Abstract
Ergotism is a rare cause of peripheral vasoconstriction with varying presentation depending on the affected vessels. Coronary vasospasm is a rare, potentially fatal manifestation of ergotism. Cytochrome P-450 isoenzyme CYP3A metabolizes ergot alkaloids and their derivatives; thus, concomitant use of ergotamine and CYP3A inhibitors significantly increases ergotism risk. The antiretroviral drug darunavir boosted with cobicistat potently inhibits CYP3A. A few deleterious interactions are described in the literature when combining ergotamine with boosted human immunodeficiency virus protease inhibitors. Herein, we describe a patient who presented to the emergency department with profound coronary vasospasm arising from ergotamine’s interaction with darunavir and cobicistat. Emergency coronary angiography revealed no critical atherosclerotic stenosis, but prolonged coronary artery spasm. After prompt cardiopulmonary resuscitation, intravenous vasodilator treatment, and 14-day extracorporeal membrane oxygenation application, the patient fully recovered and was discharged. Concomitant use of ergot alkaloids and their derivatives could be disastrous for patients treated with cobicistat-boosted darunavir.
Keywords
Introduction
Highly active antiretroviral therapy is the standard treatment for human immunodeficiency virus (HIV), and adverse drug reactions are occasionally reported. Ritonavir, a protease inhibitor came into medical use in the 1990s, and ritonavir-boosted antiretroviral agents are reported to induce deleterious drug–drug interactions due notably to their strong inhibitory effect on CYP3A, and one of the notorious drug–drug interactions is related to concomitant use with ergot alkaloid derivatives[1-3], which is often used for migraine and postpartum hemorrhage clinically due to its pharmacological effect of vasoconstriction [4,5]. Several new antiretroviral drugs and boosters have been developed during the past decades, including cobicistat, a novel CYP3A inhibitor with similar effects as ritonavir [6,7], which has the potential to induce deleterious reactions when interaction with CYP3A-metabolized drugs[8]. Here, we present a rare complication of coronary vasospasm related to cobicistat-boosted darunavir interactions with ergotamine and review the mechanism and acute management strategy.
Case report
A 39-year-old male with multidrug-resistant HIV virologically suppressed on dolutegravir (50 mg once daily) and darunavir/cobicistat (800/150 mg once daily) presented to the emergency department with increasing chest pain and headache. According to the patient’s statement and the medical records, the patient took his first dose of 1 mg ergotamine tartrate obtained from a local clinic for suspected migraine, where he did not disclose his HIV status, so the local clinic was not aware that he was under cobicistat-boosted darunavir treatment. Then, he went to sleep without specific complaints mentioned on the night before the day he visited the emergency department. On the following morning, he had headaches and increasing chest pain around 2 h after taking his second dose of 1 mg ergotamine tartrate accompanied by the antiretroviral agents. He was hemodynamically stable upon arrival, and the electrocardiogram showed sinus rhythm; modest leukocytosis was present (13.8X103/μL) with normal serum biochemistry and cardiac markers. Approximately 1 h after admission to the emergency department, abrupt ventricular fibrillation occurred, and spontaneous circulation returned after cardiopulmonary resuscitation with defibrillation for about 20 min. Emergency coronary angiography was promptly performed and revealed diffuse coronary artery spasm (Figures 1(a) and (b)) with transient vessel dilation after intra-arterial nitroglycerin injection; no critical atherosclerotic change was detected (Figures 1(c) and (d)). Coronary angiography. Coronary artery spasm over the A, left anterior descending artery (LAD), left circumflex artery (LCX), and their branches; and B, right coronary artery (RCA) and its branches in left anterior oblique (LAO) view; vessels dilated over the C, LAD, LCX, and their branches; and D, RCA and its branches after intra-arterial nitroglycerin injection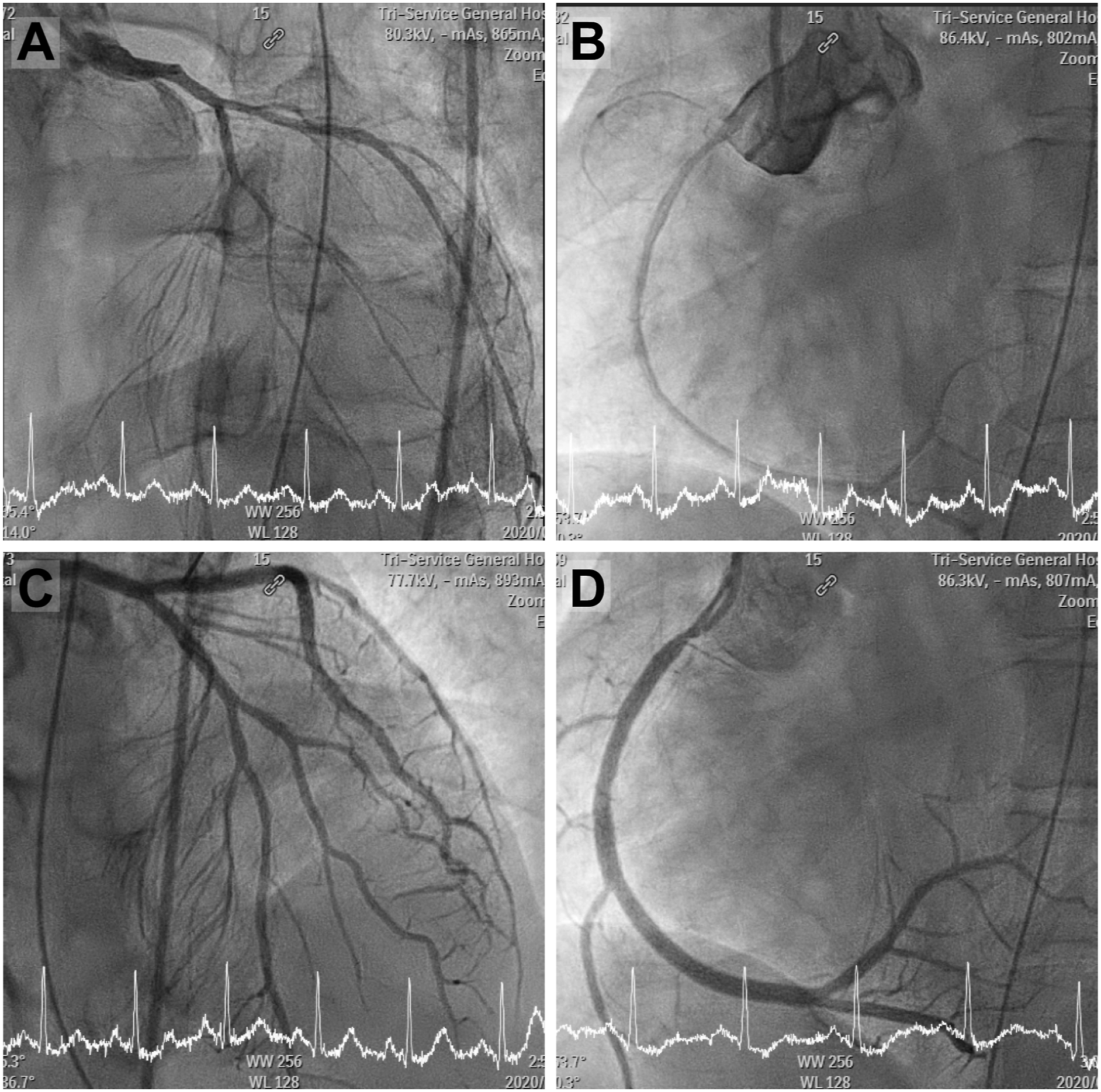
On admission to the intensive care unit, three pulseless electrical activity (PEA) episodes occurred that suspiciously correlated with prolonged coronary vasospasm and cardiogenic shock. Extracorporeal membrane oxygenation (ECMO) was initiated. Transthoracic echocardiography revealed generalized dyskinesia over the left ventricle with a 15% estimated ejection fraction.
The patient’s condition gradually stabilized during intravenous infusion of nitroglycerine, heparin, and amiodarone with ECMO support; repeated echocardiography showed restoration of the ejection fraction to 40% on day 8. ECMO was discontinued after 14 days of application; the patient was discharged uneventfully after 23 days of hospitalization. Transthoracic echocardiography 1 week after discharge from the hospital showed normal systolic and diastolic left ventricle function with a 65% estimated ejection fraction.
Discussion
Ergotism is a rare ischemic complication of vasoconstriction caused by ergot alkaloid overdose or drug interactions, mostly affecting lower extremity vessels [4,1,2]. Ergotamine-containing preparations may produce coronary vasospasm and reduce coronary microcirculatory blood flow, causing arrhythmias, cardiogenic shock, and sudden cardiac death [3,6,7]. The incidence of ergotism is low, around 0.01% (0.5/100.000 patient-years) [8], but may greatly increase when concurrent use with cytochrome P-450 isoenzyme CYP3A-inhibiting drugs, as CYP3A metabolizes ergotamine [4,5,8]. Ritonavir, a protease-inhibiting antiretroviral agent and potent CYP3A inhibitor has mostly been reported to interact with ergotamine-containing preparations [3-5]. Our patient took darunavir, a HIV protease-inhibiting antiretroviral drug primarily metabolized by CYP3A, which acts as CYP3A substrate and inhibitor [9,10], and cobicistat, a pharmacokinetic booster of darunavir and a novel CYP3A inhibitor with effects similar to ritonavir [8,11]. Coadministration of ergotamine with cobicistat and darunavir can drastically slow ergotamine metabolism, increasing its toxicity [8-11]. The calculated Naranjo score of the patient is 6, which revealed the adverse effect probably caused by ergotism and precipitated by the drug–drug interaction.
The long-lasting contractile response of ergot alkaloids in isolated human coronary arteries persists even after repeated washout in vitro [12]. This may explain our patient’s prolonged coronary artery spasm, cardiogenic shock, and repeated PEA episodes even after emergency coronary angiography with intra-arterial nitroglycerin injection. Treatments include immediate ergotamine-containing preparation discontinuation and prompt vasodilating agent administration. Pharmacological intervention includes calcium channel blockers, oral prazosin hydrochloride, heparin, and catheter-based systemic vasodilator therapy with nitroglycerin, prostacyclin, and prostaglandin E2 [4,5]. Emergency coronary angiography with intra-arterial injection of vasodilators and timely ECMO are recommended for lethal ECG presentation with hemodynamic changes. We believe this to be the first report of refractory coronary vasospasm resulting from adverse interactions between ergotamine, cobicistat, and darunavir.
Conclusion
Since protease inhibitors and boosters are widely applied antiretroviral therapies, clinicians must review HIV patients’ medication in detail and be prudent when prescribing concomitant drugs to prevent possible adverse drug–drug interactions. Clinicians and healthcare providers can use resources, such as the Liverpool HIV drug interaction website (www.hiv-druginteractions.org), to prevent and identify potential drug–drug interactions when dealing with patients treated with antiretroviral agents.
Footnotes
Author contribution
Cheng-En Wu: Conceptualization and writing original draft, Wen-I Liao: Writing-review and editing, Chun-Hsien Wu: Resources, Shih-Hung Tsai: Supervision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
