Abstract
Background:
Insomnia is common in adolescents and often comorbid with psychiatric disorders. This study evaluated changes in insomnia, sleep, and comorbid symptoms following cognitive-behavioral therapy for insomnia (CBT-I) in adolescents with comorbid psychiatric disorders and chronic pain.
Methods:
In this non-controlled clinical pilot study, participants (n = 23, 78% female) were recruited from adolescent psychiatry and pediatric pain clinics. Assessments of self-reported insomnia, sleep onset latency, wake after sleep onset, total sleep time, sleep efficiency and depression, anxiety, functional disability, and pain intensity were completed at pre- and post-intervention and 3 months follow-up.
Results:
From pre- to post-intervention, statistically significant improvements were found for insomnia symptoms (p < .001; d = 1.63), sleep onset latency (p < .001; d = 1.04), wake after sleep onset (p < .001; d = 0.38), total sleep time (p = .015; d = 0.22), sleep efficiency (p < .001; d = 1.00), depression (p < .001; d = 0.87), and anxiety (p = .001; d = 0.31). Only eight participants reported data at follow-up with maintained improvements for all measures.
Conclusion:
This study provides support that insomnia symptoms and sleep can improve following CBT-I delivered in a clinical setting and that co-occurring psychiatric symptoms can be reduced. The results should be interpreted with caution due to the uncontrolled conditions and limited sample size. Well-powered clinical trials are needed to validate the suggested effects.
Introduction
Sleep disorders, especially insomnia, are common in adolescents. Insomnia is defined as difficulties falling asleep, staying asleep, and/or suffering from early awakenings for at least three nights/week over at least 3 months with associated clinically significant functional impairment (American Psychiatric Association, 2013). Median age of onset of adolescent insomnia is 11 years with difficulties initiating sleep as the most common symptom (Johnson et al., 2006). In the general population, about 10% of adolescents meet criteria for an insomnia diagnosis according to the Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5) (Dohnt et al., 2012) and more than 33% report “at least some” symptoms of insomnia (Dohnt et al., 2012).
More than 50% of adolescents with insomnia fulfill the criteria for at least one other comorbid psychiatric disorder (Johnson et al., 2006), with anxiety disorders and major depressive disorder (MDD) being the most common (Johnson et al., 2000). In adolescents aged 7–15 years diagnosed with MDD, 77% of girls and 69% of boys report symptoms of sleep disturbances (Liu et al., 2007). In a study on sleep and depression in adolescence, 66% of individuals with insomnia also presented symptoms of depression (Amaral et al., 2017). From late childhood to early adolescence, there is an increased risk of developing sleep problems and anxiety (McMakin & Alfano, 2015) and as much as 90% of children with anxiety disorders report sleep problems (Chase & Pincus, 2011). In addition to psychiatric problems, pain is also frequently occurring with disordered sleep (Kundermann et al., 2004). Pain problems may disturb sleep, but the opposite is also true, that is, individuals with experimental disruption or defragmentation of sleep report increased pain sensitivity (Haack et al., 2009; Kundermann et al., 2004). Observational longitudinal studies show that sleep disruption predicts increased pain severity, and vice versa (Smith et al., 2009). In adolescent chronic pain patients, 60% have been reported to suffer from sleep disorders (Kanstrup et al., 2014).
Meta-analyses have shown that cognitive-behavioral therapy (CBT) for treatment of insomnia (CBT-I) in adult populations has positive effects on insomnia and several sleep parameters (van Straten et al., 2018). CBT-I refers to a protocol that aims to improve the participant’s sleep by targeting both cognitive and behavioral components of the disorder (Edinger & Means, 2005). CBT-I for adults and adolescents alike include treatment techniques such as sleep restriction, stimulus control, changing negative thoughts about sleep through cognitive interventions, and relaxation (Epstein et al., 2012; Morin et al., 1999). The intervention can be delivered in various forms, such as face-to-face individual meetings, in group format or as self-help, for example, via books or via an Internet platform. The length of the program can vary, but for adults, four sessions seem to be a minimum to obtain good effects in adult CBT-I (van Straten et al., 2018).
The evidence base for cognitive and behavioral interventions for adolescent insomnia is growing, but further empirical support is needed. A systematic review and meta-analysis of psychological treatment for pediatric insomnia in 2014 showed that interventions in both controlled and non-controlled studies yielded small to large effect sizes for several sleep outcomes (Meltzer & Mindell, 2014). However, due to the limited number of studies that could be included, the evidence was weak for older children and adolescents. Some studies have indicated a positive effect of cognitive and behavioral interventions for adolescent insomnia as well (Blake et al., 2017). A positive within-group effect on several sleep parameters, as well as on measures of sleepiness and comorbid anxiety, was found. A later meta-analysis on cognitive and behavioral interventions for insomnia in adolescents (Aslund et al., 2018) showed positive between-group effects at post-intervention on total sleep time (TST) and sleep onset latency (SOL), favoring the intervention group (effect sizes ranging from d = 0.21 to d = 0.81). At short-term follow-up, positive effects were maintained and wake after sleep onset (WASO) was significantly improved. Thus, previous studies suggest a positive effect of CBT-I on sleep and comorbid disorders in adolescents, but the effect in a naturalistic clinical psychiatric environment, where insomnia might not be the primary reason for care, needs to be examined.
This study therefore aims to test the preliminary effects of CBT-I for adolescent insomnia with comorbid MDD, anxiety disorders, and/or chronic pain. It was hypothesized that (1) insomnia symptom severity would decrease pre- to post-CBT-I, with results maintained at 3-month follow-up and (2) SOL, WASO, TST, sleep efficiency (SE), and self-rated symptoms of depression, anxiety, functional disability, and current pain intensity would improve pre- to post-intervention and be maintained at follow-up. Another aim was to explore the patient acceptability of CBT-I in a clinical setting.
Methods
Design
The study was a non-controlled clinical pilot study. All included participants were assessed pre- and post-treatment and 3 months after the treatment ended (follow-up).
Ethics and consent
The Regional Ethical Review board of Stockholm approved the study (2012/871-31/1). All participants in the study provided written informed assent and their parent(s) or legal guardian(s) provided written informed consent in accordance with the Declaration of Helsinki.
Measures
The insomnia diagnosis was confirmed with a structured interview based on Diagnostic and statistical manual of mental disorders (4th ed.; DSM-IV) diagnostic criteria for sleep disorders (American Psychiatric Association, 1994). The Mini International Neuropsychiatric Interview for Children and Adolescents (MINI-KID; Sheehan et al., 1998), a semi-structured diagnostic interview based on DSM-IV, was used to diagnose comorbid psychiatric disorders.
Primary outcome: Insomnia symptom severity is measured by the Insomnia Severity Index–adolescent version (ISI-a) (Kanstrup et al., 2014), a seven-item self-report questionnaire with scores ranging from 0 to 28, higher scores indicating more severe insomnia. The adult version of the Insomnia Severity Index (ISI) is a well-used scale with adequate psychometric properties (Cronbach’s α = .74) and sensitivity to change (Bastien et al., 2001; Blais et al., 1997). Clinical cut-off for improvement after treatment of adult insomnia is –9.9 points (marked) and –8.4 (moderate), respectively, and a score below 8 indicates remission (Morin et al., 2011). ISI-a is an adapted version for adolescents with similar psychometric properties (Cronbach’s α = .88).
Secondary outcomes: To measure SOL, WASO, TST, and SE, a self-report sleep-wake diary was used for seven consecutive days/nights (Short et al., 2017).
Anxiety symptoms were measured by the well-validated Spence Children’s Anxiety Scale (SCAS) (Spence, 1998), a self-report questionnaire with scores ranging from 0 to 114 (higher scores indicate more severe symptoms). Cut-off for screening for clinical anxiety is ⩾22 points (Olofsdotter et al., 2016).
Depressive symptoms were measured by the Center for Epidemiological Studies Depression Scale for Children (CES-DC) (Faulstich et al., 1986), a 20-item self-report questionnaire. Scores range from 0 to 60, with higher scores indicating more severe symptomatology and with scores of ⩾30 used as cut-off for probable depressive disorder (Olsson & von Knorring, 1997).
Functional disability was measured with the Functional Disability Inventory (FDI) (Walker & Greene, 1991), a 15-item self-report questionnaire with scores ranging from 0 (no disability) to 60 (severe disability).
Current pain intensity was measured by a Visual Analogue Scale (VAS) (Carlsson, 1983), a continuous scale for magnitude estimation. It consists of a line where the ends are defined as extreme limits (0 = no pain; 10 = worst possible pain) of pain experience.
Treatment acceptability was examined primarily by investigating intervention retention.
Participants
Participants (n = 23) were patients from three child and adolescent psychiatric outpatient clinics and one pediatric pain clinic in Stockholm, Sweden. Individuals eligible for participation were aged between 13 and 17 years with adequate Swedish language skills, presenting insomnia symptoms at a clinical level, defined as more than 10 points on the ISI-a (Bastien et al., 2001; Morin et al., 2011) and meeting criteria for insomnia disorder according to Diagnostic and Statistical Manual of Mental Disorders (4th ed., text rev.; DSM-IV-TR) (American Psychiatric Association, 1994). As all individuals included in the study reported sleep difficulties at least three times per week, had adequate opportunity for sleep, and reported daytime impairments due to their sleep problems (as evidenced by data from pre-assessment sleep-wake diaries and questionnaires), participants would likely meet criteria for insomnia disorder (at least episodic) according to the DSM-5. Exclusion criteria were severe comorbid psychiatric disorders (e.g. bipolar disorder, psychotic disorder, autism spectrum disorder), severe suicidality, defined as ⩾17 points on MINI-KID suicide subscale, substance use disorder, any ongoing use of psychotropic medication, or psychological treatment for insomnia or other psychiatric disorder.
Recruitment procedure
Participants were recruited and treated between April 2015 and November 2017. ISI-a was used to screen for sleep problems. Due to logistical problems, records were not kept of the screened patients who did not present with sleep problems or who opted out of treatment. When ISI-a >10 points, patients were invited to a diagnostic interview. Following the interview, consecutive participants who met the criteria for inclusion were offered study participation. Participants completed baseline assessments (sleep-wake diary and questionnaires on insomnia, psychiatric symptoms, function, and pain) 1 week before treatment start, directly after treatment, and at 3-month follow-up (see Figure 1).
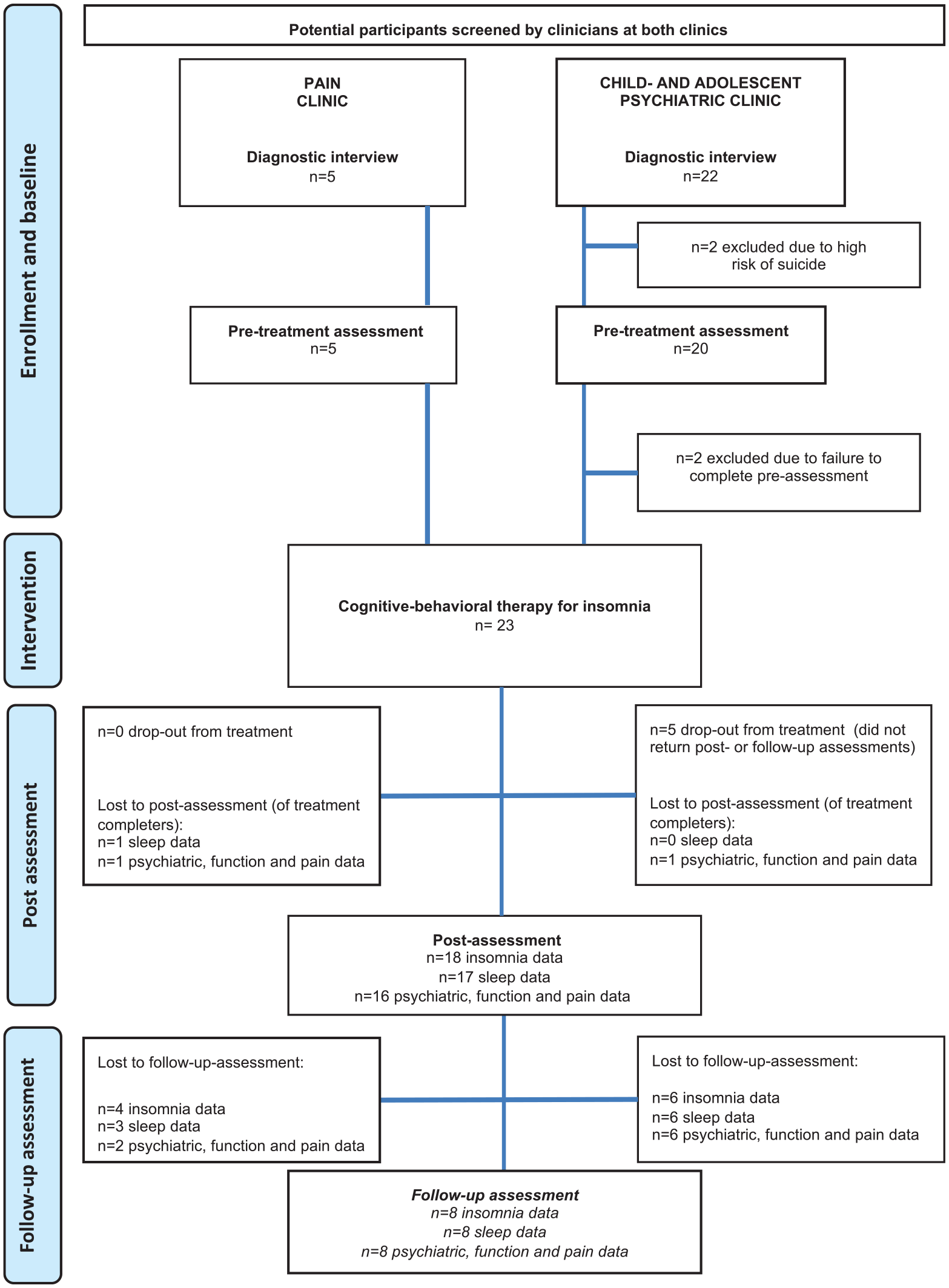
Flowchart.
Intervention: ySNOOZE
The ySNOOZE manual used in this study was based on well-supported manualized CBT-I treatment paradigms for insomnia in adults (Riemann et al., 2017) and adapted for adolescents by reducing the amount of psychoeducational information and increasing the focus on behavioral change. The ySNOOZE intervention consisted of 45-minute weekly individual face-to-face sessions with a therapist (clinical psychologist with > 18 months of CBT-training), following a pre-set structure (aim, agenda, review of homework from previous session, content of the session, new homework, introduction to next session) with specific time frames. For details, see Supplementary Table 1. Parents/caregivers were informed about the treatment, but did not take active part. A 4-hour seminar on the use of the ySNOOZE manual, as well as clinical supervision, was provided to the therapists (n = 6). Sleep restriction was introduced to all participants in session 2 and adjusted weekly based on calculated SE from sleep-wake diary registrations kept by participants throughout the treatment. Participants presenting suicidal thoughts were handled according to the regular routines of the clinic(s), as part of study procedure.
Statistical analyses
Missing data at post-treatment assessments were imputed using the multiple imputation procedure in SPSS 25 with missing values analyses performed to ensure that the missing data was random. Data were also assessed to ensure normal distribution. Paired samples t-tests were performed (intent-to-treat) to assess whether changes from pre- to post-treatment were statistically significant. The effect sizes of within-group changes between the assessment points were calculated as standardized Cohen’s d effect sizes (Cohen, 1992) with 95% confidence intervals (95% CIs). The within-group effect was considered small if d < 0.5, moderate if >0.8, and large if >1.1 (Öst, 2006).
Results
Baseline description of participants
Table 1 presents demographic information and baseline data for the 23 participants. For details, see Supplementary Table 2. Participants reported at least one, and on average 3.8 (4.4 for patients at the child-and adolescent psychiatric clinic, 1.7 for patients at the pain clinic), comorbid diagnoses according to MINI-KID, with the most common being MDD and anxiety disorders. A majority of participants (65% in total, 56% of psychiatric patients, and 100% of pain patients) reported long-standing (⩾12 months) pain with a mean pain duration of 34.4 (26.3) months. The majority (57%) of participants (80% of pain patients, 39% of psychiatric patients) reported not using prescription medication (see Table 2). Except for one subject, medication was stable between pre- and post-measurement. For details on medication use per clinic, see Supplementary Table 3.
Demographic information and baseline data for the participants.

ADHD: attention-deficit hyperactivity disorder; ADD: attention-deficit disorder.
Reported use of medication at baseline.
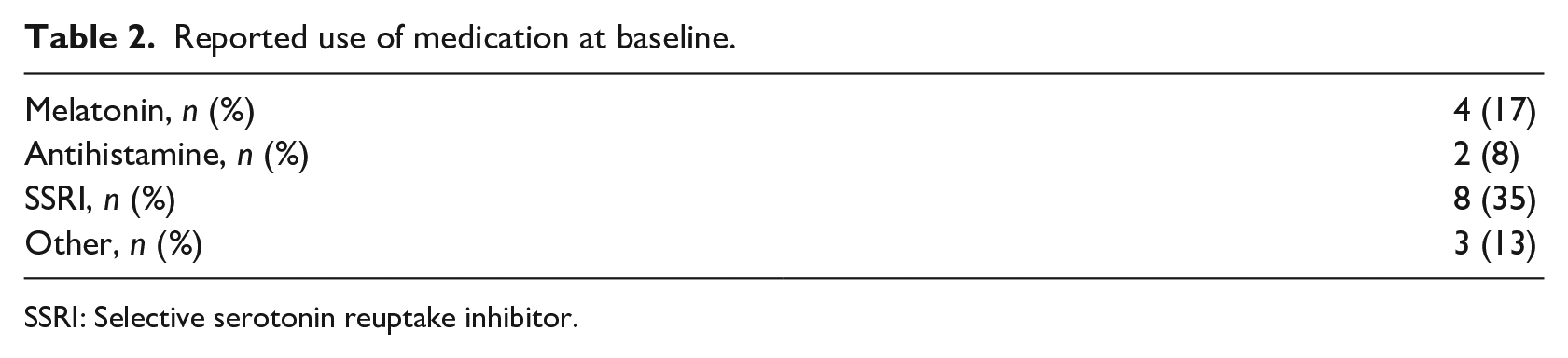
SSRI: Selective serotonin reuptake inhibitor.
Treatment acceptability and study attrition
The retention rate was 78% as 18 of the 23 participants attended all sessions of the treatment. Dropout occurred during the first three sessions of treatment (one participant after session 1, two after session 2, and two after session 3). At post-treatment, assessments were completed by 78% (insomnia), 74% (sleep), and 70% (psychiatric, function, and pain) of the original sample and at follow-up by 35% (all measures). Reporters at follow-up did not differ from non-reporters, with regard to recruitment site, age, sex, or symptom levels (insomnia, depression, anxiety, functional disability, pain) at pre- and post-assessment (data not shown). Means and standard deviations of follow-up data are reported for descriptive purposes only, due to large amounts of missing data making imputations unreliable (Schafer & Graham, 2002).
Treatment outcomes
Changes in insomnia symptom severity and sleep pre- to post-treatment
Means, standard deviations, and effect size with 95% CI for all outcomes are reported in Table 3. There was a statistically significant improvement in insomnia symptoms from pre- to post-treatment (t(132) = 18.86, p < .001, d = 1.63). For details on ISI-a scores of each participant, see Figure 2. There was also a significant reduction in SOL (t(126) = 11.71, p < .001; d = 0.99) and WASO (t(126) = 4.32, p < .001; d = 0.42) as well as a significant increase in TST (t(126) = −2.46, p = .015, d = 0.22) and SE (t(126) = −11.13, p < .001; d = 0.8).
Treatment outcome measures, estimated (post) and observed (pre, follow-up) data.
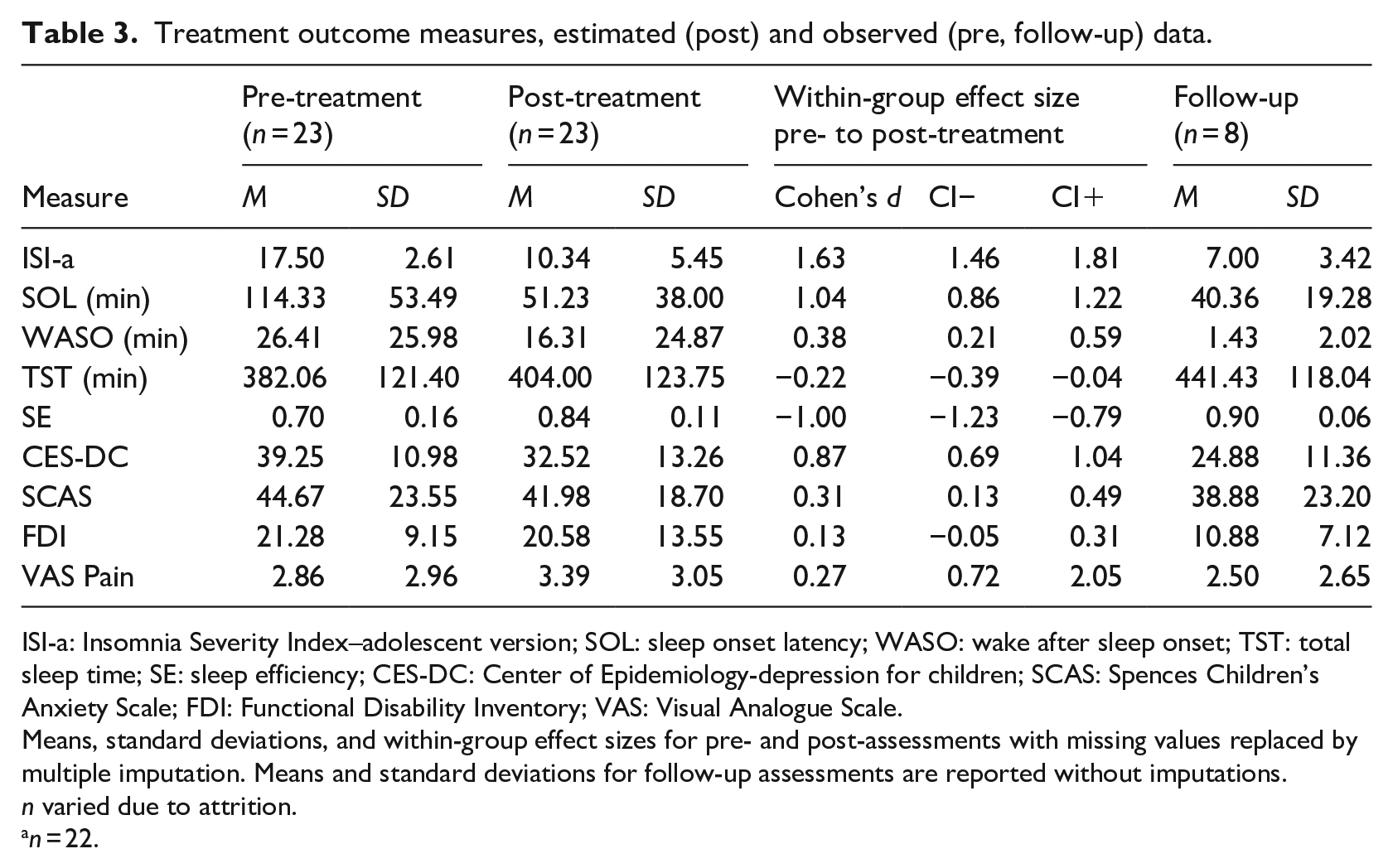
ISI-a: Insomnia Severity Index–adolescent version; SOL: sleep onset latency; WASO: wake after sleep onset; TST: total sleep time; SE: sleep efficiency; CES-DC: Center of Epidemiology-depression for children; SCAS: Spences Children’s Anxiety Scale; FDI: Functional Disability Inventory; VAS: Visual Analogue Scale.
Means, standard deviations, and within-group effect sizes for pre- and post-assessments with missing values replaced by multiple imputation. Means and standard deviations for follow-up assessments are reported without imputations. n varied due to attrition.
n = 22.
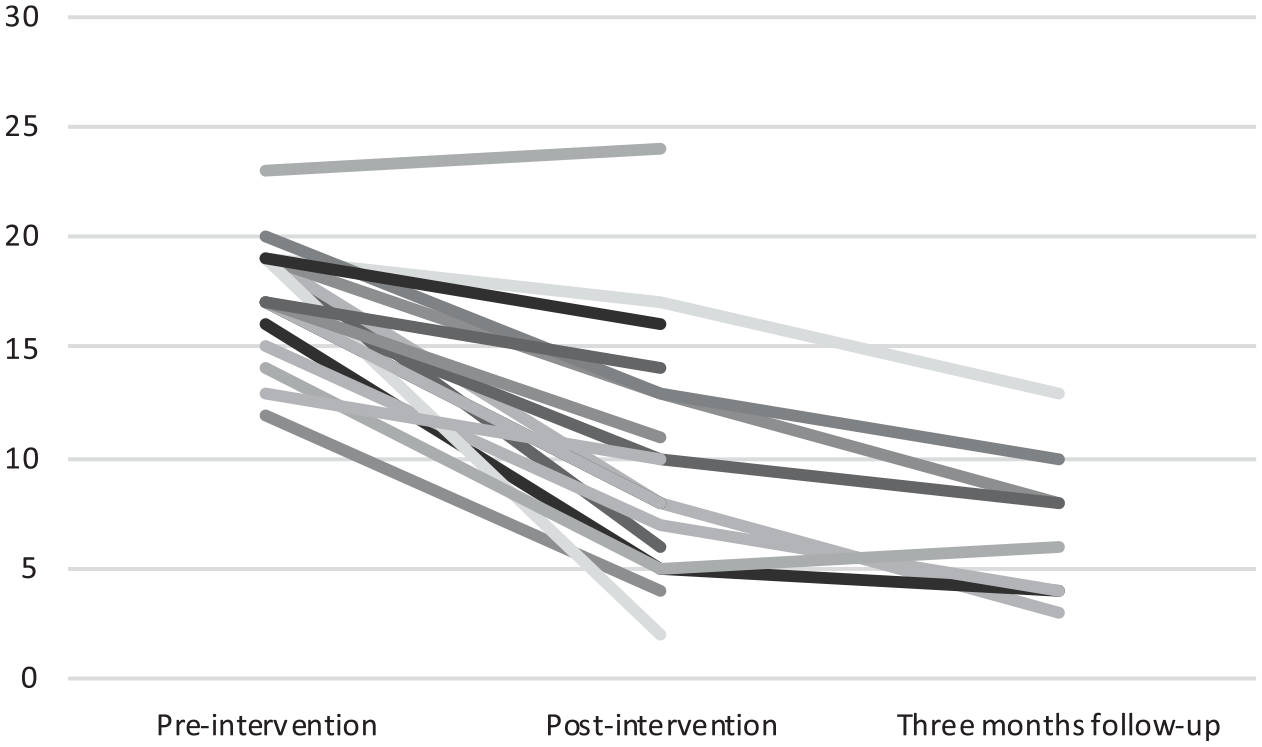
Insomnia symptom severity (as measured by the Insomnia Severity Index–adolescent version) for each participant at all three assessments.
Changes in comorbid symptoms, function, and current pain intensity pre- to post-treatment
Symptoms of depression showed statistically significant improvements from pre- to post-treatment (t(125) = 9.74, p < .001, d = 0.87) as did symptoms of anxiety (t(125) = 3.47, p = .001, d = 0.31). Scores of functional disability and current pain intensity showed no statistically significant change from pre- to post-treatment.
Changes in insomnia symptom severity, sleep, comorbid symptoms, function, and current pain intensity post to follow-up
For the eight participants who provided data, improvements were maintained at follow-up, as can be seen in Table 3, and functional disability scores were greatly reduced. There was a small numeric reduction in current pain intensity at follow-up compared to post-treatment assessment, but not compared to pre-treatment assessment.
Sensitivity analyses
To address study site variation and influence of imputations, sensitivity analyses were performed on measures of insomnia, depression, and anxiety. To test if changes in symptoms differed between the recruitment sites, independent sample t-tests on change-scores (pre to post) were performed. Differences in change-scores were not statistically significant for either measure (data not shown). To test if imputations of data influenced the results, dependent sample t-tests were performed on “last observations carried forward” values for missing post-data. Pre to post changes were statistically significant for insomnia and depression but not for anxiety (data not shown).
Discussion
This study examined changes in insomnia symptom severity after a CBT-I in patients with insomnia and comorbid psychiatric disorders and chronic pain. In addition, changes in SOL, WASO, TST, SE, as well as comorbid symptoms of anxiety, depression, functional disability, and pain were assessed. The main finding was a significant reduction in symptoms of insomnia between pre- and post-treatment, with a large effect size (d = 1.63). This is in line with previous findings in adolescents (de Bruin et al., 2015) and adults (van Straten et al., 2018). The study also showed a significant reduction of SOL and WASO, and a significant increase in TST and SE from pre- to post-treatment, with effect sizes ranging from small to medium (d = 0.22 to 1.04). In all, the results give preliminary support for the use of CBT-I in an adolescent clinical population, which is in line with previous findings on adolescents (Aslund et al., 2018). Wakefulness in bed parameters (SOL, WASO) exhibited larger changes than sleep parameters (TST), which is also in line with previous findings in this age group (Aslund et al., 2018).
Importantly, reductions in symptoms of depression and anxiety, with effect sizes ranging from small to medium, were also found after the intervention as compared to before, in line with what has previously been found in adults (Ye et al., 2015). In contrast, there was no significant improvement in functional disability. Previous studies examining interventions for adolescents with insomnia have not included measures of general function, although it has been shown that the degree of insomnia symptoms is related to functional disability, with greater reduction in function associated with higher levels of insomnia symptoms (Kanstrup et al., 2014). Regarding current pain intensity, no statistically significant changes were seen between pre- and post-assessments. A study on CBT-I in adolescents with migraine headaches showed a significant reduction in number of days with headache following treatment (Law et al., 2018) and future studies on CBT-I might include a similar measure to provide a complementary and possibly more sensitive estimate of the treatment effect.
The available data suggest that the positive changes in all measures may be retained 3 months after intervention. At follow-up, scores of functional disability were greatly reduced for those who provided data. This may indicate a delay in the improvement of general function following CBT-I. However, the continued improvement at follow-up could reflect that the participants who reported follow-up were more engaged in the treatment than non-reporters. No noticeable reduction in pain intensity was observed at follow-up when compared to pre-treatment. Although attrition analysis showed that participants who did not complete follow-up assessment were similar to those who did, the high attrition rate severely limits the generalizability of these results, and they should only be considered as hypothesis generating.
Regarding clinical significance, insomnia symptom severity showed a reduction of mean score by 7.2 points, approximating what is considered a clinically significant improvement in adults (Morin et al., 2011). Of the 18 completers, three participants (17%) reported a >8.4 point (= moderate) improvement of insomnia symptom score from pre- to post-treatment, and four participants (22%) reported a >9.9 (= marked) improvement (Morin et al., 2011). Regarding changes in sleep parameters following treatment, it should be noted that even though participants fell asleep faster after CBT-I, the average SOL at post-treatment (51 minutes) was still above the cut-off for what is considered relevant for insomnia disorder (>31 minutes) (Lichstein et al., 2003) and what is regarded as an indicator of poor sleep (>45 minutes) (Ohayon et al., 2017). Good SE (>0.85) (Ohayon et al., 2017) was obtained by nine participants after treatment, but the group average was slightly below (SE = 0.84). Regarding clinical significance of comorbid psychiatric symptoms, the mean score after treatment was still above the cut-off for depression (Olsson & von Knorring, 1997) and anxiety (Olofsdotter et al., 2016), even though the symptom reduction was statistically significant. For those reporting at follow-up, six participants (75%) reported levels of insomnia below the threshold of clinical significance (>10 points) (Morin et al., 2011). Mean SOL for the participants reporting at follow-up was 40.4 minutes, that is, <45 minutes, corresponding to an indicator of good sleep (Ohayon et al., 2017), and mean SE was 90%, indicating good SE. This could indicate that improvements in sleep may be gradual in this patient group.
Regarding comorbid symptomatology, an encouraging finding of the study is that symptoms of depression and anxiety were reduced after treatment, as previously discussed. Findings in adults suggest that patients with both diagnoses benefit more from CBT-I than from CBT for depression (Blom et al., 2015), indicating that insomnia needs specific treatment also when co-occurring with depression. A question for future research is whether the same reasoning holds true in adolescents; if CBT-I might be more beneficial than CBT for depression for adolescents with comorbid insomnia and depression.
As attrition occurred between pre- and post-assessments, imputations were performed and could possibly have masked differences between reporters and non-reporters. Sensitivity analyses indicated that this was not the case for insomnia severity and depression but possibly for anxiety. Sensitivity analyses on recruitment site differences were nonsignificant, indicating a similarity between patients rather than a difference. Other similarities are the fact that a majority of psychiatric patients also reported long-standing pain and all pain patients reported at least one comorbid psychiatric problem.
Regarding treatment acceptability, the retention rate was similar to that shown in a study of CBT-I in adolescents with comorbid traumatic brain injury (Lah et al., 2019) as 80% of participants attended all sessions. Dropout or non-adherence to CBT-I (Riedel & Lichstein, 2001) is common and particularly the sleep restriction component is often perceived as highly aversive (Kyle et al., 2011). During treatment, sleep time often decreases (Kyle et al., 2014) and patients with comorbid depression and anxiety may be sensitive to shifts in motivation to pursue long-term goals. It is possible that patients in this clinical context would benefit from more therapist support for increased motivation to adhere to treatment, as has been suggested for adults with attention-deficit hyperactivity disorder (ADHD) and sleep problems (Jernelov et al., 2019). Another format, such as internet-delivered treatment, may increase acceptability and retain a larger group of adolescents in treatment.
As noted above, the attrition rate was high from post-measurement to follow-up. In future studies, data collection could be facilitated by using online questionnaires with automatic reminders. A recommendation would also be inclusion of qualitative data to increase the understanding of the participants’ experiences and to let them inform the intervention, as has been done with adults (Blom et al., 2016). It should also be noted that no objective measure was used to assess sleep as treatment outcome in this study. However, as insomnia is diagnosed on self-reported indication of poor sleep quality, self-reported measures are often regarded as the most adequate way to measure intervention effect in adults (Lichstein et al., 2003). An important strength of this is the inclusion of participants with multiple comorbid disorders that were treated in outpatient clinics, as part of regular care.
The main limitations of this pilot study are the non-controlled design and the small sample size, and conclusions about causality and effects of the intervention cannot be drawn. However, the present findings are promising and support the notion that CBT-I may be used to treat insomnia in adolescents with high levels of comorbid symptomatology and that symptoms of depression and anxiety may improve after treatment. This is a promising base for a future randomized controlled trial. The results should be interpreted with caution, and well-powered controlled clinical trials, ideally with long-term follow-ups, are necessary to validate these preliminary findings.
Conclusion
This study provides evidence that insomnia symptoms and sleep parameters improve following CBT-I delivered in a clinical setting to adolescents with comorbid depression, anxiety, and pain. It also indicates that depression and anxiety symptoms improve following CBT-I in this population. These results are important, since adolescent patients with comorbid psychiatric conditions constitute a highly burdened clinical population. To further develop the field of CBT-I in adolescents, clinical populations should be included in large randomized controlled trials, including monitoring of both comorbid problems and measures of overall daytime function, and with a design and sample size that allow for meaningful analyses of mediating and moderating factors.
Supplemental Material
sj-pdf-1-ccp-10.1177_1359104520929376 – Supplemental material for Cognitive-behavioral therapy for insomnia in adolescents with comorbid psychiatric disorders: A clinical pilot study
Supplemental material, sj-pdf-1-ccp-10.1177_1359104520929376 for Cognitive-behavioral therapy for insomnia in adolescents with comorbid psychiatric disorders: A clinical pilot study by Lie Åslund, Mats Lekander, Rikard K Wicksell, Eva Henje and Susanna Jernelöv in Clinical Child Psychology and Psychiatry
Supplemental Material
sj-pdf-2-ccp-10.1177_1359104520929376 – Supplemental material for Cognitive-behavioral therapy for insomnia in adolescents with comorbid psychiatric disorders: A clinical pilot study
Supplemental material, sj-pdf-2-ccp-10.1177_1359104520929376 for Cognitive-behavioral therapy for insomnia in adolescents with comorbid psychiatric disorders: A clinical pilot study by Lie Åslund, Mats Lekander, Rikard K Wicksell, Eva Henje and Susanna Jernelöv in Clinical Child Psychology and Psychiatry
Supplemental Material
sj-pdf-3-ccp-10.1177_1359104520929376 – Supplemental material for Cognitive-behavioral therapy for insomnia in adolescents with comorbid psychiatric disorders: A clinical pilot study
Supplemental material, sj-pdf-3-ccp-10.1177_1359104520929376 for Cognitive-behavioral therapy for insomnia in adolescents with comorbid psychiatric disorders: A clinical pilot study by Lie Åslund, Mats Lekander, Rikard K Wicksell, Eva Henje and Susanna Jernelöv in Clinical Child Psychology and Psychiatry
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
Author biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
