Abstract
The aim of the present study was to determine the relationship between night eating symptoms (NES) and body mass index (BMI) in medical students and to examine the potential mediating role of impulsivity, depressive symptoms and sleep-related factors in this relationship. The study included 408 medical students in their first to third years of study. Participants were asked to complete the sociodemographic information form, Night Eating Questionnaire (NEQ), Pittsburgh Sleep Quality Index (PSQI), Barratt Impulsiveness Scale-11 (BIS-11), and Patient Health Questionnaire Depressive Symptom Scale (PHQ-9). The results of our study support the existence of a relationship between NES and BMI. Among the potential mediators in this relationship, a mediating role of sleep quality rather than depressive symptoms and impulsivity, was identified. Sleep latency, a component related to sleep quality, was found to have an effect on BMI. Studies are needed to evaluate the effectiveness of interventions to improve sleep hygiene for weight control in NES patients.
Introduction
Night Eating Syndrome (NES) was first described by Stunkard et al. (1955) as a clinical disorder that is characterized by morning anorexia, evening hyperphagia, and insomnia, and has been linked primarily to obesity (Stunkard et al., 1955). Although NES is more prevalent among obese individuals—with rates ranging from 6% to 16%—it also affects about 1.5% of the general population (Allison et al., 2006; Rand et al., 1997; Striegel-Moore et al., 2005. Cerú-Björk et al., 2001; Gluck et al., 2001; Howell et al., 2009). It was officially recognized as a distinct clinical diagnosis under the category of “Other Specified Feeding or Eating Disorder” in the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) in 2013.
Previous studies examining the relationship between NES and Body Mass Index (BMI) have yielded inconsistent results (Howell et al., 2009). Some studies report a significant association between NES and higher BMI (Colles et al., 2007; Gallant et al., 2012; Kucukgoncu and Bestepe, 2014), while others—especially population-based studies—do not find such a link, likely due to sample homogeneity, varying diagnostic criteria, and psychiatric comorbidities (Lundgren et al., 2010; Striegel-Moore et al., 2005, 2008).
It is known that NES is more common in individuals diagnosed with psychiatric disorders such as major depression, generalized anxiety disorder (GAD), obsessive compulsive disorder (OCD) and panic disorder (PD; Striegel-Moore et al., 2005), and that psychostressors, genetics, obesity, depression, anxiety disorders, eating disorders, diabetes and cultural factors are involved in its etiology (Allison et al., 2004, 2010; Gulsun et al., 2012). The prevalence of NES has been found to be 22.4% in the outpatient psychiatric population (Saraçlı et al., 2015), and NES has been reported to be significantly more common in patients with major depression; in addition, individuals with NES tend to report depressed mood more frequently than individuals without NES (Gluck et al., 2001) interestingly, individuals with NES have been reported to be more depressed at night, in contrast to the typical pattern observed in endogenous depression, where depressed mood can be relieved at night (Colles et al., 2007). Obese people with NES have reported higher depression scores on the Zung Depression Inventory and lower self-esteem scores on the Rosenberg Self-Esteem Scale (Gluck et al., 2001). Another study conducted in adolescents in 2024 reported that night eating and low or no physical activity were associated with increased depression and suicide risk (Tuncay and Sarman, 2024). Mood swings and sleep disturbances, which are components of the NES, may be potential factors that influence the relationship between obesity and the NES. These studies provide important information about the relationship between NES and depressed mood, but the relationship between NES and various psychiatric disorders suggests that the psychopathology of NES is not fully understood.
Evaluating the etiology of NES from different perspective suggests that sleep problems may play an active role in the onset of NES. Thus, individuals with NES can have a significantly lower sleep quality, may experience frequent awakenings related to eating urges, and have difficulty in falling asleep (Cerú-Björk et al., 2001; Rogers et al., 2006). Another study reported no statistically significant relationship between NES and sleep disorders, the use of sleep medication, sleep efficiency, subjective sleep quality, and total sleep quality score. The same study, however, reported a statistically significant relationship between NES and sleep duration, sleep latency, and daytime dysfunction (Bektaş and Garipağaoğlu, 2016). Based on the findings of the referred study, NES can be considered to primarily negatively affect sleep continuity and efficiency rather than sleep or sleep latency.
Another psychological domain that has been extensively examined in relation to BMI and obesity is impulsivity, with most studies generally reporting comparable data. For instance, individuals with obesity have been observed to exhibit higher impulsivity in self-report measures and behavioral tasks testing inhibitory capacity (Decker et al., 2021; van Den Akker et al., 2014). Similarly, another study highlighted a positive correlation between higher body weight and various aspects of impulsivity (Legenbauer et al., 2018). On the other hand, one study noted that while a relationship existed between impulsivity and body weight, the effect was small, suggesting that only certain aspects of impulsivity under specific conditions might influence obesity (Meule and Blechert, 2016).
Medical students are considered to be a vulnerable group for psychiatric disorders due the challenging nature of their education, being under stress, and experiencing sleep irregularities. Thus, medical students could be at risk for NES and various studies have examined the prevalence of NES and associated psychiatric conditions among this student population (Haneef and Almuammar, 2024; Yilbas et al., 2022).
A review of the current literature reveals numerous studies examining the relationship between NES and BMI (Colles et al., 2007; Lamerz et al., 2005; Rand et al., 1997; Stunkard et al., 1955). However, studies investigating the impact of certain psychiatric conditions on this relationship are limited. The current study was designed and conducted to directly explore the specific relationship between NES and BMI among medical students, and additionally to examine the potential mediating roles of impulsivity, depressive symptoms, and sleep-related factors in this relationship.
Methods
Participants
The sample of the current cross-sectional study consisted of medical students in their first to third years of study from the Faculties of Medicine at Selcuk University and Necmettin Erbakan University. The sample size for the study was calculated as 398 using the G-Power program, assuming the prevalence of NES in the population to be 1.9%, with a 5% margin of error and a 98% confidence interval (Allison et al., 2006). A total of 408 students aged between 18 and 30 years were included in the study. Informed consent was obtained from those who participated in the study. The inclusion criteria for the study were being between the age of 18–30 years, volunteering to participate in the study, and being a medical students in their first to third years of study. Students who were under 18 or over 30 years of age, taking medications that could lead to weight gain, diagnosed with schizophrenia or bipolar disorder, were using sedative medications, had used psychotropic drugs in the last 3 months, had any chronic disease requiring medication, and students who used substances were excluded from the study.
Ethics approval
The necessary ethics approval was obtained from the local Ethics Committee for Non-Interventional Research (IRB: 07.06.2024-199) and from the deanships of both universities.
Data collection tools
Sociodemographic form
This form inquires about the sociodemographic characteristics of the participants such as age, height, weight, gender, marital status, education level of the parents, place of residence, income status, and smoking-alcohol-substance use status, as well as their chronic illnesses, psychiatric illness status, and use of medication. BMI was calculated using self-reported height and weight data provided by the participants.
Night Eating Questionnaire (NEQ)
NES diagnostic criterias in DSM-5 defined by evening hyperphagia (where at least 25% of the total daily food intake happens after dinner) and nighttime eating episodes that occur a minimum of twice a week. Additionally, to be diagnosed with NES, patients must experience at least three of the following: a strong urge to eat between dinner and bedtime, morning anorexia, insomnia during sleep onset or maintenance, the belief that they cannot fall asleep without eating, and a depressive mood that worsens, particularly in the evening (Allison et al., 2010; American Psychiatric Association, 2013). The NEQ is a 14-item scale developed by Allison and colleagues to assess the presence and severity of NES symptoms. The NEQ comprises items specifically designed to assess evening and night eating behaviors, morning anorexia, the frequency of night awakenings accompanied by eating, sleep disturbances, emotional states, and the duration of these complaint. The validity and reliability of the Turkish scale were established by Allison et al. (2008) and Atasoy et al. (2014). The Cronbach’s Alpha reliability coefficient was reported to be 0.69 in the Turkish validity and reliability study (Atasoy et al., 2014). The questions in the scale are structured on a 5-point Likert scale, with each item scoring between 0 and 4, measuring four features of night eating including nocturnal eating, evening hyperphagia, morning anorexia, and mood/sleep. The total score of the scale ranges from 0 to 52. The commonly used cut-off score for NES is ⩾25 points (Allison et al., 2008).
Pittsburgh Sleep Quality Index (PSQI)
The PSQI is a self-report scale that assesses sleep quality and sleep disturbances over a 1-month period. The PSQI was developed by Buysse et al. (1989), and is known to have sufficient internal consistency, test-retest reliability, and validity (Buysse et al., 1989). The validity and reliability studies of the index for Turkish patients were conducted by Ağargün, Kara, and Anlar, and was determined to be suitable for the Turkish population (Ağargün et al., 1996). The Cronbach’s alpha internal consistency coefficient was found to be 0.80. In evaluating the PSQI, 18 items are scored. The PSQI has seven components: subjective sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, use of sleep medication, and daytime dysfunction. Some components are represented by a single item, while others are derived by grouping several items. Each item is scored between 0 and 3 points, and the total of the seven component scores forms the total PSQI score, which ranges from 0 to 21. A higher total score indicates worse sleep quality.
Patient Health Questionnaire-9 (PHQ-9)
The PHQ-9 is a nine-item subscale of the Patient Health Questionnaire-Somatic, Anxiety, and Depressive Symptom Scales, developed by Kroenke et al. (2010). The validity and reliability studies for the Turkish version were established by Kroenke et al. (2010) and Yazici Güleç et al. (2012). The PHQ-9 is a multiple-choice screening and assessment scale. It was designed to meet the needs of primary care health workers who can frequently encounter somatic, depressive, and anxiety symptoms. It was developed as a 3-point Likert scale (0 = Not at all bothered, 1 = Somewhat bothered, 2 = Very much bothered).
Barratt Impulsiveness Scale-11 (BIS-11)
BIS-11 is a self-report scale that is used to assess impulsivity, and consists of three non-overlapping subscales with good reliability: attentional impulsiveness, motor impulsiveness, and non-planning impulsiveness (Patton et al., 1995). The validity and reliability studies of the Turkish version of the scale were conducted by Tamam and colleagues. The Cronbach’s Alpha reliability coefficient was reported to be 0.82 in the Turkish validity and reliability study. The scale has a 4-point Likert structure, with responses ranging from never, occasionally, often, and always. The scale does not have a cut-off score; however, higher scores indicate higher levels of impulsivity (Tamam et al., 2013).
Procedure
Students who were enrolled at the two different medical faculties over a pre-determined period of time were informed about the study. As the study was conducted on a voluntary basis, the students who agreed to participate were provided the relevant data forms, and were asked to fill them out under observation, with an average of 20 minutes allotted for each form.
Statistical analysis
The data were analyzed using SPSS 18.0 for Windows (SPSS Inc., Chicago, IL, US) and JAMOVI (Version 2.5) programs in the current study. Descriptive statistical methods (frequency, percentage, mean, standard deviation) were used to evaluate the study data. The normality of the variables was examined using visual (histogram and Q-Q plots) and analytical (skewness, kurtosis values, coefficient of variation, Kolmogorov-Smirnov/Shapiro-Wilk tests) methods. The relationship between the psychometric test results of the participants was evaluated using Pearson and Spearman correlation coefficients. The mediating roles of impulsivity, depressive symptoms, and sleep quality in the relationship between night eating symptoms and BMI were analyzed using the GLM procedure. The results were evaluated at a 95% confidence interval and a p < 0.05 significance level.
Results
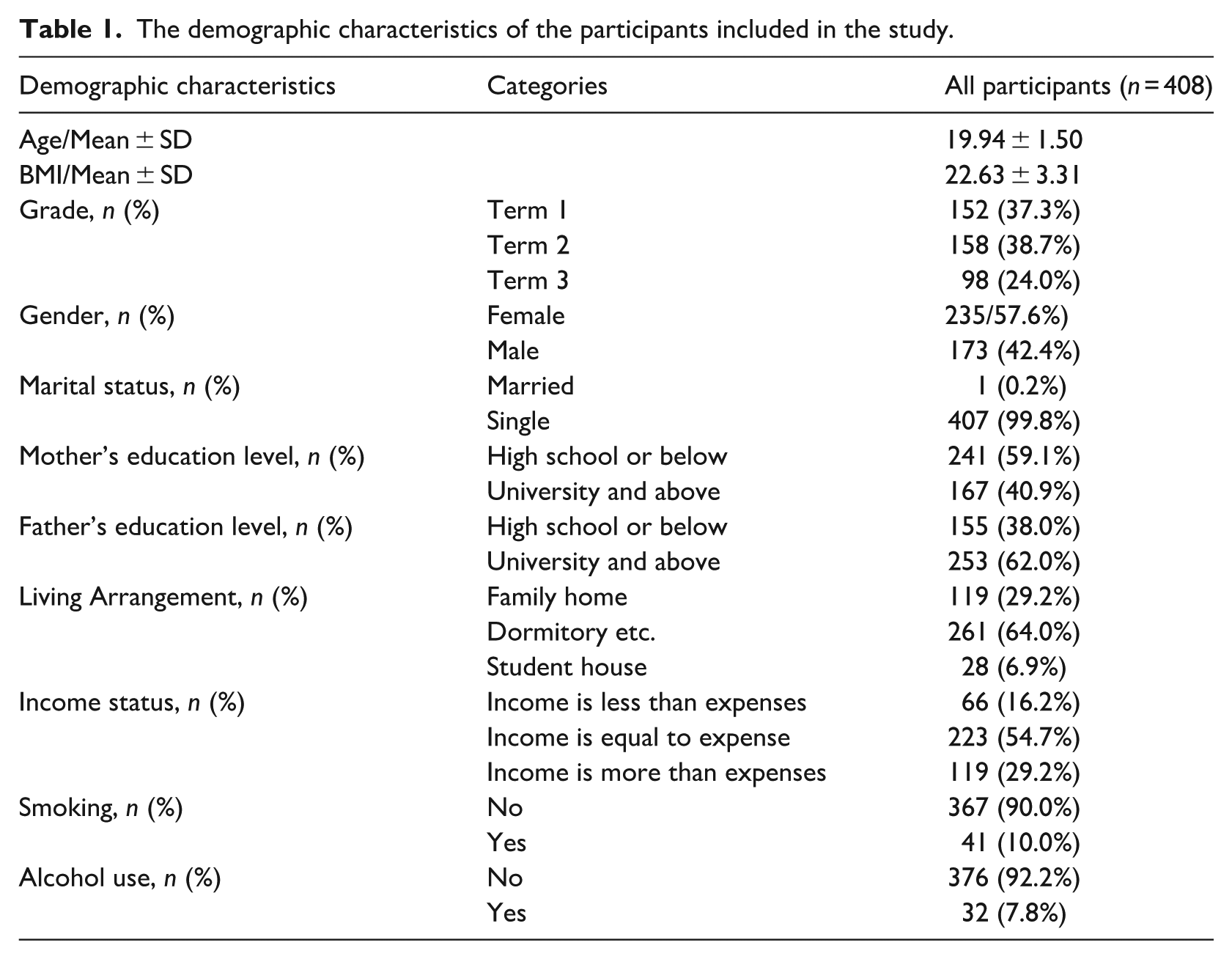
A total of 408 medical students with a mean age of 19.94 ± 1.50 years were included in the study. 57.6% of all participants were female, and 38.7% of all participants were at the second term of their education. The demographic characteristics of the participants included in the study are shown in Table 1.
The demographic characteristics of the participants included in the study.
A statistically significantly albeit modest positive correlation was identified between the NEQ score of the participants and their BMI (r = 0.146; p = 0.004). The mean NEQ score of the participants was 14.42 ± 5.55, the mean BIS score was 28.84 ± 6.26, the mean PHQ-9 mean was 10.20 ± 5.66 and the mean PSQI score was 6.92 ± 2.69. The correlations between the BIS, PHQ-9, PSQI and NEQ total scores used in the study are shown in Table 2. A statistically significant moderate correlation was identified between the NEQ score and the BIS (r = 0.328), PHQ-9 (r = 0.446), and PSQI (r = 0.503) scores. A modest correlation was also identified between the BIS score and PHQ-9 (r = 0.383, respectively) and a low correlation between BIS and PSQI (r = 0.193). A significant but moderate correlation was identified between the PHQ-9 and PSQI scores.
The correlations between the BIS, PHQ-9, PSQI, and NEQ total scores.
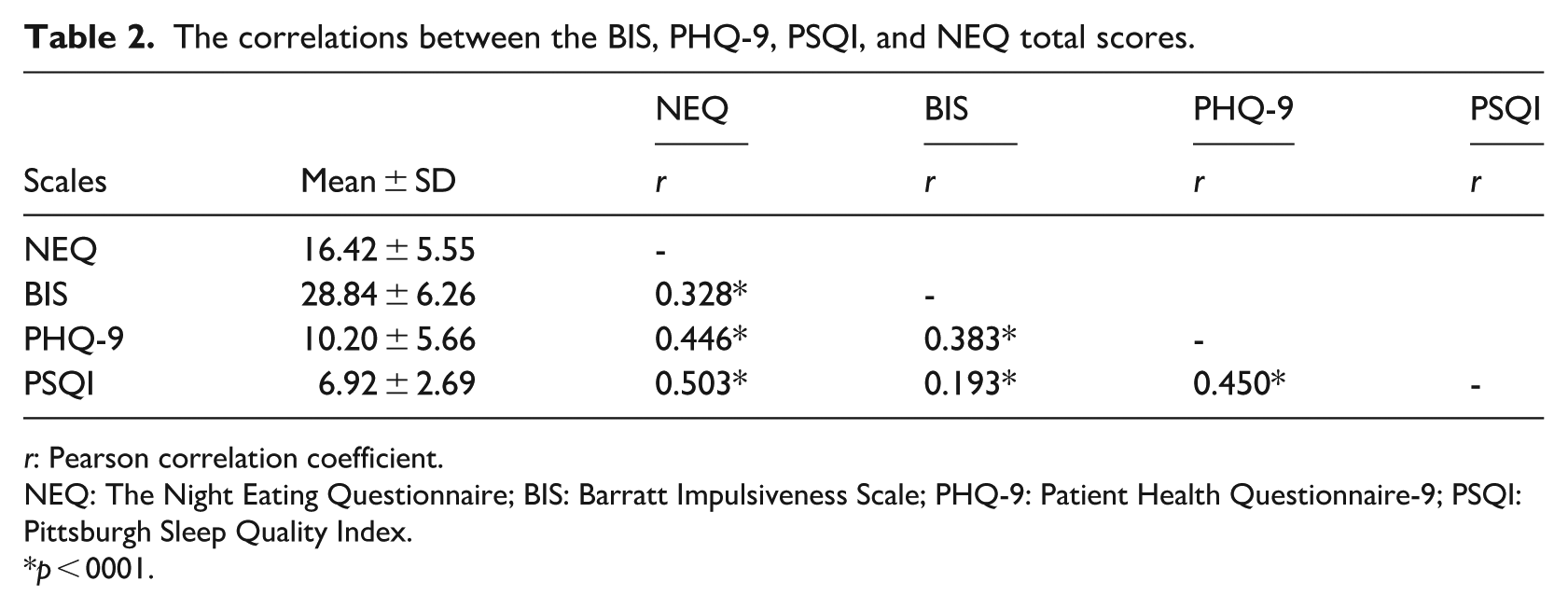
r: Pearson correlation coefficient.
NEQ: The Night Eating Questionnaire; BIS: Barratt Impulsiveness Scale; PHQ-9: Patient Health Questionnaire-9; PSQI: Pittsburgh Sleep Quality Index.
p < 0001.
The correlation coefficients between the PSQI sub-dimension scores and NEQ, BIS, and PHQ-9 of the participants are shown in Table 3. A moderate but statistically significant positive correlation was found between NEQ and the PSQI-1 and PSQI-2 scores (r = 0.418 and 0.429, respectively). In addition, a weak positive correlation was identified between the NEQ score and PSQI-5, PSQI-6, and PSQI-7 scores (r = 0.392 and 0.264, respectively; r = 0.238) while a weak positive correlation was found between the BIS score and PSQI-1 (r = 0.200). Finally, a weakly positive but statistically significant correlation was found between PHQ-9 and the PSQI-1 (r = 0.369), PSQI-2 (r = 0.311), PSQ-5 (r = 0.381), and PSQI-7 (r = 0.307) scores.
Relationship of PSQI sub-dimensions with NEQ, BIS, and PHQ-9.
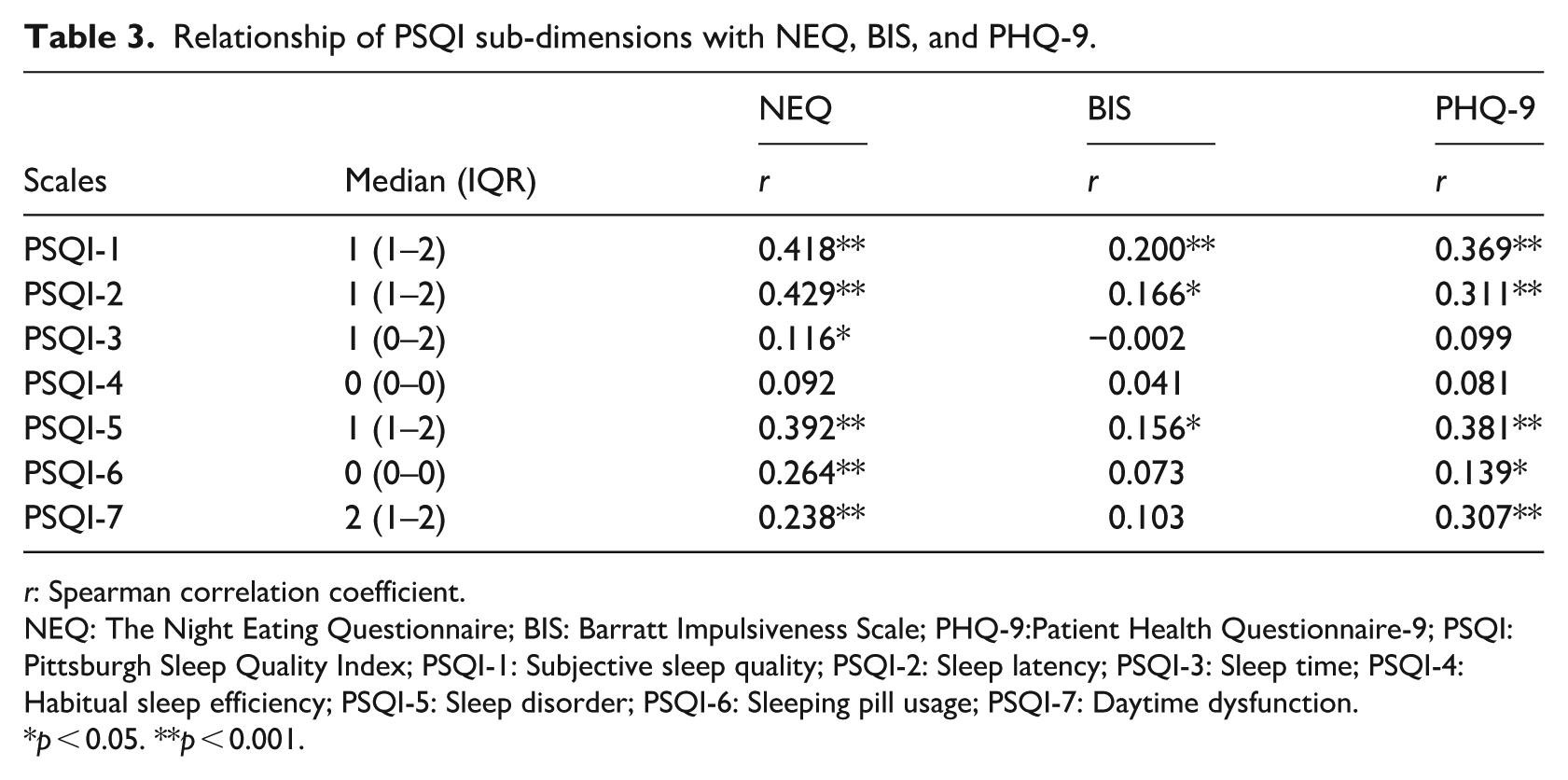
r: Spearman correlation coefficient.
NEQ: The Night Eating Questionnaire; BIS: Barratt Impulsiveness Scale; PHQ-9:Patient Health Questionnaire-9; PSQI: Pittsburgh Sleep Quality Index; PSQI-1: Subjective sleep quality; PSQI-2: Sleep latency; PSQI-3: Sleep time; PSQI-4: Habitual sleep efficiency; PSQI-5: Sleep disorder; PSQI-6: Sleeping pill usage; PSQI-7: Daytime dysfunction.
p < 0.05. **p < 0.001.
The mediating role of impulsivity (BIS total score), depressive complaints (PHQ-9 total score) in the relationship between NES and BMI did not reach statistical significance in the current study (p > 0.05). However, sleep quality (PSQI total score) was found to mediate the relationship between NES and BMI (β = −0.066, SD = 0.020, 95% CI [−0.080, −2.814]; see Table 4 and Figure 1).
Mediating role of impulsivity, depressive complaints and sleep quality in the relationship between night eating symptoms and BMI.
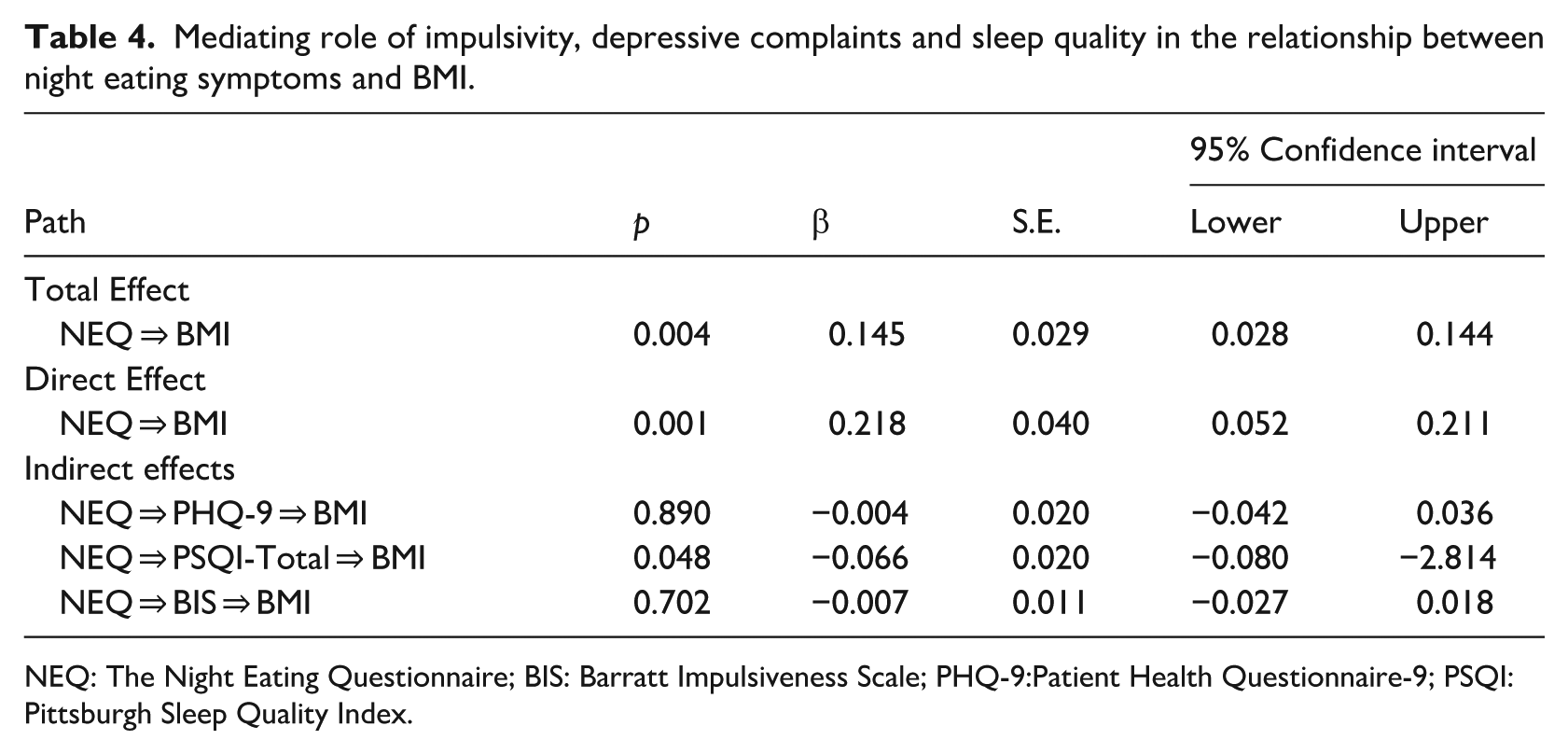
NEQ: The Night Eating Questionnaire; BIS: Barratt Impulsiveness Scale; PHQ-9:Patient Health Questionnaire-9; PSQI: Pittsburgh Sleep Quality Index.
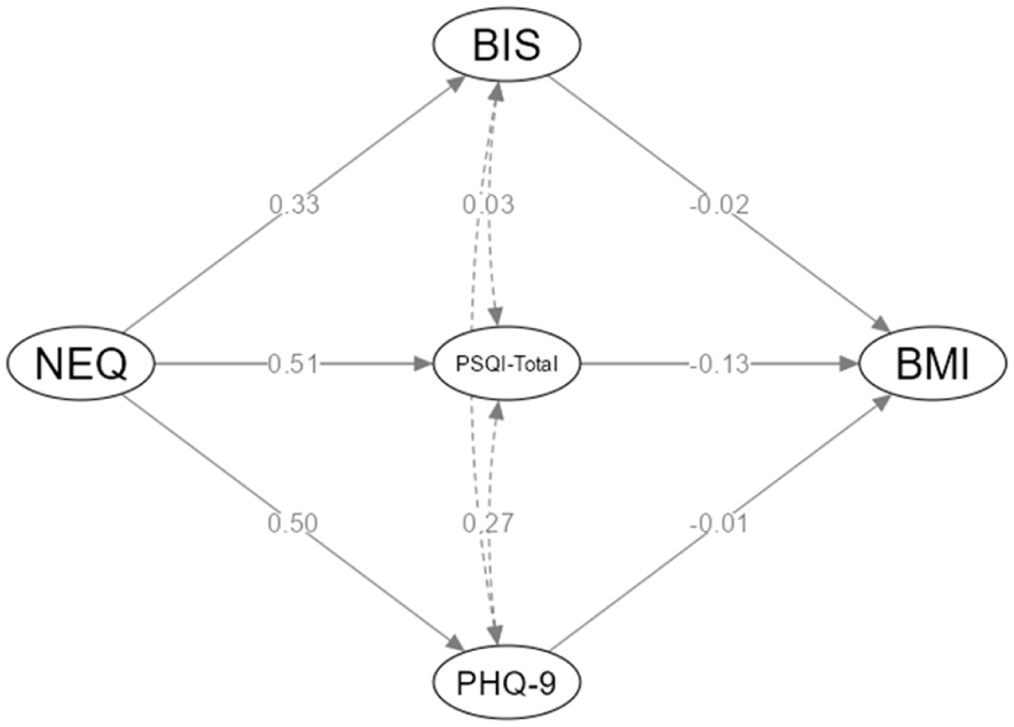
Path diagram: The mediating role of BIS, PHQ-9, and PSQI total scores in the relationship between NES and BMI.
The mediating role of the sub-dimensions of sleep quality in the relationship between NES and BMI was next examined by creating a separate model (Table 5). PGQI-2 (sleep latency) was found to play a mediating role (β = −0.051, S.E. = 0.014, 95% CI [−0.057, −0.005]), while the other sub-dimensions did not.
Mediating role of sleep quality sub-dimensions in the relationship between night eating symptoms and BMI.
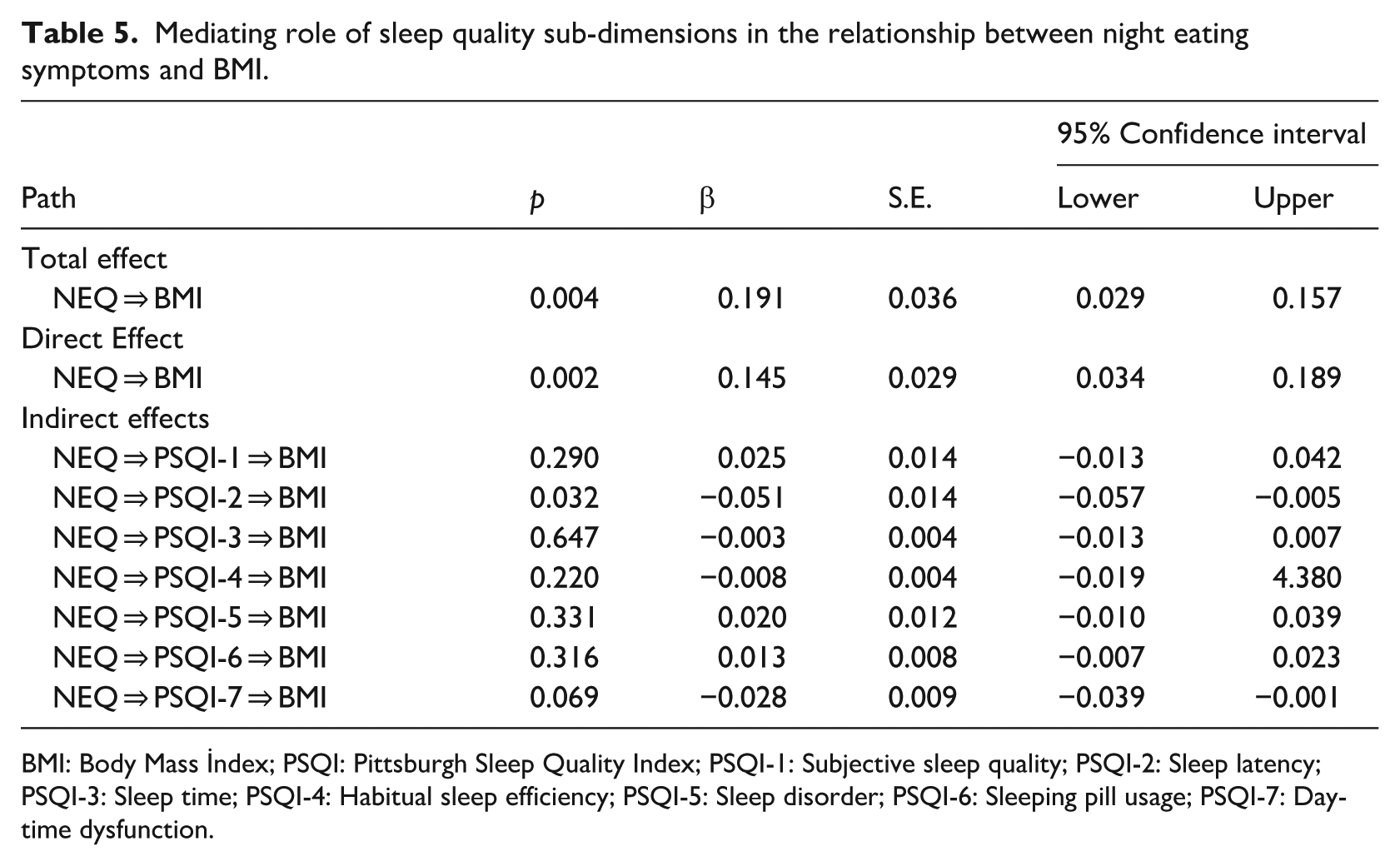
BMI: Body Mass İndex; PSQI: Pittsburgh Sleep Quality Index; PSQI-1: Subjective sleep quality; PSQI-2: Sleep latency; PSQI-3: Sleep time; PSQI-4: Habitual sleep efficiency; PSQI-5: Sleep disorder; PSQI-6: Sleeping pill usage; PSQI-7: Daytime dysfunction.
Discussion
The effects of impulsivity, depressive mood, and sleep quality on the BMI of patients with NES were evaluated in the current study using correlation and mediation regression analyses with relevant psychometric tests. The findings indicate the presence of a relationship between NES and BMI, with sleep quality emerging as a more significant mediator in this relationship than depressive mood or impulsivity. Specifically, sleep latency, a component of sleep quality, was found to have a direct effect on BMI.
Recent reviews of the association between NES and BMI (weight status) have found inconsistent findings across populations (Bruzas and Allison, 2019; Sakthivel et al., 2023). These reviews included studies across a variety of racial and clinical groups, including both healthy individuals and those with psychiatric or other medical comorbidities. While the majority of studies reported a positive association between NES and BMI, a significant number of studies, particularly those conducted in college student populations, failed to find a significant association. For example, a study by Nolan and Geliebter (2017) found a significant association between NES and BMI in a general community sample (mean age = 42.9 years), but not in college students (mean age = 18.7 years; Nolan and Geliebter, 2017). Meule et al. (2014) also reported that the NES-BMI relationship follows a U-shaped pattern across age, with significant relationships primarily found in individuals aged 31–60 years, but not in those outside this range (Meule et al., 2014). These findings suggest that age may be an important moderator of the NES-BMI relationship. Interestingly, although our sample consisted of young medical students (mean age ≈ 19 years), we found a significant relationship between NES and BMI, similar to findings reported in other university student samples such as He et al. (2019) and Riccobono et al. (2020). This suggests that factors other than age, such as differences in student populations, measurement tools, and exclusion criteria, may also influence the detection of this relationship. Gender has also been investigated as a possible moderator, but the results are inconsistent. For example, Kara et al. (2020) and Gallant et al. (2014) found that gender did not have a significant moderator effect on the NES-BMI relationship. Furthermore, it is noteworthy that most of the existing studies are cross-sectional and longitudinal data on these relationships are limited. Future studies with prospective designs are needed to better understand these moderating effects. In contrast, the association between NES and poor sleep quality appears to be more robust and is consistently replicated across studies (Bruzas and Allison, 2019; Sakthivel et al., 2023). However, despite the increasing recognition of sleep disturbances in individuals with NES, very few studies have investigated the mediating role of sleep quality or its subcomponents such as sleep latency in the association between NES and BMI. To our knowledge, our study is one of the first to address this gap by examining this mediating pathway. These findings add new insights to the existing literature in which disrupted sleep patterns may play a critical role in the NES-BMI link, especially in highly stressed populations such as medical students.
The interplay between NES, obesity, weight gain, and BMI is complex and is influenced by various factors and mechanisms. Previous research suggests that disrupted circadian rhythm, hyperphagia, and impaired ability to cope with psychological distress that are commonly observed in patients with NES may contribute to weight gain and obesity (Chisti and Navaneetha, 2024; Costa et al., 2015; Gallant et al., 2012; O’Reardon et al., 2006) In line with these findings, the current study also found a significant association between sleep disturbance, a clinical manifestation of NES, and BMI.
Research suggests that sleep disorders and weight gain have a significant relationship with inadequate sleep being associated with an increased risk of weight gain. Sleep disturbances, such as sleep apnea, insomnia, and sleep deprivation, can lead to metabolic disturbances, changes in appetite hormones, and alterations in energy expenditure, all of which can contribute to weight gain (Markwald et al., 2013; Mavanji et al., 2013; Rodrigues et al., 2021). Furthermore, a short sleep duration has been established as a risk factor for weight gain (Appelhans et al., 2013; Crönlein et al., 2015) Additionally, sleep quality has an impact on weight gain, with unfavorable sleep characteristics being linked to increased weight gain (Häusler et al., 2019). Various studies have emphasized the importance of addressing sleep disorders in the context of weight management, since improvement in sleep quality was shown to potentially reduce weight gain(Coborn et al., 2017) Sleep deprivation and prolongation of sleep latency may also affect physical activities, energy expenditure, and appetite regulation, all of which can contribute to weight gain (De Porter et al., 2017). Hormonal and physiological changes play a crucial role in the relationship between sleep disturbances and weight gain such that inadequate sleep can lead to hormonal dysregulation, ultimately contributing to weight gain (Ayala-Moreno et al., 2023; Azuara-Alvarez et al., 2023) In this context, the role of appetite-regulating hormones such as leptin and ghrelin, which are responsible for signaling satiety and hunger respectively, is of particular importance (Morelhão et al., 2018; Xiao et al., 2013). One of the key findings of our study was the mediating role of delayed sleep onset in the NES–BMI relationship, which may operate through hormonal mechanisms involving leptin and ghrelin, both of which are sensitive to disruptions in sleep timing. Insufficient sleep has been associated with imbalances in these hormones, leading to increased food intake and potentially contributing to weight gain (Morelhão et al., 2018; Xiao et al., 2013). Moreover, disruptions in the growth hormone-insulin-like growth factor-1 axis and leptin levels have been implicated in sleep disturbances and their impact on weight regulation (Lindberg et al., 2003). Sleep disturbances, such as sleep deprivation and sleep restriction, have been shown to affect the secretion and levels of these hormones, which can influence metabolic processes, and energy balance, potentially leading to weight gain (Barf et al., 2012; Lindberg et al., 2003). Furthermore, sleep disturbances have been associated with changes in the sympathetic nervous system and can result in alterations in glucose metabolism, insulin resistance, and energy regulation, all of which are factors that can contribute to weight gain (Barf et al., 2010). Therefore, our finding that sleep quality is the most influential factor in the NES-BMI relationship is supported by previous studies. While this highlights the role of sleep as a proximal mechanism, it is also important to consider the possibility of bidirectional relationships. For instance, NES is characterized by late-night eating and nocturnal awakenings, which may contribute to disrupted sleep pattern (Dixon et al., 2001; Patel and Hu, 2008; Yeh and Brown, 2014). Conversely, poor sleep quality—particularly prolonged sleep latency as indicated by the PSQI-2 dimension—may impair appetite regulation (Shigeta et al., 2001; Vorona et al., 2005) and increase vulnerability to night eating behaviors. These reciprocal influences suggest a potential feedback loop, in which poor sleep and NES mutually reinforce each other over time. Future longitudinal or experimental studies are warranted to explore this dynamic interaction and to identify whether improving sleep quality could reduce NES symptoms or vice versa
While designing the current study, we hypothesized that depressive symptoms and impulsivity may also be important in the relationship between BMI and NES. However, contrary to our expectations, our findings did not support this hypothesis. Although several studies have reported a significant relationship between depression and weight gain (Forman-Hoffman et al., 2007; Sutin and Zonderman, 2012), others suggest that this association may be mediated by sleep disturbances, a diagnostic criteria of depression (Harris et al., 2009; Hobzova et al., 2017). Indeed, depressive symptoms were associated with both impulsivity and sleep quality in the current study. When sleep quality was considered as a separate entity, other depressive symptoms did not have a significant effect on BMI. Additionally, appetite loss and weight loss are often the diagnostic criteria in the most common subtype of depression known as “melancholic depression” (American Psychiatric Association, 2013).
Although variables associated with depressive symptoms such as chronic stress, emotion dysregulation, self-esteem, or lifestyle did not emerge as significant mediators in the current study, previous research suggests that these factors may nevertheless play important roles in the development and maintenance of NES, particularly in individuals with elevated BMI. It has been observed that high levels of stress and maladaptive coping strategies may lead to NES in university students and increase NES sensitivity (Wichianson et al., 2009). Similarly, it is known that there is a relationship between experience of emotion and NES, and that the urge to satisfy unmet emotional needs can exacerbate stress-related eating attacks during the night (Shillito et al., 2018). On the other hand, in a study conducted with university students to understand symptoms compatible with NES diagnostic criteria, it was found that students who showed and did not show night eating behavior did not differ according to lifestyle characteristics such as physical activity and smoking, while NES was significantly associated with sleep duration (Yahia et al., 2017). In other words, although depressive symptoms did not contribute to the BMI-NES relationship in this study, it is important to contribute to studies examining various variables that can change depressive symptoms, such as people’s emotion regulation strategies or lifestyle characteristics. Future studies may benefit from exploring these variables using longitudinal designs or testing for interaction effects, to better understand their complex roles as potential moderators or mediators in the etiology of NES.
The relationship between impulsivity and BMI is complex, with studies presenting both supporting (Han et al., 2022; van Den Berg et al., 2011) and opposing evidence (Bal et al., 2018; Legenbauer et al., 2018). Thus, some studies have suggested a positive association between impulsivity and higher BMI (van Den Berg et al., 2011), while others have reported conflicting results (Legenbauer et al., 2018). This highlights the need for more comprehensive investigations into the specific facets of impulsivity that can contribute to the weight status.
The relatively large samples size of the current study can be considered to be its strength. However, several methodological limitations should be acknowledged. First, the cross-sectional design precludes any inference of causality. Although mediation models were applied to explore potential pathways, longitudinal studies are necessary to establish temporal precedence and clarify directionality among variables. Second, the diagnosis of NES was not based on a psychiatric examination; instead, it was determined by using cut-off scores from the NEQ. While the NEQ is a widely used and validated screening tool, reliance on a cut-off score limits diagnostic precision and may not fully capture clinical nuances. Third, all key variables—including height, weight, sleep quality, and psychological symptoms—were assessed using self-report instruments. This raises concerns about potential recall bias and social desirability effects. Furthermore, the assessment of sleep quality was entirely subjective and did not include objective measurements such as actigraphy or polysomnography. Similarly, important sleep-related parameters such as chronotype and circadian rhythm misalignment, which could meaningfully affect both NES and BMI, were not evaluated. Fourth, the study included medical students without prior psychiatric complaints or consultations, there may be limitations in generalizing the findings to the broader population. Furthermore, the presence of comorbid psychiatric conditions (other eating disorders, OCD, personality disorders) may have partially influenced the study outcomes. Finally, several potentially influential confounding factors—such as dietary intake patterns and physical activity levels, were not directly measured or statistically controlled in the present study. These variables may have influenced the observed relationships among NES, sleep quality, and BMI. Future research should aim to replicate these findings using longitudinal designs, clinical interviews for NES diagnosis, and objective sleep monitoring tools. Including diverse populations across different age groups, cultures, and educational settings, as well as incorporating lifestyle and circadian-related variables, would enhance the ecological validity and clinical applicability of future studies.
In conclusion, results of current study demonstrate that the severity of NES symptoms is significantly associated with depressive complaints, impulsivity levels, and multiple dimensions of sleep quality. Specifically, subjective sleep quality, frequency of sleep disturbances, use of sleep medication, and impairments in daytime functioning were all positively correlated with NES symptom severity. These findings support a multidimensional model of NES in which emotional regulation, behavioral inhibition, and circadian rhythm disruptions jointly contribute to symptom intensity and associated health outcomes. Furthermore, findings of present research suggest the presence of a relationship between NES and BMI, with sleep quality playing a more significant mediating role than depressive symptoms or impulsivity. Sleep latency, in particular, was identified as a key component affecting BMI. The results of our study highlight the need for future research to explore the effectiveness of interventions aimed at improving sleep hygiene or circadian regulation in controlling weight gain among NES patients.
Footnotes
Ethical considerations
The study was approved by the Non-Pharmaceutical and Medical Device Ethics Committee of Necmettin Erbakan University Faculty of Medicine with decision number 2024/5018 dated 07.06.2024.
Consent of participants
Written and verbal informed consent forms were obtained from all participants included in the study.
Consent for publication
All of the authors agree with the content of the manuscript and submission to Journal of Health Psychology.
Author contributions
Research idea: SG, MA. Design of the study: SG, EKU, ZO, MA. Acquisition of data for the study: SG, EKU, ZO, BKK, ENS, MA. Analysis of data for the study: SG, EKU, ZO, BKK, ENS, MA. Interpretation of data for the study: SG, EKU, ZO, BKK, ENS, MA. Drafting the manuscript: SG, EKU, ZO, BKK, ENS, MA. Revising it critically for important intellectual content: SG, EKU, ZO, BKK, ENS, MA. Final approval of the version to be published: SG, EKU, ZO, BKK, ENS, MA.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
