Abstract
Cidofovir is a broad-spectrum antiviral agent that has shown efficacy against skin lesions caused by human papillomavirus (HPV). We present a case of extensive verruca vulgaris lesions refractory to imiquimod that was responsive to topical cidofovir therapy, and analyze other case series in the literature of successful treatment of benign HPV-associated skin lesions with topical cidofovir. Topical cidofovir’s favorable response rate and tolerability make it a useful treatment option for patients of differing ages and immune status who have nonmalignant HPV-associated skin lesions and desire topical therapy.
Introduction
Cidofovir is a broad-spectrum antiviral agent that possesses activity against several DNA viruses. It is a potent nucleoside analog of deoxycytidine monophosphate, and its mechanism of action involves induction of apoptosis in infected cells via accumulation of pro-apoptotic proteins such as p53. 1 It has been used to treat infections with herpes simplex virus (HSV), molluscum contagiosum, and human papillomavirus (HPV). 2 Whereas systemic cidofovir has numerous side effects, topical cidofovir is generally well-tolerated, except for two cases of acute kidney injury reported in association with its use to treat resistant HSV infections. 3 However, both of these cases involved potential confounding factors, including concomitant use of medications known to induce kidney injury. As a result of its overall favorable tolerability and response rate, use of topical cidofovir for HPV-associated skin lesions refractory to other treatment modalities has gained acceptance. Herein, we present a case of large surface area plaque verruca vulgaris refractory to topical imiquimod therapy that was successfully treated with topical cidofovir, and we consider other case series in the literature employing topical cidofovir for benign HPV-associated skin lesions refractory to other treatment modalities. Taken together, this case and those reported in the literature support use of topical cidofovir for HPV-associated lesions for both immunocompetent and immunocompromised patients across a broad range of ages and lesion locations.
Case Report
A 43-year-old woman of African descent presented with multiple large verrucous plaques located on both palms and the right foot, present for 6 months. The lesions had previously been treated with multiple modalities, including cryotherapy, surgical excision, and grafting by plastic surgery. The patient presented to dermatology seeking a second opinion for non-surgical treatment options. Her past medical history was notable for chronic back pain and atopic dermatitis, and she used clobetasol propionate 0.05% ointment twice daily for dermatitis flares.
Physical examination revealed well-demarcated, hyperkeratotic, exophytic, hypopigmented verrucous plaques located on both palms and the medial arch of the right foot (Figure 1). This presentation was unusual in that the lesions were plaque-like and ranged in diameter from 3 to 9 cm, instead of the more typical nodular or papular lesions measuring millimeters in diameter. Serologic tests for human immunodeficiency virus (HIV), hepatitis B virus (HBV), and hepatitis C virus (HCV) were negative. Complete blood count, comprehensive metabolic panel, and immunoglobulin profiles were within normal limits. Verrucous plaques located on both palms (left) and the medial arch of the right foot (right) before treatment with topical cidofovir.
Three punch biopsy specimens were taken, from lesions on each palm and from the arch of the right foot. Prominent papillomatosis, compact hyperkeratosis with focal hemorrhage, hypergranulosis, and acanthosis were present, yielding findings diagnostic of verruca vulgaris. Topical imiquimod was attempted twice daily with nightly occlusion, 5 days per week for 4 months, but the lesions remained unresponsive. During a return visit, the risks, benefits, and potential side effects of systemic cidofovir, topical cidofovir, intralesional bleomycin, and topical 5-fluorouracil were discussed. The patient elected to attempt topical cidofovir 3% cream, which was prescribed for use twice daily. The 3% topical cidofovir was compounded using 0.75 g cidofovir 75 mg/mL (Vistide injection, Gilead Sciences, Inc., Foster City, CA) in 22.5 g of Eucerin cream (Beiersdorf AG, Hamburg, Germany).
After 8 weeks of treatment, the verrucous lesions had been reduced to pink granulated plaques. Cidofovir was discontinued at this point, and the patient was instructed to apply Vaseline. 15 weeks after initiation of treatment, the patient showed gross re-epithelialization of affected areas and focal repigmentation (Figure 2). 15 weeks after initiating topical cidofovir therapy, the patient demonstrates re-epithelialization and focal repigmentation.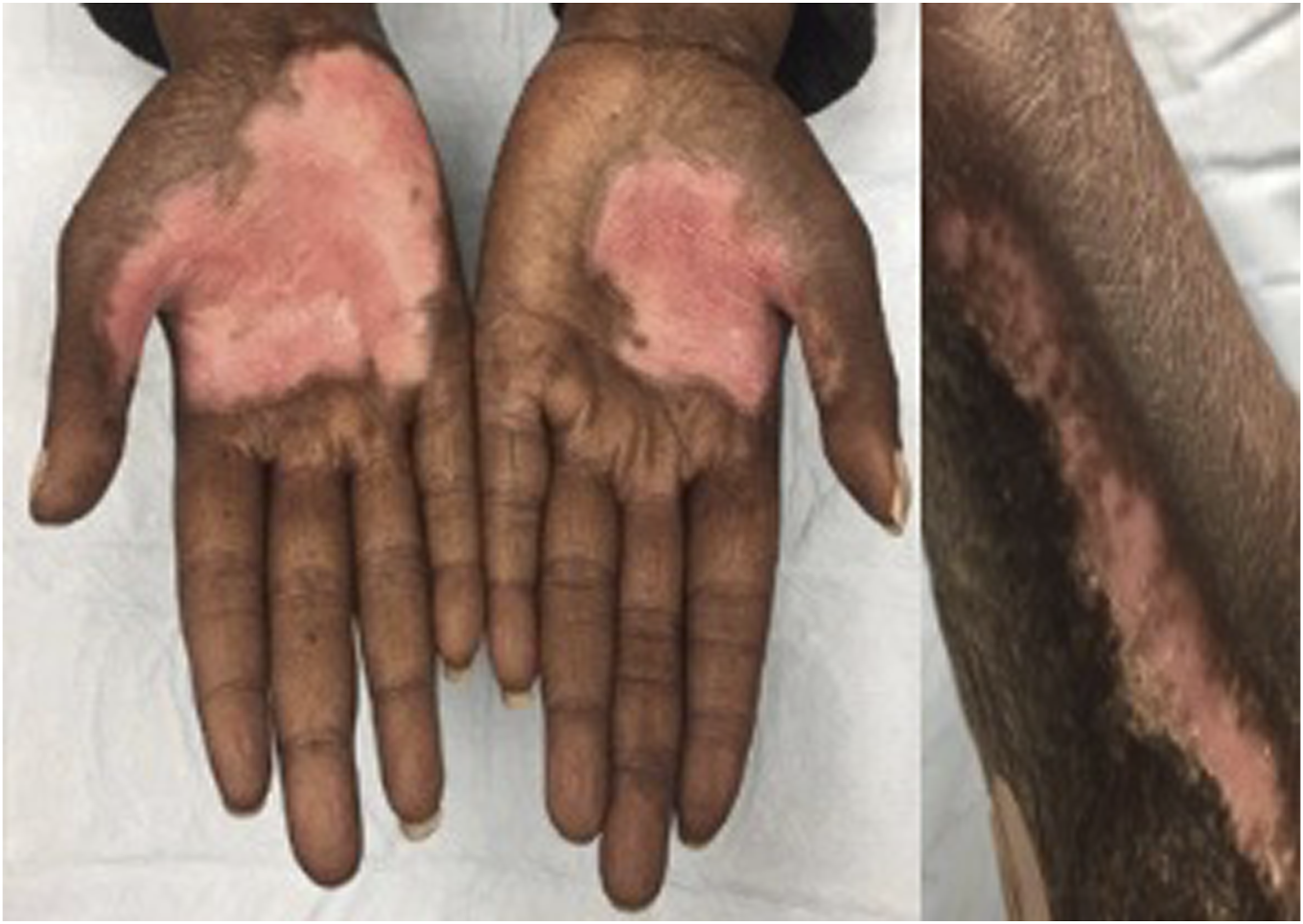
Discussion
Studies utilizing topical cidofovir for cutaneous HPV-associated lesions.

The diverse array of patients who demonstrated positive response to topical cidofovir according to the literature was notable, corroborating this as a widely applicable treatment option in patients of varying age, immune status, and lesion presentation. In conjunction with the multitude of literature-reported successes in treating HPV-associated lesions with topical cidofovir, our case substantiates use of this treatment in patients with severe refractory disease.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
