for the use of cART drug regimen among young children living with HIV in the context of scarce therapeutic options.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
