Abstract

Carotid intima-media thickness (cIMT) is considered a marker of subclinical atherosclerosis. However, due to the variability of the tunica media thickness (cMT), which primarily consists of smooth muscle cells, 1 the accuracy of cIMT has been questioned, leading to its exclusion from recent clinical guidelines as a marker of cardiovascular risk. 2 Conversely, recent ultrasound studies have demonstrated the feasibility of measuring the thickness of cMT and carotid intima (cIT), the latter being more closely associated with atherosclerosis.1,3,4
Oxidized low-density lipoprotein (oxLDL) is strongly involved in atherogenesis. 5 Though circulating oxLDL levels are independently associated with increased cIMT in various populations,6,7 this association was not found in hypertensive patients.8,9 This lack of association could be explained by the fact that cIMT increases due to elevated blood pressure (BP) may be derived from cMT growth 4 and by the common use of statins among hypertensive patients, which may influence LDL circulating levels. To tackle these issues, the present study evaluated the relationship of oxLDL with carotid wall layers in hypertensive individuals, splitting the sample according to statin use.
This study measured plasma oxLDL levels in a convenience sample of 117 consecutive hypertensive patients (38 not using and 79 using statins) aged > 18 years followed at a university outpatient clinic who underwent clinical, laboratory, and carotid ultrasound analysis. The study was approved by the institution’s Ethics Committee and all participants gave written informed consent. Plasma samples collected after 12 hours of fasting were used to assess oxLDL levels using the ELISA method (kit 10-1143-01; Mercodia, Uppsala, Sweden) 7 and routine laboratory parameters.
High-resolution images of the left and right common carotid arteries (CCA) 2 cm proximal to carotid bifurcation were obtained as previously reported by a qualified physician using a Vivid-q device (General Electric, Milwaukee, WI, USA) equipped with a linear transducer (12L-RS) set at 10 MHz1,3,4 (Supplemental Figure 1). For each image, five measurements of cIMT, cMT, and cIT were performed manually on plaque-free areas using ImageJ software (NIH, Bethesda, MD, USA), and the average of both CCA measurements was used to calculate each layer’s thickness. Carotid plaques were defined as: CCA cIMT ≥ 1.5 mm, or focal wall thickening of 0.5 mm, or encroaching into the lumen by > 50%.
Continuous data with normal and nonnormal distribution are presented as mean ± SD and median (25th, 75th percentiles), respectively, and categorical variables are presented as proportions. Significance was set at p < 0.05. Analyses were conducted in Stata 14.2 (StataCorp LLC, College Station, TX, USA).
The characteristics of the whole sample (n = 117; 59.7 ± 11.4 years, 44% men) and in individuals not using (n = 38; 55.9 ± 12.8 years, 45% men) or using statins (n = 79; 61.5 ± 10.2 years, 44% men) are shown in Supplemental Table 1. Average oxLDL, cIMT, cMT, and cIT levels were 82.2 ± 48.2 U/L, 0.731 ± 0.145 mm, 0.471 ± 0.101 mm, and 0.260 ± 0.066 mm, respectively, in statin nonusers, and 69.6 ± 36.4 U/L, 0.749 ± 0.130 mm, 0.483 ± 0.103 mm, and 0.266 ± 0.056 mm, respectively, in statin users (Supplemental Table 1).
The results of multivariable linear and logistic regression analysis adjusted for traditional proatherogenic risk factors, such as age, sex, systolic BP, body mass index, diabetes, smoking, LDL-cholesterol (LDL-C), and antihypertensive medications showed that oxLDL was associated with cIT (p = 0.007; Figure 1A) and cIMT (p = 0.019; Figure 1C), but not with cMT (Figure 1B) or carotid plaques (Figure 1D) in statins nonusers, and did not associate with carotid layers or plaques in statins users (Figure 1A–D). There was a significant interaction for the association of oxLDL with cIT (p = 0.010) but not with cIMT (p = 0.21) (Figures 1A and 1C), indicating a significant difference in the relationship of oxLDL with cIT, but not of oxLDL and cIMT, between statin users and nonusers. Conversely, LDL-C showed no independent association with carotid wall layers or plaque, whereas the association between oxLDL and cIT remained significant after the inclusion of glucose (p = 0.011) or removal of LDL-C (p = 0.007) in the model. Among statin users, 65% used low-potency statins, but multivariable regression analysis showed no association between oxLDL and cIT in this subgroup of patients (p = 0.42).
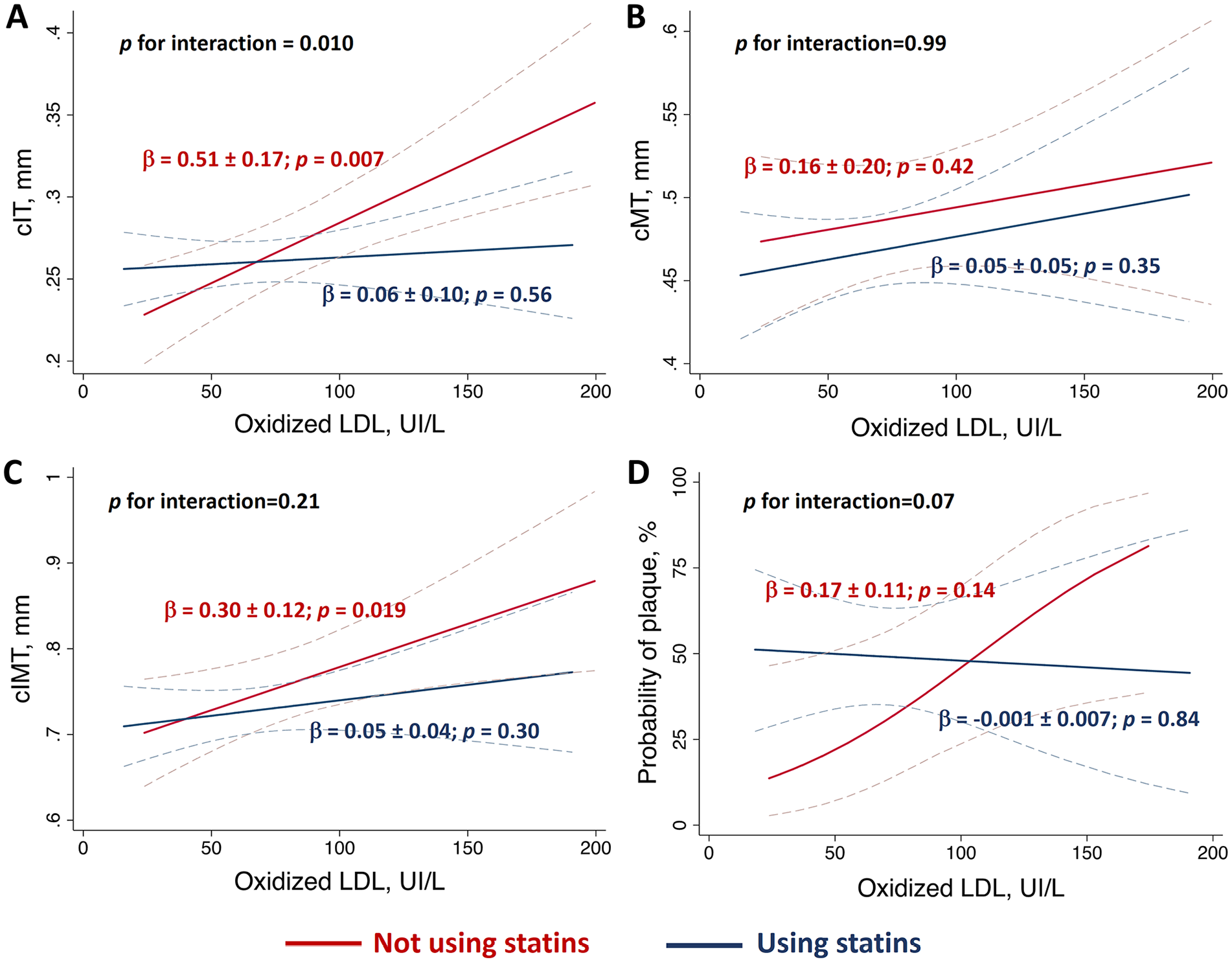
Relationship between circulating oxidized LDL levels and
The present study provides novel evidence that circulating oxLDL levels were independently related to cIT and cIMT, but not to cMT, in hypertensive patients not using statins. These data might have clinical implications. First, because cIT is a more accurate marker of atherosclerosis than cMT,1,3 they suggest that oxLDL might play a role in atherogenesis among hypertensive patients as well. In this regard, higher circulating oxLDL levels were reported in hypertensive individuals compared to normotensive ones, 9 strengthening the notion that lipoprotein oxidation is involved in the pathophysiogenesis of atherosclerosis in this population. Second, given that cMT is the major component of cIMT in hypertensive patients,1,10 the absence of relationship between oxLDL and cMT might contribute to explain the lack of association between oxLDL and cIMT reported in alternative hypertensive populations.8,9 Third, our findings suggest that statins, by lowering oxLDL levels, may confound the association of oxLDL with cIT and cIMT.
The present study has limitations. Owing to its cross-sectional design, the observed associations cannot be assumed as causal. Additionally, our sample comprised hypertensive patients followed at a tertiary hospital, thus limiting the generalizability of our results to other hypertensive populations.
In conclusion, our data demonstrated that circulating oxLDL levels were related to cIT, but not to cMT, in hypertensive patients not using statins. These results extend our understanding of the pathophysiological role of oxLDL on preclinical atherosclerosis in hypertensive patients.
Supplemental Material
sj-pdf-1-vmj-10.1177_1358863X251323516 – Supplemental material for Association of oxidized LDL with carotid wall layers in hypertensive patients
Supplemental material, sj-pdf-1-vmj-10.1177_1358863X251323516 for Association of oxidized LDL with carotid wall layers in hypertensive patients by Saturne Barthold, Eduarda OZ Minin, Luís FRS Carvalho-Romano, Edmilson R Marques, Layde R Paim, Elisangela CP Lopes, Camila FL Vegian, José A Pio-Magalhães, José R Matos-Souza, Otavio R Coelho-Filho, Andrei C Sposito, Wilson Nadruz and Roberto Schreiber in Vascular Medicine
Footnotes
Data availability statement
All data presented in this study are available upon request to the author for correspondence, and the raw data will be archived in an Institutional Data Repository.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Dr Roberto Schreiber was supported by São Paulo Research Foundation (FAPESP 2017/23563-1).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
