Abstract
This cross-sectional study investigated social appearance anxiety in individuals with Alopecia Areata (AA), a chronic autoimmune condition causing visible, non-scarring hair loss. A total of 129 AA patients and 142 age- and sex-matched healthy controls (18–65 years) completed the Social Appearance Anxiety Scale (SAAS) and the Hospital Anxiety and Depression Scale (HADS); the AA group additionally completed the Dermatology Life Quality Index (DLQI) and a Visual Analog Scale (VAS). SAAS scores were markedly higher in AA than controls (65.32 ± 8.45 vs 21.45 ± 9.32, p < 0.001). Within AA, SAAS showed a moderate positive association with perceived severity (VAS; r = 0.304, p < 0.05). HADS-Anxiety/Depression did not differ significantly between groups. In the AA cohort, DLQI averaged 12.3 ± 5.4 (median 12, IQR 9–16), with 62.8% ⩾ 10 and 10.9% ⩾ 20. DLQI correlated most strongly with perceived severity (VAS; r = 0.45, p < 0.001) and clinical visibility (r = 0.37, p = 0.002), with comparatively smaller associations with lesion count/area (r = 0.26/0.34). Taken together, these findings indicate that AA’s psychosocial burden is driven primarily by appearance- and visibility-related factors rather than generalized anxiety/depression or sheer disease extent. Routine dermatologic care should incorporate targeted psychosocial assessment—particularly for patients with visible-site involvement—to facilitate timely support.
Keywords
Introduction
Alopecia Areata (AA) is a chronic, immune-mediated dermatologic condition characterized by non-scarring hair loss that typically presents in sharply defined patches (Gilhar et al., 2012). These lesions most commonly affect the scalp, beard, eyebrows, and eyelashes, but may also extend to other body regions. With a global prevalence of approximately 1%–2%, AA follows an unpredictable trajectory, ranging from limited patchy hair loss to complete scalp (alopecia totalis) or total body involvement (alopecia universalis) (Duque-Estrada et al., 2013; Villasante et al., 2015). Although the disease is somatically asymptomatic, its visible manifestations—especially when affecting cosmetically and socially salient areas—can exert a profound psychosocial impact (Hunt and McHale, 2005).
In the context of psychodermatology, AA is increasingly recognized as a condition where psychological distress is not merely a secondary outcome but a core component of disease burden. The visibility of hair loss, particularly on facial regions, often leads to significant disruptions in self-image, social interactions, and emotional well-being (Jafferany, 2007) Unlike many other dermatologic conditions, the psychosocial impact of AA is often disproportionate to the objective severity or surface area affected. Instead, factors such as anatomical site visibility, symbolic significance of the hair-bearing region, and the individual’s subjective perception of disfigurement play a crucial role in modulating psychological responses (Boukthir et al., 2020; Picardi et al., 2000). Recent work further highlights that dermatological conditions can undermine both self-esteem and perceived social support, reinforcing the need to address psychosocial dimensions alongside cutaneous findings (Costeris et al., 2021).
To comprehensively assess these multidimensional burdens, standardized and validated psychometric instruments have become essential in clinical research. Tools such as the Social Appearance Anxiety Scale (SAAS), the Hospital Anxiety and Depression Scale (HADS), and the Dermatology Life Quality Index (DLQI) provide structured, reproducible metrics for evaluating emotional distress, appearance-related social fears, and quality-of-life impairment in dermatologic populations (Finlay and Khan, 1994; Hart et al., 2008; Nijenhuis and van der Hart, 2011). While inherently based on self-report, these instruments have demonstrated strong construct validity and are considered reliable indicators of patient-experienced psychological outcomes. Their structured nature enables reproducible quantification and facilitates comparison across studies, making them suitable for evidence-based psychosocial evaluation.
The present study aims to bridge the gap between clinical dermatologic findings and psychological outcomes in AA by integrating detailed clinical parameters—such as lesion size, number, subtype, and anatomical localization—with condition-specific psychometric assessment. Prior studies have often examined AA from either a dermatologic or psychiatric perspective in isolation, limiting the ability to contextualize psychosocial distress within concrete clinical phenotypes. By contrast, our study adopts a multimodal framework to examine how lesion visibility and topographical distribution influence levels of social appearance anxiety.
We hypothesize that involvement of highly visible or socially symbolic regions (e.g. the eyebrows or eyelashes) will be associated with significantly higher SAAS scores, regardless of total lesion burden. This hypothesis reflects a growing recognition that psychosocial impairment in dermatology is often driven more by perceived visibility and social salience than by objective disease extent. Through this approach, we aim to offer a more granular understanding of AA’s psychological impact and to inform targeted mental health interventions within dermatologic care pathways.
Materials and methods
Study design and setting
This comparative cross-sectional analysis was prospectively conducted at the Dermatology Outpatient Clinic of the Departments of Psychiatry and Dermatology, Faculty of Medicine, between March 2021 and August 2023. The study protocol followed STROBE guidelines to ensure methodological transparency.
Ethical compliance
Approval was obtained from the Non-Interventional Clinical Research Ethics Committee of Van Yüzüncü Yıl University (Protocol No: 2020/02-14; February 21, 2020). All participants provided written informed consent in accordance with the Declaration of Helsinki after receiving a comprehensive study briefing.
Sample Population and Recruitment Strategy
Participant Selection
The study enrolled 271 participants: 129 AA patients diagnosed by board-certified dermatologists and 142 age-/sex-matched healthy controls. Controls were recruited from hospital staff and community volunteers, excluding individuals with dermatologic or psychiatric histories
Stratification criteria for AA cohort
Disease subtype (patchy AA, alopecia totalis/universalis)
Lesion distribution (scalp, beard, eyebrows, etc.)
Severity metrics (lesion count, surface area, visibility grading)
Two dermatologists independently evaluated clinical parameters, with discrepancies resolved through consensus.
Inclusion/exclusion criteria
Adults aged 18–65 years
Minimum primary education literacy
Diagnosis of AA was made exclusively by a board-certified dermatologist based on clinical examination (with dermoscopy where indicated); no biopsies were required in routine cases.
Consent capacity
Major psychiatric disorders: Current or past-year major psychiatric disorders defined as: schizophrenia spectrum and other psychotic disorders; bipolar I/II disorder (current episode); major depressive disorder with psychotic features; moderate–severe substance use disorder; active suicidal ideation/behavior; psychiatric hospitalization within the past 12 months; or any condition judged to impair consent or valid questionnaire completion.
Concurrent dermatologic conditions
Recent AA treatments (3-month washout)
Pregnancy/lactation
Psychotropic medication use (6-month washout)
Assessment tools
Statistical approach
Power analysis
An a priori power calculation (G*Power 3.1.9.7) determined 128 participants/group provided 95% power to detect medium effects (Cohen’s *d* = 0.5, α = 0.05).
Analytical methods (IBM SPSS v26.0)
•
•
•
○ One-way ANOVA (SAAS across lesion locations).
○ Pearson correlations (SAAS, VAS, HADS, DLQI).
○ Moderation analysis (PROCESS Model 1: HADS-A × VAS interaction).
•
Results
Sociodemographic characteristics
The final sample consisted of 129 patients with Alopecia Areata (AA) and 142 healthy controls. As summarized in Table 1, no statistically significant differences were found between groups in terms of age, gender, marital status, education level, or occupation (p > 0.05 for all), indicating successful group matching and minimizing potential confounding variables.
Demographic characteristics of study participants.
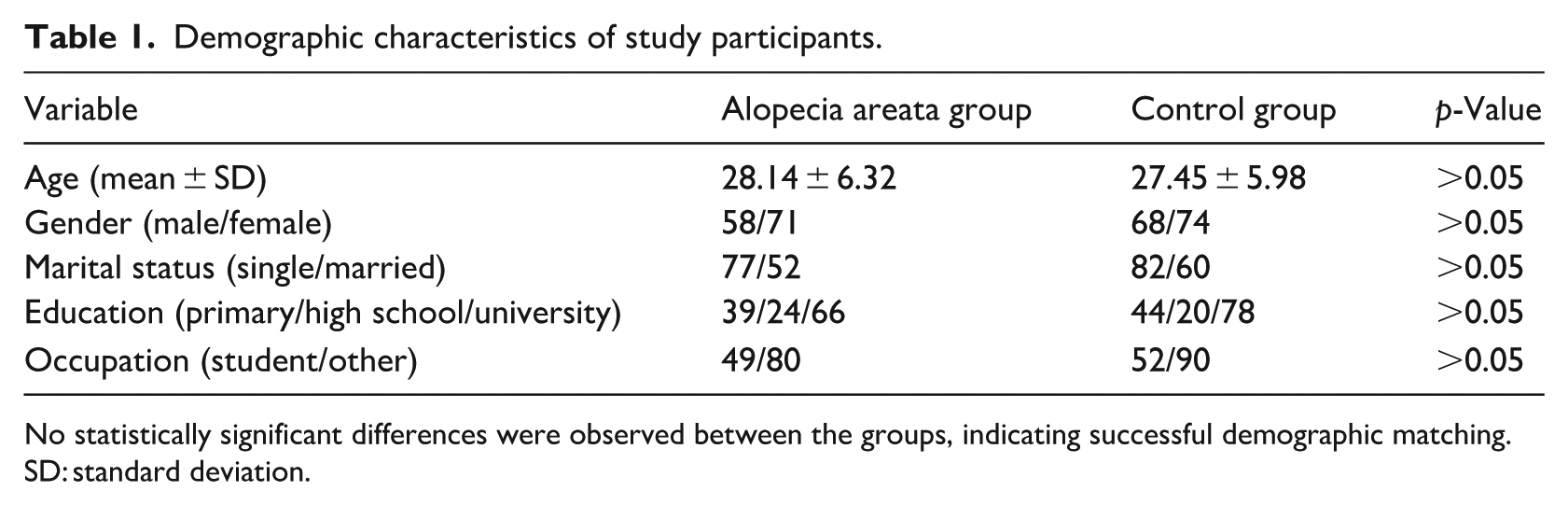
No statistically significant differences were observed between the groups, indicating successful demographic matching.
SD: standard deviation.
Comparison of psychometric scores between groups
Patients with AA exhibited significantly elevated SAAS scores (65.32 ± 8.45) compared to healthy controls (21.45 ± 9.32), with a large effect size (Cohen’s d > 3.5, p < 0.001). No statistically significant differences were found in HADS-Anxiety or HADS-Depression scores between groups (p > 0.05 for both), suggesting that the observed anxiety is appearance-specific rather than generalized. Detailed data are shown in Table 2.
Comparison of psychometric scores between alopecia areata and control groups.
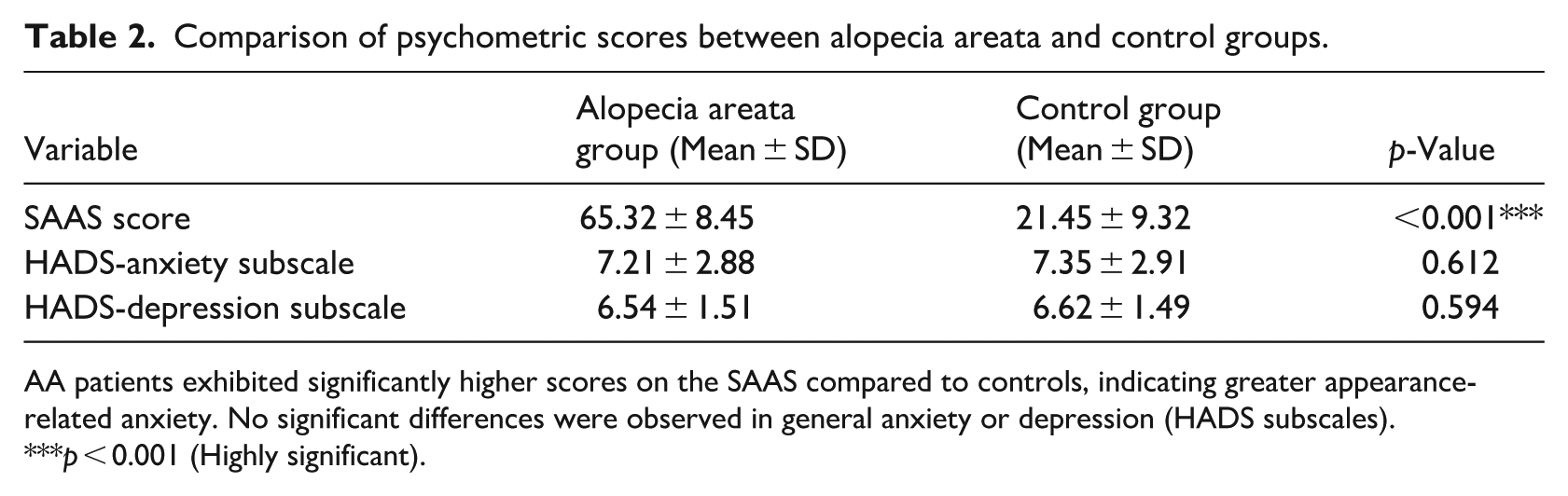
AA patients exhibited significantly higher scores on the SAAS compared to controls, indicating greater appearance-related anxiety. No significant differences were observed in general anxiety or depression (HADS subscales).
p < 0.001 (Highly significant).
Correlational analysis within the AA group
Pearson correlation analyses within the AA group revealed a statistically significant moderate positive correlation between SAAS and VAS scores (r = 0.304, p < 0.05), indicating that higher perceived disease severity—as assessed via the VAS—is associated with increased social appearance anxiety. This underscores the importance of distinguishing subjective illness experience from objective clinical indicators. However, correlations between SAAS and general anxiety/depression (HADS-A and HADS-D) were weak and non-significant, as detailed in Table 3.
Correlational analysis between SAAS and psychological/clinical measures in the alopecia areata group.
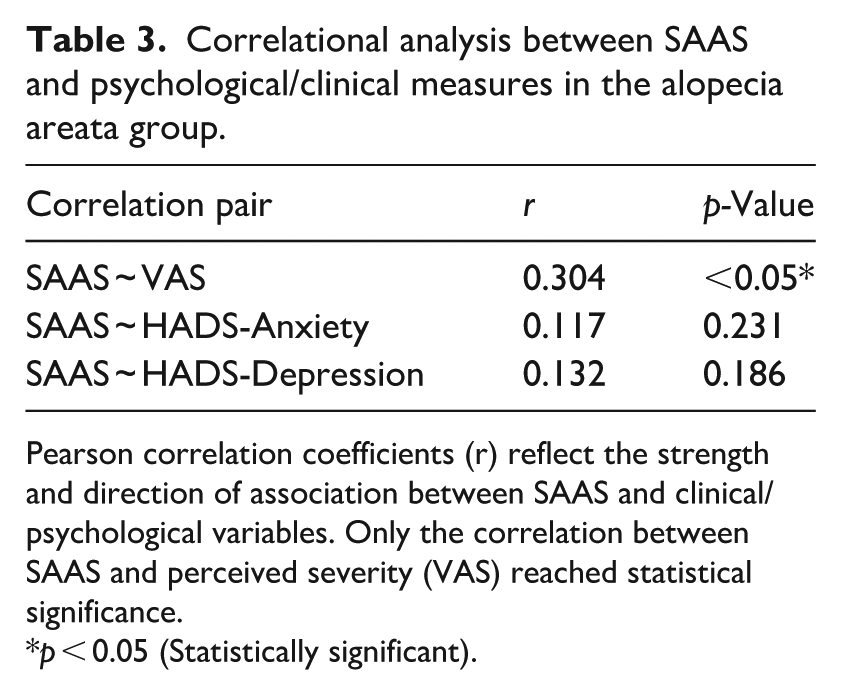
Pearson correlation coefficients (r) reflect the strength and direction of association between SAAS and clinical/psychological variables. Only the correlation between SAAS and perceived severity (VAS) reached statistical significance.
p < 0.05 (Statistically significant).
Subgroup analysis by lesion localization
One-way ANOVA revealed significant differences in SAAS scores across lesion locations (F = 5.47, p < 0.01). Post-hoc Tukey tests indicated that patients with lesions in facial areas, particularly the eyelashes (71.3 ± 5.5) and eyebrows (70.1 ± 5.9), exhibited significantly higher SAAS scores compared to patients with truncal or limb involvement. These findings highlight the psychosocial impact of lesion visibility, as shown in Table 4.
SAAS scores by lesion localization in alopecia areata patients.
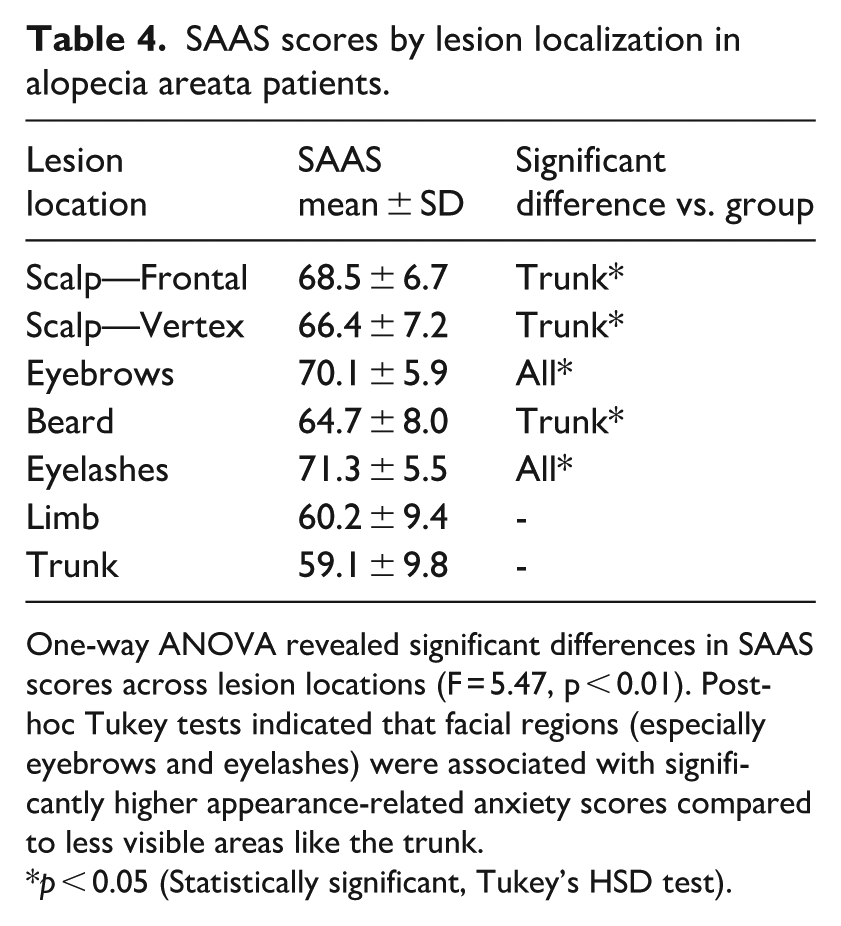
One-way ANOVA revealed significant differences in SAAS scores across lesion locations (F = 5.47, p < 0.01). Post-hoc Tukey tests indicated that facial regions (especially eyebrows and eyelashes) were associated with significantly higher appearance-related anxiety scores compared to less visible areas like the trunk.
p < 0.05 (Statistically significant, Tukey’s HSD test).
DLQI outcomes and correlations with VAS and Clinical Severity Index (AA only)
In the AA cohort (n = 129), DLQI averaged 12.3 ± 5.4 (median 12, IQR 9–16), with 62.8% scoring ⩾ 10 and 10.9% scoring ⩾ 20, indicating clinically meaningful QoL impairment in a sizeable subset. As hypothesized, higher DLQI tracked greater perceived severity on the VAS (Pearson r = 0.45, p < 0.001; Spearman ρ = 0.49, p < 0.001). Within the CSI components, visibility grade showed a moderate positive association with DLQI (r = 0.37, p = 0.002; ρ = 0.40, p = 0.001), whereas associations with lesion count (r = 0.26, p = 0.011; ρ = 0.30, p = 0.006) and total involved area (r = 0.34, p = 0.003; ρ = 0.37, p = 0.002, n = 126) were weaker. Together with the previously reported SAAS–VAS association, these findings indicate that QoL loss in AA aligns more closely with perceived and visible disease burden than with sheer lesion quantity, as shown in Table 5 and Figure 1.
DLQI descriptive outcomes and correlations with VAS and Clinical Severity Index in AA (n = 129).
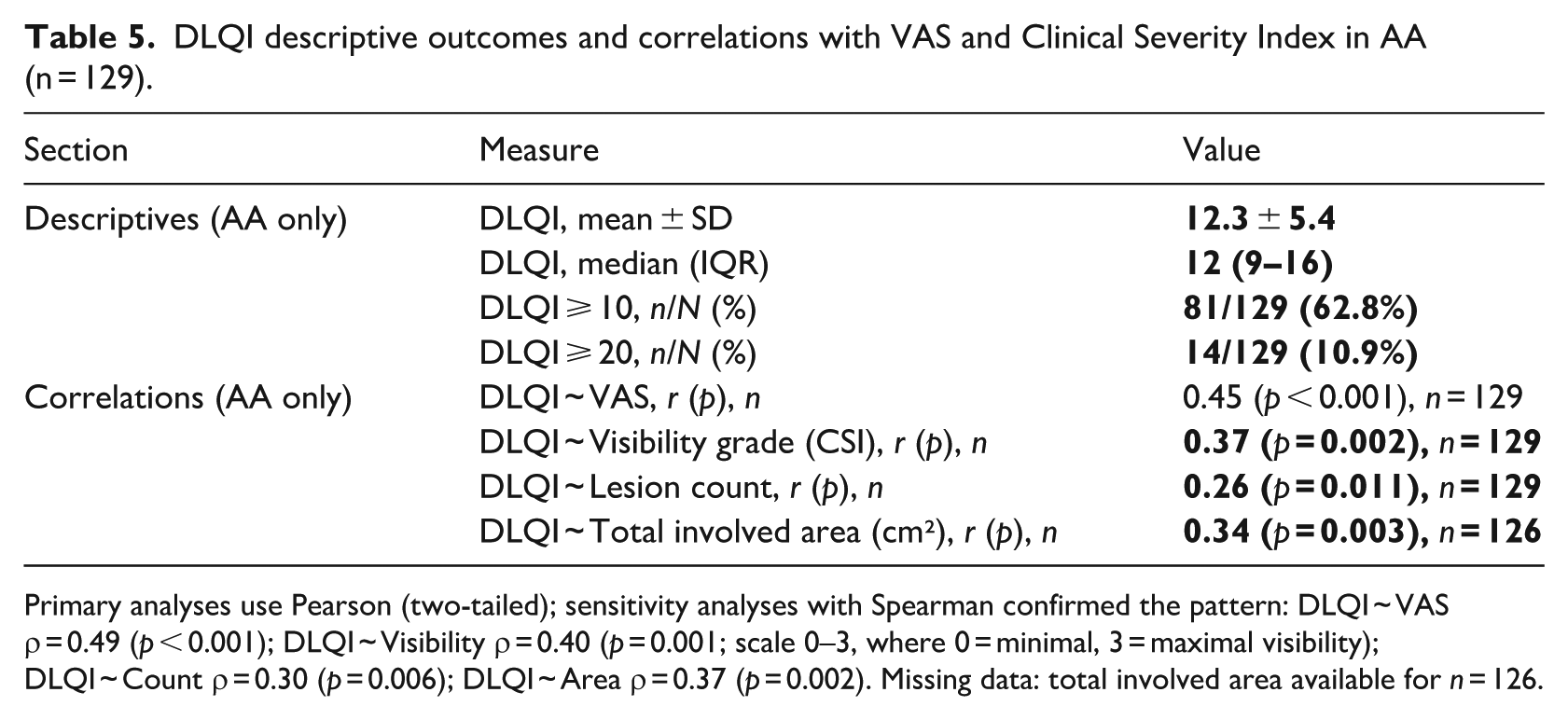
Primary analyses use Pearson (two-tailed); sensitivity analyses with Spearman confirmed the pattern: DLQI ~ VAS ρ = 0.49 (p < 0.001); DLQI ~ Visibility ρ = 0.40 (p = 0.001; scale 0–3, where 0 = minimal, 3 = maximal visibility); DLQI ~ Count ρ = 0.30 (p = 0.006); DLQI ~ Area ρ = 0.37 (p = 0.002). Missing data: total involved area available for n = 126.
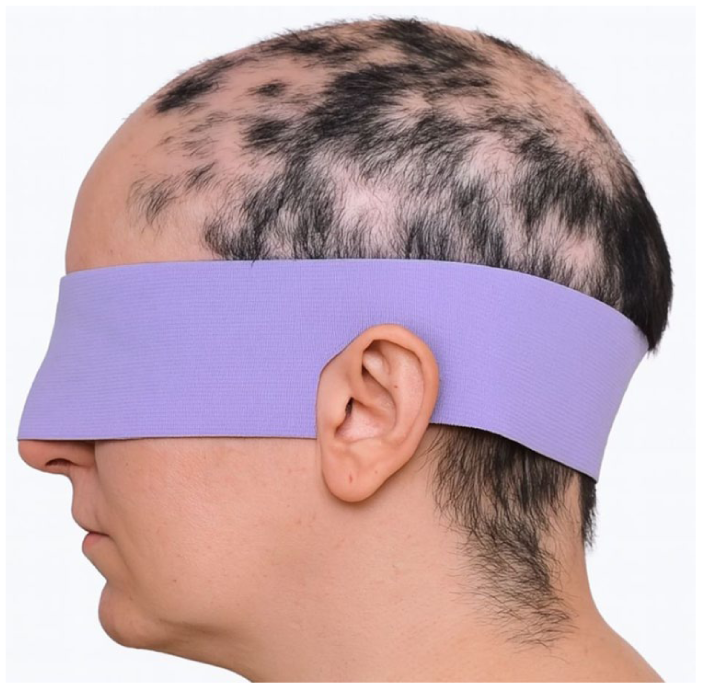
Clinical presentation of a patient with alopecia areata.
This image depicts a patient diagnosed with Alopecia Areata, presenting with patchy hair loss localized to the scalp. The image highlights the characteristic clinical features of the condition and aims to underscore the potential psychosocial impact of visible hair loss.
This bar graph illustrates the comparative mean scores of the SAAS, HADS-A, and HADS-D between patients with Alopecia Areata and healthy controls. As shown, SAAS scores were markedly higher in the AA group, reflecting elevated appearance-related anxiety. In contrast, no statistically significant differences were observed in general anxiety or depression levels between the two groups. These findings emphasize the specificity of social appearance concerns in the AA population and suggest that general anxiety and depression do not significantly contribute to the psychological burden observed in this cohort (Figure 2).
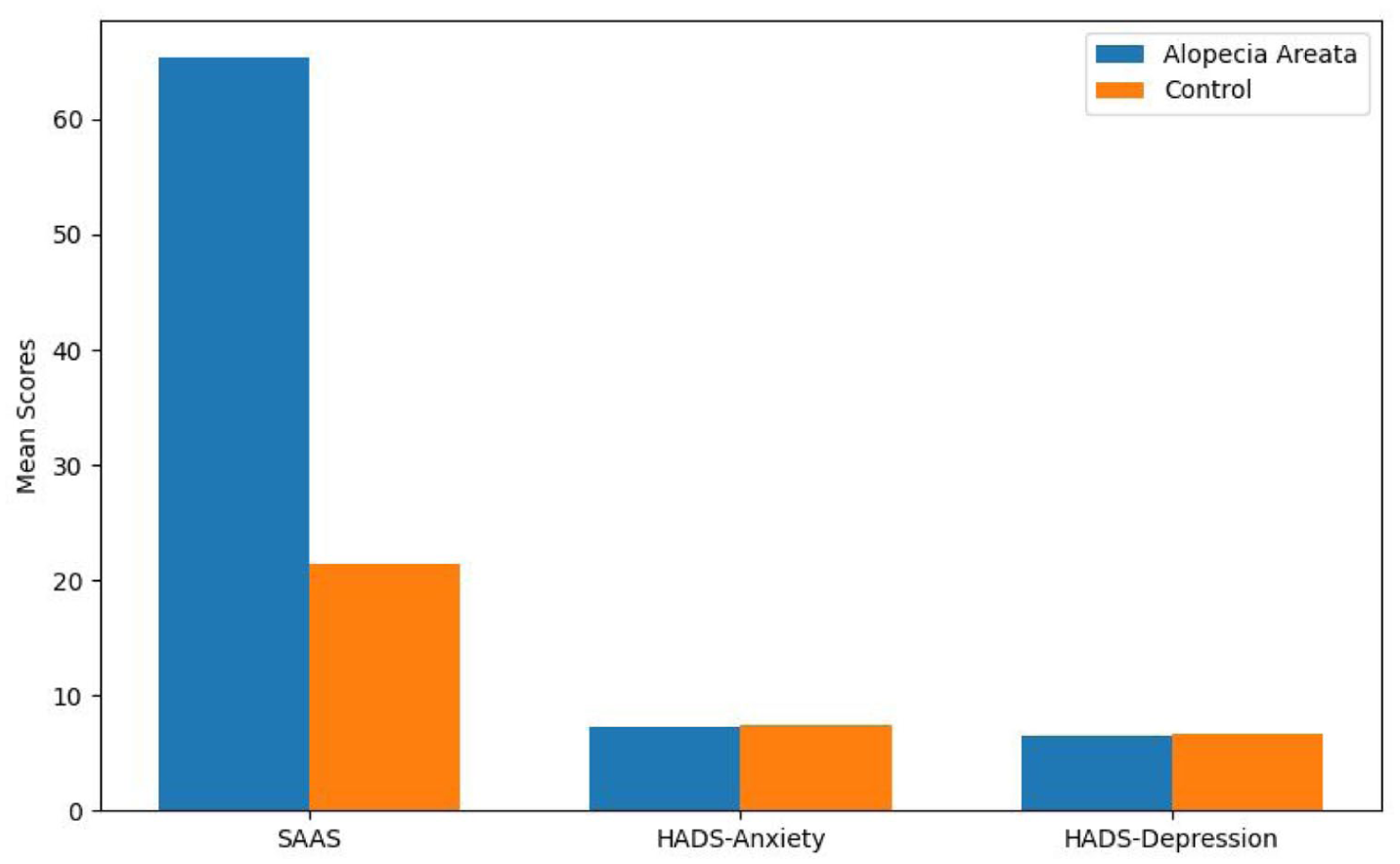
Comparison of psychometric scores between alopecia areata and control groups.
Discussion
This study provides an integrated clinical and psychometric perspective on social appearance anxiety in patients with Alopecia Areata (AA), offering novel insights into how lesion localization, visibility, and subjective illness perception jointly shape psychosocial burden. Using condition-specific measures and topographically detailed dermatologic assessment, we demonstrate that AA patients—particularly those with facial involvement—experience significantly elevated levels of appearance-related anxiety, as captured by the SAAS. Importantly, this distress is not reflected in general anxiety or depression scores, highlighting a psychosocial profile that is unique to visibility-focused dermatologic disorders.
Our findings align with and extend prior literature indicating elevated psychological comorbidity in AA. However, whereas earlier studies often relied on broad psychiatric inventories (e.g. HADS, BDI) without stratifying for lesion location or visibility (Chu et al., 2012; Muzumdar et al., 2024), our study integrates a visibility index, lesion mapping, and validated appearance-specific tools. This multimodal approach enables a more refined understanding of AA’s psychological impact and bridges a key gap emphasized in recent reviews and meta-analyses (Chu et al., 2012; Larsen et al., 2019; Muzumdar et al., 2024).
The absence of significant differences in HADS-Anxiety and HADS-Depression scores between AA patients and controls reinforces the view that generalized psychopathology may not fully capture the psychological burden in visible skin diseases. While HADS-A/D scores did not differ significantly between AA and controls in this sample, and SAAS correlated with perceived severity (VAS), our data suggest that the primary psychological burden is appearance-specific rather than generalized anxiety/depression as captured by HADS. This does not preclude contributions of clinical anxiety or depression in subgroups not captured here, especially given our exclusion of major psychiatric disorders and the screening nature of HADS. Instead, the strong positive correlation between SAAS and the Visual Analog Scale (VAS)—which reflects patients’ subjective perception of disease severity—emphasizes the primacy of perceptual salience over objective lesion extent in driving distress (Germain et al., 2021; Misery et al., 2023; Sarwer et al., 2022). This distinction supports calls within the psychodermatology community for broader implementation of condition-specific measures that better reflect the lived experiences of patients (Misery et al., 2023)
Consistent with psychodermatology models, DLQI correlated most strongly with perceived severity (VAS; r = 0.45) and with clinical visibility (r = 0.37), while relationships with lesion count and total area were comparatively smaller (r = 0.26/0.34; see Table 5). This visibility- and perception-centric profile dovetails with our SAAS findings and underscores the value of early psychosocial support in patients with facial/visible involvement.
A particularly distinctive contribution of our study is its subgroup analysis by lesion localization. Patients with lesions on socially exposed facial areas—especially the eyelashes and eyebrows—reported significantly higher SAAS scores than those with involvement limited to less visible regions such as the trunk or limbs. These findings parallel previous evidence from vitiligo and facial psoriasis, where visibility and symbolic associations of affected areas are strongly predictive of psychological morbidity (Misery et al., 2023; Thompson and Kent, 2001). Moreover, very recent psychodermatology evidence indicates that appearance satisfaction and perceived social support may mediate the link between dermatological disorders and overall psychological distress (Costeris and Petridou, 2025). However, AA presents a unique case: its often sudden onset, patchy progression, and uncertain prognosis may compound the psychological toll by introducing a dynamic threat to identity and self-image, distinct from the chronic and static nature of many other dermatoses (Duque-Estrada et al., 2013).
Additionally, the weak and non-significant correlations between SAAS and HADS subscales observed in our data challenge the assumption that general psychopathology drives appearance-related anxiety. Instead, our findings suggest that social appearance concerns in AA represent a discrete psychological domain, one that warrants independent clinical attention. As such, the SAAS—validated across dermatologic populations for its sensitivity to appearance-based social evaluation—emerges as a critical screening tool for identifying those at risk (Doğan, 2010; Thompson and Kent, 2001).
From a clinical implementation standpoint, these results carry actionable implications. First, facial lesion involvement should be treated as a psychosocial high-risk marker. Patients with visible lesions should be prioritized for mental health screening, even in the absence of generalized anxiety or depression symptoms. Second, dermatology clinics may benefit from incorporating brief SAAS evaluations into routine assessments. Early identification of socially anxious individuals can guide timely referrals to mental health professionals. Cognitive-behavioral interventions, for example, have demonstrated efficacy in reducing appearance-focused distress and enhancing self-esteem in dermatologic patients (Ferreira et al., 2024; Muzumdar et al., 2024).
Finally, our findings support a broader paradigm shift in dermatological care: from disease-centered models to patient-centered, psychosocially-informed frameworks. In this regard, we echo the recent consensus from the European Psychodermatology Task Force advocating for integrative care models that address not only visible symptoms but also the invisible burdens of chronic skin disease (Germain et al., 2021). Visibility-related distress in AA, as shown here, is not merely cosmetic or secondary—it is central to the disease experience.
Limitations
While the study is strengthened by its stratified sampling design, validated tools, and rigorous dermatological evaluation, several limitations should be noted. First, its cross-sectional nature limits causal inference. Longitudinal studies could clarify temporal dynamics between disease progression and psychosocial adaptation. Second, cultural attitudes toward appearance and hair loss may have influenced both symptom expression and psychometric responses, limiting generalizability beyond the regional population. Third, although the SAAS is validated, clinical cut-off points for pathological appearance anxiety in dermatology remain to be fully established.
Conclusion
In conclusion, this study demonstrates that appearance-related anxiety—rather than general psychopathology—is the dominant psychological burden in Alopecia Areata, particularly when lesions affect the face. By employing a clinically grounded and psychometrically nuanced approach, we offer a replicable framework for assessing psychosocial distress in dermatology. Our findings advocate for early psychosocial screening in high-visibility presentations and support the integration of targeted psychological interventions into standard dermatologic care.
Footnotes
Acknowledgements
The authors extend their deepest gratitude to all participants who generously contributed their time and experiences to this study. This work would not have been possible without their valuable involvement and willingness to share.
Ethical considerations
This study was conducted in accordance with the ethical principles of the Declaration of Helsinki. Ethical approval was obtained from the Van Yüzüncü Yıl University Ethics Committee (Approval No: 2020/02-14, Date: 21/02/2020).
Consent to participate
Written informed consent was obtained from all individual participants included in the study.
Consent for publication
Participants were informed that anonymized data may be used for publication purposes and consented accordingly. The materials, including any images, contain a photograph in ![]() that belongs to one of our study participants. Written informed consent for publication of the image was obtained on 15 July 2022, during the data collection period of the study. The patient’s eyes were masked to ensure anonymity.
that belongs to one of our study participants. Written informed consent for publication of the image was obtained on 15 July 2022, during the data collection period of the study. The patient’s eyes were masked to ensure anonymity.
Author contributions
F.K. (Faruk Kurhan) conceptualized and designed the study, conducted the data analysis, interpreted the findings, and was the primary author responsible for drafting and revising the manuscript. G.G. (Göknur Gözaydın Yavuz) contributed significantly to the development of the research methodology, supervised data collection, provided critical revisions for intellectual content, and approved the final version of the manuscript for submission. Both authors read and approved the final manuscript and agree to be accountable for all aspects of the work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
