Abstract

What is a popliteal artery aneurysm (PAA)?
The popliteal artery is an important blood vessel located behind the knee. It starts as the continuation of the superficial femoral artery (located in the thigh). Below the knee, it splits into two branches: the peroneal artery and the tibioperoneal trunk. These branches supply oxygen-rich blood to the muscles of the lower legs, ankles, and feet.
The term ‘arterial aneurysm’ refers to the abnormal weakening of the arterial walls causing the artery to balloon out to more than 1.5 times its normal diameter. 1 The normal popliteal artery measures 7–11 mm in diameter, being generally larger in men. Therefore, a PAA is typically diagnosed when the artery is larger than about 15 mm, as shown in Figure 1.
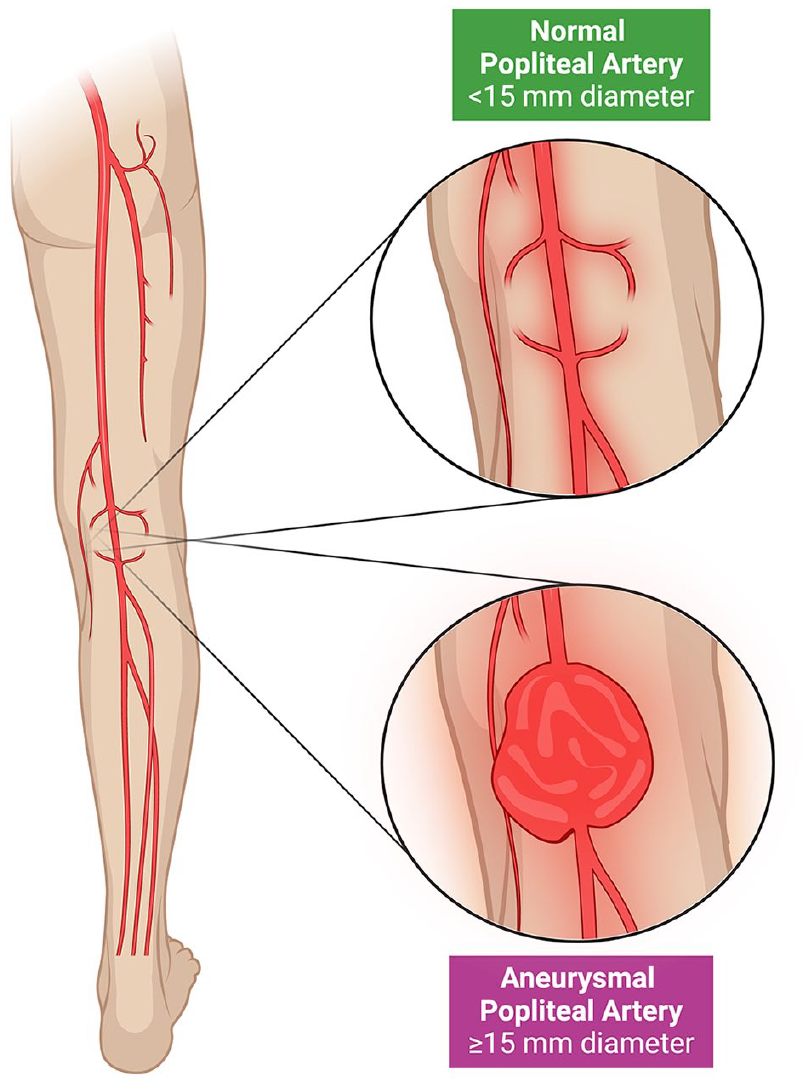
A normal popliteal artery and an aneurysmal popliteal artery.
What causes PAA and who is at risk for PAA?
Although aneurysms can occur in any artery of the body, PAAs are the most common type of aneurysm in the peripheral arteries to the limbs. PAAs are rare, present in only 0.1–1% of the general population. Studies have shown that PAAs are more common in men (95% of the cases), between the ages of 50 and 70 years old. 2
Several risk factors have been associated with the development of PAAs:
Male sex
Advanced age
Smoking
Hypertension
Presence of other aneurysms in the body, especially in the aorta
Diabetes (although present in around 15% of patients with PAAs, diabetes has also been shown to be associated with slower aneurysm growth).
These risk factors also predispose to the development of arterial aneurysms in other blood vessels. In fact, around half of patients with PAA have a PAA in both legs, and approximately 40% of patients with PAA also have an abdominal aortic aneurysm (AAA).1,2 Therefore, all pa-tients diagnosed with a PAA should also be screened for a PAA in the other leg and for AAA. These screenings are usually accomplished with vascular ultrasound.
What are the symptoms and potential complications of a PAA?
About 30–40% of people with PAAs do not experience any symptoms. These cases are typically discovered by chance during imaging tests conducted for unrelated reasons. In some cases, a doctor may detect them during a physical examination when they notice a pulsating mass behind the knee, even if the patient has no symptoms or discomfort.
Symptoms and complications from PAA can be diverse and occur by different mechanisms, 1 as shown in Figure 2.
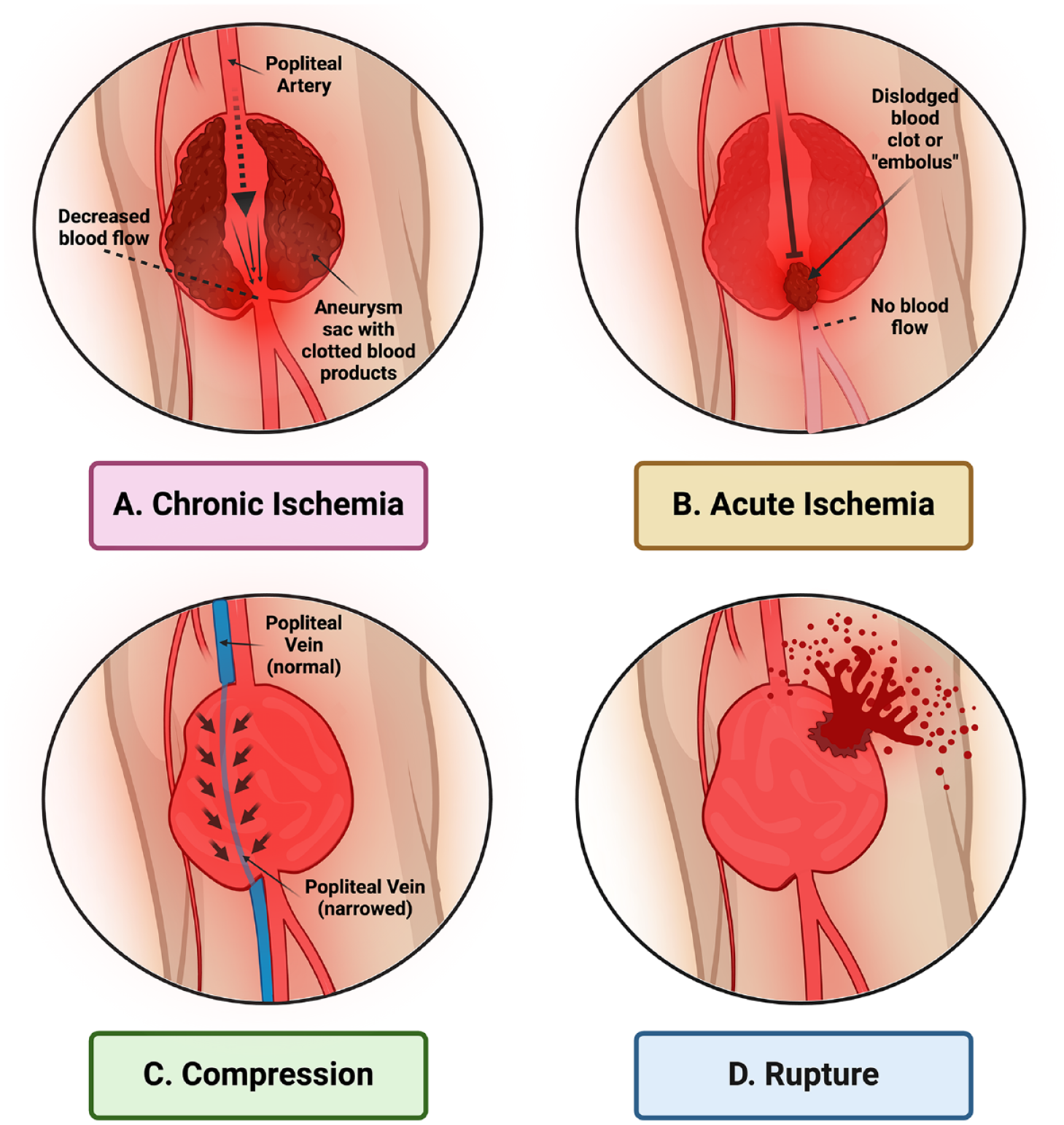
Possible complications of a popliteal artery aneurysm.
Chronic ischemic symptoms
‘Chronic ischemia’ refers to a slow decline in blood supply to tissues over a long period of time, leading to insidious symptoms. This usually occurs due to progressive accumulation of blood clots within the aneurysm (known as ‘thrombosis’), narrowing, or even completely closing the artery and blocking the passage of blood. When the amount of blood reaching the foot and calf muscles cannot keep up with activity, like walking a distance, patients often experience crampy pain known as ‘claudication’. As chronic ischemia worsens, patients may also feel pain at night even when resting, notice discoloration in their toes, or develop wounds that do not heal over time.
Acute ischemic symptoms
In contrast to chronic ischemia, ‘acute ischemia’ refers to a sudden interruption of blood supply. This scenario is more common with PAA and is a medical emergency, as the tissue has significantly less time to adapt to the abrupt lack of oxygen. Acute ischemia can result from rapid development of thrombosis (clot) within the PAA or an ‘embolus’, where part of a clot that formed within the PAA breaks off and travels downstream to block a major artery, completely cutting off its blood flow. Symptoms can include sudden pain, paleness, loss of pulses on the foot, numbness, tingling, or a cold foot.
Local compressive symptoms
When the aneurysm is very large, it can press on nearby structures behind the knee. If it compresses nerves, then patients may experience numbness or difficulty moving their feet. If it compresses nearby veins, then it can cause varicose or dilated veins and swelling in the lower leg.
Aneurysm rupture
Rupture of a PAA is a rare but serious complication. Patients typically experience sudden and severe pain and swelling behind the knee and in the surrounding area because of the accumulation of blood. The risk of rupture is higher with large aneurysms, and it can also result in symptoms of acute ischemia because the blood flow is abruptly cut off.
How is a PAA diagnosed?
Normally, the popliteal arteries are somewhat difficult to feel on physical examination, especially in patients with a large amount of underlying fatty tissue. Thus, confirmatory imaging tests are usually required. Imaging options include vascular ultrasound, computed tomography angiography (CTA), or magnetic resonance angiography (MRA).
A duplex (vascular) ultrasound is a relatively inexpensive noninvasive study that can accurately detect PAAs and monitor their size. 3 A probe with gel is placed on the skin, and sound waves are used to visualize and measure the PAA.
CTA and MRA are more complex and can provide specific anatomic details that help to plan the surgical repair of PAAs. 3 In these studies, the patient lies down on a bed that slides into a scanner. The scanner takes pictures from different angles and puts them together to create a high-quality image. However, there are some downsides, such as the need for contrast injection (which can affect the kidneys), radiation exposure with CTA, and limitations for MRA when there are metallic implants in the body.
What other conditions may affect the popliteal artery area?
Symptoms of chronic ischemia, such as claudication (leg pain with walking), nonhealing wounds, and toe discoloration, can also be caused by other conditions. Therefore, it is crucial to distinguish a PAA from these conditions as their management varies:
What are the treatment options for PAA?
Treatment of PAA includes medical management (lifestyle modification and medication), and if necessary, surgical intervention (endovascular or open approaches).
Medical management
Medical management focuses on following a healthy lifestyle and reducing the risk factors that contribute to the development of vascular disease to reduce overall cardiovascular risk:
Smoking cessation
Blood pressure control
Diabetes treatment
Cholesterol management, typically with a statin
Consideration of aspirin for heart attack and stroke prevention, depending on risk of bleeding
Heart-healthy diet
Maintaining a normal weight
At least 30 min/day of moderate-intensity exercise.
If the PAA is smaller than 2 cm and the patient has no symptoms, it is reasonable to start with medical treatment and annual ultrasound monitoring of the PAA. Many patients with PAA will develop symptoms at some point (up to 70% within 5 years), so close follow-up care is important, including follow-up ultrasound studies to monitor PAA size.3,6,7
Surgical intervention
If the PAA measures 2 cm or more, or if the patient has symptoms or complications, then surgical intervention is recommended, which may be performed as an open repair or endovascular repair (Figure 3).
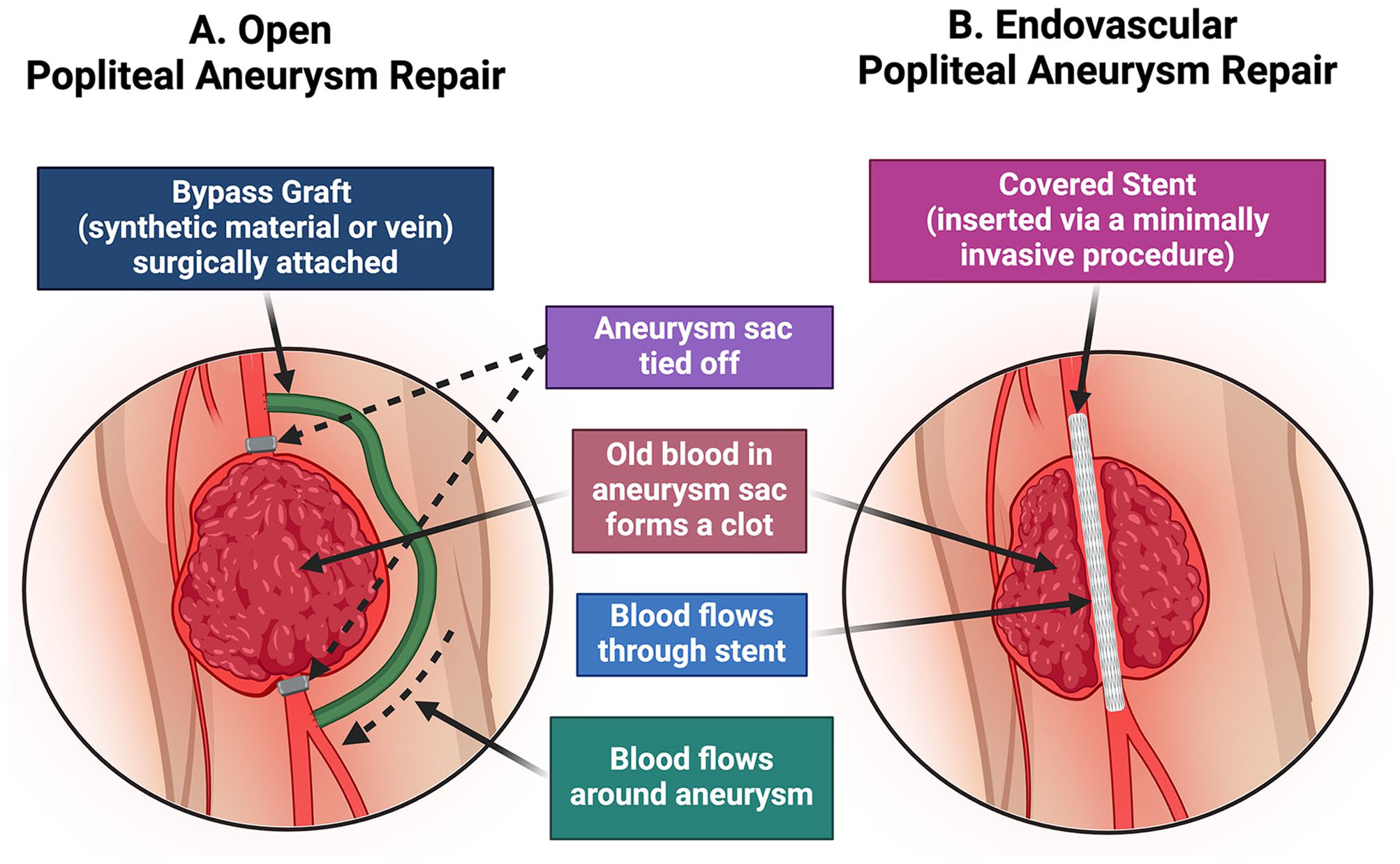
Interventional options for popliteal artery aneurysm repair.
In patients who are good candidates for open surgery, open popliteal aneurysm repair is recommended. 3 During this surgery, an incision is made on the inner aspect of the leg. The vascular surgeon creates a bypass (a connection between the artery segments that are above and below the aneurysm), skipping the dilated portion of the artery to re-route the blood flow. This ‘alternative route’ is created by using a vein graft from the same patient. If no suitable vein is available, a synthetic graft can also be used. In most cases, the aneurysm is also ligated (tied off) from above and below, blocking the blood flow to prevent further aneurysm growth. In some cases, the surgeon can opt for a different approach depending on the aneurysm characteristics. Possible risks of open repair include postoperative pain, complications of general anesthesia, bleeding, infections, hematomas (blood collection), and thrombosis (clot formation in the bypass). 6
If the patient is not a good candidate for open surgery, an endovascular popliteal aneurysm repair should be considered. 3 In this procedure, instead of making a large incision, a tiny puncture is made in the groin area. Using this entry point, the vascular specialist inserts a wire and special tubes while watching on a monitor with real-time X-rays. A special dye (intra-venous contrast) helps to identify the aneurysm. Once the aneurysm is accessed, a metallic stent is deployed, which becomes the new route for the blood flow. It is like putting a strong pipe inside a damaged one so that water does not press against the damaged walls, preventing the aneurysm from getting bigger. Of note, this approach should be avoided in younger and active patients, as repetitive knee-bending can cause stent damage over time. 6
Open repair is the approach of choice because the bypass graft is more likely than the stent to stay open and free from new blockages. 6 However, as it involves open surgery, it also has a higher risk of complications such as bleeding, infection, and issues related to general anesthesia. Surgery also requires a longer stay in the hospital. In contrast, the endovascular approach is less invasive with a shorter hospital stay, but it is less durable in the long run.
Of note, acute ischemia from PAA is an emergency situation and needs to be addressed promptly. Blood thinners (anticoagulation) should be started immediately to stop the progression of the clot. An intervention to remove the clot and to restore the blood flow is then followed by aneurysm repair to prevent these episodes in the future.
Summary
PAA is a relatively rare condition involving the dilation of the popliteal artery behind the knee. Ultrasound is often used to diagnose and monitor PAA. Medical management is crucial to slow the progression of the PAA and to reduce overall cardiovascular risk. Open surgery provides definitive treatment of the aneurysm to avoid complications.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Dr Elizabeth Ratchford’s work was supported in part by the generosity of David Kotick (1926–2021).
